Abstract
Introduction
The availability of different triptans for acute migraine treatment poses the problem of the choice. In clinical practice, the physician should be able to prescribe the triptan in the most suitable dosage and formulation for each patient, in order to optimise the treatment (1). Nevertheless, there are no dynamic or kinetic characteristics making it possible to foresee which patient will respond better to which triptan (2). All triptans have the same mechanism of action and the clinical importance of kinetic differences is not clear (3). These medications are effective and safe in the absence of contraindications, but it seems that there is no triptan that is always effective for most headache patients (4). The maximum response to triptans taken orally, measured as pain relief at two hours, is approximately 70%, with less than 2 of 3 patients responding in 3 of 3 attacks (5). A patient non-responder to a triptan can benefit from another one or from the same drug, if administered by a different route (5,6). However, if the medication is not effective the first time, it is improbable that it will be used again (7). Indeed, a large number of patients take only one prescription of triptans (8,9). To sum up, even though the physician prescribes the drug, it is the patient who decides whether to take it, and whether to continue taking it (10).
In Italy, six triptans are available: sumatriptan, zolmitriptan, and rizatriptan, which have been available for at least 10 years, and eletriptan, almotriptan, and frovatriptan, which have been available for at least five years. In clinical practice, these medications are not much prescribed as yet (11,12), while triptans are the most commonly prescribed class of acute medications in headache specialty care (10,13). The aims of our study were: (1) to examine the pharmacoepidemiology of triptans in migraine patients attending a headache centre; and (2) to analyse any differences between patients continuing to use triptans and patients who, after having taken these drugs, discontinued them.
Patients and methods
Patients
We enrolled male and females outpatients older than 18 years consecutively examined during a follow-up visit at the Headache Centre of the University Hospital of Modena from October 2008 to March 2009. We included only patients who had been diagnosed with migraine (episodic or chronic) according to the International Classification of Headache Disorders Second Edition (ICHD-II) criteria (14) at least three years before, and had used at least once in the previous three years, one or more dose of a triptan, in any formulation, or were currently using a triptan as acute treatment. The criterion to define triptan discontinuation was that the patient declared, on the questionnaire, to have discontinued triptan(s) at least three months before (which also emerged from the diaries written in the three months preceding the follow-up visit). The criterion to define current use was that the patient declared to have been using triptan(s) for at least three months (which also was recorded in the diaries written in the three months preceding the follow-up visit). Triptans could have been prescribed by the general practitioner, a physician at the headache centre or any other physician. Patients with secondary headache, tension-type or cluster headache were excluded.
All patients were asked for their written informed consent to take part in the study; none of them refused to give it. The study was approved by the Ethical Committee of Modena and conducted in compliance with the latest version of the Declaration of Helsinki.
Questionnaire and procedures
A questionnaire with both closed and open questions was prepared for the study. The questionnaire included a demographic section and a section investigating which triptans the patients had used at least once in the previous three years, in any dosage and formulation, and/or discontinued and/or continued to take, why they used or discontinued them, whether any triptan had caused adverse effects, and if so, which triptan caused adverse effects. Previous to its administration, the questionnaire had been tested among the patients seen at the ambulatories of the Headache Centre in September 2008.
The questionnaire was administered in the waiting room by a trained postgraduate student of the School of Clinical Pharmacology, who had never examined the patients before.
Data analysis
The data collected by the questionnaire, by clinical chart (diagnosis and history of migraine), and by the patients’ diaries (headache days and use of acute treatment medications in the 30 days preceding the visit) were entered into a specially prepared database. A descriptive analysis of the frequencies of all parameters collected was made, as well as a comparison between the group of patients who reported that they had been habitually using at least one triptan for three months (i.e. patients currently using triptans, information that also emerged from their diaries), and the group of patients who reported having discontinued triptans at least three months before (i.e., patients who had discontinued triptans, information that also emerged from their diaries). Moreover, we compared, among the various triptans, the percentages of patients who reported that they had used one of the drugs at least once in the previous three years and then discontinued it at least three months before the study (i.e. patients who had discontinued each triptan) and the percentages of patients reporting at least one adverse effect associated with one triptan (i.e. patients reporting adverse effects with each triptan). Finally, we compared, among the various age brackets, the percentages of patients reporting triptan-associated adverse effects and the relationship between age and adverse effects.
Statistical differences between continuous variables were analysed by Wilcoxon rank-sum test. Comparisons between percentages were made using Chi-square test or Fisher’s exact test. The relationships between age and adverse effects were determined using linear regression. All data were expressed as the mean ± standard deviation (SD). A
Results
Demographic and migraine data

SD = standard deviation. Patients currently using triptans vs. patients who had discontinued triptans: a
Only a minority of the patients who had discontinued triptans (40/97, 41%) had taken this decision because of the drugs’ ineffectiveness; the majority (57/97, 59%) had instead discontinued them because of adverse effects. On the other hand, those patients who had decided to continue using triptans did so because of their effectiveness (210/246, 85%); only very few patients (36/246, 15%) continued to use them because of the absence of adverse effects.
Distribution of patients by triptan used, discontinued, and triptan-associated adverse effects

Comparison among discontinued triptans: avs. sumatriptan 100 mg,
Comparison among triptan-associated adverse effects: avs. subcutaneous sumatriptan,
Many more patients reported adverse effects associated with subcutaneous and oral 100 mg sumatriptan than with any other triptan. Patients reporting adverse effects were fewer among those who had used/were still using zolmitriptan than almotriptan (
Distribution of patients who reported triptan-associated adverse effects according to the kind of adverse effect

Patients currently using triptans vs. patients who had discontinued triptans: a
Globally, no patient reported serious or unexpected adverse effects associated with triptans. The most common adverse effects were drowsiness, nausea, and chest symptoms. Nearly every kind of adverse effect was much more frequently reported by subjects who had discontinued triptans than by those who continued to take these drugs (Fisher’s exact test).
Distribution by age of total patients and patients who reported triptan-associated adverse effects
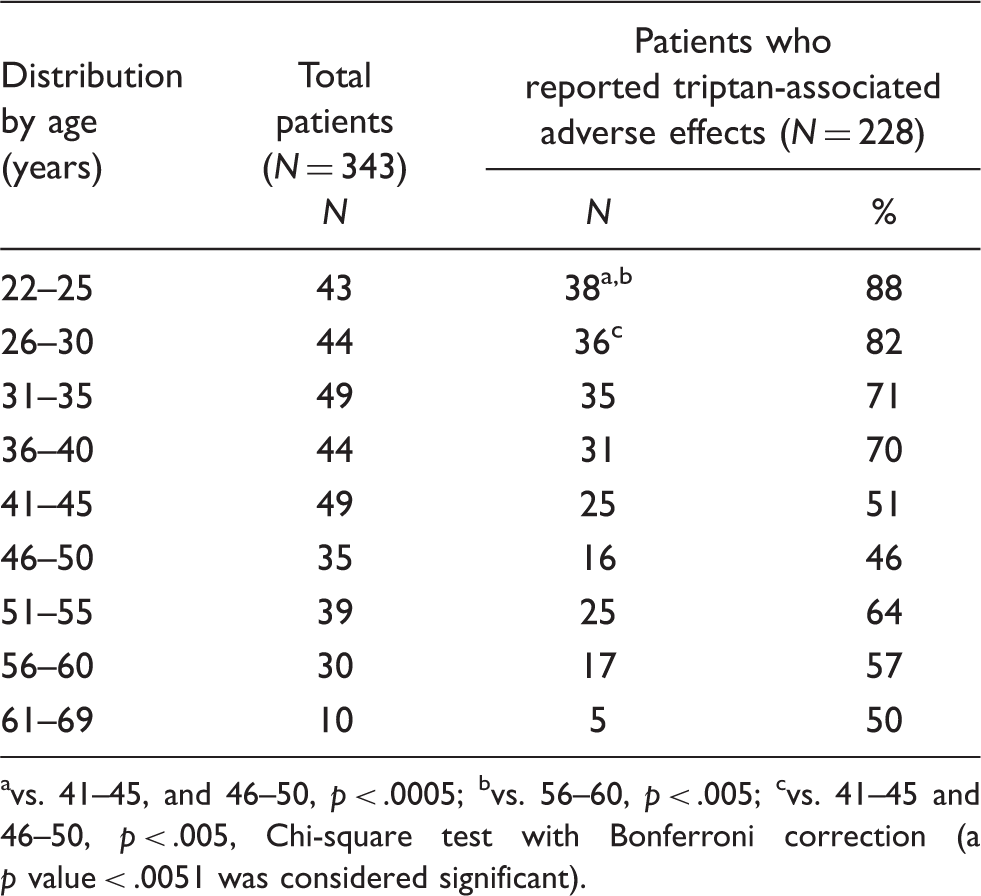
vs. 41–45, and 46–50,
Discussion
Our results show that triptans are not only the medication most prescribed to migraine patients in specialty care (10,13), but also the medication usually used by these patients. Of the 346 migraine patients attending the Headache Centre of the University Hospital of Modena for a control visit, 72% who had used triptans continued to take them; 85% of these patients continued because of the drugs’ efficacy. These patients (Table 1) had a long history of migraine and a high frequency of attacks; 54% were married and 76% were working. Triptans improve quality of life as well as reducing the loss of productivity and absenteeism from work (16,17). Therefore, it can be supposed that these medications help the patients greatly in fulfilling their family and work responsibilities.
Not all patients are satisfied with triptans (2). We found that 28% of migraine patients had discontinued triptans after having used them. This group included a higher percentage of male patients, significantly younger, with a shorter history of migraine and less frequent attacks, even if their diagnoses were similar to those who continued to use triptans. Single-time triptan users were also significantly more prevalent among men and patients younger than 30 or older than 70 years in a study carried out in Israel, in the general population, during a one-year follow-up (9).
Subcutaneous sumatriptan is the most effective acute migraine treatment (headache relief at two hours about 80%) (18). Nevertheless, migraine patients prefer oral medications (19). Our results (Table 2) confirm this preference, even if indirectly, as subcutaneous sumatriptan was the drug discontinued by most patients, discontinued significantly more than rizatriptan 5 mg, sumatriptan 50 mg, or almotriptan.
The perfect triptan, suitable for all patients, does not exist (20). Even excluding subcutaneous sumatriptan, each drug, in any dosage, had been discontinued by some patients (from 39% in the case of sumatriptan 50 mg to 75% in the case of rizatriptan 10 mg). Furthermore, our patients seemed to prefer formulations with a lower dosage. Many more patients continued to use sumatriptan 50 mg and rizatriptan 5 mg instead of double dosages of these medications (100 mg and 10 mg, respectively). This preference cannot be explained by the lower cost of formulations with lower dosage, as in Italy all triptans (except for sumatriptan nasal spray 10 mg) are completely refunded by the government health system.
Even though triptans have similar pharmacological properties, the profile of tolerability of each medication is different (21). In our study, subcutaneous and oral 100 mg sumatriptan caused adverse effects in the largest number of patients, significantly more than any other triptan. These results are consistent with a better tolerability of second- and third-generation triptans than that of sumatriptan (21).
The safety of triptans in the absence of contraindications has been proved (22). None of the 343 patients studied reported serious adverse effects. Nevertheless, triptans can cause symptoms, such as chest tightness or heaviness, which are alarming to patients and physicians because they can mimic acute cardiac events (23). These symptoms are considered to be one of the reasons for the limited use of these medications (24). Indeed, 59% of our patients who had discontinued triptans had taken this decision precisely because of adverse effects. These patients (Table 3) reported nearly every kind of adverse effect (in particular, chest symptoms: 49%) at a significantly higher rate than patients continuing to use triptans (chest symptoms 14%,
It has recently been observed that the therapeutic gain of triptans over placebo increases with age (26,27). We have found (Table 4) that the tolerability of triptans also increases with age: while 85% of patients between 22 and 30 years reported adverse effects, this percentage decreased to 42% in patients between 51 and 60. An explanation of these results might be that patients who better tolerate triptans tend to continue using them into later life. There was, however, a significant inverse relationship between patients’ age and the likelihood of reporting triptan-associated adverse effects (
Our study has some limitations. It was carried out among migraine patients at a headache centre; the patients could have suffered from more severe headaches and multiple comorbidities, and the findings cannot therefore be generalised to other patients and settings. Nevertheless, this sample reflects the population of migraine patients who seek medical advice, which is formed by those who suffer from disabling headaches (34) and are the target of prescription medications such as triptans. These patients usually have a long history of headache, have already consulted other physicians and have used various treatments (35). The medications that they take should therefore be those that they consider more useful for their migraine. Data about the use of the various triptans are influenced by the time period in which these medications have been marketed in Italy and by physicians’ preferences and prescription habits. We relied on patients’ reports and diaries; because some patients could also have used and discontinued triptans three years before taking part in our study, recall bias could have affected the results of our survey.
However, our study is not as rigorous as a randomised clinical trial. Our aim was not to compare the efficacy and safety of triptans, but simply to analyse the pharmacoepidemiology of these medications from patients’ perspective. Even if the patients’ memories do not precisely reflect reality, remembered experience influences both patient and physician decisions, as the physician in a headache clinic chooses a therapy according to patients’ subjective reports of their response or non-response to a treatment (36).
The relationship between age and adverse effects was assessed univariately; we cannot exclude the possibility that unmeasured variables, such as concomitant diseases, could have affected the results. Finally, we did not make any inferences regarding a causal relationship between the drug and the adverse effect. However, patients are generally able to distinguish adverse drug reactions from clinical events related to other factors (37).
In conclusion, only a minority of migraine patients in specialty care discontinue triptans, mainly because of adverse effects. These patients are younger and experience more adverse effects than most patients, who continue to habitually use triptans, above all for their effectiveness. In clinical practice, the physician should exhaustively inform younger patients about the adverse effects of triptans. The physician should also monitor older patients, as better tolerability does not necessarily imply greater safety, and can foster the overuse of triptans.
Footnotes
Acknowledgements
We would like to thank Prof Anna Valeria Vergoni and Dr De Gerolamo for their advice about the statistical analysis of the data.
