Abstract
Background
Cluster headache is a disorder characterized by intermittent, severe unilateral head pain accompanied by cranial autonomic symptoms. Most cases of CH are episodic, manifesting as “in-bout” periods of frequent headache separated by month-to-year-long “out-of-bout” periods of remission. Previous imaging studies have implicated the hypothalamus and pain matrix in the pathogenesis of episodic CH. However, the pathophysiology driving the transition between in- and out-of-bout periods remains unclear.
Methods
The present study provides a narrative review of previous neuroimaging studies on the pathophysiology of episodic CH, addressing alterations in brain structures, metabolism, and structural and functional connectivity occurring between bout periods.
Results
Although the precise brain structures responsible for episodic CH are unknown, major roles are indicated for the posterior hypothalamus (especially in acute attacks), the pain neuromatrix with an emphasis on central descending pain modulation, and non-traditional pain processing networks including the occipital, cerebellar, and salience networks. These areas are potentially related to dynamic transitioning between in- and out-of-bout periods.
Conclusion
Recent progress in magnetic resonance imaging of episodic CH has provided additional insights into dynamic bout-associated structural and functional connectivity changes in the brain, especially in non-traditional pain processing network areas. These areas warrant future investigations as targets for neuromodulation in patients with CH.
Keywords
Introduction
Cluster headache (CH) is a primary headache disorder that presents with periodic severe unilateral periorbital pain accompanied by cranial autonomic symptoms. CH is a rare disorder with a prevalence of less than 1%, affects mainly men, and has a typical age of onset between 20 and 40 years (1). During attacks, the autonomic symptoms accompanying CH include unilateral ptosis, miosis, lacrimation, conjunctival injection, ear fullness, rhinorrhoea, and nasal congestion ipsilateral to the side of headache pain. Based on this symptom presentation, CH is classified as a prototype of trigeminal autonomic cephalalgia according to the diagnostic criteria of the International Classification of Headache Disorders, 3rd edition, beta version (2). Individual CH attacks usually last between 15 and 180 minutes and can occur up to eight times per day during cluster periods. Typically, headaches occur with regular circannual (mainly in the autumn and spring) and circadian timing (2,3). CH is divided into episodic and chronic subtypes based on the duration and frequency of episodes. Episodic CH, which accounts for 80–90% of CH cases, manifests in bout periods lasting from seven days to 12 months, during which patients may experience daily attacks at a high frequency, and asymptomatic out-of-bout remission periods that last for months to years at a time. Chronic CH (10–20% of cases) is diagnosed when patients experience a full year without remission or if the duration of remission is less than 30 days (2,4).
The pathophysiology of CH remains poorly understood. Several possible mechanisms have been proposed, based on assumptions generated by a variety of methods including clinical and therapeutic observations as well as electrophysiological, endocrinological, and animal studies. The question of whether CH pain is of peripheral or central origin remains a matter of debate, and both possibilities are considered. Several characteristics (namely predominance of trigeminal pain, parasympathetic discharge, and sympathetic deficit) of CH attacks are consistent with the involvement of the cavernous sinus, because this is the only peripheral site where a combination of trigeminal C-fibers and sympathetic fibers could be affected by a single cause (5). However, this possibility has not been confirmed clinically. The orbital phlebography alterations within the cavernous sinus do not differ from findings in other headache forms. Furthermore, systemic signs of inflammation in CH have never been identified (6). Consequently, inflammation of the cavernous sinus can be ruled out as the source of CH pain.
Another possibility is that CH is the result of excessive activation of the peripheral trigeminal nerve. According to this model, activation of the first (ophthalmic) division of the trigeminal nerve produces severe unilateral head pain, while activation of autonomic efferents produces autonomic symptoms such as lacrimation (7). This model is supported by findings of increased concentrations of a trigeminal activation marker (i.e. calcitonin gene-related peptide) and of a parasympathetic activation marker (i.e. vasoactive intestinal peptide) in jugular vein blood ipsilateral to CH attacks (8). However, similar changes in calcitonin gene-related peptide concentration have also been documented during migraine attacks (9). Therefore, although the peripheral trigeminal nerve might be involved in CH, its activation alone cannot account for CH.
The regularity and seasonality of CH attacks in conjunction with a relapsing-remitting presentation suggest that CH may be tied to a biological clock, such as that in the hypothalamus (10). In further support of hypothalamic involvement, CH patients often show accompanying neuroendocrinological abnormalities, including low testosterone in men (11) and abnormal levels of hormones regulated by the hypothalamus including cortisol, growth hormone, thyroid-stimulating hormone, prolactin, melatonin, follicle-stimulating hormone, and luteinizing hormone (12). Furthermore, anatomical studies in rodents have revealed that the trigemino-hypothalamic tract provides a direct connection between the posterior hypothalamus and trigeminal nucleus caudalis, which could mediate autonomic symptoms (13).
Besides this, previous animal experiments have found that stimulation of the pontine superior salivary nucleus, which is the origin of cells in the parasympathetic autonomic vasodilator pathway, independently produces short term neuronal activation in the trigeminocervical complex; this activation is associated with autonomic facial symptoms, and is inhibited by oxygen treatment, in CH (14,15). The superior salivary nucleus receives a reflex connection from the trigeminal nucleus and direct projections from the paraventricular hypothalamic nucleus. In addition, trigeminovascular nociceptive processing may be modulated by manipulation of the paraventricular hypothalamic nucleus (16). Therefore, the hypothalamic nuclei may be involved in the nociceptive activation of trigeminovascular neurons and thus have the potential to trigger CH.
In addition, the sphenopalatine ganglion (SPG) has also been associated with cranial pain and autonomic symptoms as well as activation of the trigeminal-autonomic reflex in CH (17). Prior studies have suggested that activated post-ganglionic parasympathetic fibers from the SPG can release neuropeptides that produce local meningeal vessel dilation and trigeminal nociceptive fiber activation (17,18). These neuronal inputs can also trigger a reflex connection in the superior salivary nucleus, contributing to elevated cranial parasympathetic activity mediated by the SPG (17,19,20). To this end, SPG stimulation has been explored as a minimally invasive and effective treatment option for refractory CH (21–23).
Collectively, this convergence of clinical and basic research evidence supports the hypothesized involvement of the hypothalamus and trigeminovascular system in CH pathophysiology. However, basic research has not yet elucidated the pathophysiology underlying the transition between in- and out-of-bout periods. Structural and functional neuroimaging methods, including single-photon emission computed tomography (SPECT), positron-emission tomography (PET), magnetic resonance spectroscopy (MRS), structural magnetic resonance imaging, diffusion tensor imaging (DTI), and functional MRI (fMRI), could complement basic research. Hereafter, we provide a narrative review of previous neuroimaging studies on the pathophysiology of episodic CH, addressing alterations in brain structures, metabolism, and connectivity occurring during in- or out-of-bout or between bout periods.
Early brain neuroimaging studies in CH
Functional studies
During in- or out-of-bout
Functional and structural neuroimaging findings on cluster headache.
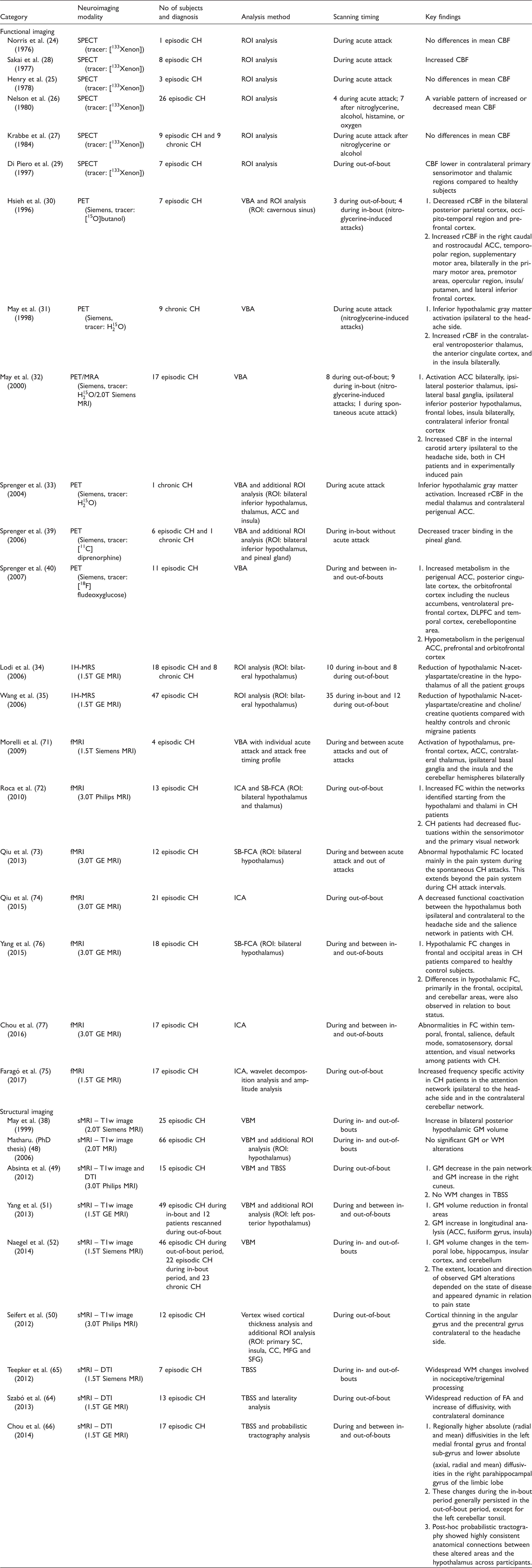
Abbreviations: ACC: anterior cingulate cortex; CC: cingulate cortex; CBF: cerebral blood flow; CH: cluster headache; DLPFC: dorsolateral prefrontal cortex; DTI: diffusion tensor imaging; FA: fractional anisotropy; FC: functional connectivity; fMRI: functional magnetic resonance imaging; FWE: family-wise error; GM: gray matter; HC: healthy controls; ICA: independent component analysis; L: left; MFG: middle frontal gyrus; MNI: Montreal Neurological Institute; MRS: magnetic resonance spectroscopy; PET: positron-emission tomography; R: right; ROI: region of interest; rCBF: regional cerebral blood flow; RSN: resting state network; SB-FCA: seed-based functional connectivity analysis; SC: somatosensory cortex; SFG: superior frontal gyrus; sMRI: structural magnetic resonance imaging; SPECT: single-photon emission computed tomography; TBSS: tract-based spatial statistics; VBA: voxel-based analysis; VBM: voxel-based morphometry; WM: white matter.
In the first positron emission tomography (PET) study of nitroglycerin-triggered CH attacks, Hsieh et al. (30) reported the activation of brain areas involved in the central nociception, in the absence of changes in the brain stem or hypothalamus. Subsequently, in a landmark series of neuroimaging studies, May et al. (31,32) observed a marked increase in activation of the ipsilateral posterior hypothalamus in CH patients experiencing nitroglycerin-triggered attacks, relative to headache-free status or control subjects. Similar hypothalamic activation was reported in a patient experiencing a spontaneous CH attack (33).
Later, proton magnetic resonance spectroscopy (MRS) studies provided additional evidence in support of hypothalamic dysfunction in CH. In 2006, both Lodi et al. (34) and Wang et al. (35) documented significantly lower hypothalamic N-acetylaspartate/creatine ratios in CH patients compared to healthy control subjects and chronic migraine patients. These MRS findings indicated neuronal dysfunction in the hypothalamus in patients with CH.
It is noteworthy that certain brain regions involved in pain processing, commonly called the pain matrix, show altered structural connectivity and activity in a broad range of acute and chronic visceral and somatic pain conditions (36,37). Implicated regions in CH include the anterior cingulate cortex, posterior thalamus, basal ganglia, insula, and cerebellar hemispheres (22,32,38). Indeed, dysfunction of the pain modulatory system has been demonstrated in CH patients using PET (39,40). Using PET with the opioidergic ligand [11C] diprenorphine in CH patients in their bout period but outside of an acute attack, Sprenger et al. found decreased tracer binding in the pineal gland, and an inverse relationship between the duration of CH and opioid receptor binding in the ipsilateral hypothalamus (39).
Between in- and out-of-bouts
Sprenger et al. (40) also used PET to identify hypometabolism in frontal areas such as the perigenual anterior cingulate and prefrontal cortices in episodic CH patients (without acute attacks). Compared to out-of-bout scans, in-bout scans showed increased metabolism in the thalamus and perigenual anterior cingulate, posterior cingulate, prefrontal, insular, and temporal cortices, suggesting that there are dynamic functional differences in the central descending pain modulatory system between in-bout (without acute attacks) and out-of-bout periods. These findings suggest that CH pathophysiology may also result from disordered descending opioidergic or pain modulation mechanisms, with significant dynamic changes between the in-bout and out-of-bout periods.
Collectively, the aforementioned functional evidence suggests that episodic CH pathophysiology involves the hypothalamus as well as other structures in the pain matrix, particularly those related to descending pain modulation. There are no currently available functional imaging studies to study the differences between episodic and chronic CH patients. However, attempts to treat intractable CH with deep-brain stimulation (DBS) of the posterior hypothalamus have met with limited success (41–43). DBS may yield improvement in about 60% of cases after weeks or months of therapy (44), but has been reported to have no effect on acute CH attacks (45). The partial effectiveness of hypothalamic DBS against CH observed thus far is consistent with the supposition that other structures play a contributory causative role in CH pathophysiology. It could therefore be plausible that the hypothalamus is involved in acute attacks, but the pathophysiology associated with in-bout/out-of-bout transitions may extend beyond the hypothalamus, and involve dynamic interactions with unidentified cortical and subcortical areas.
Recent brain structural and functional MRI findings in CH
Recent advancements in brain MRI techniques and analytical approaches can better elucidate the pathophysiology of episodic CH and dynamic changes occurring between in-bout and out-of-bout periods. The development of specialized magnetic resonance imaging (MRI) methods has enabled more refined investigations of the human brain. Such methods also permit the detection of superregional, systems-level, alterations associated with CH. In particular, gray matter (GM) macrostructural changes revealed by T1 voxel-based morphometry (VBM), white matter (WM) microstructural and structural connectivity changes on diffusion tensor imaging (DTI), and functional connectivity changes revealed by resting-state functional MRI (RS-fMRI) have provided insights into the potential mechanisms of CH pathophysiology.
Gray matter macrostructural changes revealed by VBM
VBM is an analysis framework well suited for inter-group comparative studies of regional brain volume and tissue density (46). It allows for spatial normalization through the alignment of T1-weighted volumetric images to a standard spatial template, enabling multiple anatomical MRI scans to be analyzed as a group (47). Furthermore, VBM can distinguish among GM, WM, and cerebrospinal fluid components to permit selective analyses of GM or, less commonly, WM volume (47). Tissue-segmented images can then be smoothed and submitted for further statistical analyses (46). Statistical overviews of VBM analysis results can be presented in pseudo-colorized maps of statistically significant voxels with a color scale that indicates t-statistic values for voxels violating the null hypothesis (47).
During in- or out-of-bout
Using T1 VBM, May et al. (38) found that patients with CH had increased GM volume in the hypothalamus during both in-bout and out-of-bout periods. However, this finding has yet to be reproduced. On the other hand, in a subsequent VBM study, Matharu found no differences in global or hypothalamic GM or WM volume in 66 CH patients compared to 96 age- and sex-matched control subjects (48). In a T1 VBM study of out-of-bout episodic CH patients, Absinta et al. (49) also found no changes in hypothalamic GM volume relative to control subjects, but did observe reduced GM volume in several regions related to pain processing including the thalamus, caudate nucleus head, precentral gyrus, posterior cingulate cortex, middle frontal gyrus, middle temporal gyrus, inferior parietal lobule, and insula.
In a structural MRI study for cortical surface, Seifert et al. (50) investigated cortical thickness in out-of-bout episodic CH as compared with a healthy control group. A reduction of cortical thickness was found in the angular gyrus and the precentral gyrus contralaterally to the headache side, but this reduction did not correlate with disease duration. However, cortical thickness in the primary sensory cortex correlated with disease duration, suggesting disease-related plasticity (50).
Between in- and out-of-bouts
Our research further used VBM to investigate longitudinal regional GM volume differences between in-bout and out-of-bout periods, and between CH patients and healthy control subjects, and identified structural differences in frontal pain modulatory areas (51). Accordingly, GM volume changes may reflect insufficient pain-modulatory capacity in frontal areas in patients with episodic CH.
There are no available structural imaging studies for chronic CH patients alone. In a structural imaging study of episodic and chronic CH patients, Naegel et al. (52) reported distinct regional changes in GM volume in different brain regions including the temporal lobe, hippocampus, insular cortex, and cerebellum in 91 patients with CH, including 46 episodic CH during out-of-bout, 22 episodic CH during in-bout, and 23 chronic CH. No hypothalamic changes were detected in patients with CH compared to healthy controls. The extent, location, and direction of observed GM alterations depended on the state of disease and appeared to be dynamic in relation to the pain state (52).
Collectively, recent VBM studies, mainly performed in the absence of acute CH attacks, have not identified structural GM changes in the hypothalamus, but mainly areas involved in the pain matrix (48,49,51,52).
White matter microstructural and structural connectivity changes on DTI
Over the last decade, DTI has enabled the non-invasive evaluation of microstructural alterations in brain WM (53,54). DTI uses a diffusion ellipsoid model to represent the three-dimensional characteristics of water molecule diffusion within WM structures, and allows this information to be decomposed into three mutually orthogonal eigenvectors and three eigenvalues: the diffusion coefficient along the direction of maximal apparent diffusion (axial diffusivity or λ1), and the diffusion coefficients along two orthogonal directions embedded in the plane perpendicular to the maximal diffusion direction (λ2 and λ3). The most commonly used quantitative indices are mean diffusivity (MD, the mean of the three eigenvalues = (λ1 + λ2 + λ3)/3) and fractional anisotropy (FA, the standard deviation of the three eigenvalues), which reflect the amplitude and directionality of water diffusion within a measured region, respectively. MD and FA can be influenced by myelination, coherence, packing density, axonal diameter, tract structural integrity, and fiber architectural complexity (55–58). Furthermore, directional diffusivities—that is, axial diffusivity (Dax = λ1) and radial diffusivity (Drad = (λ2 + λ3)/2)—provide additional information on the potential underlying pathophysiology of WM changes (54). Alterations in Dax are suggestive of axonal damage and/or Wallerian degeneration in the primary fiber orientation(59–61), whereas increased diffusivity perpendicular to the WM tract (Drad) signifies demyelination or dysmyelination (59,62,63). In addition to these quantitative indices, the principal orientation of diffusion can be further used to reconstruct WM pathways in vivo.
During in- or out-of-bout
DTI has recently been used to investigate potential microstructural WM changes in CH patients. One study revealed no significant differences in MD and FA values for the WM skeleton in CH patients compared to healthy control subjects (49). Two other studies showed microstructural WM changes in multiple broadly distributed brain regions, with the pain matrix well represented among them (64,65). All these studies had relatively small patient cohorts, and the discrepancy in results may be attributable to differences in the study population: Absinta et al. (49) and Szabó et al. (64) recruited only out-of-bout CH patients, whereas Teepker et al. (65) recruited both in- and out-of-bout patients, but did not assess between-group differences. Therefore, these results may not be specific for CH and should be interpreted carefully.
Between in- and out-of-bouts
Our group further used DTI with multiple (axial, radial, and mean) diffusivity indices to demonstrate WM microstructural differences in frontal and limbic areas between in-bout CH patients and healthy control subjects; these changes (except for those in the cerebellum) persisted during out-of-bout periods (66). Probabilistic tractography showed consistent anatomical connections between altered areas and the hypothalamus. These findings may explain partially why CH manifests with shifts between in- and out-of-bout periods. Further studies are warranted to determine the role of WM changes and to unravel the complexity of these connections with the hypothalamus and their association with CH pathophysiology.
Functional connectivity changes revealed by RS-fMRI
Blood oxygen-dependent (BOLD) fMRI measures changes in blood flow and tissue oxygenation (using oxyhemoglobin-to-deoxyhemoglobin ratios within brain venules and veins) as a surrogate marker of neuronal activity (67). Local shifts in blood oxygenation occur immediately after neuronal activation, enabling the assessment of localized brain activity with a time resolution in the order of seconds. Unlike task-fMRI, RS-fMRI captures baseline levels of brain activity occurring when subjects are not performing any particular task; during imaging, subjects are instructed to stay awake and not to think about anything in particular (68). Of note, resting-state brain activity accounts for >95% of the brain metabolic load, (9) yet the function of this activity remains unclear. Neuroscientists have posited that basal activity plays a role in synapse maintenance and inter-regional information transfer (70).
RS-fMRI involves the collection of so-called intrinsic connectivity data, the purpose of which is to produce a functional brain map representing regional inter-connectivity. Signal variations detected on RS-fMRI are neuronal in origin and correspond to the baseline activities of distinct networks (as opposed to activities triggered by external stimuli). Accordingly, inter-regional temporal correlations of low-frequency (<0.1 Hz) fluctuations are interpreted as indicators of functional connectivity (FC) (68,70). RS-fMRI thus enables the feasible collection of information on multiple brain networks and their functional organization even in pathological contexts. Furthermore, the examination of spontaneous brain activity may provide information about interactions among brain structures and lead to a better understanding of brain functional architecture.
During in- or out-of-bout
Using RS-fMRI, Morelli et al. (71) first investigated the pattern of cerebral activation during acute attacks in patients with episodic CH. They found activation of diencephalic regions, mainly the hypothalamus, and found trends of activation in cerebral areas involved in pain processing. Rocca et al. (72) found abnormal FC in the sensorimotor and primary visual networks of CH patients in a pain-free state, and Qiu et al. (73) observed abnormal FC between the hypothalamus and pain network areas as well as the occipital cortex during acute CH attacks. Additionally, decreased functional co-activation was detected between the hypothalamus and salience network areas in patients with CH (74). This evidence suggests that FC disruptions in pain-related as well as non-traditional pain processing areas (e.g. the occipital and salience networks) are related to CH pathophysiology. Additionally, Faragó et al. (75) further investigated the amplitude of RS-fMRI activity to find signatures of increased excitability during the out-of-bout period in patients with CH. They found increased frequency-specific activity in the attention network ipsilateral to the headache side and the contralateral cerebellar network.
Between in- and outside of bouts
Our study group further used seed-based RS-fMRI to demonstrate hypothalamic FC changes in frontal and occipital areas in CH patients compared to healthy control subjects. Furthermore, differences in hypothalamic FC, primarily in the frontal, occipital, and cerebellar areas, were also observed in relation to bout status. Additionally, annual bout frequency correlated with hypothalamic FC in the cerebellar region (76). More recently, we combined RS-fMRI with a network-sensitive independent component analysis approach and identified FC abnormalities within the temporal, frontal, salience, default mode, somatosensory, dorsal attention, and visual networks in CH patients. Dynamic FC changes in the frontal network according to bout status were strongly related to CH duration in that study (77). These findings advance the understanding of network functionality related to episodic CH, and emphasize the role of functional changes in multiple brain networks, especially non-traditional pain-processing networks.
Conclusion and implications
The findings of recent neuroimaging studies have yielded four major observations regarding CH: (a) CH appears to involve structural and functional changes in the pain matrix, especially in the descending pain modulation network; (b) disordered frontal top-down pain modulation is dynamically altered between in-bout and out-of-bout periods; (c) the anatomical and functional links between the hypothalamus and forebrain as well as cerebellar areas are altered in CH and may underlie, to some extent, its pathophysiology and clinical characteristics; and (d) the hypothalamus is likely involved in acute attacks, though the relationship may be a dynamic one (Figure 1).
Schematic brain representation summarizing the findings of neuroimaging studies of cluster headache (CH): (1) CH appears to involve structural and functional changes in the pain matrix, especially in the descending pain modulation network; (2) disordered frontal top-down pain modulation is dynamically altered between in-bout and out-of-bout periods; (3) the anatomical and functional links between the hypothalamus and forebrain as well as cerebellar and occipital areas are altered in CH and may underlie its pathophysiology and clinical characteristics; and (4) the hypothalamus is likely involved in acute attacks, with a possible tendency to reverse outside acute attacks. The blue circles indicate the anatomic locations with structural and functional changes during in- or out-of-bout (without acute attacks) episodes. The yellow circles indicate the anatomic locations with structural and functional changes between in- and out-of-bouts. The red circles indicate the anatomic locations with structural and functional changes during acute attacks.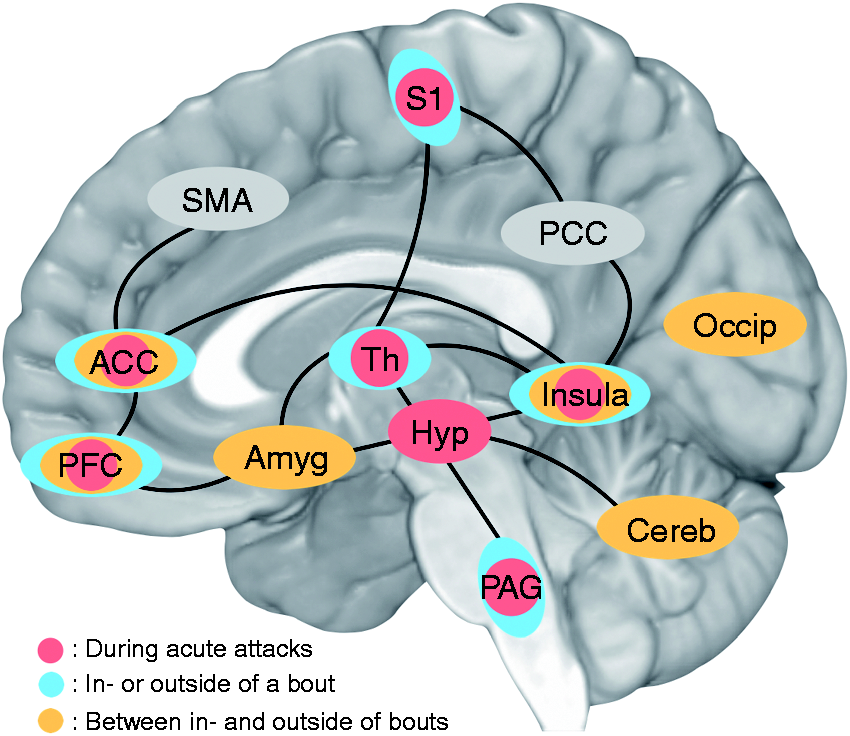
These results have several implications. First, insufficient pain modulation in frontal pain areas appears to contribute to the pathophysiology of CH. Structural neuroimaging studies have observed significant GM volume and WM microstructural differences in these areas (51,66). Accordingly, it appears that the descending pain modulation system is structurally impaired in CH patients. These structural differences may contribute to the complex pathophysiology that makes CH patients susceptible to the emergence of bout periods and headache attacks. The results also have implications for the development of new CH treatments, including the identification of targets for neuromodulation.
Second, recent seed-based RS-fMRI studies have reported significant hypothalamic FC differences in CH patients (74,76). CH-associated changes in hypothalamic FC primarily involved frontal pain modulatory, occipital, and cerebellar areas. Furthermore, bout-associated FC changes in multiple discrete cortical areas not traditionally associated with pain processing were identified (77). Additionally, an RS-fMRI study found changes in the brain functional networks of patients with episodic CH, namely altered resting state activity in the attention network ipsilateral to the headache, which might be a signature of increased cortical excitability, and might play an important role in the pathogenesis of CH (75). Collectively, it is thus plausible that, in addition to the hypothalamus, the pathophysiology of episodic CH involves multiple brain networks such as the frontal pain modulation network and non-traditional pain processing networks. However, little is known about brain network reorganization in CH patients, and much less is known about how such reorganization might vary with respect to bout status. Future studies are needed to investigate the possible roles of brain network reorganization in CH.
Third, recent DTI findings have indicated discrete WM microstructural changes in CH patients, and tractography has confirmed a structural association between the hypothalamus and clusters of regional WM changes in pain modulatory areas (64–66). These structural hypothalamic connectivity alterations may explain the clinical characteristics of CH: hypothalamic-cerebellar WM changes observed consistently between in-bout and out-of-bout periods, together with findings of significant differences in hypothalamic FC between in-bout and out-of-bout periods (primarily in cerebellar areas) (76), may underlie the mechanism of bout period transitions. Bout shifts may be triggered by an alteration of this structural and functional connection combined with the loss of cerebellar inhibition and modulation of the hypothalamus. Given the role of the hypothalamus in CH, it is reasonable to hypothesize that cerebellar-hypothalamic circuits underlie CH pathophysiology and bout period transitions, in particular. Further research is required to examine these hypotheses, and to elucidate therapeutic benefits of targeting the cerebellum with transcranial magnetic stimulation.
Finally, previous studies have reported metabolic and structural changes in the hypothalamus during spontaneous or provoked acute CH attacks. However, the VBM and DTI findings discussed above do not provide direct support for the notion that hypothalamic structural differences play a primary role in CH. It could be that the appearance of hypothalamic involvement in CH varies according to the timing of the investigation (e.g. in-bout versus out-of-bout status, or during acute attacks). Although our abovementioned studies evaluated CH patients during the in-bout period, we did not examine patients during acute attacks (51). Hypothalamic volume abnormalities might potentially reflect a dynamic process related to acute CH attacks. Furthermore, the trigeminovascular system, especially the SPG and its reflex connection (trigemino-parasympathetic reflex) to the superior salivary nucleus have also been suggested to be associated with pathophysiology of the acute CH attacks (17). Blockade of the SPG could relieve the symptoms of acute CH attacks (17,19,20). However, connectivity investigations of the structural and functional relationships between the brainstem nuclei and their projection or afferent regulatory areas (i.e. SPG and hypothalamus) remain nascent. Further optimization of neuroimaging techniques may be required for enhanced comprehension of the pathophysiology of acute CH attacks.
In conclusion, recent progress in neuroimaging has provided a critical starting point from which the structural and functional network bases of CH pathophysiology can be further investigated. Additionally, our understanding of CH is poised to benefit from new and emerging techniques that will enable more detailed analyses of structural and functional neuroimaging data.
Article highlights
Cluster headache (CH) appears to involve structural and functional changes in the pain matrix, especially in the descending pain modulation network. Disordered frontal top-down pain modulation is dynamically altered between in-bout and out-of-bout periods. The anatomical and functional links between the hypothalamus and forebrain as well as cerebellar areas are altered in CH and may underlie, to some extent, its pathophysiology and clinical characteristics. The hypothalamus is likely involved in acute attacks, though the relationship may be a dynamic one. Recent progress in neuroimaging has provided a critical starting point from which the structural and functional network bases of CH pathophysiology can be further investigated.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported in part by grants from Taipei and Taichung Veterans General Hospital [V106C-106, VGHUST105-G7-1-1, V105C-127, V105D9-001-MY2-2, V105E9-001-MY2-1, VTA105-V1-1-1, VGHUST104-G7-1-3, TCVGH-103G213, TCVGH-YM1030204], Tri-Service General Hospital, Taiwan (TSGH-C101-159, and TSGH-C106-068); Ministry of Science and Technology of Taiwan [MOST 105-2314-B-016-004-, MOST 106-2321-B-010-009-, MOST 104-2314-B-010-015-MY2, MOST 103-2321-B-010-017-, 104-2218-E-010-007-MY3, 106-2321-B-010-011-], Academia Sinica [Grant No. IBMS-BM10601010026, IBMS-CRC103-P04, AS-104-TP- B10], National Health Research Institutes (NHRI-EX106-10611EI), Brain Research Center, National Yang-Ming University, Ministry of Health and Welfare, Taiwan [MOHW 103-TDU-B-211-113-003, MOHW 104-TDU-B-211-113-003, MOHW 105-TDU-B-211-113-003], and a grant from the Ministry of Education, Aim for the Top University Plan.
