Abstract
Background
Rimegepant was safe and well tolerated in a multicenter, open-label, phase 2/3 trial for acute treatment of migraine administered as 75 mg every other day (EOD) and as needed (PRN; EOD + PRN) for 12 weeks or PRN for 52 weeks.
Methods
Post-hoc analysis of this long-term safety study in adults was conducted according to five subgroups based on current and historical triptan use: current triptan use and no historical discontinued triptans; no current triptan use and no historical discontinued triptans (triptan-naïve); and history of =1, ≥1 and ≥2 discontinued triptans. Triptan failure was defined as discontinuation of any triptan, for any reason, including insufficient response and/or tolerability.
Results
The proportion of participants with ≥1 on-treatment adverse event (AE) was 57.6–66.3% across triptan subgroups. Rimegepant-related AEs (17.7–23.2%), treatment discontinuations (1.6–3.8%) and the most common AE (upper respiratory tract infection, 7.7–9.5%) were consistent across subgroups. After long-term treatment, the proportion of participants who preferred rimegepant to their previous medication was >75% in all triptan subgroups.
Conclusions
Long-term acute treatment of migraine with rimegepant 75 mg up to once daily was safe and well tolerated in triptan-naïve participants, current triptan users and those with single or multiple historical triptan discontinuations.
Trial Registration
NCT03266588.
This is a visual representation of the abstract.
Introduction
Triptans are serotonin 5-HT1B/1D receptor agonists and have been among the few specific acute treatment options for migraine for decades (1). Triptans, including sumatriptan, can be an effective acute treatment for migraine, providing pain relief by as quickly as two hours; however, some people with migraine may not have an initial response to triptans and 30–40% may experience a relapse within 24 hours of the initial dose or discontinue treatment due to tolerability (2,3). Triptan prescription can be further complicated as triptans are contraindicated in certain cardiovascular conditions (1). With multiple triptan formulations available and several with multiple routes of administration, it is not uncommon for people with migraine to switch from one triptan to another (4–6). While some may benefit from switching to a different triptan, switching triptans has also been associated with increased primary care costs and no improvement in headache-related disability (5,6). People with migraine who do not respond to, or tolerate, oral triptans may benefit from acute treatment with agents that have a novel mechanism of action (1,7).
Recently, medications that target the calcitonin gene-related peptide (CGRP) receptor have emerged as an alternative and specific acute treatment option for migraine. Rimegepant is an oral, CGRP receptor antagonist approved in the USA, EU and UK for the acute treatment of migraine at a daily dose of 75 mg as needed (at a maximum of 75 mg per 24-hour period). A single dose of rimegepant 75 mg has been shown to be effective and safe for the acute treatment of migraine in multiple clinical studies (8–12). The safety and tolerability of rimegepant 75 mg for the acute treatment of migraine has also been demonstrated in a long-term clinical trial (13,14). In the multicenter, long-term, open-label safety study of 1800 adults, rimegepant 75 mg administered every other day (EOD) and as needed (PRN; EOD + PRN) for 12 weeks or PRN for up to 52 weeks was safe and well tolerated (13).
This post-hoc analysis of the multicenter, long-term, open-label safety study (NCT03266588) aimed to explore the safety and effectiveness of rimegepant 75 mg according to the participants’ historical and current triptan experience. As a substantial proportion of people with migraine who use triptans often express dissatisfaction with their acute treatment (including the speed of relief, recurrence of pain and need for a second dose) and approximately 80% would be willing to try another acute medication (15), participant preferences and satisfaction were also explored.
Methods
Ethical considerations
The clinical trial described herein was conducted in accordance with the ethical principles of Good Clinical Practice, according to the International Conference on Harmonisation Harmonised Tripartite Guideline and all local laws and regulations. Before any study-related procedures were undertaken, investigators at each study center obtained Institutional Review Board/Independent Ethics Committee approval of the protocol and study-related materials, and participants provided written informed consent.
Study design
This was a post-hoc subgroup analysis of the prospective, phase 2/3, one-year open-label safety study of rimegepant in the acute treatment of migraine conducted at 103 sites in the USA (Study 201, NCT03266588) (13). The study comprised a screening visit and 30-day Observation Period before participants were provided with study medication and commenced a long-term treatment period of up to 12 months (52 weeks). During the long-term treatment period, participants visited the study site approximately every two weeks during the first month and then every four weeks until week 12 or week 52. A final follow-up visit was scheduled 14 ± 2 days after the end of treatment.
Participants were sequentially enrolled into three treatment cohorts. Cohort 1 (PRN 2–8) comprised participants with a self-reported history of two to eight moderate to severe attacks/month and were assigned to take rimegepant 75 mg PRN up to once daily to treat migraine attacks of any severity, for up to 52 weeks. Cohort 2 (PRN 9–14) comprised participants with nine to 14 self-reported moderate to severe attacks/month and were assigned to take rimegepant 75 mg PRN up to once daily to treat migraine attacks of any severity, for up to 52 weeks. Cohort 3 (EOD + PRN), designed to collect data during higher frequency dosing, comprised participants with a history of four to 14 moderate to severe attacks/month. They were assigned to take rimegepant 75 mg EOD irrespective of migraine attacks, and PRN on other days to treat migraine attacks of any severity, up to once daily for up to 12 weeks.
Participants
Full participant eligibility criteria have been previously described (13). Briefly, eligible participants were males and females aged ≥18 years, with at least a one-year history of migraine (with or without aura), migraine onset prior to age 50 years, at least two to 14 migraine attacks of moderate or severe pain intensity per month within the last three months prior to screening, and an average duration of untreated attacks of 4–72 h. Participants with contraindications for triptan use were eligible provided they met all other study criteria. To avoid confounding safety data, which would have affected the primary endpoint, use of triptans was only permitted during the Observation Period and had to be discontinued at the baseline visit. Key exclusion criteria were a history of basilar or hemiplegic migraine; current evidence of uncontrolled, unstable or recently diagnosed cardiovascular (CV) disease; myocardial infarction, acute coronary syndrome, percutaneous coronary intervention, cardiac surgery, stroke or transient ischemic attack during six months prior to screening; uncontrolled hypertension (>150 mmHg systolic or 100 mmHg diastolic after 10 minutes of rest) or uncontrolled diabetes (hypertension and/or diabetes that was stable for three months prior to screening was permitted); body mass index (BMI) ≥30 kg/m2; HbA1c ≥6.5%; and history of HIV disease.
Assessments and endpoints
Primary safety and tolerability endpoints included the frequency of adverse events (AEs) occurring in ≥5% of treated participants, serious AEs and AEs leading to study drug discontinuation. The severity of AEs and their relationship to the study drug were determined by the investigator. Secondary safety endpoints included the frequencies of concurrent elevations of aspartate aminotransferase (AST) or alanine aminotransferase (ALT) >3 × upper limit of normal (ULN) with total bilirubin >2 × ULN, hepatic-related AEs and hepatic-related AEs leading to study drug discontinuation. Additional safety endpoints included the frequencies of AEs related to study drug and other prespecified AEs of special interest (potential drug abuse AEs, cardiovascular AEs and suicidality AEs).
Exploratory self-reported assessments of satisfaction, disability and quality of life included changes relative to baseline in migraine-related disability, migraine-related quality of life, preference of medication, satisfaction with medication and clinical global impression of change. Migraine-related disability was assessed using the Migraine Disability Assessment Scale (MIDAS). MIDAS is a retrospective, self-reported questionnaire that measures headache-related disability (16). The MIDAS total score defines the level of disability: 0–5 (little/no disability), 6–10 (mild disability), 11–20 (moderate disability) and ≥21 (severe disability). The MIDAS was completed on a paper form on site. The impact of treatment on subject-reported quality of life was assessed using the Migraine-Specific Quality of Life questionnaire (MSQoL v2.1) and was completed on a paper form on site (17). Participant preference was assessed using Migraine Preference of Medication (PoM), which comprises a brief five-point scale that captures the participants’ perception of whether the medication they are taking has had a greater benefit compared with previous medications to treat their pain (13). Participant satisfaction with medication was assessed using the seven-point Satisfaction with Medication questionnaire (SM) (13). The PoM and SM were completed using the participant eDiary (13). The Clinical Global Impression of change (CGI-c) scale is a brief observer-rated seven-point scale that rates participant total improvement relative to the investigator's past experience with other participants with the same diagnosis, with or without collateral information (13,18). The CGI-c was administered by the investigator and completed on a paper form on site. For summary purposes, the seven change scores were collapsed to three: improved, no change, or worse.
Statistical analysis
The statistical methods used in the original clinical trial have been previously described (13). All summaries reported herein were performed on the treated population (took any amount of rimegepant). This post-hoc analysis explored the impact of triptan use (none, current, or prior failure) in the acute treatment of migraine with rimegepant in five subgroups based on historical and current triptan experience: current triptan use and no historical discontinued triptans (triptan-using); no current triptan use and no historical discontinued triptans (triptan-naïve); and history of =1, ≥1 and ≥2 discontinued triptans (=1, ≥1 and ≥2 triptan-failure). Triptan treatment failure was defined as a self-reported history of prior triptan discontinuation for any reason, including insufficient response (lack of efficacy) and/or tolerability, and included any medication in the triptan class (any formulation of almotriptan, eletriptan, frovatriptan, naratriptan, rizatriptan, sumatriptan, or zolmitriptan, or the combination sumatriptan–naproxen) (19). The number of historical triptan discontinuations was based on the number of unique triptan medication names associated with triptan failure, regardless of formulation.
Summary statistics of the various reported measures were produced overall, as well as for the cohorts and subgroups, with no formal statistical comparisons performed.
Results
Participant demographics
Of 1800 treated participants, 1008 (56.0%) had no history of discontinued triptan use (triptan-using, n = 373; triptan-naïve, n = 635). Of participants who had historical triptan discontinuation (triptan-failure; n = 792), the majority (n = 546) had discontinued only one triptan (=1 triptan-failure), whereas 246 participants had discontinued ≥2 triptans (≥2 triptan-failure). The most frequently noted reasons for previous triptan discontinuation included: unreliable efficacy from attack to attack; slow onset of pain relief; inadequate relief of associated symptoms; and treatment-associated AEs.
Baseline participant demographics and clinical characteristics are summarized in Table 1. Overall, baseline demographics were generally consistent across cohorts and historical triptan experience subgroups. Participants who had ≥2 triptan-failure tended to be older (mean [SD] age, 46.4 [12.1] years) than those who were triptan-naïve (39.8 [12.1] years) (Table 1). The median (range) time since migraine onset was generally shorter in triptan-naïve participants (16.0 [0–58] years) compared with triptan-using participants (22.0 [1–58] years), as well as those with triptan-failure (=1 = 22.8 [2–76] years; ≥1 = 23.7 [2–76] years; ≥2 = 26.1 [2–62] years) (Table 1). Overall, participants experienced a comparable number of moderate to severe migraine attacks per month, regardless of historical triptan experience (6.5–7.3 attacks per month). Triptan-naïve participants tended to have a shorter average duration of untreated migraine attacks (mean [SD], 28.1 [20.6] hours) than triptan-using participants (35.9 [21.7] hours) and those with triptan-failure (36.9–37.8 hours) (Table 1). There were few participants with true contraindications to triptans, but more participants with underlying risk factors (20).
Baseline demographics and clinical characteristics.
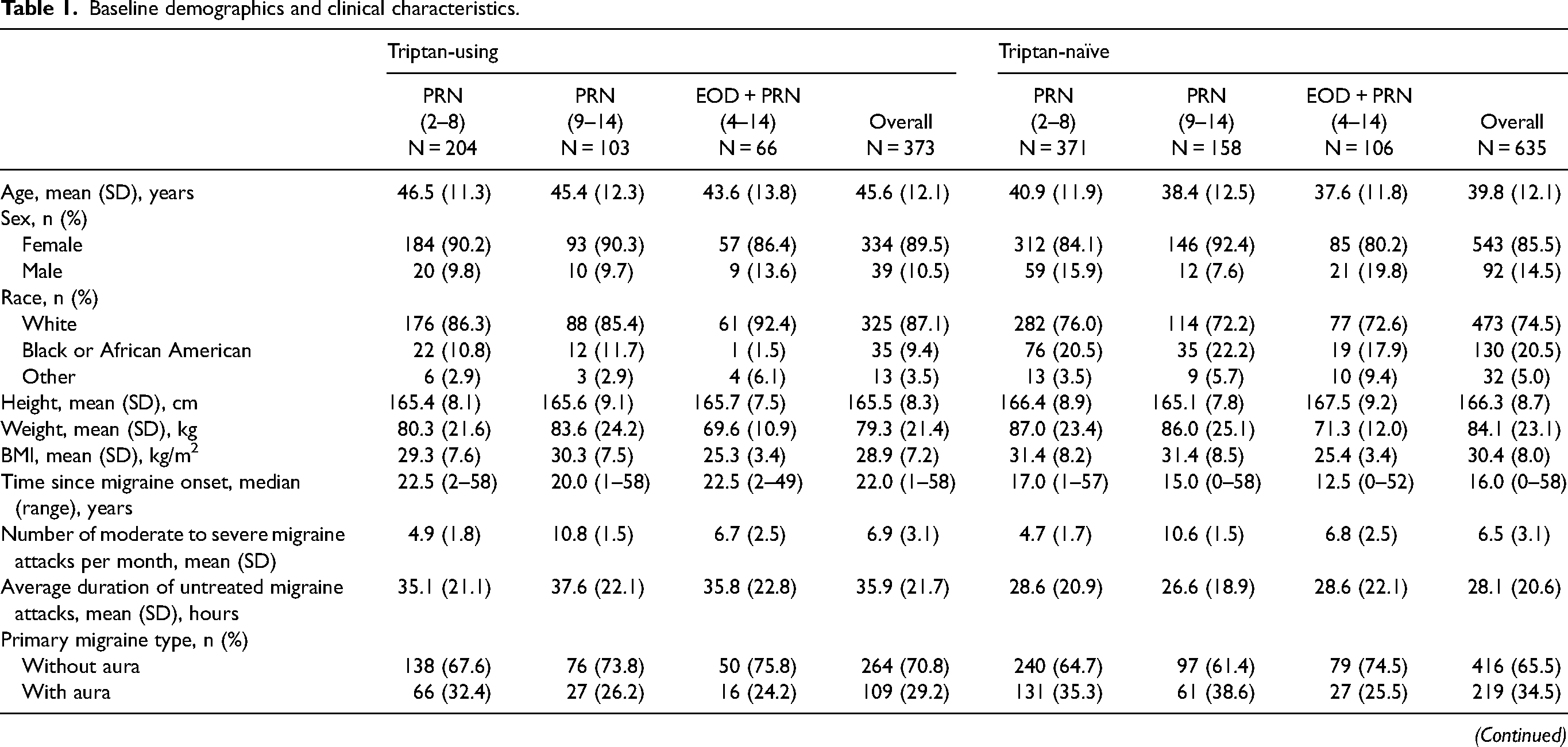

Triptan-using: current triptan use and no historical discontinued triptans.
Triptan-naïve: no current triptan use and no historical discontinued triptans.
BMI, body mass index; EOD, every other day dosing; PRN, as needed dosing; SD, standard deviation.
Safety and tolerability
On-treatment AEs are summarized in Table 2. Overall, the proportion of participants who experienced any AEs tended to be nominally higher in those with ≥2 triptan-failure (n = 163; 66.3%) compared with those who were triptan-naïve (n = 366; 57.6%) and triptan-using (n = 220; 59.0%). The number of participants who experienced any AEs was 339 (62.1%) and 502 (63.4%) for participants with =1 triptan-failure and ≥1 triptan-failure, respectively. A similar pattern was observed for rimegepant-related on-treatment AEs. The proportion of participants with on-treatment AEs deemed to be related to rimegepant by the investigator was nominally lower in participants with no historical discontinuation of triptans (17.7–19.2%) compared with those who had discontinued triptans (21.1–23.2%) (Table 2). The proportion of participants who experienced an SAE was low across cohorts (0–4.9%) and triptan experience subgroups (1.7–3.7%) (Table 2).
Summary of on-treatment AEs.


All data are n (%).
Triptan-using: current triptan use and no historical discontinued triptans.
Triptan-naïve: no current triptan use and no historical discontinued triptans.
AE, adverse event; EOD, every other day dosing; PRN, as needed dosing; SAE, serious adverse event.
The proportion of participants who discontinued rimegepant treatment due to on-treatment AEs was generally comparable regardless of historical triptan experience (Table 2). Of participants with =1, ≥1 and ≥2 triptan-failure, the overall number of participants who discontinued rimegepant due to on-treatment AEs was nine (1.6%), 14 (1.8%) and five (2.0%), respectively. For participants who were triptan-naïve and triptan-using, the number of participants who discontinued rimegepant due to on-treatment AEs was 24 (3.8%) and 10 (2.7%), respectively.
The proportion of participants with on-treatment hepatic-related AEs and additional AEs of special interest (potential drug abuse AEs, cardiovascular AEs and suicidality AEs) was generally consistent across cohorts and triptan experience subgroups (Table 2). The proportion of participants who discontinued rimegepant due to hepatic-related AEs was <1.5% in any cohort and triptan experience subgroup.
On-treatment AEs of any severity occurring in ≥5% of participants (any cohort or subgroup) are summarized in Table 3. The most common on-treatment AE occurring in ≥5% of participants was upper respiratory tract infection, which was generally consistent across cohorts (0–12.2%; n = 0–55) and triptan experience subgroups (7.7–9.5%; n = 22–74) (Table 3). On-treatment AEs that occurred in ≥5% of participants only in a cohort with historical triptan discontinuation were neck pain, back pain, diarrhea, bronchitis and influenza (Table 3).
On-treatment AEs of any severity occurring in ≥5% of participants (any cohort or subgroup).


All data are n (%).
Triptan-using: current triptan use and no historical discontinued triptans.
Triptan-naïve: no current triptan use and no historical discontinued triptans.
AE, adverse event; EOD, every other day dosing; PRN, as needed dosing.
Low frequencies of liver function test elevations were observed and were comparable regardless of current triptan use and number of discontinued triptans (Table 4). One participant had concurrent on-treatment elevations of ALT or AST >3 × ULN and total bilirubin >2 × ULN.
Liver function test elevations by current triptan use and number of previously discontinued triptans.
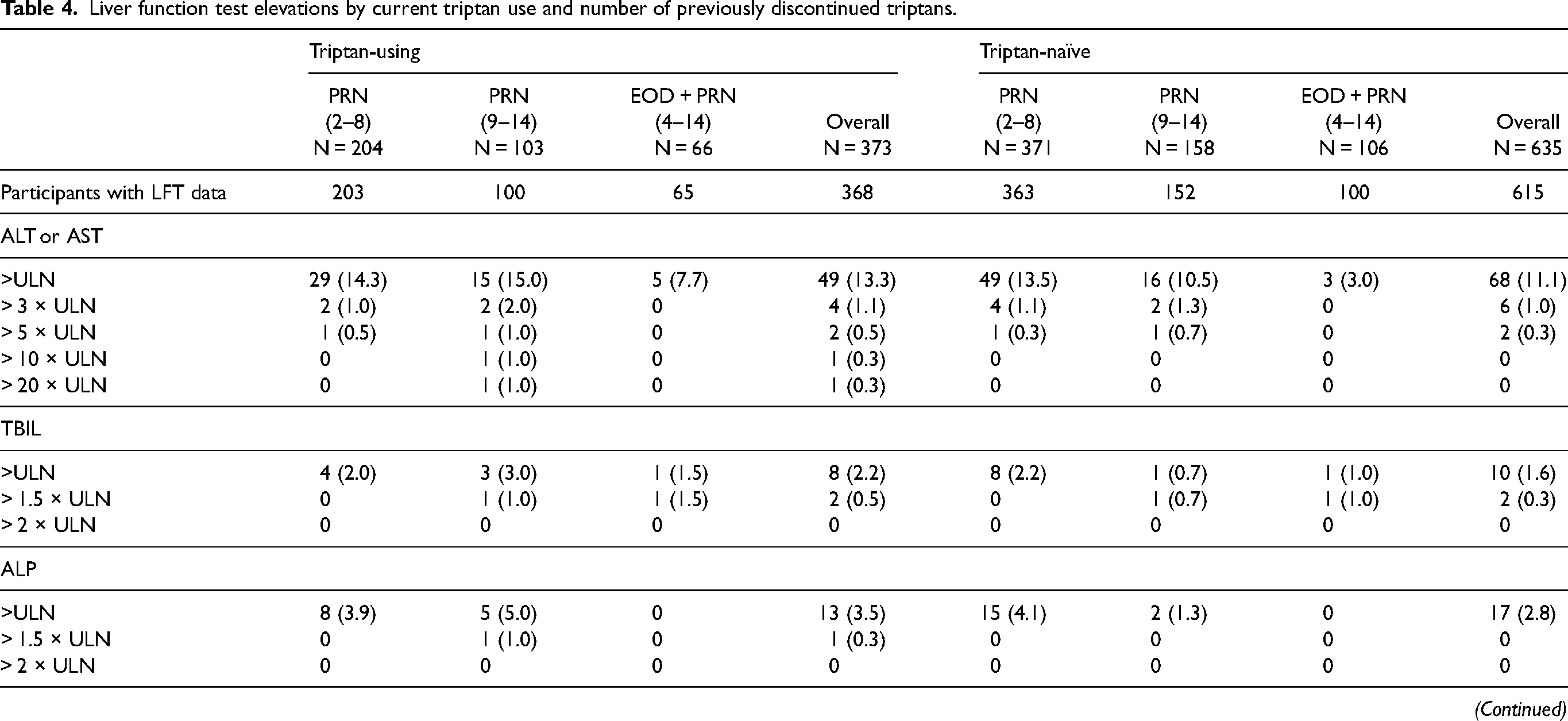
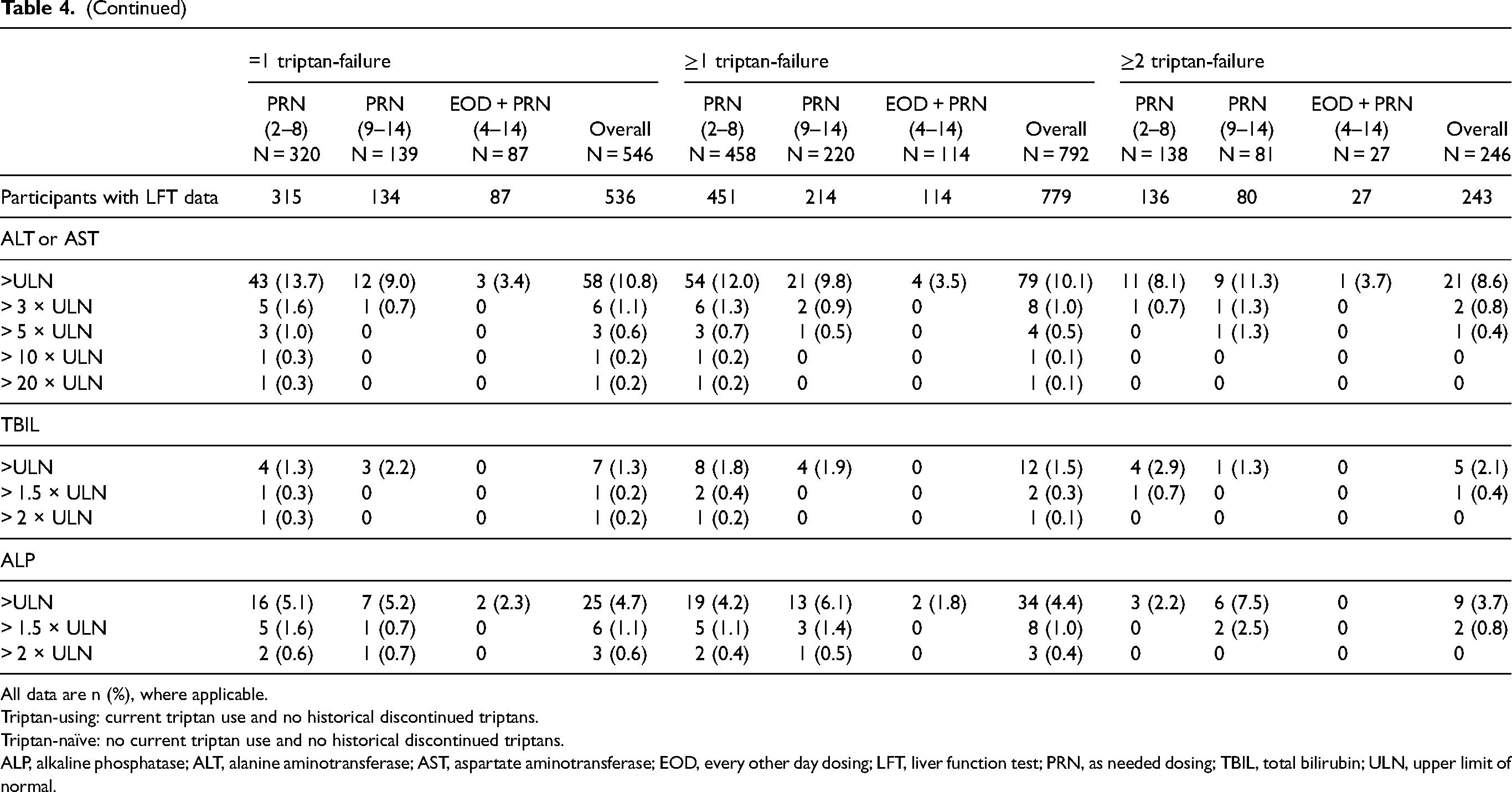
All data are n (%), where applicable.
Triptan-using: current triptan use and no historical discontinued triptans.
Triptan-naïve: no current triptan use and no historical discontinued triptans.
ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; EOD, every other day dosing; LFT, liver function test; PRN, as needed dosing; TBIL, total bilirubin; ULN, upper limit of normal.
Low frequencies of clinically significant (Grade 3–4) on-treatment laboratory abnormalities were recorded. The most frequently recorded clinically significant on-treatment laboratory abnormality by triptan experience subgroup was with low-density lipoprotein (LDL)-cholesterol (fasting) in 0–4.0% and 0–4.3% of triptan-using and triptan-naïve participants, respectively. For participants with =1, ≥1 or ≥2 triptan-failure, the most frequently recorded clinically significant on-treatment laboratory abnormality in any cohort was with LDL-cholesterol (1.2–4.8%), LDL-cholesterol (fasting; 2.2–5.1%) and LDL-cholesterol (fasting; 0–10.0%), respectively.
Patient-reported outcomes
Clinically meaningful changes in total MIDAS, absenteeism and presenteeism scores were observed in most cohorts and triptan experience subgroups at week 12 and were sustained through to week 52 (PRN cohorts) (Figure 1). At week 12, the overall mean (SD) change from baseline in total score was least pronounced in triptan-naïve participants at −11.1 (28.7) compared with triptan-using participants (−15.2 [31.0]) and triptan-failure participants (−12.0 to −12.5 [28.8 to 29.6]). At week 52, the overall mean (SD) change from baseline in total score was most pronounced in triptan-naïve participants at −16.8 (27.0) compared with triptan-using participants (−13.4 [31.9]) and triptan-failure participants (−13.2 to −14.2 [29.8 to 31.3]).

Mean (SD) change from baseline in MIDAS scores at week 12 and week 52 (PRN) by cohort and triptan experience subgroup. Triptan-using: current triptan use and no historical discontinued triptans. Triptan-naïve: no current triptan use and no historical discontinued triptans. The dashed line represents the minimal important difference or change in MIDAS score. EOD, every other day dosing; MIDAS, Migraine Disability Assessment Scale; PRN, as needed dosing; SD, standard deviation.
Numeric improvements from baseline were observed for all three MSQoL domains (emotional function, preventive role function, restrictive role function) at week 12 and week 52 (PRN) for all cohorts and triptan experience subgroups (Figure 2). Overall, improvements in MSQoL domains were of a comparable magnitude regardless of triptan subgroup.

Mean (SD) change from baseline in Health-related quality of life (MSQoL domain scores) at week 12 and week 52 (PRN) by cohort and triptan experience subgroup. Triptan-using: current triptan use and no historical discontinued triptans. Triptan-naïve: no current triptan use and no historical discontinued triptans. EF, emotional function; EOD, every other day dosing; MSQoL, Migraine-Specific Quality of Life questionnaire; PR, preventive role function; PRN, as needed dosing; RR, restrictive role function; SD, standard deviation.
In all cohorts and triptan experience subgroups, the majority of participants indicated a preference for rimegepant over their previous acute treatment medication at week 52 (Figure 3). Overall, at week 12, the proportion of participants who preferred rimegepant was greatest in triptan-naïve participants (81.9%; 77/94) and the proportion of participants who preferred rimegepant tended to decrease as the number of historical triptan discontinuations increased (63.4% [64/101] and 50.0% [18/36] for =1 and ≥2 triptan-failure, respectively). Overall, at week 52, the proportion of participants who preferred rimegepant was comparable across all triptan experience groups (Figure 3). A similar trend was observed for participant satisfaction (Figure 4). Overall, at week 12, the proportion of participants who were completely or very satisfied with rimegepant was greatest in triptan-naïve participants (71.3%; 67/94) and the proportion of participants who were completely or very satisfied tended to decrease as the number of historical triptan discontinuations increased (54.9% [56/102] and 40.5% [15/37] for =1 and ≥2 triptan-failure, respectively). Overall, at week 52, the proportion of participants who were completely or very satisfied with rimegepant was comparable across all triptan experience groups (Figure 4).

PoM at week 12 and week 52 (PRN) by cohort and triptan experience subgroup. Triptan-using: current triptan use and no historical discontinued triptans. Triptan-naïve: no current triptan use and no historical discontinued triptans. CI, confidence interval; EOD, every other day dosing; PoM, Preference of Medication; PRN, as needed dosing.

Satisfaction with medication at week 12 and week 52 (PRN) by cohort and triptan experience subgroup. Triptan-using: current triptan use and no historical discontinued triptans. Triptan-naïve: no current triptan use and no historical discontinued triptans. CI, confidence interval; EOD, every other day dosing; PRN, as needed dosing.
Overall, the majority of participants were rated as improved at week 12 and week 52 compared with their baseline visit based on the CGI-c scale (Figure 5). The improvement in CGI-c was consistent regardless of triptan experience subgroup. Overall, at week 52, the proportion of participants rated as improved was 90.3% (195/216), 92.1% (303/329), 89.9% (267/297), 90.3% (392/434) and 91.2% (125/137) for triptan-using, triptan-naïve, and =1, ≥1 and ≥2 triptan-failure participant subgroups, respectively (Figure 5).

CGI-c at week 12 and week 52 (PRN) by cohort and triptan experience subgroup. Triptan-using: current triptan use and no historical discontinued triptans. Triptan-naïve: no current triptan use and no historical discontinued triptans. CI, confidence interval; CGI-c, Clinical Global Impression of change; EOD, every other day dosing; PRN, as needed dosing.
Discussion
This post-hoc analysis of a prospective, phase 2/3, one-year open-label safety study explored the safety, tolerability, and patient-reported outcomes of rimegepant in the acute treatment of migraine based on participants’ current and historical experience with triptans. This analysis demonstrated that long-term acute treatment of migraine with rimegepant 75 mg up to once daily as PRN (up to 52 weeks) and EOD + PRN (up to 12 weeks) was safe and well tolerated in participants who were either triptan-naïve, triptan-using or had single or multiple triptan-failure. A nominally higher rate of on-treatment AEs was observed in participants with ≥2 triptan-failure relative to triptan-naïve participants. Other safety measures including discontinuation due to on-treatment AEs, hepatic-related AEs and liver function test elevations were low and of a comparable magnitude among triptan experience subgroups. The safety and tolerability results of this analysis are generally consistent with those of the original clinical trial (13) and other published rimegepant studies (8–12).
In this analysis, triptan-failure participants generally had comparable baseline demographics and clinical characteristics to those who were triptan-naïve and triptan-using. Participants with ≥2 triptan-failure tended to be older and have a longer median time since migraine onset than those who were triptan-naïve; however, this may reflect the duration for which they had received acute treatment, rather than a potential indicator of triptan failure. People with triptan-insufficient efficacy and/or tolerability have previously been reported to have more frequent and severe headaches at baseline (3,21). In this analysis, participants experienced a comparable number of moderate to severe migraine attacks per month, regardless of historical triptan experience; however, participants with historical discontinuation did experience a longer average duration of untreated migraine attack compared with those who were triptan-naïve and triptan-using, indicative of a greater need for optimization of acute treatment in these individuals.
There is a continued need for new acute treatment options for people with migraine to help address unmet medical needs (3,22–24). It is important to note that triptans are also underused, and, for some people, triptans may be the most favorable option in terms of overall efficacy and safety for the acute treatment of migraine (25). However, triptans are not universally effective nor suitable in all people with migraine. Therefore, alternative therapies with different modes of action represent additional options for such individuals. In a two-year longitudinal analysis of people with migraine using triptans based in Italy, almost 40% of people with migraine had unmet medical needs (23). In the 2017 MAST (Migraine in American Symptoms and Treatment) study of almost 4000 respondents who were using oral acute prescription medication, a total of 95.8% (3765/3930) of respondents had at least one unmet acute treatment need and attack features including rapid headache onset, moderate to severe disability, inadequate two-hour pain freedom and headache recurrence within 24 hours were particularly noteworthy areas of sub-optimal treatment response (24). These unmet needs may account for the dissatisfaction many people have with their acute treatment, leading to approximately 80% willing to try a new treatment (15). The emergence of gepants, including rimegepant, as an effective acute treatment for migraine therefore marks an important therapeutic opportunity. Notably, rimegepant has been shown to be fast-acting, providing pain relief within one hour post-dose (8). Coupled with the favorable safety and tolerability profile described herein, rimegepant may be of particular benefit to those with a history of triptan discontinuation. For people who receive acute treatment with triptans and do not experience meaningful pain-relief at two hours post-dose, there is evidence that re-challenging or switching to a different triptan may result in subsequent pain-relief for some (3). However, there is concern that cycling through triptans, or other pain-relief treatments, in an attempt to achieve adequate pain-relief can lead to overuse, and also add to the greater migraine burden due to cycling through ineffective or poorly tolerated treatments (26). Furthermore, switching triptans may result in increased primary care costs and no improvement of headache-related disability (5,6). In this analysis, rates of potential medication abuse AEs were low and of a comparable magnitude across triptan experience groups.
In this post-hoc analysis, clinically meaningful numeric improvements in migraine-related disability, quality of life and CGI-c scores were coupled with high levels of medication satisfaction and preference for rimegepant for both PRN (up to 52 weeks) and EOD + PRN (up to 12 weeks) dosing for all triptan experience subgroups. This suggests that people who may have experienced an insufficient response, or lack of tolerability, to triptans may benefit from switching to rimegepant, an agent with a distinct mechanism of action. This notion is supported by another post-hoc analysis that pooled results from three randomized, placebo-controlled, phase 3 rimegepant clinical efficacy trials, which found that rimegepant was effective in the acute treatment of migraine in participants who had insufficient response to one triptan or ≥2 triptans, as well as in those who were current triptan users (9). Furthermore, it has been suggested that the unmet medical needs in people with migraine receiving triptans may be exacerbated by insufficient use of effective preventive migraine treatments (23). In a multicenter, phase 2/3, randomized, double-blind, placebo-controlled trial, rimegepant 75 mg taken EOD was effective for preventive treatment of migraine (27). In this post-hoc analysis, results from the EOD + PRN cohort, which could be considered as combined preventive and acute treatment, were consistent with the overall and PRN cohorts, suggesting that preventive dosing with rimegepant may also be effective in people with any prior triptan experience, including in those who have discontinued previous treatments.
The present analysis has some limitations. This study was primarily designed for safety/tolerability, and not effectiveness. As this analysis was based on an open-label, non-comparative clinical trial, there is the potential for bias, especially for patient-reported outcomes. The lack of active comparator, specifically triptans, limits the understanding of the potential benefits of switching to an alternative acute treatment following triptan failure.
Conclusions
Long-term acute treatment of migraine with rimegepant 75 mg up to once daily as PRN (up to 52 weeks) and EOD + PRN (up to 12 weeks) was safe and well tolerated in participants who were triptan-naïve, triptan-using, and in those who had single or multiple historical triptan discontinuations. Improvements in migraine-related disability, quality of life and CGI-c scores, together with high levels of medication satisfaction and preference were associated with rimegepant in participants with any historical triptan experience.
Article highlights
A post-hoc analysis of a phase 2/3, open-label study of rimegepant in the acute treatment of migraine according to current and historical triptan use
Rimegepant 75 mg administered up to once per day as PRN (52 weeks) or EOD + PRN (12 weeks) was safe and well tolerated in participants with any triptan experience, including those with multiple discontinuations
Footnotes
Acknowledgments
Medical writing support was provided by Leon Adams, PhD, and Anne Marie McGonigal, PhD, CMPP, of Engage Scientific Solutions and was funded by Pfizer.
Data availability
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JA: Consulting (Honoraria): Abbvie, Aeon, eneura, Dr Reddy, Eli-Lilly, GlaxoSmithKline, Lundbeck, Linpharma, Ipsen, Merz, Pfizer, Neurolief, Gore, Satsuma, Scilex, Vectura Fertin, Theranica, Tonix Clinical Trials (Grant to institution): Parema, Ipsen, Lundbeck, Abbvie Editorial Boards/Steering Committee: Medscape, SELF magazine (medical editor). JMP: Research support: NIH; Consultant/advisory board/honoraria: Allergan / AbbVie, Haleon, and Pfizer. TF and GCP are employed by and own stock/stock options in Pfizer Inc. GCP also owns stock in AbbVie.
Ethical statement
The Institutional Review Board/Independent Ethics Committee approved of the protocol and study-related materials and participants were provided written informed consent.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Biohaven, which was acquired by Pfizer in October 2022.
