Abstract
Background
Several studies have applied resting-state functional MRI to examine whether functional brain connectivity is altered in migraine with aura patients. These studies had multiple limitations, including small sample sizes, and reported conflicting results. Here, we performed a large, cross-sectional brain imaging study to reproduce previous findings.
Methods
We recruited women aged 30–60 years from the nationwide Danish Twin Registry. Resting-state functional MRI of women with migraine with aura, their co-twins, and unrelated migraine-free twins was performed at a single centre. We carried out an extensive series of brain connectivity data analyses. Patients were compared to migraine-free controls and to co-twins.
Results
Comparisons were based on data from 160 patients, 30 co-twins, and 136 controls. Patients were similar to controls with regard to age, and several lifestyle characteristics. We replicated clear effects of age on resting-state networks. In contrast, we failed to detect any differences, and to replicate previously reported differences, in functional connectivity between migraine patients with aura and non-migraine controls or their co-twins in any of the analyses.
Conclusion
Given the large sample size and the unbiased population-based design of our study, we conclude that women with migraine with aura have normal resting-state brain connectivity outside of migraine attacks.
Introduction
Migraine with aura is a common and disabling neurological disorder characterized by attacks of transient neurological symptoms, most often in the form of gradually developing visual disturbances (1). The aura symptoms are commonly followed by migraine headache and associated symptoms of nausea and photo- or phonophobia. It has been established that migraine with aura reflects dysfunction of the cerebral cortex, most likely the well-characterized phenomenon of cortical spreading depolarization (CSD) (2). CSD has been demonstrated in humans following various types of cortical damage but little is known about how CSD might arise in the healthy-appearing cortex of migraine patients. Also, it is not yet clear how this cortical dysfunction leads to subsequent headache.
Resting-state functional magnetic resonance imaging (rs-fMRI) is a non-invasive technique that uses the spatiotemporal pattern of changes in the blood oxygen level dependent (BOLD) signal to detect correlated activity among remote brain areas, i.e. functional resting-state brain connectivity (3). In migraine patients with visual aura, rs-fMRI might reveal specific patterns of functional resting-state brain connectivity involving the visual cortical areas (4). Alterations in functional connectivity associated with migraine with aura might also be related to cortical properties conveying susceptibility to CSD and subsequent headache.
Previous studies applying rs-fMRI to the study of interictal migraine patients with aura revealed conflicting findings. Several studies reported abnormal connectivity patterns involving different cortical areas and networks (5–12), while one study found no differences in functional connectivity between aura patients and controls (13). All previous studies had several limitations, including a clinic-based design and relatively small numbers of participants. Further, between-group analyses did not take possible confounding lifestyle and health factors into account.
Here we set out to re-examine if migraine with aura alters functional resting-state brain connectivity patterns including the occipital visual cortex. To this end, we performed a set of well-established rs-fMRI analyses in a large population-based sample of female twins with and without migraine with aura.
Materials and methods
We recruited subjects through the Danish Twin Registry, the oldest and most complete national twin registry worldwide (14). We identified female twins born between 1931 and 1982 who, based on earlier responses to a previously used questionnaire (15), were classified as: screen positive for migraine with aura, co-twins to females screen positive for migraine with aura, or screen negative for migraine of any type. We enrolled females from these three groups in our study by a two-step procedure. First, the twins received a brief questionnaire on health and lifestyle issues (sent out in waves from February 2011 to April 2014). Nonresponders received one reminder. Second, we invited eligible questionnaire responders to participate in a semi-structured phone interview conducted by five trained physicians and focused on headache history. Based on interview responses, each subject’s headaches were classified according to the International Classification of Headache Disorders, 3rd edition beta version (16). Twins with definite migraine with aura and their co-twins were invited to participate, if eligible according to study criteria (17). We only recruited potential controls among screen negative twins that were also classified as participants without migraine according to the telephone interview with a single exception (a twin originally classified as co-twin to a sister with migraine with aura, where interview revealed that neither of the twins had ever suffered from any form of migraine). Any information revealing an exclusion criterion led to exclusion at any stage of the study. Eligible subjects were invited to participate in the MRI part of the study at the Danish Research Centre for Magnetic Resonance, Copenhagen University Hospital Hvidovre.
Information on potential confounders and other covariates was collected through the initial postal questionnaire, the telephone interview, and a second brief questionnaire the twins completed on the day of their brain scan. Information on the twins’ zygosity—classified as monozygotic, dizygotic same sex, and dizygotic opposite sex—was provided by the Danish Twin Registry (14). Education level was categorized into ‘low’ in females who reported <12 years of schooling and <3 years of vocational training. Other sociodemographic characteristics were likewise dichotomized (residency in capital and environs (Greater Copenhagen), cohabitation, currently employed). Smoking history was defined as ‘ever’ or ‘never’. For ever-smokers we also calculated pack-years of exposure. The number of alcoholic drinks per week was classified as follows: 0, 1–4, 5–9, and 10+. Ever-use and number of years of oral contraceptive use were recorded. On the day of the brain scan we recorded the twins’ height and weight, their blood pressure (measured in the upper arm before and after the MRI), and withdrew a blood sample to determine cholesterol and glycated haemoglobin values. The height and weight were used to calculate body mass index (BMI; weight in kilograms divided by the square of height in metres). The lowest of the two blood pressure measures was used. Hypertension was defined as systolic blood pressure of ≥160 mmHg or a diastolic blood pressure of ≥95 mmHg, or current use of antihypertensive drugs. Diabetes was defined as self-reported physician diagnosed diabetes, or a measurement of glycated haemoglobin of 44 mmol/mol (6.5%) or higher. Twins were classified as having a history of coronary artery disease if they reported a history of physician diagnosed angina or myocardial infarction.
Throughout this manuscript, “patient” refers to a participant with a definite diagnosis of migraine with aura, “co-twin” refers to a twin sibling to a patient who does not have a diagnosis of migraine with aura (but may have migraine without aura), and “control” refers to a participant with no diagnosis of migraine. On average, patients were scanned six months and controls four months after completing the physician-conducted headache interview (17). Patients were scanned in the interictal phase, i.e. outside of migraine attacks only. Detailed information regarding participant participation is provided in a previous publication (17). Importantly, analyses comparing participants and non-participants indicated no major selection biases. Participants received no financial reward for participating. The study was approved by the Ethics Committee of the Region of Southern Denmark and the Danish Data Protection Agency. Written informed consent was obtained from all participants. The study is registered at clinicaltrials.gov (NCT02047695).
MRI acquisition
Structural and functional images were acquired on a 3T Siemens (Erlangen, Germany) Magnetom Verio MR scanner equipped with a 32 channel receive head coil. Resting-state T2*-weighted functional MRI was acquired using a gradient echo-based echo planar imaging sequence with a repetition time (TR) of 2150ms, 26ms echo time, flip angle 78 degrees, 64 × 64 acquisition matrix, 192 × 192 mm2 in plane field of view, 42 slices of 3mm thickness without slice gap. In-plane readout acceleration with a factor of two was accomplished using the generalized autocalibrating partial parallel acquisition (GRAPPA) using 24 reference lines. The first two volumes were excited without readout to limit effects of T1 equilibrium. In total, 280 volumes were acquired across a ten-minute scan session where participants were instructed to rest with their eyes open. In order to limit the effects of motion, the participants were instructed to refrain from any voluntary movement and their heads were stabilized via foam cushions.
Structural imaging was accomplished with a T1-multi-echo 3D flash sequence with the following parameters: TR=18.7 ms, echo time = 2.20, 4.70, 7.20, 9.70, 12.20 and 14.70 ms, flip angle 20°, field of view = 256 mm, 176 sagittal slices with two factor in-plane acceleration via GRAPPA with 18 reference lines. For segmentation purposes, a volume with contrast comparable to a standard T1-weighted sequence was created by averaging across all echoes acquired in the sequence.
MRI data analysis
The interleaved slice acquisition order was compensated by temporal sinc interpolation with spatial resampling using 4th order spatial b-spline interpolation. For each participant, five volumes with reversed phase-encoding direction were acquired, which enabled us to estimate and compensate for geometric distortions caused by field inhomogeneity. To this end we used the topup tool from the FSL software package (18). The volumes were then realigned to the mean volume of each participant in a two-step procedure involving a six-parameter rigid body transformation with a least square cost function to estimate within session movement parameters. Despiking was performed with the d4 Daubechies mother wavelet using the wavelet despike toolbox version 1.1 (19). Additional nuisance effects were projected out using voxel-wise multiple linear regression. This regression model included a discrete cosine transform (DCT) based high pass filter with a cut-off frequency of 1/128 Hz. The model also included the mean signal from two regions of no interest; the lateral ventricles according to the Harvard-Oxford atlas (20) (reverse normalized to the individual participant), and white matter as estimated from the anatomical scan using the SPM12 unified segmentation procedure with posterior probability threshold of 0.5 eroded two times to eliminate partial gray matter voxels. Residual effects of movement were modelled using a 24 parameter Volterra expansion of the estimated rigid body realignment parameters.
In addition, individual volumes with severe motion effects were detected according to a framewise displacement threshold of 1 mm (assuming a head radius of 50 mm) or changes in the temporal derivative of the full volume RMS (DVARS) exceeding 1%. These volumes were individually modelled thereby effectively disregarding them in further analysis. The model of nuisance effect included a voxel-wise mean, but this was reintroduced in the resulting time series to enable typically masking procedures applied during independent component analysis (ICA). The volumes were then normalized to MNI space (using the SPM12 EPI template) and resampled to 3 mm isotropic resolution by affine registration followed by 1176-parameter low dimensional non-linear warping based on spatial DCT functions utilizing the unified segmentation procedure in SPM12. As a final pre-processing step, the volumes were spatially smoothed with a 6mm full-width-at-half-maximum isotropic Gaussian filter.
All of the rs-fMRI analyses described in the following sections were performed by one of the authors (K.H.M.), an expert in fMRI analyses who was blinded with regard to participant group status (i.e. whether participants were migraine with aura patients, non-migraine controls, or aura-free co-twins).
Independent component analysis
We then identified 25 spatially independent ICA components using the Group Independent Component analysis toolbox version 4.0c across all participants. During ICA modelling, 3 additional initial volumes were excluded to ensure T1 equilibrium and the infomax ICA algorithm (21) with default settings was applied to determine maximally spatially independent sources using a sigmoid non-linearity, which favours super-Gaussian sources.
We visually inspected components to censor any non-neuronal components caused by participant motion, cardiac or respiratory cycles or other nuisance signal contributions. For the remaining components we identified components that spatially resembled components typically observed in resting-state fMRI studies. In particular, components representing either the primary and secondary visual (Brodmann area 17 and 18) were identified via spatial correlation with templates presented in a previous publication (22).
We identified individual expressions of the component maps using back-reconstruction and used a two-sample t-test with age as a covariate to test for differences between migraine patients and healthy controls for each of the identified components. Inference was assessed using random permutation testing through the Permutation Analysis of Linear Models (PALM) toolbox (23) with correction for multiple comparisons using the threshold-free-cluster-enhancement with E = 0.5, H = 2, neighbourhood connectivity of 6 and a cluster significance threshold of P < 0.05. For these tests, we did not consider correction for multiple comparisons across the components. To assess if covariates might influence the results, we did a separate test where we, in addition to age, corrected linearly for body mass index, hypertension, weekly alcohol consumption, and a voxel-wise estimate of the gray matter density. The latter was included as we found structural visual cortical differences between groups in a previously published analysis of this dataset (24).
Next, we investigated differences within twin pairs, where one of the twins had migraine with aura and the co-twin did not have migraine with aura. This was done using paired t-tests, since this matched-pair analysis reduces the influence of several unmeasured confounders, and thus relies on the assumption of discordance in exposure and outcome. We abstained from within-pair analyses stratified by zygosity due to the small number of discordant twin pairs (14 monozygotic and 16 dizygotic pairs) that participated in the study.
Seed based region-of-interest analysis
In addition to identifying spatial components via ICA, we extracted the average signal within areas of interest in particular within the visual cortex. These regions were extracted using the SPM Anatomy Toolbox (25) and included the primary visual areas BA17 and BA18, the dorsal extrastriate cortex (hOC3d and hOC4d) and the primary somatosensory cortex (areas 1, 2, 3a and 3b). Visual area V3A was extracted using the PALS-B12 anatomical atlas (26). These probabilistic cytoarchitectonic maps were thresholded at 0.5. We additionally investigated binary, spherical regions of interests of the hypothalamus and the pons as described in a previous publication on rs-fMRI in migraine with aura (27).
These average time series were then correlated to the signal in all brain voxels to extract measures of functional connectivity between the area of interest and other brain regions. The correlation values were Fisher Z-transformed and entered further analysis via the same procedures as the identified independent components.
Network analyses
To investigate changes in functional network integration, we also estimated functional connectivity networks from the resting-state fMRI data for patients and controls. To this end, adjacency matrices to assess between parcel functional connectivity were constructed by calculating the pairwise Fisher transformed Pearson correlation coefficient between the mean signal in each parcel. A combination of Nilearn (version 0.9.2), NetworkX toolbox (version 2.8.4), scikit-learn (version 1.1.2) and statsmodels 0.13.2 running in Python 3.9.13 and custom scripts were then used to assess various graph metrics and attempt to classify migraine status directly based on the upper triangular adjacency values.
Networks were constructed by adaptively thresholding the adjacency values such that a network sparsity level of 90% was reached for each participant. In the resulting network, we calculated several global network metrics including global efficiency, local efficiency, average clustering and average node connectivity. As these network metrics are highly sensitive to the thresholding procedure applied when forming the graph, we checked several additional thresholding procedures post-hoc to investigate to what extent this affected group differences including thresholding based on a 95% sparsity level, Bonferroni correction across all possible connections and a false discovery rate of q < 0.05 considering positive correlated values. For each of these networks and thresholds we tested for between-group (cases vs. non-migraine controls) differences using individual two-sided two-sample t-tests with pooled variance estimates and considered differences significant if the null-hypothesis could be rejected at a false positive rate of α < 0.05 with Bonferroni-Holm correction among the four tested metrics. We did not consider multiple comparisons across networks or thresholds.
For classification based on functional connectivity, we used a support vector classifier with a linear kernel and 10-fold stratified cross validation (CV) to assess classification performance using area under the receiver operator characteristic curve (AUC) in an outer cross validation loop and estimated the optimal regularization (C-parameter) in a nested inner loop also based on 10-fold stratified CV with AUC as score function. Two different types of feature representations were considered for this classification, one based on within and between network covariance and one based on correlation. The between parcel covariance was estimated using a shrinkage covariance estimator and in the covariance case the upper triangular values (representing between network connectivity) and the diagonal (within parcel variance) was extracted and used as features directly. In the correlation case, the upper triangular Fisher Z-transformed correlation coefficients as estimated using the same shrinkage estimator was used as features in the classification. Significance of the classification performance was assessed used a random permutation-based test where labels of the original data where permuted 10,000 times across participant to form an empirical null distribution using the exact same nested cross validation procedure. Initially, the 268-parcel atlas presented in a previous publication (28) and applied in a previous study (29) was used for parcellation. However, we also investigated parcellations with substantially fewer parcels post-hoc to ensure that negative results were not a result of noise and overfitting due to over-parametrization. These parcellation atlases included the 17-network functional parcellation described in a previous publication (30) and the 90-region cortical anatomical parcellation presented previously (31) as these are widely used in earlier studies (32,33). Additionally, we tested several soft parcellation schemes including the Allen and MIALAB ICA probabilistic atlas (34), the 64-region version of the DIFUMO atlas (35), and the 122-region deterministic multiscale functional brain parcellation. For these probabilistic or weighted parcellations the signal within each parcel was determined as a weighted average of the relevant voxels.
Amplitude of low frequency fluctuations (ALLF)
The fractional amplitude of low frequency fluctuations was estimated using the DPABI Matlab toolbox (version 6.1_220101) with the low and high edges of the pass band defining low frequency fluctuations set to 0.01 Hz and 0.08 Hz, respectively. Significance of the between-group effects for cases vs. non-migraine controls was assessed using the exact random permutation procedure with TFCE as for the ICA components. Similarly, a paired t-test was used to test for differences between affected and unaffected cotwins also with inference via random permutation test and TFCE. We performed a similar analysis of the non-fractional amplitude of low frequency fluctuations post-hoc to reveal any potential differences for this measure.
Regional homogeneity (ReHo)
Also using the DPABI toolbox we estimated the regional homogeneity based on Kendall's coefficient of concordance (KCC) for clusters based on 26-voxel neighbourhood of each voxel. Note that the images were not spatially smoothed prior to calculating the KCC as this would have inflated the values considerably. Otherwise, pre-processing steps remained the same and like for calculation of ALFF, the data was pass-band filtered between 0.01 Hz and 0.08 Hz for this analysis. Once again, we tested for differences between cases and non-migraine controls and between affected and unaffected cotwins using voxel-wise random permutations tests with correction for multiple comparisons via TFCE.
Data availability
The obtained ethical and data protection approvals prevent us from sharing the raw neuroimaging data. We are happy to share derived measures such as extracted components and networks upon request.
Results
A total of 172 migraine patients with aura, 34 co-twins and 139 controls were scanned. Due to imaging quality issues, 12 scans from patients, four scans from co-twins, and three scans from controls were excluded. Thus, the results are based on data from 160 patients, 30 co-twins, and 136 controls. Patients were similar to controls with regard to age, and several lifestyle characteristics, although 33.8% of patients were classified as hypertensive, compared with 22.1% of controls (P = 0.03) and patients on average consumed 3.7 alcoholic drinks per week compared with 5.0 alcoholic drinks per week on average for controls (P = 0.03) (Table 1). Headache characteristics of patients corresponded to our expectations based on previous population-based epidemiological studies of migraine with aura (36).
Characteristics of study participants.

Means ± standard deviation, unless otherwise specified.
Patients versus controls. Generalized linear mixed models (binomial for proportions and Gaussian for means) were used to test the null hypothesis: there is no difference between patients and controls. Random intercepts for twin pairs were included to adjust for twin pair cluster effects.
Patients versus co-twins. Generalized linear mixed models (binomial for proportions and Gaussian for means) were used to test the null hypothesis: there is no difference between patients and co-twins. Random intercepts for twin pairs were included to adjust for twin pair status.
Education level was defined as low in women with less than 12 years of schooling and less than 3 years of vocational training.
Body Mass Index, the weight in kg divided by the square of the height in meters.
Hypertension was defined as systolic blood pressure of 160 mmHg or higher, or diastolic blood pressure of 95 mmHg or higher, or current use of antihypertensive drugs for hypertension.
Measured on the day the MRI was performed.
Data on systolic and diastolic blood pressure missing for five controls.
Low density lipoprotein. Not measured in three cases due to high triglyceride level (44 mmol/l).
Diabetes was defined as self-reported physician diagnosed diabetes in postal questionnaire, or glycated haemoglobin of 44 mmol/mol (6.5%) or higher.
Self-reported physician diagnosed angina pectoris or myocardial infarct in postal questionnaire.
Self-reported in postal questionnaire. Data on ever use of oral contraceptive use were missing for three cases, one co-twin, and one control. Data on number of years of oral contraceptive use were missing in seven cases, two co-twins, and four controls.
In 12 months before physician-conducted phone interview, establishing headache diagnoses according to International Headache Society classification criteria.
Information missing for 1 patient.
Per protocol, co-twins with migraine without aura could be included in the study.
Potential controls with >30 tension-type headache days in 12 months prior to interview were excluded by design.
Participants completed questionnaire information on headaches within last 48 hours on the day they were scanned and were subsequently phone-interviewed by a physician 48-72 hours after the scan to verify this information and to ascertain presence of headaches during or after the study scan.
Participants reported exclusively migraine without aura attacks.
Two of the 160 patients were on daily preventive medication for migraine (metoprolol and topiramate, respectively). In addition, 13 patients were on daily antihypertensive medication. Of these, one took bendroflumethiazide and amlodipine in combination and the remaining 12 were on monotherapy with bendroflumethiazide (n = 5), propranolol (n = 2), candesartan (n = 1), enalapril (n = 1), amlodipine (n = 1), or an unknown drug (n = 2). Four patients were on antidepressive medication (monotherapy with citalopram (n = 3) or venlafaxine (n = 1)). None of the patients were on antiseizure medication for other indications than migraine.
Of 136 controls, six were on daily antihypertensive medication. Of these, two took combination therapy of amlodipine/enalapril and lercanidipine/enalapril, respectively, while the remaining four were on monotherapy with amlodipine (n = 2), losartan (n = 1), or an unknown drug. None of the controls were on antidepressive or antiepileptic medication.
Resting-state fMRI findings
Spatial independent component analysis revealed plausible networks typically seen in analysis of resting-state data. In particular, the sorting of components according to the templates of the visual cortex revealed two visual components (see Figure 1). These components were extremely similar for cases and controls and testing revealed no significant differences between the cases and non-migraine controls or between affected and unaffected co-twins. Similarly, we identified default-mode components by spatially correlating the average components with the DMN_ICA_REST template (37), which revealed that the two most correlated components represented an anterior and posterior part of the default mode network respectively as displayed in Figure 2. In addition, we identified a component related to the primary motor and somatosensory cortex as displayed. Again, there were no significant differences between the groups or co-twins for any of these components. Similarly, we also tested for differences in two independent components that closely resembled networks 9 and 10 in the 20 component decomposition reported by Smith et al. (38), the so-called “executive control network”, as differences in this network was previously reported in migraine with aura (6). These analyses also did not reveal any significant differences in these networks. Also, analyses which additionally corrected for BMI, hypertension, weekly alcohol consumption and voxel-wise gray matter volume showed no significant group differences. While none of these tests of the expression of independent components revealed significant migraine-related differences, several of the components showed significant age-related changes when investigated with a similar test for linear effects of age. As an example, Figure 3 shows regions with significant negative correlation with age in purple for the component resembling the posterior default mode network.

Spatial representations of the visual independent components. Panels A and B show average component z-scores of the two visual components for cases whereas panels C and D show the corresponding average z-scores for controls. The expressions of these components are very similar and no significant differences between them were detected.
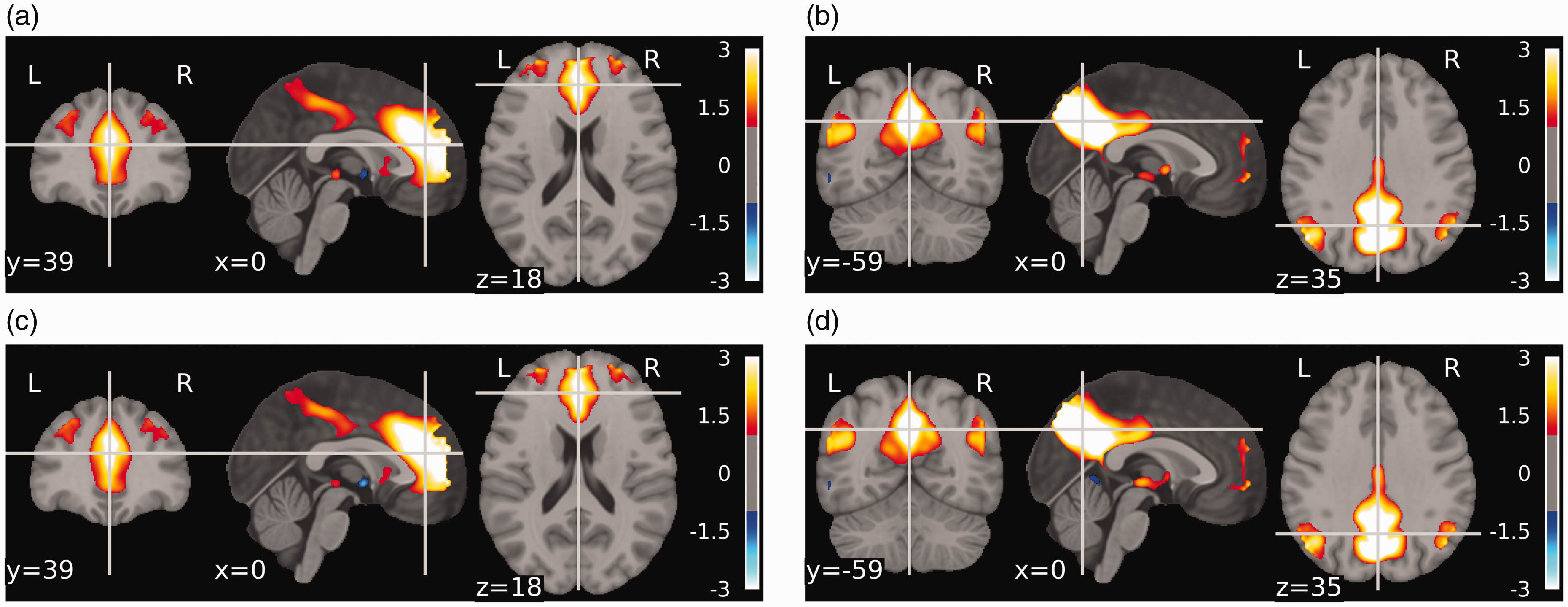
Spatial representations of the default mode components. Panels A and B show average component z-scores of the anterior and posterior default mode components for cases, whereas panels C and D show the average z-scores for corresponding components in controls. The expressions of these components are very similar and no significant differences between them were detected.
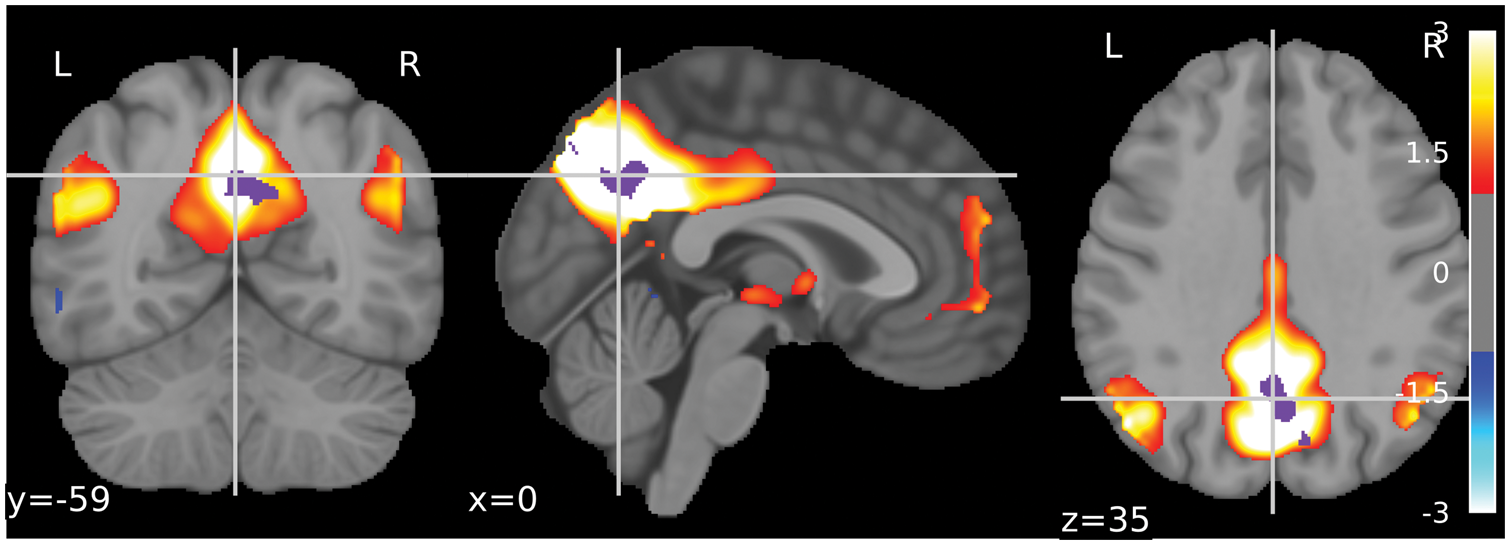
Age related effects in the posterior default mode network. The figure shows the average z-scores of the posterior default mode network with regions exhibiting significant negative correlation with age marked in purple. Like for the other tests inference and correction for multiple comparisons was performed using random permutation test and TFCE.
Voxel-wise analyses of seed region based functional connectivity maps also did not reveal significant differences between any of the regions of interest, neither for cases vs. controls nor between the affected and unaffected cotwins.
For the test for global network metrics there were no significant differences between the local network efficiency of migraine patients and matched migraine-free controls (lowest P = 0.12). The distributions of these metrics between the groups are displayed in Figure 4A revealing that they are extremely similar across the groups. Also, none of the other global network metrics showed significant differences and none of the post hoc tests for different network densities and parcellation atlases revealed significant differences between these measures. Note that all these results remain negative even though no correction for multiple comparisons across networks or thresholds is performed.

Results of graph-based analyses. Panel A shows the distribution of the global network metrics based on Shen et al.’s 2013, 268 region parcellation with a 90th percentile threshold. There is a small but weakly significant difference in the local efficiency between the groups that did not survive correction for multiple comparisons. Panel B shows that the graph features (upper triangular Fisher-Z transformed correlation values) provide a significant prediction of participant age (p<0.01).
Classification of migraine patients against matched migraine-free controls based on functional connectivity did not reach significance (accuracy = 0.555, AUC = 0.553, F1-score = 0.502) in the random permutation test. However, prediction of age based on these connectivity measures using support vector regression showed a significant (P < 0.01, mean absolute error = 4.6 years) prediction. These results are displayed in Figure 4B. None of the post-hoc tested parcellations showed significant separation between groups. Anecdotally, the highest classification performance was obtained for the 90 regions cortical anatomical parcellation with an AUC score of 0.587 but this remained insignificant in the random permutation test. Neither fractional ALFF or ReHo revealed significant differences between migraine patients and matched migraine-free controls or affected and unaffected co-twins. An illustration of the resulting spatial maps for migraine patients and matched migraine-free controls are displayed in Figure 5.

Fractional amplitude of low frequency fluctuations and regional homogeneity. Panels A and B show the estimated fractional amplitude of low frequency fluctuations and regional homogeneity for cases respectively. The same is displayed for controls below in panels C and D. Note that the expression of these are extremely similar across the groups and no differences were revealed via the random permutation test.
Discussion
We performed the largest study to date of resting-state connectivity using functional MRI in migraine patients. We studied a clinically well-characterized population-based sample of women with migraine with aura outside of migraine attacks, matched migraine-free controls and migraine aura free co-twins. Following extensive data analysis using several different approaches, we found no evidence of abnormal functional resting-state brain connectivity in migraine patients with aura.
These neutral findings are in line with a previous clinic-based study of 40 migraine patients with aura compared to 40 age and gender-matched controls using a seed-based analyses of 27 different seed locations as well as ICA, also showing no between-group differences for any analyses (13). Two of the authors of this previous study (A.H. and M.A.) contributed to the conception, design, and interpretation of the present study but, to avoid any suspicion of confirmation bias, did not carry out analyses of the present rs-fMRI data.
Several other previous rs-fMRI studies in migraine with aura patients have reported abnormal resting-state brain connectivity. An overview of previous rs-fMRI studies in migraine with aura, including characteristics of their participants, their methods, and a summary of their findings, is provided in Table 2. We evaluated previous studies of rs-fMRI in migraine with aura compared to non-migraine controls only and, thus, we did not consider results of stimulus- or task-driven fMRI studies of studies applying other neuroimaging modalities than fMRI, since these would not be comparable to the findings of the present study. All of the previous studies reporting abnormal rs-fMRI in migraine aura investigated clinic-based patient groups with a sample size ranging from 14 to 26 MA patients. These studies were not directly comparable due to differences in data analyses and no reported findings of any study were reproduced by another. None of the studies aimed to replicate previous findings or publicly pre-registered their analysis methods.
Overview of previous resting state fMRI studies in migraine with aura.

MA: Migraine with aura, MO: migraine without aura, HC: healthy controls, ICA: Independent component analysis, DMN: Default mode network,
subjects are identical,
not further specified,
median values provided.
Although we applied similar analytical methods, we were not able to reproduce any of these previously reported findings. Given previously reported effect and sample sizes we believe that our study was sufficiently powered to be able to demonstrate relevant effects. While the nature of the analyses applied here does not exclude the existence of differences, we believe that such effects are likely small in light of the large variability of resting-state networks reported in the literature (39) and differences, which can be expected due to, for example, caffeine intake (40), cortisol levels (41), genetic factors (42) and age (43,44). The notion that our study was strongly powered is corroborated by the demonstration of clear effects of age on the expression of resting-state networks despite the relatively narrow age range of our participants. This was exemplified by the significant prediction of age based on connectivity features (Figure 4B) and decreased involvement of the posterior default mode network (Figure 3), which is consistent with earlier reports (44–46). While differences in processing and analyses between previous studies make it difficult to directly compare these studies to the present study, we believe that the processing and analysis steps applied here are appropriate and sufficiently general to capture any network reconfiguration related to migraine with aura.
Potential limitations of the present study
It should be considered whether the discrepancy between the previous studies and the present study could be due to differences in participant characteristics. Migraine patients in the previous studies were younger (approximately 33 years of age on average, see Table 2) compared to the patients of the present study who had a mean age of 48.3 years. All the participants in the present study were women while this was not the case in any previous study, although the majority of patients in previous studies were women, reflecting the gender distribution of migraine patients in the general population (male:female ratio of 1:3) (36). The mean number of migraine attacks with aura per year in the previous, clinic-based studies ranged from nine to 61. These numbers are high compared to the attack frequency of migraine patients in general. In the general population, 75% of migraine patients (with and without aura) have 0–7 migraine days per year and 15% have 8–14 migraine days per year. Note in this regard that a migraine attack may last up to three days, by definition (47). In addition, migraine patients with aura experience migraine attacks less frequently compared to patients without aura (48). It could be speculated that only patients with a high attack frequency exhibit resting-state brain connectivity abnormalities that can be detected using rs-fMRI between attacks. Notably however, most of the previous studies investigated whether the magnitude of abnormal connectivity correlated with migraine aura attack frequency (5,6,9–12) and none of the studies found such a correlation.
Another potential limitation of this study is that we did not record any occurrence of premonitory or postdromal symptoms among the migraine patients during scan sessions. Subtle symptoms of migraine attacks may be associated with changes in resting-state brain connectivity that would increase the variability of the acquired fMRI data. In addition, we did not record the time from the last occurrence of migraine aura or migraine headache to the time of scanning. This information would have allowed for valuable additional analyses of patients with and without recent migraine attacks.
The main strengths of the present study were the use of the population-based Danish Twin Registry, which enabled us to identify a large number of patients and matched controls, and detailed physician interviews and questionnaires that allowed us to take clinical features and potentially confounding lifestyle characteristics into account. Also, we were able to perform paired analyses of twin pairs discordant for migraine with aura, an approach that strongly reduced or eliminated potential confounding effects of genes and common environment. While it is clear that migraine patients participating in clinic-based observational studies do not reflect the general population of patients, another potential limitation of the clinic-based design is the comparison of patients recruited from a specialized centre to healthy participants. Healthy participants are often sampled from populations (e.g. university students or health-care professionals) who may have a general health status that differs from that of the patient population. If health and lifestyle factors of the participants are not recorded and controlled for in the between-group analyses results may be confounded.
The challenge of a large methodological heterogeneity and a low reproducibility of results is not limited to rs-fMRI studies of migraine with aura. A systematic review of 28 studies applying rs-fMRI to investigate migraine patients with and without aura concluded that the results of the studies had a poor level of reproducibility and that the studies collectively had not revealed findings that could be considered specific of migraine (49). Methodological studies on rs-fMRI building on meta-analyses of large datasets indicate that small sample sizes could be a main reason for this lack of reproducibility (50,51), with one study of rs-fMRI in major depressive disorder suggesting that at least 400 participants should be included in each group for between-group analyses (51).
In conclusion, our results indicate that female migraine patients with aura have normal resting-state brain connectivity when being studied outside of migraine attacks. We did not reproduce previous findings of clinic-based rs-fMRI studies in migraine with aura patients. We recommend that any future studies on this topic include large samples of well-characterized participants, preferably in a population-based design.
Article highlights
Previous, relatively small studies using resting-state functional MRI have reported abnormal brain connectivity in migraine with aura patients but with conflicting findings.
In this large, population-based functional MRI using an extensive series of data analyses, no evidence of abnormal brain connectivity was found in migraine with aura patients and previously reported findings could not be replicated.
Resting-state functional MRI studies should include large samples of well-characterized participants, preferably in a population-based design.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Anders Hougaard has received honoraria for lecturing and/or writing from Allergan, Novartis, Teva, Lundbeck, and Eli Lilly. He serves as Assistant Editor of Headache.
David Gaist has received honoraria for lecturing from Bristol Myers Squibb and Pfizer. He received a research grant from Novo Nordisk Foundation.
Messoud Ashina has received personal personal fees from AbbVie, Amgen, Eli Lilly, GlaxoSmithKline, Lundbeck, Novartis, Pfizer and Teva. He has participated in clinical trials as the principal investigator for AbbVie, Amgen, Eli Lilly, Lundbeck, Novartis, Pfizer and Teva. He received a research grant from Lundbeck Foundation, Novo Nordisk Foundation, and Novartis. He has no ownership interest and does not own stocks of any pharmaceutical company. He serves as associate editor of Cephalalgia, associate editor of the Journal of Headache and Pain, and associate editor of Brain.
Hartwig R. Siebner has received honoraria as speaker and consultant from Lundbeck AS, Denmark, and as editor-in-chief and associate editor (Neuroimage Clinical) and senior editor (NeuroImage) from Elsevier Publishers, Amsterdam, The Netherlands. He has received royalties as book editor from Springer Publishers, Stuttgart, Germany, Oxford University Press, Oxford, UK and from Gyldendal Publishers, Copenhagen, Denmark.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study received support from the Lundbeck Foundation (R93-A8392), Novo Nordisk Foundation and Fabrikant Vilhelm Pedersen and Hustrus legat, Fonden til Lægevidenskabens Fremme, and A.P. Møller og Hustru Chastine Mc-Kinney Møllers Fond til Almene Formaal. D.G. is funded by Odense University Hospital. M.A. was funded by Novo Nordisk Foundation (1014333). Hartwig R. Siebner holds a 5-year professorship in precision medicine at the Faculty of Health Sciences and Medicine, University of Copenhagen which is sponsored by the Lundbeck Foundation (Grant Nr. R186-2015-2138).
