Abstract
Introduction: The objective of this study was to compare the prevalence of primary headaches in systemic lupus erythematosus (SLE) versus healthy subjects, and to determine whether headaches in SLE are associated with MRI- or cerebrospinal fluid (CSF) abnormalitites.
Patients and methods: The case-control study included MRI- and CSF investigations. Headache was classified according to the International Classification of Headache Disorders. Depression and fatigue were measured with Beck Depression Inventory (BDI) and Fatigue Severity Scale (FSS) respectively.
Results: Twenty-four out of 67 SLE patients and 13 out of 67 age- and gender matched healthy subjects had migraine (36% vs 19%, P = 0.03). Nine (13%) SLE patients had migraine with aura vs 4 (6%) in healthy subjects, P = 0.14. The prevalence of tension type headache was equal (60% in patients vs 58% in controls). There was no association between migraine and SLE disease activity, biochemical or immunological markers, cerebral white matter hyperintensities, interleukin-6 in CSF, impairment of the blood-brain barrier, or intrathecal immunoglobulin production. SLE patients had higher BDI- and FSS scores compared with healthy control subjects, and SLE patients with migraine had higher BDI scores than lupus patients without migraine.
Conclusions: Migraine is more prevalent in SLE patients, associated with depression like in the general population, but not associated with disease activity or abnormalities detected on cerebral MRI, in CSF, or any SLE characteristics except from SLE photosensitivity. The inclusion of the migraine item in SLE disease activity instruments remains questionable.
Introduction
Systemic lupus erythematosus (SLE) is a chronic, inflammatory autoimmune disease that can affect multiple organs, including the central nervous system (CNS). CNS involvement may vary from subtle disturbances of cognition and mood to the more dramatic and life-threatening conditions of psychosis, seizures, or cerebral infarctions (1). Headaches are commonly reported by SLE patients, and have traditionally been considered to be part of the disease spectrum. This fact is reflected in the American College of Rheumatology (ACR) criteria for neuropsychiatric SLE (NPSLE) in which headache is classified as one of the 19 neuropsychiatric syndromes thought to be manifestations of the disease (2). This view also includes headache as a criterion in measures for disease activity, including the SLE disease activity index (SLEDAI), where the presence of a severe, persistent headache that ‘may be migrainous, but must be nonresponsive to narcotic analgesia’ has a significant effect on the score (3).
Whether primary headaches are truly more prevalent in lupus patients than in the general population is still a matter of discussion; divergent views may be due to methodological issues in previous trials, such as small study samples, lack of control groups, lack of adequate criteria for headache, and selection of patients that do not correctly reflect the unbiased lupus population. One meta-analysis including studies published from 1975 to 2002 revealed no difference in headache prevalence between SLE patients and healthy subjects (4). Since then, two controlled studies came to seemingly contradictory results: one reporting a higher prevalence of migraine in SLE patients (5) and the other found more headache in general among SLE patients but no significant differences for any primary headache, including migraine (6). One Norwegian study applying the 1988 IHS-criteria (7) described the characteristics and prevalence of headache in SLE patients, reporting that headaches occur frequently in patients with SLE, but are probably not more prevalent than in a comparable non-SLE population (8).
The aim of the present study was to estimate the true prevalence and types of headache in Norwegian patients with SLE, in a geographically different area than the previous study. We applied the latest International Classification of Headache Disorders, 2nd edition (ICHD-II) criteria (9), included an age- and gender-matched control group of healthy subjects, and performed cerebral magnetic resonance imaging (MRI) for evaluation of structural abnormalities and white matter hyperintensities (WMH). In addition, we studied immunological or inflammatory disturbances of the CNS as revealed by increased interleukin (IL-6) in the cerebrospinal fluid (CSF), intrathecal immunoglobulin production, or impairment of the blood–brain barrier (BBB) to detect any association with headache in general or its variants.
Patients and methods
Patients
Stavanger University Hospital is the only hospital in the southern part of Rogaland County, Norway, and all patients with SLE in this region are referred to this hospital. The hospital provides local and regional services to a population of 310,000 inhabitants. Medical records of all in- and out-patients with a diagnosis of SLE from 1980 to 2004 were reviewed. Eighty-six patients, all Caucasian, fulfilled the 1982 revised American College of Rheumatology (ACR) criteria for SLE (10). Seventy patients (81%) gave informed consent to be included in the study, which was approved by the Regional Research Ethics Committee and complied with the Helsinki Declaration. Two patients withdrew their consent after inclusion and one patient was diagnosed with a brain tumour and subsequently excluded. Sixty-seven patients – 58 women (87%) and 9 men (13%) – were thus included in the study and subjected to a 2-day stay in the hospital exclusively for the purpose of investigation. All laboratory and other investigation procedures were performed during these 2 days or, in some instances, within a few weeks after discharge. Mean age was 43.4 ± 13.3 years (median, 42.4 years; range, 19.6–75.9 years) and mean disease duration was 12.3 ± 8.6 years (median, 11 years; range, 0.5–32.0 years). Disease activity was mild, with a mean SLE disease activity index (SLEDAI) (11) of 3.7 ± 3.9 (median, 2.0; range, 0–26). Cumulative organ damage assessed by the Systemic Lupus International Collaborating Clinics/ACR damage index (SLICC) (12) was 2.2 ± 2.1 (median, 2.0; range, 0–11).
Due to remission or very low disease activity, 12 of the 67 patients (18%) were not taking any drug for SLE. Forty-four patients (66%) used corticosteroids, 34 (51%) used antimalarial medications, and 35 patients (52%) received immunosuppressants (mostly azathioprine or cyclophosphamide) at the time of investigation.
Healthy subjects
Sixty-seven healthy age- and gender-matched individuals without neurological, immunological, or malignant disease served as controls. Age was matched within ±2 years, giving a mean age of 43.4 ± 13.3 years (median, 42.4 years; range, 21.2–76.7 years). The healthy subjects were recruited among hospital staff, neighbours of the hospital staff, as well as friends and neighbours of the patients. The control group was subjected to a 2-day investigation in the hospital similar to the SLE group.
Years of education for SLE patients were (median) 13.0 years; range, 7.0–20.0 years, vs the controls (median) 15.0 years; range, 10.0–25.0 years (P = 0.001). Working status was not systematically recorded.
Clinical evaluation
All patients and healthy control subjects underwent clinical examination by an experienced internist (EH) and a neurological examination by an experienced neurologist (ABT). Headache was assessed by the neurologist, using structured interview and classified according to the ICHD-II criteria (9). A neutral question about having had headache within the previous year was used as screening question. The structured interview included a detailed neurological and headache history covering the relevant criteria to make a headache diagnosis according to ICHD-II. The ICHD diagnostic criteria for primary headaches are symptom-based and classify patients according to the phenomenology of their headaches. Secondary headaches considered to be due to any disease other than SLE were excluded. The terms ‘migraine’ and not ‘migraine-like headache’ were used. This decision was based on headache description fulfilling ICHD migraine criteria A–D in a context outside disease flares and not attributed to another disorder.
Headache status was defined as one or more headache attacks during the last 12 months.
To assess depression, we used the Beck Depression Inventory (BDI) (13). The Fatigue Severity Scale (FSS) (14) and a fatigue visual analogue scale (VAS) were used to evaluate fatigue.
Laboratory evaluation
Routine haematological, biochemical, and immunological tests were analysed in the hospital’s laboratories. The HEp-2000 assay (Immunoconcepts, Sacramento, CA, USA) was used for detection of antinuclear antibodies (ANA) and Nova Lite dsDNA Crithidia luciliae 708200 indirect immunofluorescence assay (NOVA Diagnostics, San Diego, CA, USA) for anti-dsDNA antibodies. To detect lupus anticoagulant (LA), activated partial thromboplastin time and dilute Russell’s viper venom time (Dade Behring, Marburg, Germany) were used. QUANTA Lite™ ACA IgM and IgG ELISA (Inova Diagnostics, San Diego, CA, USA) were used for screening anti-cardiolipin IgM and IgG antibodies. Complement factors C3 and C4 were detected by nephelometry.
CSF samples were obtained by lumbar puncture in 52 patients between 1 pm and 2 pm. All samples were immediately placed on ice and centrifuged at 4°C at 3000 g for 10 min; the supernatant was then immediately aliquoted and frozen at –70°C until analysis. IL-6 was measured with the Luminex 100 (Luminex Corp., Austin, TX, USA) using IL-6 UltraSensitive™ AB Bead Kit, together with human extracellular buffer kit (BioSource™, Invitrogen Corp., Carlsbad, CA, USA). The sensitivity for IL-6 was <1.0 pg/ml (range, 0.84–614 pg/ml). Acquired data were analysed using the StarStation software v2.3 (Applied Cytometry, UK). Measurements of IgG and albumin in CSF and serum was performed according to the manufacturer’s instructions with the Cobas Integra Immunoglobulin G (Turbidimetric) and Tina-quant a Albumin Gen.2, respectively (Roche Diagnostics, Mannheim, Germany). Impairment of the blood–brain barrier (BBB) was defined by a CSF/serum albumin index (albuminCSF [mg/l])/albuminser [g/l]) >9. Intrathecal immunoglobulin production was defined as an IgGCSF/albuminCSF ratio (IgGCSF [mg/l]/albuminCSF [mg/l]) >0.27 and an IgG index (IgGCSF [mg/l] × albuminser [g/l]/IgGser [g/l]/albuminCSF [mg/l]) >0.7 (15).
Cerebral MRI
MRI examinations were performed with a 1.5-T Philips Gyroscan NT Intra Release 10 (Philips Medical Systems, Best, The Netherlands). The interval between clinical and MRI examinations was 12 ± 11 days for SLE patients and 28 ± 24 days for healthy subjects. Two experienced radiologists (MB and OJG) rated each scan independently in a blinded manner. If disagreement occurred, a consensus was reached by discussion.
The MRI scan protocol was as previously described (16). To assess WMH scores, four regions of cerebral white matter were evaluated using the semi-quantitative scale of Scheltens: the periventricular region (PV); the subcortical and deep white matter of the frontal, parietal, temporal, and occipital areas (FPTOs); the basal ganglia (BG); and the infratentorial region (IT) (17). The method takes into account the spatial localisation, size, and number of lesions. The inter- and intrarater reliabilities for this procedure were shown to be good to excellent in a previous study with the same radiologists (18). The total WMH sum score (range 0–84) as a measure of WMH burden has shown good correlation with WMH lesion volume (19).
EEG
All EEG recordings were performed with digital EEG machines (Walter Graphtec GMBH, Lübeck, Germany and Galileo NT, Florence, Italy). EEG electrodes were positioned according to the international 10–20 system (20) and standard recording were performed for 20 min during relaxed wakefulness, with 3 min of hyperventilation, and with photic stimulation. All EEG recordings except for one were reported by the same electroencephalographer (AH).
Statistical analysis
Results are reported as mean ± SD when normally distributed, otherwise as mean ± SD with median and range. Chi-squared test was used comparing pair-wise categorical data when expected frequencies was more than 5, otherwise Fisher’s Exact test was used. Unpaired t-test was used for continuous data when normally distributed and Mann–Whitney U-test when not. Logistic regression was used when appropriate. P < 0.05 was considered significant.
Results
Headaches
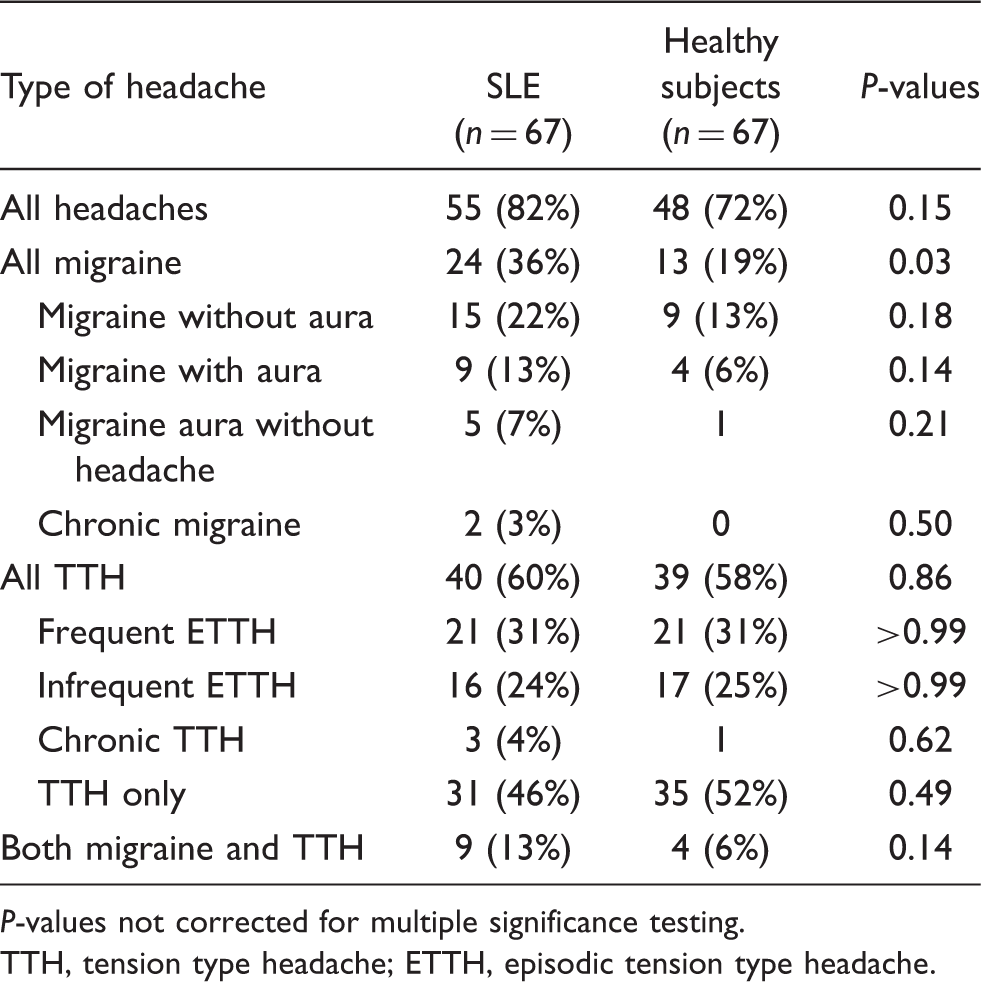
Twenty-four of the SLE patients (36%) and 13 of the healthy subjects (19%) had migraine attacks during the last 12 months (P = 0.03). Nine patients (13%) had migraine with aura including five who had attacks also fulfilling the criteria for migraine aura without headache. Four (6%) of the healthy subjects had migraine with aura including one subject who had migraine aura without headache. The prevalence of tension-type headache did not differ between lupus patients and healthy subjects (Fig. 1, Table 1). One SLE patient had classical trigeminal neuralgia, and one healthy subject reported primary stabbing headache.
Headache prevalence in 67 patients with SLE and 67 age- and gender-matched healthy subjects. SLE, systemic lupus erythematosus; TTH, tension type headache; h.a., headache; ETTH, episodic tension type headache. Prevalence of headache types in 67 patients with systemic lupus erythematosus (SLE) and 67 age- and gender-matched healthy subjects
P-values not corrected for multiple significance testing. TTH, tension type headache; ETTH, episodic tension type headache.
Cerebral MRI
Sixty-two SLE patients completed MRI scan of the brain. Eleven patients (18%) had cerebral infarcts. Of those, three patients (27%) had one infarct and eight (73%) had two or more. Three patients (28%) had cortical infarcts, four (36%) had lacunar, and four patients (36%) had combinations of cortical and lacunar infarcts. All the 67 healthy subjects were examined by MRI. Two of these (3%) had cerebral infarcts, in both consisting of two or more lacunar infarcts.
White matter hyperintensities
Total white matter hyperintensities (WMH) and different headache categories in 62 patients with systemic lupus erythematosus (SLE)

P-values not corrected for multiple significance testing.
TTH, tension type headache.
EEG
All 67 SLE patients and 65 of the healthy subjects underwent EEG examination. Twenty-eight patients (42%) had abnormal EEGs of whom 11 (39%) had focal abnormalities and 15 (54%) diffuse abnormalities; two (7%) had epileptic activity. Five of the 12 patients without headache (42%) had EEGs with focal pathology compared with six out of 55 with headache (6%; P = 0.02). No other differences regarding headache and EEGs were detected.
Twelve (18%) of the healthy subjects had abnormal EEGs consisting of focal abnormalities in nine (15%) and diffuse abnormalities in three (5%). None had epileptic activity. SLE patients had more abnormal EEGs (42% vs 18%; P = 0.004) and more diffuse abnormalities compared to healthy subjects (22% vs 5%; P = 0.003) while no differences were revealed regarding focal abnormalities (16% vs 14%; P = 0.68).
Laboratory tests
No differences appeared in relation to headaches in general or any category of headache regarding haemoglobin, leukocytes, platelets, erythrocyte sedimentation rate, ANA, anti-nDNA antibodies, lupus anticoagulant, anti-cardiolipin antibodies, anti-β2-glycoprotein 1 antibodies, or complement factor C4. Complement factor C3 was lower in patients without headache compared with those with any headache (median, 0.83; range, 0.59–1.37 vs median, 1.09; range, 0.46–1.57; P = 0.01), although within normal ranges. This correlation was not present for subtypes of headache. The significance of this finding is uncertain and probably coincidental.
CSF
Five out of the 52 patients (10%) in whom spinal puncture was performed, all with TTH only, had elevated CSF/serum albumin index, indicating impairment of the BBB. None of the 29 patients without TTH only had BBB impairment (P = 0.01). Three of these five patients had infrequent ETTH and two had frequent ETTH. Patients with any headache, any migraine, or migraine with aura showed no differences compared with patients without these headaches. There was no difference regarding the indices measuring intrathecal IgG synthesis. Four had a slightly increased IgGCSF/albuminCSF ratio and only one had an increased IgG index. The one patient with positive IgG index had no headache in the previous year. There was no association between IL-6 concentrations in CSF and any headache (median, 3.09; range, 0.00–9.94 vs median, 3.15; range, 0.0–8.45) or category of headache (data not shown).
Relation of headache to other variables in SLE patients
Twenty-three of the 24 SLE patients with migraine (96%) reported SLE photosensitivity compared with 31 of the 43 non-migraine patients (72%; P = 0.02). No associations were found between any type of headache and the use of any type of medication for SLE, SLEDAI, SLICC, disease duration, thrombo-embolic episodes, or hypertension.
Fatigue
SLE patients had higher FSS scores than healthy subjects (median, 4.3; range, 1.3–7.0 vs median, 2.0; range, 1.0–5.3; P < 0.0001). The same applied to fatigue VAS in patients vs controls (median, 49.0; range, 1.0–98.0 vs median, 12.0; range, 1.0–72.0; P < 0.0001). SLE patients with any headache had higher FSS scores (median, 4.8; range, 1.3–7.0) than those without headache (median, 3.5; range, 1.3–5.7; P = 0.03). The same was true regarding migraine with aura (median, 6.3; range, 4.0–7.0 vs median, 4.3; range 1.3–6.6; P = 0.007). Patients with all migraine had higher scores than those without migraine, although not significant (median, 4.7; range, 1.3–7.0 vs median, 4.3; range, 1.3–6.6; P = 0.06). Patients with TTH only did not have significantly higher scores compared to those without this headache (median, 4.8; range, 1.3–6.6 vs median, 4.3; range, 1.3–7.0; P = 0.89). Fatigue VAS was significantly higher in SLE patients (median, 57.0; range, 1.0–98.0) with any headache compared with those without headache (median, 16.5; range, 4.0–79.0; P = 0.01), but no differences were apparent in the headache subgroups.
Healthy subjects with migraine did not have higher FSS scores (median, 2.0; range, 1.3–4.0) than those without migraine (median, 2.1; range, 1.0–5.3; P = 0.66). The same applied to FSS scores in healthy subjects with migraine with aura (median, 1.8; range, 1.4–3.6) compared with those without this headache subtype (median, 2.1; range, 1.0–5.3; P = 0.81).
Depression
Becks Depression Inventory scores and different headache categories in 63 patients with SLE

P-values not corrected for multiple significance testing.
TTH, tension type headache.
Discussion
There are some important data from this study. First, migraine prevalence in lupus patients was nearly twice that of healthy subjects. Second, migraine was not associated with disease activity or any disease characteristics except from SLE photosensitivity. Third, tension type headache was not more prevalent in the patients than in the healthy subjects.
Whether migraine headaches occur more frequently in SLE patients than healthy controls has been a subject of debate for many years. An alternative explanation for high migraine prevalence in SLE is that the epidemiology of both migraine and SLE shows a considerable female preponderance and that the age-related peak prevalence of migraine and SLE patients are almost identical (8). This controlled study reveals significantly more migraine in the SLE patients compared with healthy subjects of the same age and gender. The reported migraine prevalences in SLE patients as well as healthy controls are in agreement with data reported by others in Scandinavia (8),(21),(22). Concerning migraine with aura, one meta-analysis revealed a significant increase in migraine with aura in SLE patients compared with controls: 13% versus 5%, P < 0.05 (4). These prevalences are identical to our study (i.e. 13% vs 6%), the difference not reaching significance, probably because of a smaller number of subjects.
There were no associations between migraine or migraine with aura and any disease associated or demographic variable, including disease activity or duration, organ damage, any biochemical or immunological variables, load of WMH on cerebral MRI, IL-6 in CSF, impairment of the BBB, or intrathecal immunoglobulin production. Focal EEG abnormalities were more common in SLE patients without headache. This is illogical and similar findings were not evident in the controls. This observation is, therefore, probably coincidental.
Migraine is a complex neurobiological disorder susceptible to several trigging factors (23). Immunological mechanisms in SLE may render the brain of SLE patients more susceptible to triggers and thus migraine. In that sense, higher prevalence of migraine could indirectly indicate SLE-related dysfunction in the brain. This is interesting in as much as SLE patients with migraine had significantly more SLE photosensitivity than patients without migraine, and it is a clinical experience that sunlight exposure to photosensitive SLE patients may lead to dermatitis, and even initiate a general disease flare.
It is well documented that CSF IL-6 is increased in patients with active cerebral SLE (24), but its presence did not influence headache presentation in our study. Breakdown of the BBB in SLE patients has recently been hypothesised as a necessary prerequisite for an immune attack on the CNS (25). Our results do not indicate such a mechanism for migraine in SLE.
SLE patients in our study had more depression and fatigue than the healthy subjects. Within the SLE group, patients with migraine had higher BDI scores than those without migraine, but the difference in FSS scores did not reach statistical significance (P = 0.06), probably due to low statistical power. However, SLE patients with the subtype migraine with aura had more fatigue than patients without aura. In the general migraine population, more depression is reported (26). More depression and fatigue in the SLE patients may thus be responsible for some of the increased migraine prevalence compared to healthy subjects. TTH only in SLE patients were not associated with depression and fatigue.
There has been much debate over the inclusion of the headache item in the SLEDAI because of disagreement concerning its relevance and also because of its weighting. SLEDAI is a construct not necessarily reflecting what is basically the ‘disease activity’. There are more than 60 other instruments meant to measure disease activity in SLE. Items chosen for inclusion have been the subject of consensus between experts. The individual items are not a function of each other. As far as migraine could be a cerebral manifestation of SLE, one may argue that it defends its place because it reflects SLE disease processes influencing neuronal function and stability. Our findings, however, do not support its prominent impact on the SLEDAI score.
Further, TTH is regarded as the most frequent primary headache disorder, but until lately has not received much attention in epidemiological studies. Our study contributes to the body of knowledge concerning TTH in SLE.
Study limitations
There are some limitations to this study such as small sample size and no correction performed for multiple statistical testing. Further, it could be argued that the control group could have been more optimally selected to reflect the general population. It is possible that the selection performed has drawn more healthy and educated subjects, and thus introduced a bias that has increased the differences. Some headache specialists consider migraine with aura a different entity than migraine without aura being more biological in origin. Our study did not have statistical power to address this question. The use of the term ‘migraine’ instead of ‘migraine-like headache’ is debatable due to ICHD migraine criterion E;(9) as it contextually should be ‘not attributed to another disorder’ (e.g. SLE). Another limitation to the study is the retrospective reporting of headaches and no use of headache diaries.
Strengths of the study include a case-control design with age- and gender-matched subjects, including approximately 80% of all SLE patients identified in our geographical region. We applied the latest criteria for headache disorders, performed clinical examinations, structured interviews, as well as cerebral MRI and CSF investigations. The same neurologist performed a personal interview and neurological examination which is considered the gold standard for making headache diagnosis (27). An argument in favour of the control group representing the general population is that prevalence data are in accordance with findings in large epidemiological headache studies (22),(27).
Footnotes
Acknowledgement
The authors thank Ingeborg Kvivik for assistance with data collection and laboratory work.
