Abstract
Introduction
The objective of this study was to estimate and to characterize the actual patterns of triptan use and overuse in France using a drug reimbursement database.
Methods
We included all people covered by the French General Health Insurance System (GHIS) from the Provence-Alpes-Côte-d’Azur (PACA) and Corsica administrative areas who had at least one dispensed dose of triptans between May 2010 and December 2011. All dispensed doses of triptans, migraine prophylactic treatment and psychotropic medications were extracted from the GHIS database. Triptan overuse was defined as triptan use >20 defined daily doses (DDD) per month on a regular basis for more than three consecutive months. Risk of overuse was assessed using logistic regression adjusted for gender and age.
Results
We included 99,540 patients who had at least one prescription of a triptan over the 20 months of the study. Among them, 2243 patients (2.3%) were identified as overusers and received 20.2% of the total DDD prescribed. Twelve percent of overusers and 6.9% of non-overusers were aged more than 65 years (OR: 1.81). Overusers did not have a greater number of prescribers and pharmacists than non-overusers. They were more frequently prescribed a prophylactic medication for migraine treatment (56.8% vs 35.9%, OR: 2.36), benzodiazepines (69.9% vs 54.7%, OR: 1.93) and antidepressants (49.4% vs 30.2%, OR: 2.33).
Conclusions
This work suggests that triptan overuse may be due to insufficient prescriber awareness of appropriate prescribing. The off-label prescription of triptans among the elderly necessitates investigating their cardiovascular risk profile in this sub-group.
Introduction
Triptans, selective 5-HT1B/D agonists, are specific and effective therapy for migraine attacks recommended by all international guidelines on migraine management (1–3). Seven compounds are available worldwide but, rather than attempting to highlight clinical differences among triptans, efforts need to be focused on the optimal use of these drugs (4). In order to avoid medication-overuse headache, it is of particular importance to prevent triptan overuse, which is defined as the regular use of triptans on 10 days/month or more on a regular basis for more than three months according to the International Classification of Headache Disorders (5,6). Triptan overuse was observed early after its availability on the market (7) and described by several clinical and pharmacoepidemiological studies (8–17).
All triptans available worldwide are marketed in France and only by medical prescription. Because none are approved as over-the-counter (OTC) drugs in pharmacies like in United Kingdom (UK) and Germany, all dispensed triptans are reimbursed and recorded in the French Health insurance database. Use of such a database has become generalized in France, allowing large population-based drug-use studies. As far as we are aware, no study has recently assessed triptan use and overuse using this database (13,15).
The objective of this study was to estimate and to characterize the actual patterns of triptan use and overuse in France using a drug reimbursement database, in two administrative areas that represent 4 million people.
Methods
Main French reimbursement database
The General Health Insurance System (GHIS) covers approximately 80% of the French population (other systems cover specific population such as farmers, soldiers and railway workers). The reimbursement database exhaustively records reimbursed medications prescribed but does not inform directly about the medical indication of each reimbursement (18,19).
Study population and period
Data were extracted from the French General Health Insurance System (GHIS) in two administrative areas (Provence-Alpes-Côte-d’Azur (PACA) and Corsica), covering 4,143,644 people. This population was chosen on the basis of a 10-year collaboration between the pharmacology department of Marseille Hospitals and the PACA and Corsica section of the GHIS. This collaboration ensures rapid and easy access to high-quality databases.
For this study, we included all patients who had at least one dispensed dose of triptan between May 2010 and December 2011. This corresponds to the selection of prevalent triptan users over the study period. This 20-month period was the longest period available at the regional level.
Data collected from the reimbursement database
Included medications were all triptans, medications frequently used as prophylactic treatment in migraine, opioids, and other reimbursed drugs as indicators of associated comorbidities such as benzodiazepines and antidepressants. All medications were identified by their code in the anatomical therapeutic chemical (ATC) classification. For more details on which drugs were included, see online Supplementary Table 1.
The following variables were also extracted: the patient’s anonymous number, age (by 10-year categories), sex, universal complementary health insurance (CMU-C, which is free health insurance coverage for people on a low income), health insurance coverage for a chronic disease, the total length of hospitalizations, and the total length of sick leaves (the GHIS takes charge of the remuneration of employees who are on sick leaves) over the study period.
Triptan dose calculation
Drug quantities in each triptan prescription were converted into defined daily dose (DDD) using the ATC/DDD 2012 index. DDD is defined by the World Health Organization Collaborating Centre for Drug Statistics Methodology as the assumed average maintenance dose per day for a drug used for its main indication in adults. This allows the aggregation of all triptans’ international nonproprietary name (INN). One DDD then corresponds to one standard day of treatment of acute migraine (20).
Classification of patient groups according to their triptan use
We defined two groups of patients according to their duration of exposure:
“ Occasional triptan users” who had dispensed doses spread over less than three months (date of first dispensed dose – date of last dispensed dose <3 months). “ Regular triptan users” who had dispensed doses spread over more than three months (date of first dispensed dose – date of last dispensed dose >3 months). Regular triptan users were then divided into “non-overusers” and “overusers” (Figure 1). Triptan user groups (number and percentages).

Triptan overuse was assessed with a definition adapted from the International Classification of Headache Disorders, second edition (ICHD-2) International Headache Society (IHS) criteria. We defined triptan overuse as the use of more than 20 DDD of triptans (any formulation) per month on a regular basis for more than three consecutive months. We used a threshold of 20 DDD per month to account for possible double intakes per day in case of recurrent attack in 24 hours if the patient has responded to the first dose, as recommended in the prescribing information for all triptans.
Statistical analysis
Categorical data are presented as number of individuals and percentage. Continuous variables are presented as mean ± standard deviations, median and quartiles. To assess the effect of each variable on the risk of overuse, we calculated the odds ratio (OR) adjusted for gender and age (as categorical binary variable: <65 years old (y.o.) vs. > 65 y.o.) with their 95% confidence interval (CI) using logistic regression models with overuse vs. non-overuse as the dependent variable. We calculated two-tailed
Ethical considerations
Ethics approval was not required for this study since the analysis was carried out on an anonymized database.
Results
During the study, we identified 95,540 patients (2.3% of the population covered) who had at least one prescription of triptan, including 53,675 occasional users (56.2%) and 41,865 regular users (43.8%) (Figure 1).
A total of 384,546 dispensed doses of triptans were recorded. A single dispensed dose of triptan was recorded for 45,143 patients (47.3%). Zolmitriptan was the most frequently prescribed triptan (42.6% of triptan users had at least one dispensed dose of zolmitriptan)
Women represented 75.6% of occasional users and 81.9% of regular users. Triptans users aged more than 65 accounted for 6.2% of global triptans users (5.5% in occasional users, and 7.1% in regular users).
During the study, 2243 patients (5.4% of regular users and 2.3% of the total number of users of triptan) were identified as overusers (Figure 1). Their total triptan dispensed doses (1,005,554 DDD) accounted for 23.4% of the total triptan doses dispensed to regular users. Their characteristics are presented in Tables 1, 2 and 3.
Characteristics of overusers compared to non-overusers within regular triptan users. Data are “number of individuals (percentage)” or “mean (±standard deviation) median (lower quartile–upper quartile).”

y.o.: years old.
Triptans that had a greater risk of overuse were eletriptan (OR: 1.41, 95% CI: 1.28–1.55), naratriptan (OR: 1.20, 95% CI: 1.06 – 1.36), rizatriptan (OR: 1.17, 95% CI: 1.02–1.33) and sumatriptan (OR: 1.52, 95% CI: 1.32–1.75).
Characteristics of prescribing of triptans and associated medications in overusers compared to non-overusers within regular triptans users. Data are “number (percentage)” or “mean (±standard deviation) median (lower quartile–upper quartile).”
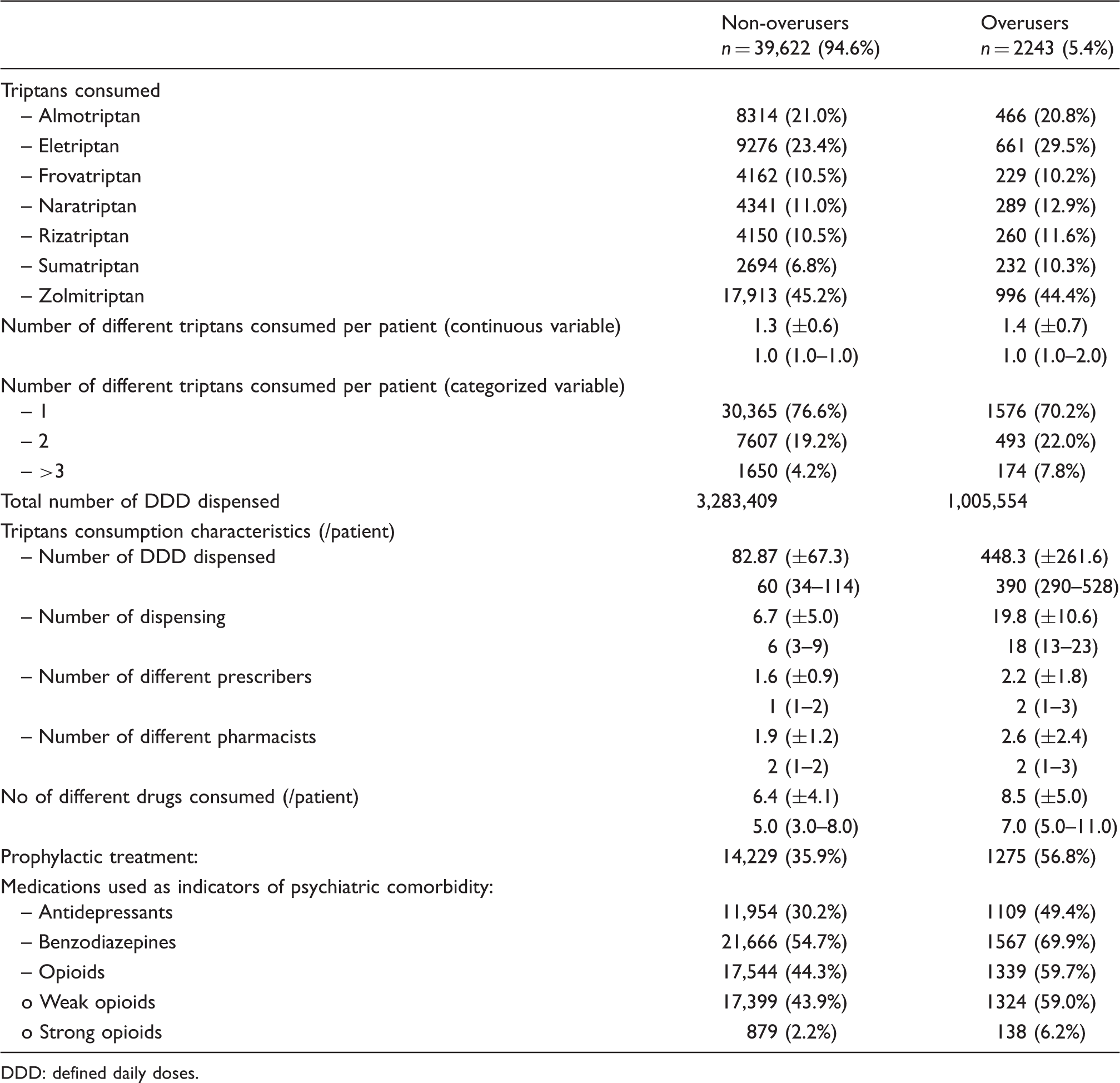
DDD: defined daily doses.
Non-overusers vs. overusers: odds ratio (OR) adjusted for sex and age (two classes: <65 y.o. and >65 y.o.), with their 95% confidence interval.

DDD: defined daily doses.
Overusers had more frequently consulted a neurologist than non-overusers (13.2% vs 8.7%, respectively,
Triptans users who had a higher number of DDD dispensed, higher number of dispensed doses, consulted a higher number of different prescribers and a higher number of different pharmacists were at a higher risk of triptan overuse (
Discussion
Our study found 2.3% of triptan users within the source population. A total of 56.2% were occasional users and 43.8% were regular users. Among regular users, 5.4% were overusers of triptans.
As we worked with a large population of nearly 95,000 triptan users, which is the exhaustive population of triptan users among the PACA and Corsica regions’ inhabitants covered by the GHIS, our study had high statistical power to detect low differences between groups. Thus, we considered the difference between statistical significance and clinical relevance while interpreting our results.
General characteristics of the population
In our study, we found 56.2% of occasional triptan users, which means that only half of triptan users continue to use triptans. Occasional dispensed doses are well known when working on a reimbursement database (21). It has also been described previously with triptans (10,13,22).
Several hypotheses may be suggested to explain this occasional use. Firstly, it is not possible to determine whether this is actually an occasional “use” or only a dispensing that will never be consumed by the patient. Secondly, drugs for migraine treatment are dispensed in anticipation of a future acute treatment, i.e. the occurrence of a migraine headache. Thirdly, this occasional use may be related to adverse events and/or a lack of efficacy that leads the patient to discontinue the treatment. Some authors already highlighted a high turnover in the population of triptan users (22,23).
Even though triptans are still not recommended in patients aged more than 65 years old (age >65 years was an exclusion criteria in pre-marketing clinical trials, resulting in poor cardiovascular risk assessment in this sub-population), the proportion of patients aged 65 and older among occasional users was estimated to be 5.5% in our study. Among the regular users and triptan overusers group, this proportion increases to 7.1% and 12%, respectively. These results confirm data available in a real-life setting in France and in other countries showing that triptans are frequently prescribed for people aged more than 65 years, with estimates ranging from 3% to 10% of triptan users (10,15,24). Triptans may cause coronary, cerebrovascular and peripheral vasoconstriction possibly leading to serious complications such as myocardial infarction, ischemic stroke and ischemic colitis, mostly in patients with cardiovascular diseases or risk factors (2). Since the approval of sumatriptan for the treatment of migraine attack, the cardiovascular risk of triptan use is still debated. In 2004, the conclusions of the Triptan Cardiovascular Safety Expert Panel, which was composed of a multidisciplinary United States group of experts in neurology, primary care, cardiology, pharmacology, women’s health, and epidemiology, were reassuring. The incidence of serious cardiovascular events with triptans both in clinical trials and clinical practice appears to be extremely low, and the overall risk-benefit profile of triptans favors their use in the absence of contraindications (25). Even if the overall risk-benefit profile of triptans for migraine is good for the whole population, the cardiovascular safety profile of triptans has to be assessed specifically in the elderly and in real-life settings, as this population is always excluded from randomized clinical trials. Moreover, a recent study on the adverse events database of the Food and Drug Administration (FDA) shows that cardiovascular adverse events are disproportionately reported after triptan use (16). However, this signal has to be confirmed by cohort studies, and a specific vascular safety study among older patients exposed to triptans will be useful.
In this article we chose not to consider cardiovascular medications as a proxy of cardiovascular outcome. In fact, these medications can have several medical indications and our reimbursement database does not provide information on the medical indication of a prescription. Any conclusions regarding cardiovascular safety based only on these medications would be highly biased.
Triptan overuse
We found 2243 overusers (5.4% of regular triptans users and 2.3% of all triptans users) with a threshold of 20 DDD per month for at least three consecutive months. With this threshold, we chose to be more specific so as to increase specificity of the detection of triptan overuse. The total cost of triptan dispensed doses for these overusers (2.3% of all triptan users) was 3,518,952 euros (22.3% of the total cost of 15,761,994 euros for the total dispensed doses of triptan over the study period).
Several arguments indicate a more serious illness in patients overusing triptans: They are more often hospitalized, they have a lower socioeconomic status (as approached by CMU-C), they had more frequently consulted a neurologist, and they are users of prophylactic migraine treatments and psychotropic medications more frequently than non-overusers.
We had access to data on duration of sick leave for each patient over the study period. To our knowledge, few studies were able to measure sick leave. Our analysis found that non-overusers had more frequently had a period of sick leave than overusers. This result was surprising at first sight but we hypothesize that overusers may be more often unemployed than non-overusers. The estimation of triptan-overuse prevalence varies in different studies. In our study, we chose to define overuse as the dispensing of at least 20 DDD/month for three consecutive months. This criterion is more stringent than the criteria used in previous studies. However, it allows a more specific and minimal estimation of the prevalence of overuse. Association between migraine and mixed anxiety and depressive disorders is well known (26). In our study, we used benzodiazepines and antidepressants as proxies for anxiety or depression. We found an important consumption of benzodiazepines (51.1% of triptan users also had at least one reimbursement of benzodiazepine). This proportion was even higher in the subgroup of overusers, reaching 69.9%. In comparison, the French Drug Agency reported that, in 2010, 20% of the French population affiliated with the GHIS had used at least one benzodiazepine (27).
Table 2 shows a low number of different prescribers and pharmacists for triptan overusers, indicating that triptan users do not consult multiple prescribers and pharmacists. Furthermore, triptan overusers have more and higher doses dispensed than non-overusers. These results suggest that overuse of triptans may be due to insufficient prescriber awareness of appropriate prescribing of triptans (prescription of too high doses, lack of patient’s prescription follow-up, intentional prescription of doses leading to overuse). Policies to prevent the overuse of triptans may require information campaigns on best practice and guidelines aimed at health workers involved in the management of migraine.
Limitations
Our study suffers from limitations inherent to the characteristics of the available database.
The first one is due to the medico-administrative database that does not include medical diagnosis leading to prescription. However, the main trends observed in the study are consistent with those of the literature on the epidemiology of migraine (13,15,28), which supports the external validity of our results.
In addition, it is impossible to determine whether the dose dispensed is actually consumed by the patient. It has been shown by Noize et al. that the reimbursement database of the health Insurance system agreed with consumption data (29). We also consider that a patient has consumed a triptan when a physician renewed the dispensed dose.
Owing to the study period, it is possible that patients are labeled occasional users while they actually are regular users (if they had dispensed doses before May 2010 or after December 2011). In pharmacoepidemiology studies, this issue is sometimes fixed by the selection of incident users rather than prevalent users. However, we did not choose this option in our study for several reasons: i) triptans are an acute treatment for migraine attacks. It would require a long washout period to detect only incident users. Gaist and Da Cas used a washout period of one year in their studies (11,17). Our data (20 months) would not allow us to take such a long washout period; ii) more important, selecting only incident users would allow us to detect only incident overusers, i.e. triptan users who overuse very early. Our study was aimed at quantifying overuse, and we would greatly lower our estimation by including only incident users; iii) our method is interesting since it gives a minimal but strong estimation of the overuse of triptans over the study period.
We estimated the “triptan overuser” status using only the pharmacological criteria of the IHS definition of triptan-overuse headache because medical data are not available in the reimbursement database. Therefore, patients identified as triptan overusers may not suffer from triptan-overuse headache. In the same way it is impossible to be sure that a medication identified as “prophylactic treatment” was actually prescribed as a prophylactic treatment of migraine attacks when this medication has several indications. This can result in an overestimation of the consumption of prophylactic treatment. However, this overestimation may be the same in overusers and non-overusers groups and may not introduce a differential bias in our results.
Conclusion
Our study found 2.3% of triptan overusers receiving 20% of the total quantity of triptans dispensed.
We also observed that overusers had three-fold higher prescribed doses than non-overusers
In addition, our data have shown that triptans are frequently prescribed for people aged above 65 years, a population in which triptans are not recommended. Because of the vasoconstrictive potential of triptans, a specific vascular safety study among older patients exposed to triptans will be useful.
Clinical implications
The study describes patterns of triptan use and overuse in a French administrative area using an exhaustive reimbursement database covering 4 million inhabitants. About 2.3% of triptan overusers are identified, receiving 20% of the total quantity of triptans dispensed. Twelve percent of overusers and 6.9% of non-overusers were aged more than 65 years. Increasing clinician awareness is essential in helping prevent triptan overuse while continuing to provide effective treatment.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflicts of interest
Dr Braunstein has nothing to declare. In the five past years, Dr Donnet has received fees for consulting and clinical research activity with Allergan, Almirall SAS, Astellas, AstraZeneca Pharmaceuticals, Grunenthal, Lilly, Menarini, MSD, Pfizer, and Zambon. Dr Vincent Pradel has nothing to declare. Dr Sciortino has nothing to declare. Dr Allaria-Lapierre has nothing to declare. In the five past years, Dr Lantéri-Minet has received fees for consulting and clinical research activity with Allergan, Almirall SAS, Astellas, AstraZeneca Pharmaceuticals, ATI, BMS, Boehringer, Boston Scientific, CoLucid, Convergence, Glaxo-SmithKline, Grunenthal, Lilly, Johnson & Johnson, Medtronic, Menarini, MSD, Pierre Fabre, Pfizer, ReckittBenckiser, Saint-Jude, Sanofi-Aventis, UCB, and Zambon. Dr Micallef has nothing to declare.
Acknowledgments
We thank Dr François Natali for his technical help with data extraction. This work was part of David Braunstein’s studies toward a master’s degree at Marseille University and was presented in oral communication form at the 8th French Annual Meeting of Pharmacology and Therapeutics (22 and 23 April 2013).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
