Abstract
Background
Migraine is a highly prevalent primary headache disorder. Despite a high burden of disease, key disease mechanisms are not entirely understood. Functional magnetic resonance imaging is an imaging method using the blood-oxygen-level-dependent signal, which has been increasingly used in migraine research over recent years. This systematic review summarizes recent findings employing functional magnetic resonance imaging for the investigation of migraine.
Methods
We conducted a systematic search and selection of functional magnetic resonance imaging applications in migraine from April 2014 to December 2021 (PubMed and references of identified articles according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines). Methodological details and main findings were extracted and synthesized.
Results
Out of 224 articles identified, 114 were included after selection. Repeatedly emerging structures of interest included the insula, brainstem, limbic system, hypothalamus, thalamus, and functional networks. Assessment of functional brain changes in response to treatment is emerging, and machine learning has been used to investigate potential functional magnetic resonance imaging-based markers of migraine.
Conclusions
A wide variety of functional magnetic resonance imaging-based metrics were found altered across the brain for heterogeneous migraine cohorts, partially correlating with clinical parameters and supporting the concept to conceive migraine as a brain state. However, a majority of findings from previous studies have not been replicated, and studies varied considerably regarding image acquisition and analyses techniques. Thus, while functional magnetic resonance imaging appears to have the potential to advance our understanding of migraine pathophysiology, replication of findings in large representative datasets and precise, standardized reporting of clinical data would likely benefit the field and further increase the value of observations.
Introduction
Migraine is a primary headache disorder classified according to criteria set forth by the International Headache Society (1). Globally, migraine has been estimated to afflict more than one billion patients, corresponding to a prevalence of higher than 13 percent and ranking it the sixth most prevalent disease according to the Global Burden of Disease Study (2–4). Moreover, migraine represents a major cause of disability, accounting for roughly 5.6 percent of global years lived with disability and 1.9 percent of disability-adjusted life years (2,4). Within the neurological diseases, only stroke contributes a higher number of global disability-adjusted life years (4). Despite the resulting high interest in therapy and research, the current understanding of both migraine pathophysiology as well as treatment options is incomplete.
One of the modalities that has been employed in service of deepening the understanding of migraine is functional magnetic resonance imaging (fMRI). This method is a non-invasive imaging approach for measuring fluctuations of the blood-oxygen-level-dependent (BOLD) signal (5). This signal serves as an indirect measure of neuronal activity via the process of neurovascular coupling (6). This in turn allows for the identification of regional neuronal activation both during resting-state (rs) and/or specific task-based paradigms, thus enabling interrogations regarding the functional role of specific brain areas and their interconnection (5,7). Methods such as independent component analysis (ICA) have been used to identify functional networks, considered as spatially distributed areas that correlate regarding their BOLD signal and are therefore presumed to work in concordance with each other, and where an ICA Z-score can quantify the degree to which a specific voxel is concordant with the wider network activity (8,9). Important and consistently replicable functional networks (9,10) include the default mode network (DMN; active during rs, associated with mind-wandering and deactivated during performance of many specific tasks [11]), salience network (SN; involved in regulation of activity of other networks, associated with task switching), executive control network (ECN; involved in cognitive control tasks and working memory), dorsal attention network (DAN; involved in attention allocation), visual network (VN; related to the processing of visual information), auditory networks (AN; related to processing of auditory information), sensorimotor network (SMN; involved in processing of somatosensory information and motor behavior), and the fronto-parietal network (FPN, sometimes referred to as central executive network [CEN] in the literature; involved in cognitive tasks) (8,12,13).
As such, fMRI has seen widespread applications in neurosciences in general and migraine specifically for the investigation of neuronal processes (14). A PubMed search for fMRI and migraine search terms revealed a considerable increase in published literature within the last two decades (Figure 1). One important aspect in this regard is the variety of measures researchers can extract from fMRI data aside from purely regional contrasting (Figure 2). Examples include:
PubMed search results for functional magnetic resonance imaging (fMRI)/migraine search terms.

Schematic display of functional magnetic resonance imaging (fMRI) metrics.
Functional connectivity (FC), a measure estimating the correlation of BOLD signal fluctuations in separate voxels. Analyses of FC enable researchers to identify which areas experience similar fluctuations of BOLD signal across time (e.g., “Does the BOLD time course in location x look similar to the time course in location y?”), whereby a high correlation is often interpreted as these areas working together in a functional sense (8).
Dynamic functional connectivity (dFC), a related method that separates the entire BOLD time course into smaller time windows and investigates changes in FC across time (e.g., “Does the BOLD time course in location x always look similar to the time course in y or only sometimes?”) (15).
Effective connectivity (EC), which analyzes how BOLD fluctuations in one area influence the subsequent signal in other areas and is thus utilized to infer causal relations between signal changes in remote areas (e.g., “Does a peak at location x lead to a subsequent peak at location y?”) (16).
Amplitude of low-frequency fluctuation (ALFF), an index aiming to quantify spontaneous local brain activity via measurements of BOLD fluctuations (e.g., “How strongly does the BOLD fluctuate at location x?”) (17).
Regional homogeneity (ReHo), a metric quantifying the concordance of BOLD fluctuations in neighboring voxels (e.g., “Does the BOLD time course at location x look like the time course of all its neighbors?”) (18).
Additionally, derived analyses such as methods of graph theory can be employed to model brain networks and measures such as degree centrality (DC) from BOLD time courses in order to further elucidate connectivity patterns (19). Importantly, while fMRI studies can certainly be informative of cerebral processes, many caveats apply regarding both data analysis and result interpretation, most importantly the facts that the BOLD signal is an indirect measure of neuronal activity and subject to high amounts of noise, that it is not reflective of specific neuronal subpopulations (e.g., an increase in inhibitory activity or excitatory activity could both lead to a higher BOLD signal), and that results are dependent on the specific statistical methods employed (5,7). Nonetheless, due to the limited understanding of migraine pathogenesis and treatment response, the presented variety of fMRI-associated metrics could potentially offer an option to better understand cerebral mechanisms associated with migraine.
This systematic review aimed to summarize fMRI-based findings published within the last seven years (i.e., publications following a review article published in 2015 [14]), with special attention on findings correlating with clinical parameters of migraine, including disease duration, pain intensity, and attack frequency as hallmarks of disease manifestation related to disability. For this, we have undertaken a literature search according to a previously registered PROSPERO protocol (CRD42021238822) and Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (20), in order to identify all research in the respective interval in which migraine phenomena were investigated with fMRI. Due to the high methodological variability within the identified studies, we focused our assessment of anatomical structures on repeatedly emerging areas of interest with relatively uniform nomenclature, including insula, brainstem, limbic system, hypothalamus, thalamus, and multiple functional networks.
Methods
Search strategy
Prior to conducting the literature search, we registered the respective protocol in the PROSPERO database (CRD42021238822) as per the PRISMA guidelines (20). Initially, we conducted a literature search on PubMed (www.pubmed.ncbi.nlm.nih.gov) using search terms related to fMRI and migraine (see online Supplementary Material 1) and filtering for articles published between April 2014 and January 2021. The present review article thus follows up on a publication from 2015 that covered earlier articles, which focused on pain-paradigm fMRI studies in migraine (14).
The articles that passed our inclusion/exclusion criteria (Figure 3 and online Supplementary Material 1) underwent a screening of all references to identify literature relevant to the topic that might have gone unnoticed in the initial PubMed search. In order to keep our review as current as possible, we then extended our PubMed search to also include articles published until December 2021 (and their respective references). While the initial protocol as published in the PROSPERO database (CRD42021238822) also aimed to review arterial spin labelling (ASL) studies in migraine, due to the high volume of fMRI literature and relative lack of ASL literature, we decided against reporting the latter findings in this manuscript for a stronger thematic focus.

Literature selection.
Data extraction
Each publication was screened for compliance with published fMRI reporting guidelines (21): clear definition of studied populations, clear description of the respective task (if applicable for task-based fMRI), documentation of the region of interest (ROI) selection process, detailed documentation of statistical analyses, whether all results reported in the respective section were supported by testing, and whether certain quality control steps (e.g., correction of movement artifacts) were documented. Additionally, since statistical adjustment for multiple comparisons is a relevant concern in most fMRI investigations (22), we have extracted the respective levels of statistical significance and correction procedures for imaging findings in each study (Tables 1–9). Further, we extracted information regarding sample size (initial sample and after study dropouts), sex distribution, scan timing in relation to ictality, available information regarding headache medication, and the main findings as relevant to the topic of this review (Tables 1–9).
Studies related to insula.

Abbreviations: acupuncture (ACP), anterior insula (aINS), amplitude of low-frequency fluctuations (ALFF), amygdala (Amy), auditory network (AN), blood-oxygen-level-dependent (BOLD), cutaneous allodynia (CA), cingulate cortex (CC), cluster headache (CH), clinical parameters (ClPa), chronic migraine (CM), migraine with complex aura (cMwA), cuneus (Cu), dorsal attention network (DAN), degree centrality (DC), disease duration (DD), dynamic functional connectivity (dFC), dorsolateral prefrontal cortex (dlPFC), default mode network (DMN), effective connectivity (EC), episodic migraine (EM), functional connectivity (FC), functional connectivity density (FCD), functional connectivity homogeneity (FCHo), false detection rate (FDR), family-wise error (FWE), healthy controls (HC), hippocampus (Hip), hypothalamus (HyTh), independent component analysis (ICA), insula (INS), interval not specified (Int ns), inferior parietal lobule (IPL), inferior temporal gyrus (ITG), locus coeruleus (LC), lingual gyrus (LiG), multiple comparison (MC), middle cingulate cortex (mCC), medial prefrontal cortex (mPFC), middle temporal gyrus (MTG), migraine with aura (MwA), migraine without aura (MwoA), nucleus tractus solitarii (NTS), caudate nucleus (NuCau), orbitofrontal cortex (OFC), posterior cingulate cortex (pCC), prefrontal cortex (PFC), pain intensity (PI), putamen (Put), resting state (rs), rostral ventromedial medulla (RVM), primary somatosensory cortex (S1), sensorimotor network (SMN), migraine with simple aura (sMwA), nucleus spinalis nervi trigemini (SpV), trigeminocervical complex (TCC), threshold free cluster enhancement (TFCE), thalamus (Th), vestibular migraine (VM), visual network (VN).
Studies related to brainstem.

Abbreviations: anterior cingulate cortex (aCC), acupuncture (ACP), attack frequency (AF), acute headache (AH), anterior insula (aINS), amplitude of low-frequency fluctuations (ALFF), auditory network (AN), nucleus accumbens (ANuc), advanced visual processing network (AVN), blood-oxygen-level-dependent (BOLD), cutaneous allodynia (CA), cingulate cortex (CC), cluster headache (CH), clinical parameters (ClPa), chronic migraine (CM), cuneus (Cu), dynamic functional connectivity (dFC), dorsolateral prefrontal cortex (dlPFC), default mode network (DMN), effective connectivity (EC), episodic migraine (EM), fractional amplitude of low-frequency fluctuations (fALFF), functional connectivity (FC), false detection rate (FDR), functional network (FN), family-wise error (FWE), gaussian random field (GRF) , healthy controls (HC), hippocampus (Hip), hypothalamus (HyTh), independent component analysis (ICA), insula (INS), interval not specified (Int ns), inferior parietal lobule (IPL), multiple comparison (MC), middle cingulate cortex (mCC), medication overuse (MedOv), middle frontal gyrus (MFG), migraine with aura (MwA), migraine without aura (MwoA), nitroglycerin (NTG), nucleus tractus solitarii (NTS), caudate nucleus (NuCau), periaqueductal grey (PAG), posterior cingulate cortex (pCC), prefrontal cortex (PFC), pain intensity (PI), precuneus (PrCu), putamen (Put), nucleus raphe (RaNuc), regional homogeneity (ReHo), nucleus ruber (ReNuc), resting state (rs), rostral ventromedial medulla (RVM), primary somatosensory cortex (S1), superior parietal lobe (SPL), nucleus spinalis nervi trigemini (SpV), substantia nigra (SuN), transcutaneous auricular vagus nerve stimulation (taVNS), trigeminocervical complex (TCC), thalamus (Th), ventrolateral periaqueductal grey (vlPAG), vestibular migraine (VM), visual network (VN).
Studies related to limbic areas.

Abbreviations: advanced visual processing network (AVN), amygdala (Amy), anterior cingulate cortex (aCC), anterior insula (aINS), attack frequency (AF), auditory network (AN), blood-oxygen-level-dependent (BOLD), chronic migraine (CM), cingulate cortex (CC), clinical parameters (ClPa), cluster headache (CH), cuneus (Cu), cutaneous allodynia (CA), default mode network (DMN), degree centrality (DC), distant functional connectivity density (dFCD), dorsolateral prefrontal cortex (dlPFC), dorsolateral superior frontal gyrus (dlSFG), dynamic functional connectivity (dFC), effective connectivity (EC), episodic migraine (EM), executive control network (ECN), external trigeminal neurostimulation (eTNS), false detection rate (FDR), family-wise error (FWE), functional connectivity (FC), functional connectivity density (FCD), functional connectivity homogeneity (FCHo), healthy controls (HC), hippocampus (Hip), hypothalamus (HyTh), independent component analysis (ICA), inferior frontal gyrus (IFG), inferior parietal lobule (IPL), insula (INS), interval not specified (Int ns), lingual gyrus (LiG), local functional connectivity density (lFCD), medial prefrontal cortex (mPFC), medication overuse (MedOv), middle cingulate cortex (mCC), middle frontal gyrus (MFG), middle occipital gyrus (MOG), migraine with aura (MwA), migraine without aura (MwoA), multiple comparison (MC), nucleus accumbens (ANuc), nucleus spinalis nervi trigemini (SpV), nucleus tractus solitarii (NTS), pain intensity (PI), paracentral lobule (PCL), periaqueductal grey (PAG), persistent posttraumatic headache (PPTH), posterior cingulate cortex (pCC), posterior thalamus (pTh), precentral gyrus (PrCG), precuneus (PrCu), prefrontal cortex (PFC), primary somatosensory cortex (S1), regional homogeneity (ReHo), resting state (rs), rostral ventromedial medulla (RVM), salience network (SN), somatosensory cortex (SSC), superior frontal gyrus (SFG), superior temporal gyrus (STG), supplementary motor area (SMA), thalamus (Th), threshold free cluster enhancement (TFCE), transcutaneous auricular vagus nerve stimulation (taVNS), ventrolateral periaqueductal grey (vlPAG), vestibular migraine (VM), visual network (VN).
Studies related to hypothalamus.

Abbreviations: acute headache (AH), anterior cingulate cortex (aCC), anterior insula (aINS), auditory network (AN), blood-oxygen-level-dependent (BOLD), chronic migraine (CM), cingulate cortex (CC), clinical parameters (ClPa), cluster headache (CH), cuneus (Cu), cutaneous allodynia (CA), default mode network (DMN), dorsal raphe nucleus (dRaNuc), dorsal visual network (DVN), dorsolateral prefrontal cortex (dlPFC), effective connectivity (EC), episodic migraine (EM), false detection rate (FDR), family-wise error (FWE), functional connectivity (FC), healthy controls (HC), hippocampus (Hip), hypothalamus (HyTh), independent component analysis (ICA), insula (INS), interval not specified (Int ns), lingual gyrus (LiG), medication overuse (MedOv), migraine with aura (MwA), migraine without aura (MwoA), multiple comparison (MC), nucleus accumbens (ANuc), nucleus raphe (RaNuc), nucleus spinalis nervi trigemini (SpV), nucleus tractus solitarii (NTS), pain intensity (PI), pain matrix (PMat), periaqueductal grey (PAG), precuneus (PrCu), prefrontal cortex (PFC), resting state (rs), rostral ventromedial medulla (RVM), thalamus (Th), vestibular migraine (VM), visual network (VN).
Studies related to thalamus.
Abbreviations: amplitude of low-frequency fluctuations (ALFF), anterior cingulate cortex (aCC), anterior insula (aINS), auditory network (AN), blood-oxygen-level-dependent (BOLD), chronic migraine (CM), cingulate cortex (CC), clinical parameters (ClPa), cluster headache (CH), cuneus (Cu), cutaneous allodynia (CA), dorsolateral prefrontal cortex (dlPFC), dynamic functional connectivity (dFC), effective connectivity (EC), episodic migraine (EM), false detection rate (FDR), family-wise error (FWE), fractional amplitude of low-frequency fluctuations (fALFF), functional connectivity (FC), gaussian random field (GRF) , granger causality (GC), healthy controls (HC), hippocampus (Hip), independent component analysis (ICA), inferior frontal gyrus (IFG), inferior parietal lobule (IPL), insula (INS), interval not specified (Int ns), lateral geniculate nucleus (LGN), lingual gyrus (LiG), middle frontal gyrus (MFG), migraine without aura (MwoA), multiple comparison (MC), nucleus spinalis nervi trigemini (SpV), nucleus tractus solitarii (NTS), power spectra density (PSD), precuneus (PrCu), prefrontal cortex (PFC), primary somatosensory cortex (S1), regional homogeneity (ReHo), resting state (rs), rostral ventromedial medulla (RVM), superior parietal lobe (SPL), supplementary motor area (SMA), thalamus (Th), threshold free cluster enhancement (TFCE), trigeminocervical complex (TCC), vestibular migraine (VM).
Studies related to functional networks.

Abbreviations: acupuncture (ACP), advanced visual processing network (AVN), amplitude of low-frequency fluctuations (ALFF), anterior cingulate cortex (aCC), anterior cingulate cortex (aCC), anterior insula (aINS), attack frequency (AF), auditory network (AN), blood-oxygen-level-dependent (BOLD), chronic migraine (CM), cingulate cortex (CC), clinical parameters (ClPa), cluster headache (CH), cuneus (Cu), cutaneous allodynia (CA), default mode network (DMN), disease duration (DD), dorsal attention network (DAN), dorsal attention system (DAS), dorsal premotor cortex (dPM), dorsal raphe nucleus (dRaNuc), dorsal visual network (DVN), dorsolateral prefrontal cortex (dlPFC), dynamic functional connectivity (dFC), effective connectivity (EC), episodic migraine (EM), executive control network (ECN), false detection rate (FDR), family-wise error (FWE), fronto-parietal network (FPN), functional connectivity (FC), functional network (FN), granger causality (GC), healthy controls (HC), hypothalamus (HyTh), independent component analysis (ICA), inferior frontal gyrus (IFG), inferior parietal lobule (IPL), inferior temporal gyrus (ITG), insula (INS), interval not specified (Int ns), lingual gyrus (LiG), medication overuse (MedOv), middle frontal gyrus (MFG), middle temporal gyrus (MTG), migraine with aura (MwA), migraine with complex aura (cMwA), migraine with simple aura (sMwA), migraine without aura (MwoA), multiple comparison (MC), nucleus accumbens (ANuc), nucleus raphe (RaNuc), nucleus tractus solitarii (NTS), pain intensity (PI), pain matrix (PMat), postcentral gyrus (PoCG), precentral gyrus (PrCG), precuneus (PrCu), prefrontal cortex (PFC), premotor cortex (PrMC), primary motor cortex (M1), primary motor cortex (M1), primary somatosensory cortex (S1), regional homogeneity (ReHo), resting legs syndrome (RLS), resting state (rs), restless legs syndrome (RLS), salience network (SN), sensorimotor network (SMN), somatosensory cortex (SSC), superior parietal lobe (SPL), superior temporal gyrus (STG), supplementary motor area (SMA), supramarginal gyrus (SMG), thalamus (Th), ventral attention network (VAN), ventral premotor cortex (vPM), vestibular migraine (VM), visual network (VN).
Studies related to emotional and cognitive processing.

Abbreviations: auditory network (AN), blood-oxygen-level-dependent (BOLD), chronic migraine (CM), cingulate cortex (CC), clinical parameters (ClPa), cuneus (Cu), cutaneous allodynia (CA), effective connectivity (EC), episodic migraine (EM), family-wise error (FWE), functional connectivity (FC), healthy controls (HC), independent component analysis (ICA), inferior frontal gyrus (IFG), locus coeruleus (LC), middle temporal gyrus (MTG), migraine without aura (MwoA), multiple comparison (MC), nucleus tractus solitarii (NTS), pain intensity (PI), resting state (rs), thalamus (Th).
Studies related to therapy.
Abbreviations: acupuncture (ACP), advanced visual processing network (AVN), amplitude of low-frequency fluctuations (ALFF), amygdala (Amy), anterior cingulate cortex (aCC), attack frequency (AF), auditory network (AN), blood-oxygen-level-dependent (BOLD), chronic migraine (CM), cingulate cortex (CC), clinical parameters (ClPa), cluster headache (CH), cuneus (Cu), cutaneous allodynia (CA), default mode network (DMN), dorsolateral superior frontal gyrus (dlSFG), effective connectivity (EC), episodic migraine (EM), external trigeminal neurostimulation (eTNS), false detection rate (FDR), family-wise error (FWE), fronto-parietal network (FPN), functional connectivity (FC), functional magnetic resonance imaging (fMRI), healthy controls (HC), independent component analysis (ICA), inferior parietal lobule (IPL), insula (INS), interval not specified (Int ns), locus coeruleus (LC), menstrual migraine without aura (MMoA), middle cingulate cortex (mCC), middle frontal gyrus (MFG), migraine without aura (MwoA), multiple comparison (MC), nucleus accumbens (ANuc), nucleus raphe (RaNuc), nucleus tractus solitarii (NTS), pain intensity (PI), paracentral lobule (PCL), periaqueductal grey (PAG), precuneus (PrCu), regional homogeneity (ReHo), respiratory-gated auricular vagal afferent nerve stimualtion (RAVANS), resting state (rs), superior frontal gyrus (SFG), supplementary motor area (SMA), thalamus (Th), transcutaneous auricular vagus nerve stimulation (taVNS), ventral attention network (VAN), ventrolateral periaqueductal grey (vlPAG), visual network (VN).
Studies related to machine learning.
Abbreviations: acupuncture (ACP), amplitude of low-frequency fluctuations (ALFF), anterior cingulate cortex (aCC), area under the curve (AUC), attack frequency (AF), auditory network (AN), chronic migraine (CM), cingulate cortex (CC), clinical parameters (ClPa), cluster headache (CH), convolutional neural network (CNN), cuneus (Cu), cutaneous allodynia (CA), effective connectivity (EC), episodic migraine (EM), false detection rate (FDR), functional connectivity (FC), functional magnetic resonance imaging (fMRI), healthy controls (HC), independent component analysis (ICA), interval not specified (Int ns), migraine with aura (MwA), migraine without aura (MwoA), multiple comparison (MC), nucleus tractus solitarii (NTS), putamen (Put), regional functional corellation strength (RFCS), regional homogeneity (ReHo), resting state (rs), thalamus (Th).
Results
Out of 224 studies identified, 114 studies remained for the synthesis of this review (Figure 3). On average, 62 ± 36 participants (median 52 participants) were included per study. Of the identified studies, 68 studied groups with migraine without aura (23–90), 15 migraine with aura (30,31,35,36,40,44–46,49,75,84,91–94), 25 episodic migraine without further specification (95–119), 4 vestibular migraine (79,120–122), 23 chronic migraine (28,31,43,54,81,97,99,100,102,106,108,110,112–114,116,123–129), 6 chronic migraine with medication overuse (87,95,102,117,127,128), and 8 migraine without further specification (120,130–136). In 102 studies healthy controls (HC) served as a reference population.
In 91 studies, acquisition of fMRI took place during the interictal interval, which was however inconsistently defined (e.g., 72 h pre scan – 24 h post scan, 48 h pre scan – no interval post scan; Tables 1–9). Nine studies collected data at least partially during ictal states. Preventative, acute and non-migraine-related medication were controlled and documented to varying degrees, with 24 studies not controlling medication in any way.
Anatomical structures and functional networks of interest
Insula
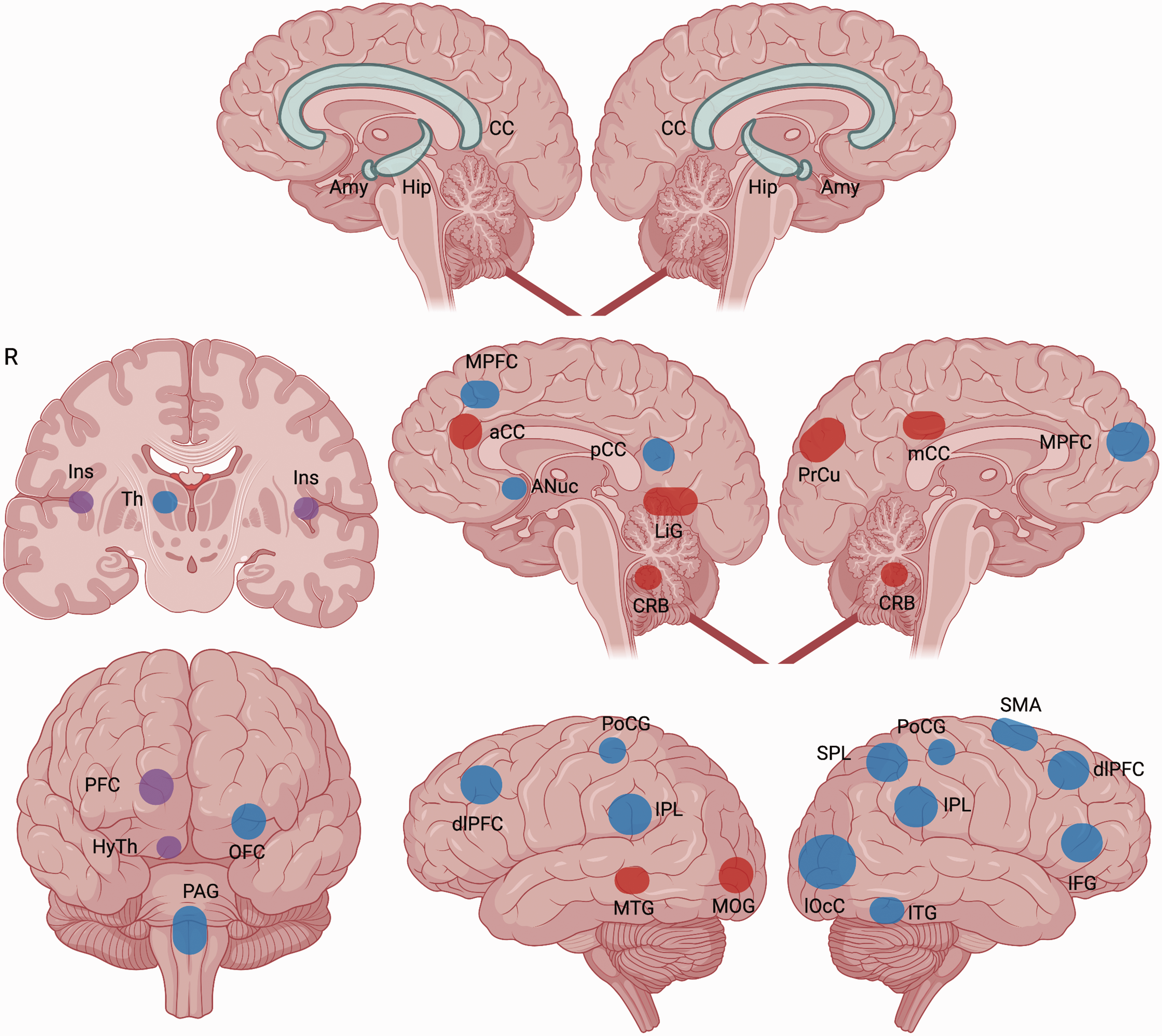
The insula is a region folded within the depths of the Sylvian fissure and consists of up to 13 subregions (137). Evidence suggests that the insula plays a role in (among other functions) pain processing and that it acts as an important hub within the SN involved in attention direction (137). Insular regions repeatedly emerged as areas of interest in migraine patients (Table 1), both compared to HC (27,50,51,55,61,62,66,71,86,108,115,122) as well as to other types of migraine (e.g., migraine with vs. without aura) (40,46,84).
Alterations in functional magnetic resonance imaging measures
In subjects with migraine compared to HC, studies reported heightened rs-FC of insula to medial prefrontal cortex and inferior parietal lobule (50), Heschl gyrus, pons, calcarine cortex and amygdala (61), cuneus (132), supplementary motor area, postcentral gyrus, temporal lobe, fusiform gyrus, cerebellum (86), and multiple other brain regions (62). This was contrasted by reports of lowered rs-FC to anterior cingulate cortex (27), occipital regions (in migraine with aura but not migraine without aura) (46), and multiple other brain regions (62,71). In the two rs-FC studies that reported altered rs-FC in high numbers of brain regions (58 and 54 regions, respectively) in migraine without aura compared to HC, most connections were lowered (instead of heightened) in migraine without aura compared to HC (43/58 and 54/54 regions, respectively) (62,71). Importantly, FC patterns were observed to differ between six insular subregions (hypergranular insula, ventral agranular insula, dorsal agranular insula, ventral dysgranular and granular insula, dorsal granular insula, and dorsal dysgranular insula) (62). Other studies yielded that nodal centrality (71), short-range FC density (FCD) (55), and FC homogeneity (FCHo) (27) of insula were decreased in migraine without aura compared to HC. ALFF values (66) and BOLD signal variability (108) of insula were reported to be increased in patients with migraine compared to HC. Pain-related BOLD activity differences between patients with migraine and HC in the insula were reported at an uncorrected level but did not survive statistical correction in one study (134). Outside the more often examined migraine types, the insula has also been implicated in vestibular migraine, where afflicted patients demonstrated heightened rs-FC between the left insula and both primary sensory cortex as well as inferior parietal lobe compared to HC (122).
Aside from differences to HC, insula FC contributed to a discrimination process between complex and simple forms of migraine with aura as defined by the migraine aura complexity score (cutoff for simple vs. complex: score > 1) (84). Furthermore, migraine with aura demonstrated heightened rs-FC compared to migraine without aura in insula regions (40).
Interestingly, simultaneous abnormalities in insula and other areas of interest have been reported. In migraine without aura patients compared to HC, rs-FCHo was reduced both in insula as well as limbic areas (cingulate cortex) and thalamus, and FC between insula and anterior cingulate cortex was reduced (27). In another study investigating the interplay between spinal trigeminal nucleus, hypothalamus and insula during tactile stimulation, habituation behavior of insula was altered in episodic migraine compared to HC (115). In a similar vein, altered rs-ALFF values between migraine without aura and HC were demonstrated in insula areas as well as brainstem areas (trigemino-cervical complex and rostral ventromedial medulla) (66).
Associations with clinical parameters
Regarding correlations with clinical parameters, disease duration correlated with FC of insula subregions (62). Also, while not a direct correlation, insula regions emerged as part of a disease duration-dependent network in a graph-based analysis of migraine without aura (51). Pain intensity as another clinical parameter of interest was found to correlate with pain-related BOLD activity (134) and FC between insula and medial prefrontal cortex (50), as well as FC between insula and occipital areas (46). Additionally, attack frequency correlated with pain-related insular BOLD activity (134).
Summary
Overall, the insula appears to be an important node in migraine pathophysiology, with many studies indicating altered rs-FC to other brain areas (Figure 4). One contributing factor to this could potentially be reflected in its status as a key area within the SN, a network involved in the integration of multimodal sensory input. Despite this, direct replication studies did not appear in the identified literature, which corresponds to a clear unmet need.

Resting-state functional connectivity alterations (rs-FC) of insular regions in migraine patients.
Brainstem
The brainstem is a structure consisting of the midbrain, pons, and medulla oblongata. It serves as both an important relay station for ascending and descending neural pathways, and also includes a wide variety of nuclei involved in governing a multitude of body functions, such as cranial nerve nuclei (138). A number of brainstem nuclei have been implicated as areas of interest in different types of migraine (Table 2), including nucles ruber (28), substantia nigra (28), periaqueductal grey (31,42,98,112,113,133), spinal trigeminal nucleus (31,95,108,113–115,133), rostral ventromedial medulla (31,82,133), pontine areas (36,41,70,92,113), raphe nucleus (42), and trigemino-cervical complex (66,70,82).
Alterations in functional magnetic resonance imaging measures
Trigeminal areas
Trigeminal areas such as the spinal trigeminal nucleus and trigemino-cervical complex were among the structures most often found to be relevant in the examined literature. A FC study comparing rs-FC of episodic migraine and chronic migraine with medication overuse found that FC was present between spinal trigeminal nucleus and hypothalamus in chronic migraine with medication overuse, but not in episodic migraine with a significant difference between both groups (95). In subjects with migraine, the spinal trigeminal nucleus displayed hyperactivity both during noxious trigeminal stimulation (133) and visual stimulation (114) compared to HC. Importantly, spinal trigeminal nucleus response to noxious stimulation was heightened in patients with migraine only right before an attack took place (133). When the relation to attack cycle was controlled to interictal, another study discovered no difference in spinal trigeminal nucleus response to non-painful tactile stimulation between subjects with migraine and HC, but did demonstrate that spinal trigeminal nucleus amplification in insula and hypothalamus was higher in episodic migraine compared to HC (115). Other studies demonstrated that BOLD signal variability (108), ReHo (70), and power of infra-slow fluctuations (113) in the spinal trigeminal nucleus were increased in patients with migraine compared to HC.
Rostral ventromedial medulla
Rostral ventromedial medulla also emerged as an area of interest. In pre-ictal states, the rostral ventromedial medulla demonstrated reduced FC both to the spinal trigeminal nucleus (133) as well as to hypothalamus (31) compared to HC. Interictally, rostral ventromedial medulla in migraine without aura demonstrated increased fractional ALFF compared to HC (82).
Periaqueductal grey
Regarding the periaqueductal grey, subjects with migraine compared to HC were shown to have reduced periaqueductal grey rs-FC to medial prefrontal cortex, orbitofrontal cortex, anterior cingulate cortex (58), and a variety of additional brain areas (98), while other studies found heightened rs-FC of periaqueductal grey to raphe nucleus (42), rostral ventromedial medulla (133), adjacent periaqueductal grey (58) and (exclusively pre-ictally) to hypothalamus and thalamus (113). Pre-ictally, subjects with migraine demonstrated heightened ReHo and power of infra-slow oscillations in the periaqueductal grey (and hypothalamus) (113). A heat-based trigeminal pain paradigm was used in a study of periaqueductal grey connectivity and patients with migraine demonstrated both increased (to supplementary motor area, sensorimotor cortex face area, insula, thalamus, and pons) and decreased (to superior medial frontal gyrus, cerebellum, middle occipital gyrus, temporal pole, and superior parietal areas) periaqueductal grey FC during pain compared to HC (103). Subjects with migraine further showed a heightened BOLD response to ammonium-based trigeminal nociception within the periaqueductal grey (as well as cerebellar areas) compared to controls, which was also modulated by ictality phase (112).
Other pontine areas
Some studies reported changes in pontine areas with less defined nomenclature. One study reported increased FC of left pons and primary somatosensory cortex (corresponding to face) as well as superior parietal lobule in ictal vs. interictal state of migraine with aura (92). Another study investigating changes between headache states found that nitroglycerin-triggered attacks led to changes in FC between pons and cingulate/frontal areas (during premonitory phase), as well as pons and cerebellar/medullar areas (during attack) in migraine patients (36).
More generally, one report found decreased FC of pons to superior parietal lobule, middle temporal gyrus and middle frontal gyrus in migraine without aura compared to HC (41). Regarding ReHo, both increased (70) and decreased (41) ReHo of left pons was described in migraine without aura compared to HC. Power of infra-slow BOLD fluctuations was found increased in migraine patients compared to HC in dorsal pons prior to a migraine attack (113).
One undefined (within the respective publication) brainstem cluster demonstrated altered ReHo in migraineurs without aura compared to HC, while another cluster showed reduced ReHo in subjects with migraine six weeks after the initial scan (77).
Associations with clinical parameters
In terms of clinical parameters, ALFF of trigemino-cervical complex and rostral ventromedial medulla correlated with pain intensity (66). Correlation of spinal trigeminal nucleus BOLD signal variability with pain intensity was attempted in another study but did not remain significant after multiple comparison correction (108). Pain intensity also correlated with rs-FC of periaqueductal grey to anterior cingulate cortex/medial prefrontal gyrus (58).
Disease duration correlated with rs-FC of periaqueductal grey in interictal episodic migraine (98). This finding was not replicated in another study, where instead a correlation of FC between periaqueductal grey and cingulate cortex with attack frequency was found (83). Disease duration was also reported to correlate with fractional ALFF values of brainstem areas (82) and FC of substantia nigra (28).
Furthermore, attack frequency correlated with the BOLD response to nociception within the periaqueductal grey (and cerebellum) (112), as well as FC to a number of areas during nociception (103).
Summary
Overall, different areas of the brainstem appear to play a role in migraine pathophysiology (Figure 5). The involvement of trigeminal nuclei corresponds well to other evidence emphasizing the importance of the trigemino-cervical complex in migraine (139,140), while the involvement of pain-modulating regions such as the periaqueductal grey and rostral ventro medial medulla could be interpreted as either part of causative or adaptive mechanisms. Regarding replication, while some aspects of the literature point towards similar directions such as trigeminal pathway hyperactivation in migraine (114,133), direct replication studies were not identified, which complicates the interpretation of seemingly contradictory findings such as, for example, both heightened and lowered pontine ReHo.

Resting-state functional connectivity (rs-FC) alterations of brainstem regions in migraine patients.
Limbic areas
The limbic system is a functional system and works as an umbrella term for a set of anatomical structures (generally taken to include the cingulate gyri, amygdalae, hippocampi, hypothalamus, and septal areas) that function in close relation to each other and are involved in governing of many vegetative, emotional, and cognitive functions (141). Various components of the limbic system were identified as relevant in migraine (Table 3), with a strong focus on the amygdala (26,55,85,100,127,130), cingulate cortex (27,40,48,55,69,70,83,130,132), and hippocampus (26,31,55,81,108,123).
Alterations in functional magnetic resonance imaging measures
Hippocampus
The rs-FC of hippocampus was decreased to some areas (hypothalamus [31], right nucleus accumbens [119], inferior parietal gyrus and supplementary motor area [26]) and increased to other areas (hypothalamus [53], cerebellar and occipital areas [26]) compared to HC. rs-FCD of hippocampus was found increased in chronic migraine (123) as well as decreased in migraine without aura (55) compared to HC. The hippocampus was found to show altered rs-EC with frontal areas, visual areas, insula, and cerebellum between migraine without aura, chronic migraine, and HC (81). ReHo of a hippocampal cluster (among other areas) was reported as altered (direction not specified) in subjects with migraine without aura compared to HC (77). Subjects with migraine also demonstrated greater rs-BOLD signal variability in hippocampus compared to HC (108).
Amygdala
In subjects with migraine, the amygdala displayed increased rs-FC to middle occipital gyrus (26), middle cingulate cortex (100), right insula (61), and left precuneus (100) compared to HC. Decreased amygdala rs-FC compared to HC was found with orbitofrontal cortex (26), right nucleus accumbens (119), inferior occipital gyrus, and middle occipital gyrus (100). Altered amygdala rs-FC was also demonstrated between different types of migraine: between episodic migraine and chronic migraine (for a number of connected areas) (100), as well as between chronic migraine with and without medication overuse (for FC between bilateral amygdala) (127). The left and right amygdala demonstrated different FC patterns in a rs-fMRI study (100) as well as one therapeutic study (52). Other studies in migraine without aura patients demonstrated altered rs-EC between amygdala and superior temporal gyrus, precentral gyrus, and inferior frontal gyrus (85), as well as decreased FCD in amygdala compared to HC (55).
Cingulate cortex
Overall, findings regarding rs-FC of cingulate cortex were heterogeneous. While some studies found heightened rs-FC of anterior cingulate cortex (to lingual gyrus [132], middle temporal gyrus and right frontal pole [131]) in subjects with migraine compared to HC, other studies demonstrated reduced rs-FC of anterior cingulate cortex (to anterior insula [27], right nucleus accumbens [119], periaqueductal grey [58], prefrontal cortex [69], and posterior cingulate cortex [69]) in migraine without aura compared to HC. For the posterior cingulate cortex, some studies showed decreased rs-FC (to medial prefrontal cortex [27], primary somatosensory cortex, temporal, parietal, and frontal areas [132]) in migraine without aura compared to HC. Another study found that rs-FCHo of posterior cingulate cortex was reduced in interictal migraine without aura compared to HC (27).
rs-FCD of the anterior cingulate cortex was reported as decreased in migraine without aura compared to HC (55). ReHo was reported as heightened in posterior cingulate cortex (70), as well as altered (direction not specified) in other cingulate clusters (among other areas) (77) in migraine without aura compared to HC. EC was also reported as altered in migraine, with EC between insula and anterior cingulate cortex significantly decreased in migraine without aura compared to HC (69). Aside from connectivity, the anterior cingulate cortex demonstrated a higher BOLD response to painful heat stimulation in migraine without aura compared to HC in one study (78). Related to this, another study found no overall group difference in BOLD response to painful forearm heat stimuli between migraineurs and HC after multiple comparison correction (134). The authors however demonstrated reduced pain-related BOLD activity in individuals with migraine compared to HC in an anterior midcingulate cluster (as well as frontal, parietal, and temporal clusters) when differentiating between different pain levels (134). Analogously, one study observed that the BOLD response during moderately painful trigeminal heat stimulation in an anterior cingulate cluster was higher in migraine without aura with cutaneous allodynia compared to HC, as well as higher within the middle frontal gyrus in migraine without aura with cutaneous allodynia compared to patients without cutaneous allodynia and HC; for more painful stimuli, group differences were observed in somatosensory areas (72).
Associations with clinical parameters
Disease duration was correlated to FC of posterior cingulate cortex to pons (132) as well as EC between anterior cingulate cortex and insula (69). It also correlated with rs-FC between amygdala and middle occipital gyrus (26) as well as with rs-EC between amygdala and precentral gyrus (85).
Pain intensity correlated with FC between anterior cingulate cortex and posterior cingulate cortex (69), as well as FC of the posterior cingulate cortex to the primary somatosensory cortex and visual areas (132), rs-dFC between amygdala and posterior cingulate cortex (130) and DC of amygdala (86).
Attack frequency correlated with posterior cingulate cortex FC to posterior parietal cortex (132), rs-EC of hippocampus and medial prefrontal cortex as well as rs-EC of hippocampus and cerebellum (81).
Summary
Overall, the current literature yields major evidence regarding the general involvement of various areas of the limbic system (Figure 6). The amygdalae appear to differ in their functional roles in migraine based on laterality, but overall their involvement in a disease related to headache is not unexpected given the wider evidence of their role in pain (142). Cingulate area processing also appears to be altered in migraine compared to HC. Notably, both the anterior cingulate and the posterior cingulate serve as important nodes of the SN and DMN, respectively. Therefore, a differentiated look upon the role of the cingulate areas appears to be necessary based on different findings of altered activity within these areas. While the hippocampus has been involved in migraine and chronic pain even with modalities other than fMRI (143,144), as well as being associated with DMN processing (145), the available fMRI literature on its distinct role in migraine remains inconclusive. Regarding replicability, while once again some results point to analogous conclusions, such as asymmetries in amygdala processing, no direct replication attempts have been identified.
Resting-state functional connectivity (rs-FC) alterations of limbic regions in migraine patients.
Hypothalamus
The hypothalamus is an area of the brain adjacent to the inferior border of the thalamus, which performs central regulatory functions within the vegetative nervous system (146). As some of the results above have already indicated, the hypothalamus is implied to play a role in migraine pathophysiology by the fMRI literature (Table 4).
Alterations in functional magnetic resonance imaging measures
In the hypothalamus of patients with migraine compared to HC, studies indicated both enhanced rs-FC (to medial prefrontal cortex, parietal lobules, visual areas [124], precentral gyrus, hippocampus, pontine areas and multiple other areas [53]) as well as decreased rs-FC (to hippocampus, anterior cingulate cortex, periaqueductal grey, spinal trigeminal nucleus, rostral ventromedial medulla, pontine areas [31], precentral gyrus, frontal pole, superior frontal gyrus, fusiforme gyrus, and lingual gyrus [53]). A study investigating episodic migraine, chronic migraine and HC found that the BOLD signal response to painful trigeminal stimulation in a the hypothalamus cluster was higher in chronic migraine compared to both HC and episodic migraine (106). Furthermore, the hypothalamus demonstrated heightened FC to a functional network labeled as “pain matrix” in chronic migraine compared to episodic migraine (97).
In this context, as mentioned with the brainstem results, another study showed that chronic migraine with medication overuse patients demonstrated significant FC between the hypothalamus and spinal trigeminal nucleus, which was not the case in episodic migraine (95). This connection was again highlighted in another study demonstrating that in interictal episodic migraine, spinal trigeminal nucleus signal was amplified stronger in insula and hypothalamus compared to HC (115).
Associations with clinical parameters
For clinical parameter correlations related to hypothalamic investigations, pain intensity was correlated to FC of the hypothalamus to medial prefrontal cortex (124).
Summary
Overall, the hypothalamus as a control center of vegetative functions appears to be relevant in the interplay of other migraine-involved areas (Figure 7), specifically regarding its connectivity with trigeminal and limbic areas. Available evidence suggests that hypothalamic activity is involved in the symptom cycle of migraine. While once again, no direct replications were identified, the current literature is somewhat more consistent regarding some aspects of hypothalamic activity in migraine, such as e.g., abnormal connectivity between the hypothalamus and the trigeminal system. However, the exact nature and circumstances of these alterations were again inconsistent across reports, illustrating the need for replication studies.

Resting-state functional connectivity alterations (rs-FC) of hypothalamus in migraine patients.
Thalamus
The thalamus includes nuclei and associative fibers positioned between midbrain and telencephalon. It serves as an important relay station for much of the sensory information entering the telencephalon (147). Multiple studies reported thalamic regions as areas of interest (Table 5).
Alterations in functional magnetic resonance imaging measures
Altered rs-FC in migraine without aura compared to HC was found in two thalamic subregions: lowered rs-FC between anterior dorsal thalamic nucleus and left precuneus as well as between ventral posterior nucleus and precuneus, inferior parietal lobule and right middle frontal gyrus (39). Pre-ictally, one study found increased rs-FC between thalamus, and periaqueductal grey in migraine patients compared to HC (113). In one comparison of thalamic rs-FC between ictal and interictal migraine without aura, the authors found both increased (to left superior parietal lobule, insula, primary motor cortex, supplementary motor area, and orbitofrontal cortex) and decreased (to right primary somatosensory cortex and premotor cortex) rs-FC of right thalamus in the ictal compared to the interictal state (47). One study specifically investigated rs-FC differences of the lateral geniculate nucleus, a part of the thalamus that serves as a relay for visual information, between subjects with migraine without aura and HC (37). The authors described heightened rs-FC of the left lateral geniculate nucleus (to left cerebellum and right lingual gyrus) and the right lateral geniculate nucleus (to left inferior frontal gyrus) in patients with migraine compared to HC (37). rs-dFC between thalamus and primary somatosensory cortex was found stronger and less variable in chronic migraine compared to HC (108), and the amount spent in specific dFC states was observed to differ between migraine without aura and HC (29). FCHo was found reduced in the left thalamus of patients with migraine without aura compared to HC (27). rs-EC between posterior thalamus and prefrontal cortex, cingulate cortex, cuneus as well as precuneus was altered in migraine without aura compared to HC (74). rs-EC between posterior thalamus and prefrontal cortex as well as limbic areas has further been found to differ between migraine without aura, with and without cutaneous allodynia (64). In patients with migraine, thalamic BOLD signal variability was found increased compared to HC (108). Low-frequency oscillations in the thalamus differed significantly between episodic migraine and HC (109), and fractional ALFF of thalamus clusters was significantly higher in migraine without aura compared to HC (82). Thalamic clusters (among other areas) also demonstrated reduced ReHo compared to HC in migraine without aura compared to HC, as well as a further reduction six weeks after the initial scan; yet, these changes did not take place in the same cluster (77). Regarding network topological metrics, thalamic network global and local efficiency were found to be lower in migraine without aura compared to HC (29).
Two studies investigated thalamus activity in the rarer population of vestibular migraine. One of these found that the thalamic BOLD signal response to vestibular stimulation was significantly higher in vestibular migraine compared to HC and episodic migraine in a mediodorsal cluster (79). Another study demonstrated altered rs-FC to anterior cingulate cortex, insula, supplementary motor area, superior parietal lobule, precuneus and middle frontal gyrus in vestibular migraine compared to HC (121).
Associations with clinical parameters
Regarding correlations with disease duration, rs-FC between ventral posterior nucleus and inferior parietal lobule (39), right thalamus and left insula (121), as well as rs-EC between posterior thalamus and prefrontal areas emerged as significant (74). For pain intensity, correlations with rs-FC between ventral posterior nucleus and inferior parietal lobule (39), as well as with rs-EC of posterior thalamus to prefrontal areas (74) have been reported. Attack frequency was described to correlate with the power spectrum density of thalamus low-frequency oscillations (109), as well as thalamus connectivity (to precuneus and middle occipital gyrus) within specific dFC states (29). In vestibular migraine, attack frequency correlated with thalamic BOLD response to vestibular stimulation (79).
Summary
Overall, the multi-faceted fMRI findings (Figure 8) of thalamic activity alterations in migraine may be interpreted in light of the thalamus’ central nature as a sensory relay station, which would in turn emphasize the aspect of dysfunctional sensory integration in migraine pathophysiology. Once again however, a broad spectrum of different analyses and methodologies was present without literature attempting direct replication of specific findings.

Resting-state functional connectivity alterations (rs-FC) of thalamus in migraine patients.
Functional networks
Investigations by fMRI have revealed consistently demonstrable sets of remote areas working in temporal coherence, often associated with specific cognitive functions, generally described as functional networks (8). A variety of functional networks identified by ICA or defined by seed placement was explored, most prominently the DMN, SN, ECN, DAN, VN, AN, SMN, and the FPN (Table 6) (9–12).
Alterations in functional magnetic resonance imaging measures
Default mode network
The DMN demonstrated enhanced FC to the hypothalamus (124) and DAN (125), contrasted by reduced FC to the ECN (125) as well as internally within the DMN (126,128,129) in chronic migraine compared to HC. In episodic migraine, nodes of the DMN demonstrated reduced FC to sensory networks compared to HC (104). In migraine without aura, FC between DMN and a visuospatial network was reduced compared to HC (59). Another study in migraine without aura identified increased FC (to multiple brain areas) as well as decreased FC (to right cerebellum and left frontal lobe) of DMN seeds during a PACAP38-induced migraine attack compared to baseline (56). DMN FC was also linked to endogenous pain modulation in migraine without aura (135).
Fronto-parietal network
For the FPN, a study in chronic migraine with and without medication overuse found FPN connectivity strength significantly lowered compared to HC (128). In migraine without aura, the FPN was reported as having reduced FC to precentral gyrus, supramarginal gyrus, inferior parietal lobule, postcentral gyrus (65), and a high number of other brain areas (57), as well as altered EC to the ECN compared to HC (68).
Salience network
Regarding the SN, decreased FC was reported between key SN nodes (insula) and visual areas in migraine with aura but not migraine without aura (46). In episodic migraine, FC of sensory networks to nodes of the SN was lowered compared to HC (104). Further, the SN in migraine with aura patients demonstrated altered intra-network dFC (specifically of the anterior cingulate cortex) compared to both HC and migraine without aura (35). In chronic migraine with and without medication overuse compared to HC, the SN showed lowered internal coherence of both groups compared to HC, without a difference between chronic migraine with and without medication overuse (128). Another study however demonstrated shared abnormalities of SN FC distinguishing both groups from HC, as well as FC alterations differentiating chronic migraine and chronic migraine with medication overuse (127). Furthermore, subjects with chronic migraine with medication overuse demonstrated heightened FC of the SN to bilateral habenulae compared with episodic migraine and HC (with no difference between episodic migraine and HC), which correlated with medication overuse duration (117). In an investigation of PACAP38-triggered migraine attacks, the authors demonstrated increased connectivity of SN seeds to right and left inferior frontal gyri during provoked attacks compared to baseline (56). Aside from FC differences, one study identified differences in BOLD responses within the “salience matrix” to unspecific salience stimuli (temperature stimuli on the foot) between ictal and interictal migraineurs without aura, chronic migraine with medication overuse, and HC (87).
Visual network
The advanced visual processing network (AVN) was reported as hyperresponsive to painful trigeminal stimulation in migraine with aura compared to migraine without aura (30). Related to this finding, migraine with aura demonstrated higher intra-network FC of VN (within the lingual gyrus) compared to HC and migraine without aura, as well as higher VN ICA Z-scores in complex aura compared to simple aura (84). A similar finding regarding VN connectivity was observed in another study published by the same group (49). The VN further showed reduced FC to precuneus and occipital cortex in episodic migraine compared to HC in a seed-based analysis (104). The dorsal visual network (DVN) demonstrated higher FC to hypothalamus in chronic migraine compared to HC (124).
Related to visual areas, one study investigated visual discomfort and BOLD response to visual stimuli in patients with migraine with aura, migraine without aura, and HC (75). The authors observed heightened visual area BOLD response to visual stimuli in migraine with aura (which correlated with visual discomfort) but not in migraine without aura compared to HC (75). Thematically adjacent, one study investigated visual area BOLD responses to visual stimulation during baseline and induced hypoxia and observed that hypoxia-related BOLD signal decreases of visual areas during stimulation were stronger in migraine with aura patients compared to HC (91). In another visual stimulation paradigm conducted in subjects with migraine with side-fixed aura, the authors described a visual processing network comprised of inferior parietal lobule, inferior frontal gyrus and superior parietal lobule of the symptomatic hemisphere, which was more active in subjects with migraine compared to HC during stimulation (94). Additionally, a small mixed cohort of 10 patients with and without aura was reported to possess altered visual area activation during a visually cued movement task compared to HC (107).
Sensorimotor network
The SMN of interictal migraine without aura demonstrated higher ICA Z-scores in postcentral gyrus, lower ICA Z-scores in middle cingulate cortex, and altered EC to a number of areas compared to HC (80). During PACAP38-induced migraine attacks, patients with migraine demonstrated heightened FC of SMN seeds to the right premotor cortex and reduced FC to the left visual cortex compared to baseline (56). Another study demonstrated reduced ReHo, DC, and ALFF in parts of the SMN (primary somatosensory cortex and right premotor cortex), as well as reduced rs-FC of primary somatosensory cortex to a variety of other brain areas in migraine without aura compared to HC (73). Somatosensory cortex was implicated in another investigation, in which the authors described that the BOLD response to strongly painful trigeminal heat stimulation was lower in migraine patients compared to HC, as well as lower in patients without cutaneous allodynia compared to patients with cutaneous allodynia; additionally, said response correlated with pain intensity (72). Furthermore, in vestibular migraine, intra-network rs-FC of the SMN was lowered compared to HC (120).
Other networks
The DAN was reported to have lowered FC to the ECN and enhanced FC to the DMN in chronic migraine compared to HC (125). In migraine without aura and migraine with aura, a DAN seed demonstrated increased FC to temporal areas compared to HC (46).
In chronic migraine, AN ICA Z-scores were heightened in superior parietal gyrus and decreased in superior frontal gyrus compared to HC (129). Heightened AN ICA Z-scores were also found in insula, postcentral gyrus, and superior temporal gyrus of migraine without aura compared to HC (88). In vestibular migraine, rs-FC of AN was reduced to parts of the DMN compared to HC and heightened to the SN compared to patients with other migraine subtypes (120).
The ECN was reported to have lower intra-network FC (in anterior cingulate cortex and middle frontal gyrus) in both migraine with aura and migraine without aura compared to HC (45). Additionally, in ictal migraine without aura, FC between ECN and the DAN as well as ventral attention network (VAN) was lowered compared to HC (60). In chronic migraine, the ECN ICA Z-score in the angular gyrus was heightened compared to HC (129).
In one study of chronic migraine and episodic migraine, a functional network labelled as “pain matrix” (dorsolateral prefrontal cortex, anterior cingulate cortex, anterior insula, thalamus, precuneus) demonstrated higher DC, stronger FC to hypothalamus, and lower FC to raphe nucleus in chronic migraine compared to episodic migraine (97). Another study also made reference to a “pain matrix” (specifically regarding insula and thalamus activity) in discussing findings of altered ReHo in a high number of brain areas when comparing migraine without aura both to HC as well as to other individuals (43).
Aside from specific networks, subjects with migraine were also observed to possess reduced coupling of structural connectivity and FC compared to HC (54). This specific study further demonstrated other differences for the structural connectome between migraine patients and HC (54). Furthermore, one whole-brain FC study identified reduced connectivity between a variety of nodes belonging to different functional networks in migraine compared to HC, as well as between migraine groups depending on the presence of restless legs syndrome (101).
Interestingly, FC analysis was multiple times reported to demonstrate no differences between subjects with migraine and HC (32,93,96) or even between interictal and ictal states in migraine after multiple comparison correction (92). While in one of these studies, conventional FC analysis yielded only limited significant differences, dFC analysis demonstrated alterations in seven interictal and four peri-/interictal ICA-derived functional networks between episodic migraine and HC (96).
Associations with clinical parameters
In terms of clinical parameter correlations, disease duration correlated with FC between primary motor cortex and middle temporal gyrus as well as Z-scores of somatosensory cortex and temporal areas (33), DMN-DAN interaction (35), and EC of postcentral gyrus to precuneus (80). Pain intensity correlated with FC between primary somatosensory cortex and superior temporal gyrus (33), FC between FPN and precuneus, anterior cingulate cortex/medial prefrontal cortex, superior frontal gyrus and inferior temporal gyrus (57), DAN and ECN maximal Z-score (125), ECN Z-scores in middle frontal gyrus (45), FC between SN nodes and visual areas (46), and regarding dFC state transitions (129). Attack frequency correlated with dFC measures of prefrontal cortex (35), FC between a DAN seed and anterior cingulate cortex (46), ECN ICA Z-score (60), FPN and SN connectivity strength (128), and regarding SMN ICA Z-score in the postcentral gyrus (80).
Summary
Overall, the wide variety of functional network alterations observed in migraine patients supports the view that migraine is a brain state (148–150). Interestingly, network alterations were not only observed during the migraine attack, but also interictally. The involvement of the DMN and SN could be interpreted in light of their respective roles in direction of attention and integration of sensory stimuli. Importantly, other discussed areas such as the cingulate gyrus and insula are major components of these networks. Visual areas were found hyperexcitable and mostly with stronger FC in migraine patients compared to HC and, while the observed evidence was heterogeneous, somatosensory areas were found to demonstrate lowered activity and connectivity to a number of areas, again emphasizing sensory integration aspects of migraine.
Regarding replication of studies, internal DMN hypoconnectivity in chronic migraine compared to HC was one of the rare consistent findings across multiple comparable studies within the identified literature. Other convergent evidence speaks to a hyperactivity of the VN in migraineurs compared to HC. Nonetheless, as discussed above, data failed to demonstrate significant FC differences after strict statistical correction procedures had been applied. Again, more studies with the goal to replicate specific analyses are required to contextualize partially contradictory findings.
Emotional and cognitive processing
A number of studies used fMRI in explorations of emotional and cognitive processing in subjects with migraine (Table 7).
Emotional processing
In one study, BOLD signal response to faces displaying different affects (happy, fearful, neutral, or sad) was altered compared to HC: subjects with migraine showed increased response to fearful faces in middle frontal gyrus compared to HC, and the authors reported that attack frequency correlated with the BOLD response to fearful faces in precentral gyrus and postcentral gyrus, as well as with the BOLD response to happy faces in basal ganglia (34).
Affective stimuli were used in two other studies (110,111). In both studies, subjects with migraine compared to HC displayed greater BOLD response to negative affective pictures in a number of brain areas, with a notable overlap in precuneus, cuneus and cerebellum between both studies (110,111). Furthermore, in one of the studies, subjects with migraine displayed increased BOLD response to neutral pictures in visual areas compared to HC (111).
Cognitive functions
Aside from emotional processing, cognitive functions were also investigated. In a visuospatial task, subjects with migraine compared to HC demonstrated higher BOLD activity in insula, orbitofrontal cortex and middle frontal gyrus, as well as decreased BOLD activity in posterior cingulate cortex; yet, activations of insula, orbitofrontal cortex and posterior cingulate cortex correlated with task performance (118). Related to visuospatial functions, one study investigated responses to a visual self-motion paradigm (realized via a virtual roller-coaster ride); here, subjects with migraine exhibited heightened BOLD activity in occipital, pontine and cerebellar clusters, as well as lower activity in other cerebellar and frontal clusters compared to HC, and subjects with migraine demonstrated stronger dizziness and motion sickness compared to HC, which (quantified via questionnaire) partially correlated with BOLD activity in the mentioned clusters (136).
Another study investigated subjects with migraine in a monetary reward/loss paradigm, and observed decreased BOLD activation in the inferior frontal gyrus during the reward consumption stage of the task compared to HC (89). Furthermore, subjects with migraine demonstrated reduced task-induced deactivation in the dorsolateral prefrontal cortex, cerebellum and anterior cingulate cortex compared to HC in a modified attentional network test. Subjects with migraine also demonstrated reduced task-related activity in the premotor cortex compared to HC. The introduction of a painful heat stimulus modulated task-based activity differently in migraineurs and HC (116).
Summary
Overall, the identified studies employed heterogeneous designs to investigate various cognitive processes in migraine patients. There appears to be evidence from multiple studies suggesting that migraine patients demonstrate altered processing of affective stimuli with heightened BOLD responses to negative affective stimuli. The studies do however differ on the exact location of altered BOLD responses. We can also not exclude the possibility of having missed studies of affective processing in migraine that have not employed fMRI, which weakens attempts at evidence synthesis.
Functional imaging in migraine therapy
Acupuncture
Over recent years, fMRI has been employed in a number of studies to investigate effects of various migraine therapies. Many of the previous publications employed acupuncture as treatment (Table 8). In one investigation, both verum and sham (i.e., positioned at locations not corresponding to known acupoints) acupuncture led to clinical improvement of attack frequency in participants, but only verum acupuncture led to changes in a previously established machine learning-based connectome disease marker, which in turn correlated with improvements in attack frequency (24).
In another study, patients with chronic migraine demonstrated decreased intra-network FC of the DMN (decreased DMN ICA Z-score in superior prefrontal gyrus and precuneus) compared to HC; yet, after 3 months of acupuncture, the previous difference in DMN FC was not demonstrable anymore (126). The changes in DMN ICA Z-scores correlated with changes in “immediate visual analogue scale scores” and monthly amount of acute headache medication, but not with metrics of attack frequency or mean pain intensity (126).
Another acupuncture therapy study demonstrated decreased rs-FC of the FPN to a high number of brain areas in patients with migraine compared to HC, and FPN FC to some of these areas correlated with pain intensity (57). In terms of clinical parameters, verum acupuncture led to reductions in pain intensity and attack frequency, but did not differ significantly from sham (not corresponding to known acupoints) acupuncture (57). In a pooled analysis of verum and sham acupuncture, significant post-treatment FPN FC changes correlated with a reduction in pain intensity (57). A similar study investigated effects of acupuncture on periaqueductal grey connectivity: again, verum acupuncture led to improvements in pain intensity, but did not differ significantly from the effects of sham (not corresponding to known acupoints) acupuncture. In another pooled analysis of verum and sham data, the authors observed that changes in ventrolateral periaqueductal grey rs-FC correlated with pain intensity changes (58). A smaller study in 12 patients and 12 matched controls without sham control found that a number of rs-FC differences between subjects with migraine (pre-acupuncture) and HC were partially attenuated after acupuncture treatment; however, no correlation of fMRI metric changes to clinical parameter changes was conducted (63).
Instead of FC, one acupuncture study analyzed ReHo in a self-control design and found multiple areas with changed ReHo in a comparison of pre- vs. post-treatment for both verum and sham acupuncture: both verum and sham (not corresponding to known acupoints) acupuncture resulted in improvements (pain intensity, attack frequency, questionnaire scores), with a significant difference in pain intensity favoring verum acupuncture in a direct comparison (76). Two ReHo changes after verum (anterior cingulate cortex and insula) and one ReHo change after sham (anterior cingulate cortex) acupuncture correlated with pain intensity decrease (76). Moreover, both verum and sham (not corresponding to known acupoints) acupuncture led to changed ALFF and ReHo values in some areas in another study, as well as significant improvements in pain intensity and emotional parameter questionnaires in menstrual migraine without aura (90). ALFF changes after verum acupuncture in the left anterior cingulate cortex correlated with improvements in emotional questionnaires; however, this study did not employ groups outside verum and sham acupuncture in patients (such as, for example, HC or no-intervention groups) (90).
While our search strategy was not specifically tailored to the identification of acupuncture studies, the identified publications have mostly shown clinical improvements of migraine symptoms following both verum and sham acupuncture, while at the same time failing to demonstrate significant differences between both. Some studies however describe sham/verum differences in fMRI parameters, thereby highlighting the potential use of fMRI as a supplementary outcome measure to better understand potential nonspecific or placebo effects in therapy studies of migraine.
Neurostimulation
Neurostimulation approaches employ a treatment strategy for migraine by stimulating the brain, nerves, or muscular tissue in an attempt to modify cerebral mechanisms contributing to migraine (Table 8) (151,152). Employed strategies, as summarized in a recent review, include, occipital, supraorbital, vagal or neuromuscular stimulation (151). Transcutaneous auricular vagus nerve stimulation (taVNS) was another therapeutic strategy tracked via fMRI. In a comparison of two groups of migraine without aura randomized to either verum or sham taVNS over four weeks, real taVNS led to heightened connectivity (post-treatment vs. pre-treatment) between different thalamus seeds and anterior cingulate cortex/medial prefrontal cortex, as well as lowered connectivity between other thalamus seeds and supramarginal gyrus, superior parietal lobe, postcentral gyrus, and precuneus compared to sham taVNS (25). The authors reported a significant correlation of FC change (pre-taVNS to post-taVNS; thalamus seed and postcentral gyrus) to reduction in attack frequency for the verum group, but not for the sham group (25). taVNS was used in another study as well, in which the stimulation (applied with 1 Hz) increased rs-FC between periaqueductal grey and middle cingulate cortex, cuneus, precuneus and middle frontal gyrus compared to pre-stimulation, and the authors also described differences in rs-FC between different types of taVNS (1 Hz and 20 Hz) (83). An additional study investigated immediate effects of taVNS on FC of amygdala in a single-blind crossover design with scanning during stimulation (52). The authors found reduced FC of both left and right amygdala (to a variety of brain areas) during real taVNS compared to sham taVNS, as well as a correlation between FC of left amygdala and right supplementary motor areas during taVNS to attack frequency; relating back to hemispheric asymmetry, both amygdalae differed in their FC responses to taVNS (52). In a similar study by the same group, the authors described significant alterations in BOLD signal compared to baseline both during verum as well as sham taVNS, with no significant difference when testing verum directly against sham (23). In a seed-to-voxel analysis however, the locus coeruleus was observed to exhibit increased rs-FC to a variety of brain areas after verum taVNS compared to sham taVNS: FC changes to locus coeruleus after taVNS in the right temporoparietal junction and the left secondary somatosensory cortex correlated with disease duration and attack frequency, respectively (23). Additionally, FC between locus coeruleus and the left amygdala was significantly higher during verum taVNS compared to sham taVNS (23).
Another method of vagus nerve stimulation employing fMRI was identified in the form of respiratory-gated auricular vagal afferent nerve stimulation (RAVANS). One study identified different brain FC changes between the application of inhalatory-gated RAVANS, expiratory-gated RAVANS, and sham RAVANS (105).
Aside from vagus nerve stimulation, one study also reported the use of external trigeminal neurostimulation (eTNS): BOLD response to a painful trigeminal heat stimulus in the anterior cingulate cortex was higher in migraine without aura compared to HC pre-eTNS, and significantly lowered post-eTNS compared to pre-eTNS; additionally, the authors observed a correlation between eTNS-related response changes in the anterior cingulate cortex and attack frequency after eTNS (78).
One study investigated the rs-FC responses of subjects with migraine compared to HC to the application of placebo analgesia in a painful stimulation paradigm (131). The authors demonstrated a significant group × condition interaction, with HC demonstrating heightened FC of ventral striatum and precentral gyrus when on placebo, which was significantly weaker in subjects with migraine (131). HC on placebo also demonstrated lowered FC of middle frontal gyrus compared to subjects with migraine (131).
Overall, analogously to our findings regarding acupuncture studies, neuromodulatory therapy studies identified within the context of our fMRI-specific review have been demonstrated to be capable of altering some fMRI metrics in migraine patients. Regarding clinical value we can make no strong statement in this review due to not tailoring our search to this specific focus.
Machine learning and advanced statistical modelling
Different studies employed machine learning in analyses of fMRI data (Table 9). In one study, fMRI data of initially 70 migraine without aura patients and 46 HC were used to select 192 rs-FC features that best discriminated both groups, which in turn were used in conjunction with a support vector machine classifier to accurately (accuracy 91%, area under the curve [AUC] 0.97 in original dataset; accuracy 84%, AUC 0.91 in independent dataset) classify fMRI data of subjects with migraine without aura and HC (24). Importantly, the classifier was unable to distinguish HC from other chronic pain disorders (chronic lower back pain and fibromyalgia) but was somewhat accurate in distinguishing migraine without aura from the chronic pain disorders (accuracy 73%) (24). As reported above, acupuncture-related changes to the rs-FC marker correlated with clinical improvement after treatment (24).
Classification of HC and subjects with migraine was also performed in a study using rs-FC features of 33 predefined regions, principal component analysis (PCA), and diagonal quadratic discriminate analysis (99). The best classifier yielded an accuracy of 86% based on rs-FC of middle temporal gyrus, insula, middle cingulate cortex, amygdala, and prefrontal regions (99). Interestingly, data from patients with longer disease duration were more accurately classified than shorter disease duration (99).
Another study employed a multivariate logistic regression model to classify migraine without aura patients into two groups according to their attack frequency based on fMRI features (38). The authors found that the best model utilized eight FC features for an accuracy of 82% and AUC of 0.91 in the original dataset and accuracy of 75% as well as AUC of 0.79 for the validation dataset (38).
Deep learning was applied in one study, which utilized convolutional neural networks for the classification of migraine without aura, migraine with aura, and HC, and the authors here utilized ALFF, ReHo, and regional functional correlation strength (RFCS) as input (44). RFCS emerged as the best performing input, with a maximum accuracy of 99% and AUC of 0.99 in classifying HC vs. any type of migraine (44). Classification of migraine without aura vs. migraine with aura using RFCS was achieved with a maximum accuracy of 97% (44). Notably, the number of patients per cohort was somewhat lower than some of the other machine learning studies reviewed (migraine without aura: n = 21; migraine with aura: n = 15; HC: n = 28) (44).
ALFF, ReHo, and RFCS were again used in a multi-kernel support vector machine approach to classify migraine without aura and HC based on a combination of fMRI and structural MRI (grey matter value) features of 116 predefined regions (67). Different input combinations of the mentioned features were compared, with the optimal combination combining functional and structural data for an accuracy of 84% and AUC of 0.83 (67).
Overall, machine learning has shown promising results in identifying fMRI-based markers of migraine. Considering the high dimensionality of fMRI data, as well as the variety of clinical phenotypes inherent in migraine, machine learning approaches applied to a standardized, homogenized and sufficiently clinically described dataset could likely advance our understanding of migraine pathophysiology.
Discussion
We identified and summarized 114 studies published between April 2014 and December 2021 (following a previous review of migraine-related fMRI literature [14]), employing fMRI in heterogeneous approaches to investigate a variety of aspects of migraine. A recent systematic review has summarized a specific aspect of the literature, namely FC studies (153). Another recent non-systematic article summarized literature findings to characterize fMRI changes over the migraine cycle (154). In our review, we employed a wider systematic search strategy to capture all literature employing fMRI in migraine for a broader overview over fMRI applications, including a total of 114 studies with a wide range of applications such as FC, EC, ALFF, ReHo, as well as stimulation, therapeutic, and machine learning approaches in diverse migraine subgroups.
In our current review, we focused on specific anatomo-functional brain structures and networks, namely insula, various brainstem nuclei, thalamus, hypothalamus, limbic areas, as well as a number of functional networks. This selection does naturally not reflect the full scope of all reported alterations in the literature in an exhaustive manner. While other areas were demonstrated as relevant in some studies, such as the cerebellum or various frontal and occipital areas, the selection we decided on was found to be both of interest on a regular basis and subject to consistent nomenclature, which we perceived as advantageous when dealing with the high methodological variability observed in the literature.
Current evidence supports the understanding of migraine as an alteration of brain state rather than a more localized disorder of pain (148,150). A wide variety of functional network alterations differentiating migraine patients from HC has been observed, not just during migraine attacks, but also in the interictal interval. Overall, the current literature suggests that the activity of a migraine patient’s brain fundamentally differs from the brain activity in healthy humans. Herein, fMRI might be considered a well-suited modality to investigate migraine pathophysiology, since the need for information regarding individual cell or small-scale network (e.g., tens to hundreds of cells) activity (which fMRI cannot provide) is less of an issue when looking at large-scale network effects arising from cumulative cell activity.
At this point however, we have to consider the caveats that are obvious in the current literature. Notably, some of the extracted results appear to oppose each other. For example, separate studies indicated increased (26) and decreased (100) rs-FC of amygdala to the middle occipital gyrus. This is likely a result of a known issue in fMRI literature, specifically the multitude of researcher degrees of freedom (7); in this specific example, the populations were different (chronic migraine vs. migraine without aura) and the seed placement was different (bilateral vs. unilateral). This illustrates an issue apparent in the literature as a whole: findings from different studies are often not directly comparable, at least for the literature identified in this review. Reporting standards and definitions of relevant vocabulary (e.g., “interictal”) were found to differ strongly between different studies. Migraine can be considered a term encompassing many entities (e.g., with and without aura, chronic, chronic with medication overuse, vestibular, menstrual), which themselves decompose into further subcategories (e.g., aura complexity, aura lateralization). As detailed in this review, multiple imaging studies demonstrated that different entities of migraine differ with regard to fMRI metrics. Thus, in order to facilitate replicability and inter-study comparability, future research should take the high dimensionality of this disease into account whenever possible. Upcoming studies would likely benefit from a set of consensus reporting standards regarding fMRI usage in migraine, which is currently not present, as well as a strict adherence to general fMRI reporting principles (7,21), in order to create greater homogeneity of data.
Another set of issues could be present in the high amount of different fMRI metrics available and the oftentimes low statistical power of group comparisons, which are known factors weakening evidence in fMRI literature (7). Recent evidence suggests that robust brain imaging results require much higher sample sizes than the median of 52 participants we observed within the current literature landscape (155), which are often further parcellated into rather small subgroups according to group assignments or other factors. Additionally, individual findings were hardly ever directly replicated. Thus, while the sheer volume of evidence regarding the regions detailed above certainly appears to implicate them in the pathophysiology of migraine, the construction of reliable mechanistic models of migraine seems to lie outside the limits of current literature.
The need for larger sample sizes naturally posits the issue of increasing costs, time, and resources in generating reliable datasets for fMRI studies in migraine. One potential solution to this problem could be found in data sharing databases, such as, OpenfMRI (156) (https://openfmri.org/) or the ENIGMA initiative (157) (https://enigma.ini.usc.edu/). Currently however, despite the high burden of disease for migraine, it appears that there is no specific register for migraine-related fMRI data sharing. The collection and sharing of imaging data, especially if combined with strict reporting standards as detailed above, would allow for the generation of a higher volume of imaging data, which in turn would likely improve the robustness of findings. Another aspect that should be considered are device-dependent differences in imaging data, the reduction of which falls under the label of data harmonization (158–161). Inter-scanner variability is an important factor complicating comparability of individual imaging data including fMRI, and different methods have been proposed to reduce inhomogeneity for fMRI (158,159). If successful, data sharing and harmonization between centers and scanner systems could enable big data approaches to data analysis, most notably also relevant for applications of machine learning.
Machine learning algorithms have been used successfully in the analysis of fMRI data in migraine (24,44,99). The potential of these approaches may partially lie in pattern recognition in high-dimensional data and representation of complex features (162). Thus, the previously mentioned complexity of migraine entities, as well as patient-specific factors such as age, sex, individual medication, or clinical parameters could be taken into account in the analysis of imaging data. The potential use cases of these algorithms are manyfold: they could contribute to early and accurate diagnosis of migraine via the identification of discriminatory fMRI features between HC and different types of migraine (44,67,99), find use as a tool for therapy monitoring (24), or identify targets for new therapeutic interventions.
Regarding treatment, fMRI has shown promise as a supplementary tool in therapeutic studies for the investigation of central mechanisms of action. However, relatively few studies have currently made use of fMRI to investigate or pinpoint treatment effects, often but not exclusively investigating acupuncture. Interestingly, pharmacological studies have been absent from the surveyed literature after filtering, implying that such studies rarely investigate central mechanisms of action using fMRI. Acupuncture studies were able to demonstrate altered fMRI responses between verum and sham therapies, which could bear interesting potential for the distinction of placebo and intervention-specific effects in future studies. Aside from acupuncture, neuromodulatory therapeutic approaches have been investigated in different forms. While studies have investigated taVNS, RAVANS, and eTNS, many other neuromodulatory bottom-up approaches exist for migraine treatment (e.g., exploiting the trigemino-cervical complex by performing peripheral muscular stimulation to alleviate headache symptoms [151,163–167]), for which fMRI investigations currently appear to be lacking. Here, fMRI could potentially help to elucidate central mechanisms of action. Interestingly, multiple studies reported associations between changes in fMRI metrics and clinical improvements (24,25,57,126), which could serve as motivation to investigate potential causal effects underlying these correlations.
Conclusion
In our review of 114 studies of fMRI applications in migraine, we identified a number of brain areas repeatedly implied in the pathophysiology of migraine. Study results were however rarely replicated and often not directly comparable in terms of the investigated populations or fMRI metrics. Future research could benefit from consensus-based imaging acquisition, post-processing, and reporting standards, strict adherence to general fMRI quality assurance standards, and data sharing-based extension of the cohort size. The latter could enable more powerful and widespread applications of machine learning for improvements in diagnosis and treatment of migraine.
Clinical implications
Functional MRI can contribute to the understanding of migraine pathophysiology and is increasingly applied. Repeatedly emerging structures of interest included the insula, brainstem, limbic system, hypothalamus, thalamus, and functional networks. Functional MRI may help to identify target structures for individualized therapy and for patient phenotyping. Advanced data analysis including machine learning may help to isolate imaging patterns common to different patient populations.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024221128278 - Supplemental material for Functional magnetic resonance imaging in migraine: A systematic review
Supplemental material, sj-pdf-1-cep-10.1177_03331024221128278 for Functional magnetic resonance imaging in migraine: A systematic review by Severin Schramm, Corinna Börner, Miriam Reichert, Thomas Baum, Claus Zimmer, Florian Heinen, Michaela V Bonfert, Nico Sollmann in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The work of NS is supported by the Dr.-Ing. Leonhard Lorenz Foundation, the German Migraine and Headache Society (DMKG), and the Joachim Herz Foundation. The work of MB is supported by the Bavarian Gender Equality Grant of the Free State of Bavaria, the German Migraine and Headache Society (DMKG), and the ZNS-Hannelore-Kohl Stiftung. FH received a grant “Innovationsfonds” of the joint federal committee of health insurance companies (GBA) for a nation-wide study on an early multimodal intervention program for children with migraine. The Division of Pediatric Neurology and Developmental Medicine, Dr. von Hauner Children’s Hospital, LMU Hospital, Munich, Germany is provided by an emFieldPro magnetic stimulator by Zimmer MedizinSysteme GmbH (Neu-Ulm, Germany).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
