Abstract
Background
Retinal migraine is a diagnosis of exclusion and is characterized by repeated episodes of transient monocular blindness associated with migraine. We report a case of systemic lupus erythematosus with acute episodes mimicking retinal migraines.
Case report
A 46-year-old woman with a history of migraine with aura since her 20s and Evans syndrome presented with episodic transient monocular blindness. Retinal migraine was considered as the cause, and migraine prophylaxis initially reduced its frequency. After 5 months, the frequency increased, with chilblain-like lupus lesions on her extremities. Laboratory testing revealed lymphopenia and hypocomplementemia, fulfilling the diagnostic criteria for systemic lupus erythematosus, which may have caused Evans syndrome and transient monocular blindness, mimicking retinal migraines. After intravenous methylprednisolone and rituximab therapy, the transient monocular blindness episodes did not recur.
Conclusion
Given the clinical presentation, systemic lupus erythematosus should be considered as a cause of transient monocular blindness and should be distinguished from retinal migraine.
Keywords
Introduction
The International Classification of Headache Disorders, 3rd edition (ICHD-3) defines retinal migraine as fully reversible attacks of transient monocular blindness (TMB) associated with migraine headaches (1). In patients with symptoms of retinal migraine, other causes of TMB should be excluded. Although the etiopathogenesis of retinal migraine remains largely unknown and controversial, proposed mechanisms include vasospasm in the retinal or ciliary vasculature and a spreading depolarization (SD) of retinal neurons similar to that in the cerebral cortex (2). Additionally, retinal migraine is a diagnosis of exclusion, requiring exclusion of other disease including amaurosis fugax and vasculitis (3).
Evans syndrome (ES) comprises Coombs-positive idiopathic autoimmune hemolytic anemia (AIHA) and immune thrombocytopenia (ITP) and is managed with immunosuppressive therapy. It can be a primary or secondary condition depending on its association with other diseases such as hematological malignancies and systemic lupus erythematosus (SLE) (4). Here, we describe a rare case of SLE with TMB episodes. Our case was initially misdiagnosed as retinal migraine as a result of a history of migraine with aura (MwA) and, unexpectedly, underlying SLE was the cause.
Case report
A 46-year-old woman with a history of MwA since her 20s presented with recurrent TMB. The aura involved repeated visual field scintillation for 20–30 minutes. This was followed by a pressure-like, constricting and sometimes pulsatile headache, which was usually unilateral and temporal, and accompanied by nausea, photophobia and phonophobia. Symptoms usually lasted for 3–4 hours and were responsive to acetaminophen. She also had amblyopia in the right eye, ITP for more than 10 years, previous venous thromboembolism (VTE) and two episodes of imminent miscarriages.
The first episode of TMB, which occurred suddenly at 46 years of age, was associated with complete visual loss in her right eye, lasted for 30 minutes and was not accompanied by a headache. By alternately covering each eye during the attacks, she confirmed that vision loss was only present in the right eye. In the same month, the patient had an acute exacerbation of anemia with jaundice and was diagnosed with AIHA, leading to the diagnosis of ES. This was treated by increasing the daily dose of prednisolone for ITP from 5 mg to 50 mg.
Three months after the diagnosis of AIHA, repeated episodes of TMB occurred. According to her 14-month headache diary, there were 73 episodes of TMB, 21 of which were accompanied by headache. However, there were 22 episodes of bilateral scintillating scotoma, 18 of which were associated with headache. The episodes usually had sudden onset with appearance of a dark gray spot in the upper visual field that resolved gradually within 5–30 minutes (≤90 minutes). Occasionally, symptoms gradually manifested as monocular vision loss, and some episodes were accompanied or followed by an ipsilateral headache within 1 hour of TMB onset. Six episodes occurred in her left eye with graying her upper left visual field and were accompanied by left orbital pain in two episodes. TMB was never associated with other aura types.
Physical, ophthalmological and neurological exams were normal, and tests for coagulation disorders were all negative. As a result of her medical history of VTE, antiphospholipid syndromes were previously ruled out, and the prothrombin time international normalized ratio was therapeutic with warfarin treatment. Cerebrospinal fluid studies revealed an opening pressure of 10 cmH2O, normal leukocyte count (polynuclear cells, <1; mononuclear cells, 1) and slightly elevated protein levels (48 mg/dl). No abnormalities were noted on head computed tomography angiogram/venogram, brain magnetic resonance imaging (MRI)/angiography or carotid ultrasonography. Transthoracic echocardiogram with microbubble study did not detect a patent foramen ovale. The electroencephalogram showed no abnormality. Fundus examination was normal even during TMB and fluorescein angiography revealed no abnormalities including retinal spasm or embolism, ruling out important vascular causes including retinal artery occlusion and vasculitis. Therefore, based on the ICHD-3 criteria, we initially diagnosed her with definite retinal migraine.
Figure 1 summarizes the initial TMB course. Warfarin was continued based on her medical history of VTE. Lomerizine, valproate and topiramate were individually trialed as migraine prophylactics. Lomerizine 10 mg/day was taken for 1 month and followed by valproate 400 mg/day for 1 week, although they were both ineffective and discontinued. Topiramate 50 mg/day was used for 1 month and reduced the frequency of TMB but was discontinued as a result of intolerable side effects: nausea and dizziness.
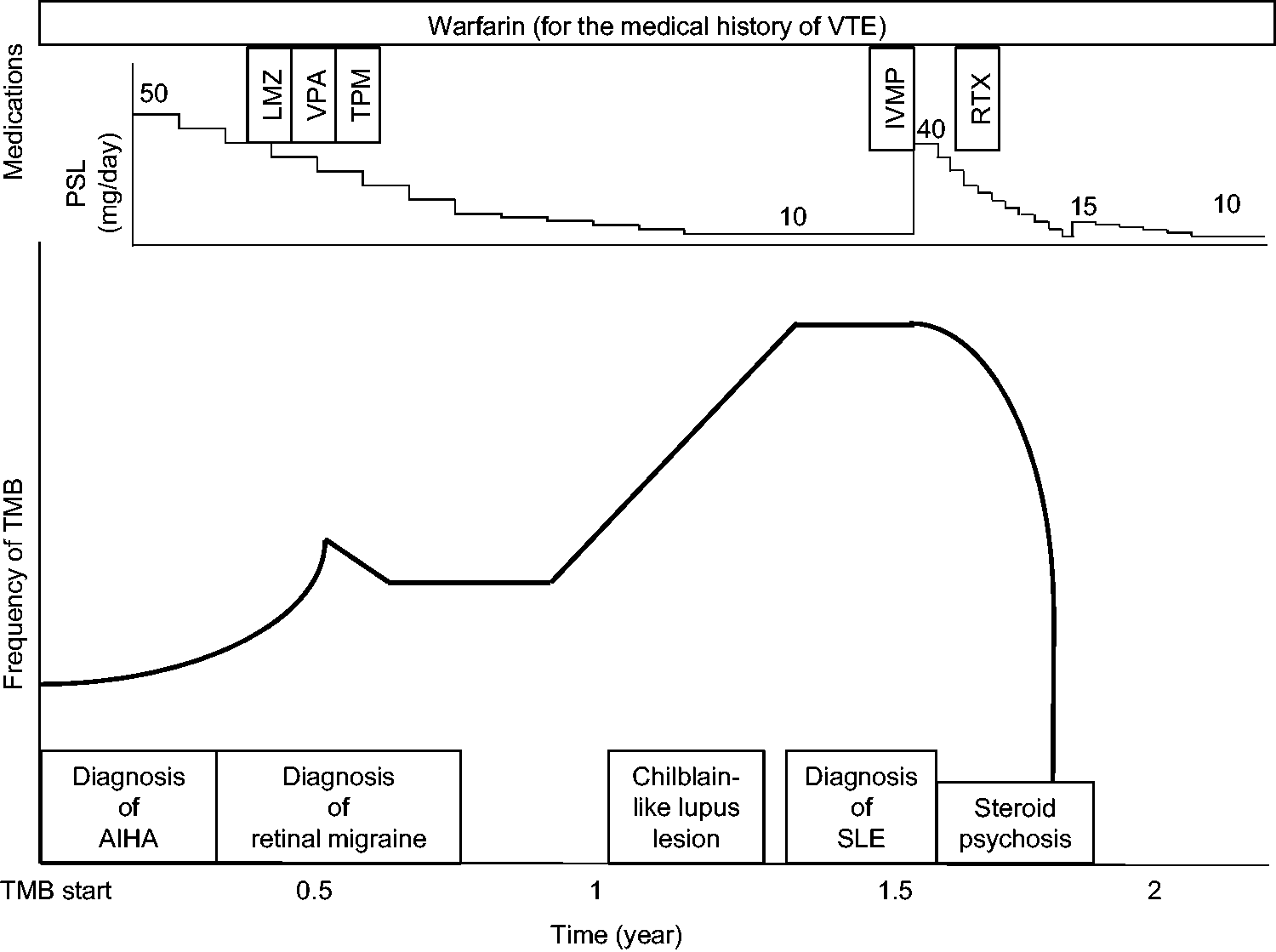
Clinical course observed in this case involving transient monocular blindness as a manifestation of systemic lupus erythematosus, mimicking retinal migraines. Clinical course showing the frequency of transient monocular blindness episodes, prednisolone (PSL) dose, and the timing of each event and treatment. AIHA, autoimmune hemolytic anemia; IVMP, intravenous methylprednisolone; LMZ, lomerizine; RTX, rituximab; SLE, systemic lupus erythematosus; TMB, transient monocular blindness; TPM, topiramate; VPA, valproate; VTE, venous thromboembolism.
Five months later, the frequency of TMB episodes increased, and she complained of pain and chilblain-like lupus lesions on her left hallux (Figure 2a). Skin perfusion pressure in her left dorsum pedis was 41 mmHg (normal: 80–90 mmHg). On biopsy, the epidermis was normal but mild perivascular lymphocytic infiltrates and mucous changes were observed in the dermis, along with microthrombi in the venules (Figure 2b). Immunofluorescence staining showed immunoglobulin M and C3c deposition in the endothelial walls of superficial dermal vessels. Lymphocyte and complement levels were also decreased and anti-cardiolipin antibody was positive. With consideration for the recently diagnosed AIHA and ITP in addition to these new findings, the diagnostic criteria for SLE were fulfilled (5).

Clinical images of systemic lupus erythematosus (SLE) mimicking retinal migraine. Micro-embolism is observed in multiple locations, aiding the diagnosis of SLE. (a) Macroscopically, erythema and edema are observed from the lateral nail fold to the tip of the left hallux. The skin lesion is diagnosed as chilblain lupus, suggesting a diagnosis of SLE. (b) Thrombi are observed within the blood vessels of the dermis (arrowheads) on hematoxylin and eosin staining from skin biopsy, a finding that supports the etiology of micro-embolism. Scale bar = 200 µm and (c, d) Brain magnetic resonance imaging showing a minor cerebral infarction of the frontal and occipital cortex on diffusion-weighted imaging when intravenous methylprednisolone is administered, and the fibrinolytic system becomes disordered.
It was deduced that SLE-associated hypercoagulability and retinal inflammation may have resulted in retinal spasm or retinal SD, thus mimicking retinal migraine. After initiation of intravenous methylprednisolone (IVMP) in combination with rituximab for SLE and ES, the frequency of TMB significantly decreased.
Unexpectedly, she abruptly experienced bilateral blurred vision that was different from the previous TMB episodes, and head MRI detected a minor frontal and occipital cortex cerebral infarction when IVMP was administered (Figure 2c,d). She also developed steroid psychosis that improved gradually throughout the corticosteroid taper. TMB has not recurred for approximately 3 years since initiating IVMP and rituximab for SLE, suggesting that SLE was the cause of episodic TMB.
Discussion
Retinal migraine is a debatable diagnosis, and revisions to the diagnostic criteria have been proposed. Definite retinal migraine is an exceedingly rare cause of TMB. Hill et al. (6) reported only five patients in the literature up to 2006 that met the ICHD-2 criteria for definite retinal migraine. Moreover, a recent review identified only 12 reported cases in the last 15 years (7). In our case, we initially diagnosed retinal migraine as the cause of TMB and initiated migraine prophylactic medications because there were no abnormal findings. We were able to diagnose SLE as the cause of TMB based on more recent findings and treated the patient with IVMP and rituximab, which abated the TMB attacks.
In general, MwA is linked to an increased risk of ischemic stroke, and migraine can also affect posterior ocular structures, resulting in retinal ischemia (8). In the process of ischemic change, the trigeminovascular system may also be activated and sensitized, initiating a series of neural, vascular and inflammatory events. As for the pathophysiology of retinal migraine, two main theories have been proposed: vasospasm and retinal SD.
Retinal or ciliary circulation vasospasm can cause retinal or optic nerve ischemia, leading to TMB. Several reports have shown funduscopic evidence of retinal vasoconstriction, lasting 1–10 minutes (9–11). Additionally, retinal vasospasm may be associated with underlying systemic diseases such as SLE (12). Although vasoconstriction was not detected in the ophthalmological examinations, TMB may be caused by spasm of the retinal microvasculature, which cannot be visualized by fundoscopy.
The second theory implicates SD of retinal neurons, which is a phenomenon that has been demonstrated in chick retina. SLE is sometimes associated with antiphospholipid antibody, which increases the risk of thrombosis. Moreover, cortical SD can be triggered by micro-embolisms, explaining the higher prevalence of migraine in patients with SLE (1314). Migraine-like headache may not be associated with SLE severity and the frequency of flares (15). However, 29% of patients with migraine and SLE exhibit headache exacerbation during SLE flares (16). Evidence suggesting that specific cautions are necessary for treating primary headaches in patients with SLE is lacking (15). In vivo data support the role of topiramate, a migraine prophylactic, for the suppression of SD (17). Although we could not evaluate the presence of retinal SD in our case, TMB episodes may be related to retinal SD triggered by SLE and suppressed by topiramate, thereby alleviating the frequency of TMB.
ES is a rare autoimmune disease that presents with AIHA and ITP. Workup for ES must exclude differential diagnoses and secondary causes of ES. To the best of our knowledge, ocular complications of ES are very rare and only one pediatric case has been reported (18). However, a previous study showed that the frequency of TMB among patients with SLE was at least 158 per 100,000 per year, compared with 14 in the normal population (19).
Generally, retinal occlusive disease and TMB are common in SLE, occurring in approximately 10% of patients, ranging from mild and asymptomatic retinopathy to severe localized vasculitis associated with blindness (20,21). Additionally, retro-chiasmatic lesions in SLE can also present with headache and TMB (20). In our case, SLE could have caused ES and also led to thromboembolism and autoimmune inflammation, resulting in disordered retinal microcirculation, similar to the vascular findings on skin biopsy. We speculate that the presence or absence of unilateral migraine-like headache accompanying each TMB episode may depend on activation of the trigeminovascular system by disordered retinal microcirculation. Monotherapy with warfarin for a history of VTE was not sufficiently effective to prevent TMB, and immunotherapy with IVMP and rituximab for SLE finally stopped the TMB episodes. The unconventional aspects of TMB episodes, including sudden onset and the absence of headache, should be considered as “red flags” to suspect secondary causes. The high frequency of right-sided TMB may be related to the pre-existing ipsilateral amblyopia, but further follow-up will be needed to understand the etiology.
In summary, we report a rare case of retinal migraine mimics associated with SLE. Initially, a misdiagnosis of retinal migraine from the viewpoint of the headache specialist was made based on a history of MwA and normal initial findings. In our case, TMB may have been caused by disordered retinal microcirculation, associated with microthrombi and inflammation in the setting of SLE. Therefore, other causes mimicking retinal migraine must be considered in such cases.
Clinical implications
Systemic lupus erythematosus can manifest as both migraine and retinal disorders; this provides clues to the pathophysiology of retinal migraine. Clinicians should suspect retinal migraine mimics and be cautious of transient monocular blindness as a manifestation of systemic lupus erythematosus. Unconventional aspects of transient monocular blindness, including sudden onset and the absence of headache, can be “red flags” to suspect the presence of secondary causes beyond retinal migraine.
Footnotes
Acknowledgments
We thank Professor Mi Ji Lee, MD, PhD, at the Department of Neurology, Seoul National University Hospital, Seoul National University College of Medicine, Seoul, Republic of Korea, for insightful discussions about this case.
Author contributions
TTa was involved in the study concept and design. TTe, MShi, HH, TK, KA, MSa and TTa were involved in the clinical care of the patient and acquired data. All authors interpreted the patient data. TTe and TTa drafted the manuscript. All authors revised the manuscript for intellectual content. All authors read and approved the final version of the manuscript submitted for publication.
Data availability
No datasets were generated or analyzed for this case report.
Declaration of conflicting interests
TTa is a consultant/advisor and/or serves on an advisory board for Otsuka, Eli Lilly, Amgen, Pfizer and Teijin. TTa received speaker honoraria from Otsuka, Eli Lilly, Daiichi Sankyo, Amgen, Kowa, Kyowa Kirin, Eisai, UCB Japan, Takeda and Santen Pharmaceutical, as well as research funding from Eli Lilly and Tsumura outside the submitted work. JN received honoraria and research scholarships from Amgen and Daiichi Sankyo.
Ethical statement
Written informed consent was obtained from the patient for publication.
Funding
The publication of this article was supported by JSPS KAKENHI (grant number 19K16989 and 22K15693 to TTa) and Keio University Global Research Institute (KGRI) Start-up grant to TTa.
