Abstract
Background
We aimed to assess the effects of preventive migraine treatment with atogepant vs. placebo on patient-reported quality of life and functioning.
Methods
Analyses of patient-reported outcomes from three 12-week, randomized, placebo-controlled trials evaluating preventive migraine treatment with atogepant 60 mg once-daily: ADVANCE (low-frequency episodic migraine [LFEM], 4–8 monthly migraine days [MMDs] and high-frequency episodic migraine [HFEM], 8–14 MMDs), PROGRESS (chronic migraine, CM) and ELEVATE (episodic migraine in those previously failed by two to four classes of oral preventive treatments).
Results
Least squares mean differences (95% confidence interval (CI)) in change from baseline were greater (p < 0.05) for atogepant vs. placebo for Migraine-Specific Quality of Life questionnaire Role Function–Restrictive domain scores at week 12 (ADVANCE: LFEM 12.0 (95% CI = 6.0–18.0), HFEM 9.9 (95% CI = 3.4–16.4); PROGRESS: 6.2 (95% CI = 2.5–9.8); ELEVATE: 17.7 (95% CI = 13.1–22.3)), for Headache Impact Test-6 total scores at week 12 (ADVANCE: LFEM −4.7 (95% CI = −6.7 to −2.7); HFEM −3.4 (95% CI = −5.5 to −1.2); PROGRESS: −2.8 (95% CI = −4.1 to −1.4); ELEVATE: −6.5 (95% CI = −8.3 to −4.7)) and for Activity Impairment in Migraine–Diary-Performance of Daily Activities scores across 12 weeks (ADVANCE: LFEM −2.3 (95% CI = −3.9 to −0.7), HFEM −4.5 (95% CI = −6.9 to −2.2); PROGRESS: −3.4 (95% CI = −5.3 to −1.5); ELEVATE: −4.7 (95% CI = −6.4 to −3.1)).
Conclusions
Preventive migraine treatment with atogepant 60 mg once-daily vs. placebo improved measures of migraine-related quality of life and functioning among participants with different headache frequencies and histories of previous treatment failure.
This is a visual representation of the abstract.
Introduction
Migraine is a debilitating, chronic neurologic disease that causes detriments to individuals’ overall health, quality of life (QoL), daily functioning, relationships and career/financial status, increasing with headache frequency (1,–3). People with chronic migraine (CM) (≥15 monthly headache days (MHDs) with ≥8 qualifying as migraine days (4)) have a greater disease burden, including higher health care costs, lower employment, more frequent anxiety and depression, and greater disability, than those with episodic migraine (EM) (<15 MHDs) (2,5,8–). However, even among individuals meeting EM criteria, those with higher MHDs may experience greater negative effects (9); the burden of high-frequency EM (HFEM) (8–14 MHDs) is greater than that of lower-frequency EM (<8 MHDs) and comparable to CM (10,–12). Migraine burden is also affected by prior treatment response. Individuals with migraine not responding to preventive treatment use more health care resources and report severe decrements in QoL and professional and social functioning (3,13). These effects increase correspondingly with additional prior treatment failures (3,13). These data reveal substantial unmet needs among people with migraine experiencing more frequent MHDs and/or multiple preventive migraine treatment failures.
Atogepant is an oral antagonist of the calcitonin gene-related peptide (CGRP) receptor approved in the USA, Europe and other regions as a preventive migraine treatment in adults (14,15). The efficacy and safety of atogepant have been demonstrated across the migraine spectrum, including in a 12-week, phase 3 trial (ADVANCE) (16) and a 52-week long-term safety trial (17) in EM. Two subsequent 12-week, phase 3 trials showed that atogepant is also efficacious and safe for treating CM (PROGRESS) (18) and EM among individuals previously failed by two to four classes of conventional oral migraine preventives (ELEVATE) (19). In PROGRESS and ELEVATE, participants who received atogepant 60 mg once-daily experienced significantly greater reductions in monthly migraine days (MMDs) over 12 weeks vs. placebo (18,19).
While reduced migraine frequency is a critical treatment efficacy benchmark recommended by regulatory agencies (20,21), migraine's functional impact encompasses a range of illness features, including sensory, cognitive and emotional elements. To evaluate treatment effects that may not be adequately captured by conventional metrics, several validated patient-reported outcome measures (PROMs) have been developed that quantify health information collected directly from patients (22). In migraine, PROMs, including the Migraine-Specific Quality of Life questionnaire version 2.1 (MSQv2.1) (23), Headache Impact Test-6 (HIT-6) (24) and Activity Impairment in Migraine–Diary (AIM-D) (25), are routinely used to measure treatment-related changes in disease burden across migraine frequencies. While some components of these instruments overlap, they each quantify distinct domains and, together, provide a holistic profile of migraine treatment response that is relevant to patients’ experiences (22,26).
The objective of these analyses was to assess the effect of atogepant 60 mg once-daily vs. placebo on complementary measures of migraine-related QoL and daily functioning (MSQv2.1, HIT-6 and AIM-D) among different populations of migraine patients across several randomized controlled trials (RCTs). Although data evaluating improvements in some PROMs with atogepant in EM and CM have been previously published (18,27,28), these analyses provide additional context for the effect of atogepant across the migraine spectrum, including patients with particular treatment challenges (i.e. more frequent headaches or previous preventive treatment failures).
Methods
Study design and participants
ADVANCE (NCT03777059), PROGRESS (NCT03855137) and ELEVATE (NCT04740827) were phase 3, multicenter, double-blind RCTs conducted to evaluate the safety and efficacy of atogepant for the preventive treatment of migraine among participants with a ≥1-year history of EM regardless of treatment failure history (ADVANCE), ≥1-year history of CM (PROGRESS), or EM (≥1 year) previously failed by two to four classes of preventive migraine treatment (ELEVATE) (see supplementary material, Figure S1). Detailed methods for ADVANCE (conducted in the US [12/14/2018–6/19/2020]), PROGRESS (conducted in North America, Europe, and Asia [3/11/2019–1/20/2022]) and ELEVATE (conducted in North America and Europe [3/5/2021–8/4/2022]) were published previously (16,18,19). In ADVANCE, participants with 4–14 MMDs in the three months before visit 1 and 4–14 migraine days during the 28-day baseline period were randomized 1:1 to atogepant 10 mg, 30 mg or 60 mg or placebo once-daily for 12 weeks. In PROGRESS, participants with ≥15 MHDs in the past 3 months and ≥15 headache days (with ≥8 qualifying as migraine days) during the 28-day baseline period were randomized 1:1 to atogepant 30 mg twice-daily, atogepant 60 mg once-daily or placebo for 12 weeks. ADVANCE and PROGRESS excluded participants previously failed by more than four preventive migraine treatments, including two or more with different mechanisms. In ELEVATE, participants with 4–14 MMDs in the previous three months and 28-day baseline period, <15 MHDs in the three months prior to visit 1, and previously failed by two to four classes of conventional oral preventive migraine treatment were randomized 1:1 to atogepant 60 mg once-daily or placebo for 12 weeks. Treatment failure was defined as no meaningful reduction in migraine frequency after two or more months of treatment at accepted therapeutic doses during the seven years prior to screening, or discontinuation due to adverse effects at any time.
For all three trials, participants were excluded if they had clinically significant disease (e.g. cardiovascular, cerebrovascular, endocrine, neurological) or migraine history accompanied by diplopia or decreased level of consciousness, retinal migraine, current diagnosis of new persistent daily headache, trigeminal autonomic cephalalgia or painful cranial neuropathy, per the International Classification of Headache Disorders, 3rd edition. Barbiturate use was not permitted from 30 days before screening through the end of each trial.
Trials complied with the International Council on Harmonisation Good Clinical Practice Guideline and the Declaration of Helsinki. Investigators obtained approval from an independent ethics committee or local institutional review board (16,18,19). Participants provided their written informed consent prior to study initiation.
Patient-reported outcomes measures
Migraine-Specific Quality of Life questionnaire v2.1
The MSQv2.1 quantifies the effect of migraine on daily functioning in the past four weeks across three domains to assess limitations in daily social and work-related activities (Role Function–Restrictive (RFR)), how migraine attacks prevent these activities (Role Function–Preventive (RFP)) and the emotional impact of migraine attacks (Emotional Function (EF)) (29). Domain scores range from 0–100; higher scores indicate better QoL. Minimally important differences (MIDs) for within-group change in each domain are ≥10.9 points, ≥8.3 points and ≥12.2 points for RFR, RFP and EF, respectively (30). The between-group MIDs for MSQ-RFR, MSQ-RFP, and MSQ-EF are 3.2, 4.6, and 7.5 points, respectively (31). Change from baseline in MSQv2.1 domain score at weeks 4, 8 and 12 and the proportions of responders at week 12 (i.e. the proportion whose change from baseline met the MID) for each domain were assessed.
Headache Impact Test-6
The HIT-6 assesses the adverse impact of headache on individual functioning at work, home and social situations (24). HIT-6 total score ranges from 36–78 and is interpreted as headache having little or no impact (≤49), some impact (50–55), substantial impact (56–59) or severe impact (60–78), with between-group MIDs of −1.5 for EM and −2.3 for CM (32,33). Change from baseline in HIT-6 total score at weeks 4, 8 and 12 and the proportions of responders (i.e. the proportion with a ≥5-point reduction from baseline, based on the within-patient MID (32)) at week 12 were assessed.
Activity Impairment in Migraine–Diary
The AIM-D is a daily diary measure evaluating the level of difficulty experienced during the previous 24 hours in performance of daily activities (PDA) and physical impairment (PI) (25). Raw domain scores are transformed to a 100-point scale; higher scores indicate greater burden. Change from baseline in mean score for each domain across weeks 1–4, 5–8, 9–12 and 1–12 were assessed. Because the AIM-D is a relatively new instrument, no MID has been established.
Safety
Safety and tolerability were assessed based on adverse events, laboratory results, electrocardiograms, vital signs, physical exams and suicidal ideation at each study visit.
Statistical analysis
Data from the atogepant 60 mg once-daily and placebo arms of each individual trial were analyzed separately (i.e. not pooled). For ADVANCE, participants within treatment groups were stratified into migraine frequency groups: low-frequency EM (LFEM, 4 to <8 baseline MMDs) and high-frequency EM (HFEM, 8–14 baseline MMDs). Because MMDs for participants with fewer than 28 (but >20) days of baseline eDiary data were prorated to a 28-day month, some MMDs included fractions; those four or more and up to (but not including) eight were categorized as LFEM. Participants with <20 days of baseline data were not evaluated. Intent-to-treat populations for each trial included all randomized participants. Unless otherwise specified, data are presented for modified intent-to-treat (mITT) populations (all randomized participants receiving one or more atogepant dose with evaluable data during the baseline and treatment periods). To support European submissions, efficacy analyses in PROGRESS and ELEVATE were also conducted in off-treatment hypothetical estimand (OTHE) populations (all randomized participants receiving one or more dose of study intervention with evaluable data during the baseline and treatment periods). Safety analyses were conducted for safety populations of each trial (all participants receiving one or more dose of study intervention).
Baseline characteristics and safety outcomes were analyzed descriptively. Change from baseline scores in MSQv2.1 domains, HIT-6 total and AIM-D domains were analyzed using mixed-effects models for repeated measures. For responder rate analyses, proportions of participants with improvements in MSQv2.1 domain and HIT-6 total scores meeting or exceeding the MID were analyzed using generalized mixed models for repeated measures. Covariates for all models included baseline score for the corresponding PROMs, treatment group, visit, interactions of treatment group + visit and baseline score + visit and study-specific stratification factors (ADVANCE: prior exposure to preventive migraine medication [yes/no]; PROGRESS: geographic region, baseline acute medication overuse, concurrent preventive medication use [yes/no], number of prior treatment failures; ELEVATE: number of prior treatment failures [2 and >2], number of MMDs during the screening/baseline period [4 to <8 and ≥8]).
Secondary efficacy endpoints were alpha controlled at the 0.05 level using a graphical approach. They included change from baseline scores in MSQ-RFR domains at week 12 in PROGRESS and ELEVATE, HIT-6 total at week 12 in PROGRESS and ELEVATE (varies by region), and both AIM-D domains across weeks 1–12 in PROGRESS and ELEVATE (varies by region; see supplementary material, Table S1). All additional efficacy endpoint analyses (prespecified or post hoc, including all LFEM and HFEM analyses in ADVANCE) were performed at the nominal significance level without adjusting for multiplicity. p values are nominal unless otherwise stated.
Results
Participants and baseline characteristics
In ADVANCE, 910 participants were randomized; 873 had evaluable follow-up data and were included in the mITT population (placebo, n = 214; atogepant 10 mg once-daily, n = 214; atogepant 30 mg once-daily, n = 223; atogepant 60 mg once-daily, n = 222). We evaluated participants who received placebo or atogepant 60 mg once-daily, stratified by LFEM (n = 119 and 113, respectively) and HFEM (n = 95 and 108, respectively); one participant in the atogepant 60 mg once-daily group was excluded from this analysis because they had 16 baseline MMDs. The mean age was in the range 40.0–43.1 years and 85.2%–89.9% of participants were female (Table 1). The proportions of participants who were previously failed by two or more preventive treatments for placebo and atogepant, respectively, were 8.4% and 14.2% among those with LFEM and 17.9% and 17.6% among those with HFEM.
Baseline demographic, clinical and migraine characteristics in the ADVANCE trial by headache frequency (mITT population).
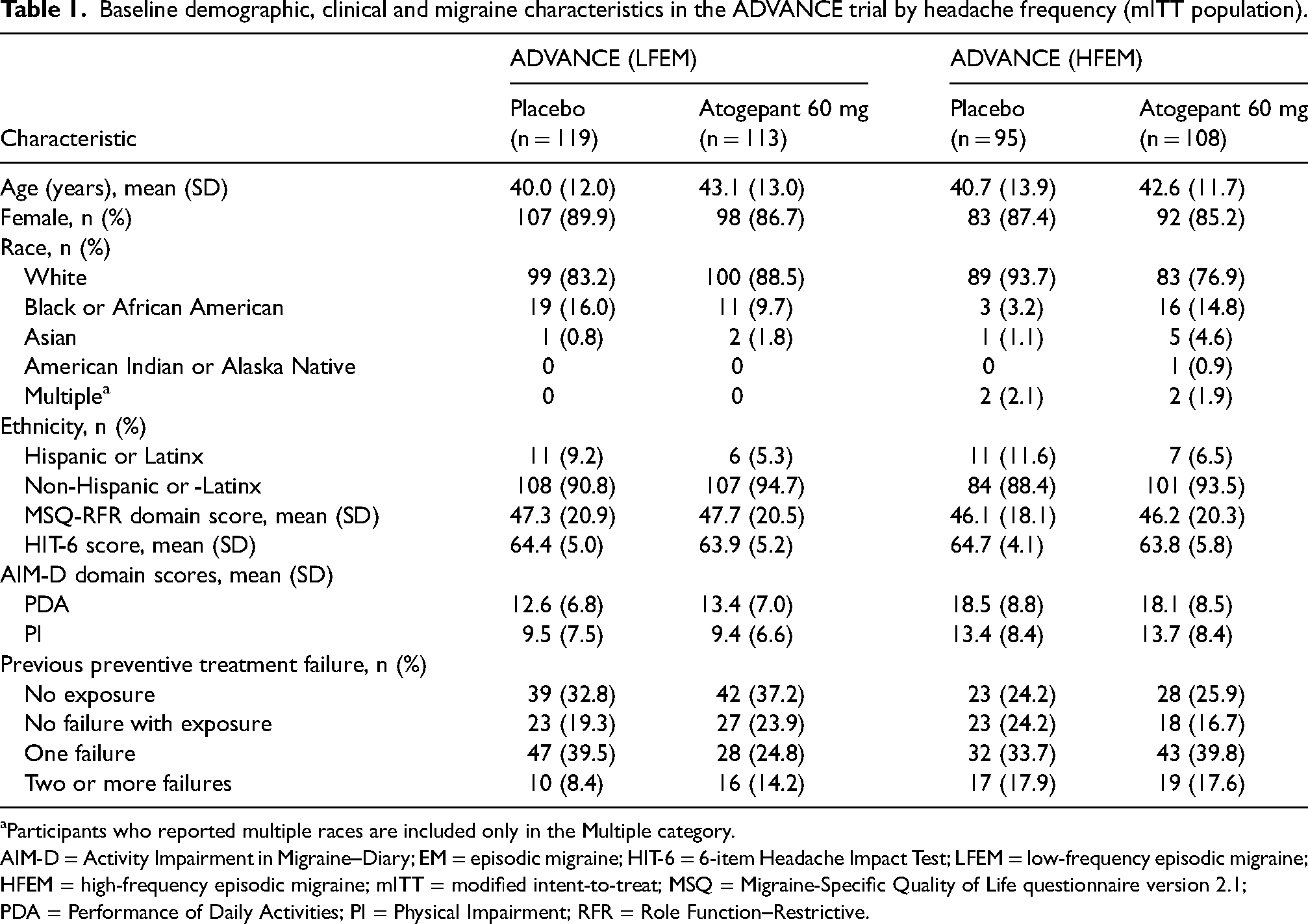
Participants who reported multiple races are included only in the Multiple category.
AIM-D = Activity Impairment in Migraine–Diary; EM = episodic migraine; HIT-6 = 6-item Headache Impact Test; LFEM = low-frequency episodic migraine; HFEM = high-frequency episodic migraine; mITT = modified intent-to-treat; MSQ = Migraine-Specific Quality of Life questionnaire version 2.1; PDA = Performance of Daily Activities; PI = Physical Impairment; RFR = Role Function–Restrictive.
In PROGRESS, 778 participants were randomized; 755 had evaluable follow-up data and were included in the mITT population (placebo, n = 246; atogepant 30 mg twice-daily, n = 253; atogepant 60 mg once-daily, n = 256). We evaluated participants who received placebo or atogepant 60 mg once-daily. The mean age was in the range 41.5–42.2 years and 86.3%–88.2% of participants were female (Table 2). The proportions of participants who were previously failed by two or more preventive treatments with different mechanisms were 37.1% for placebo and 39.7% for atogepant 60 mg once-daily.
Baseline demographic, clinical and migraine characteristics in the PROGRESS and ELEVATE trials (mITT population).
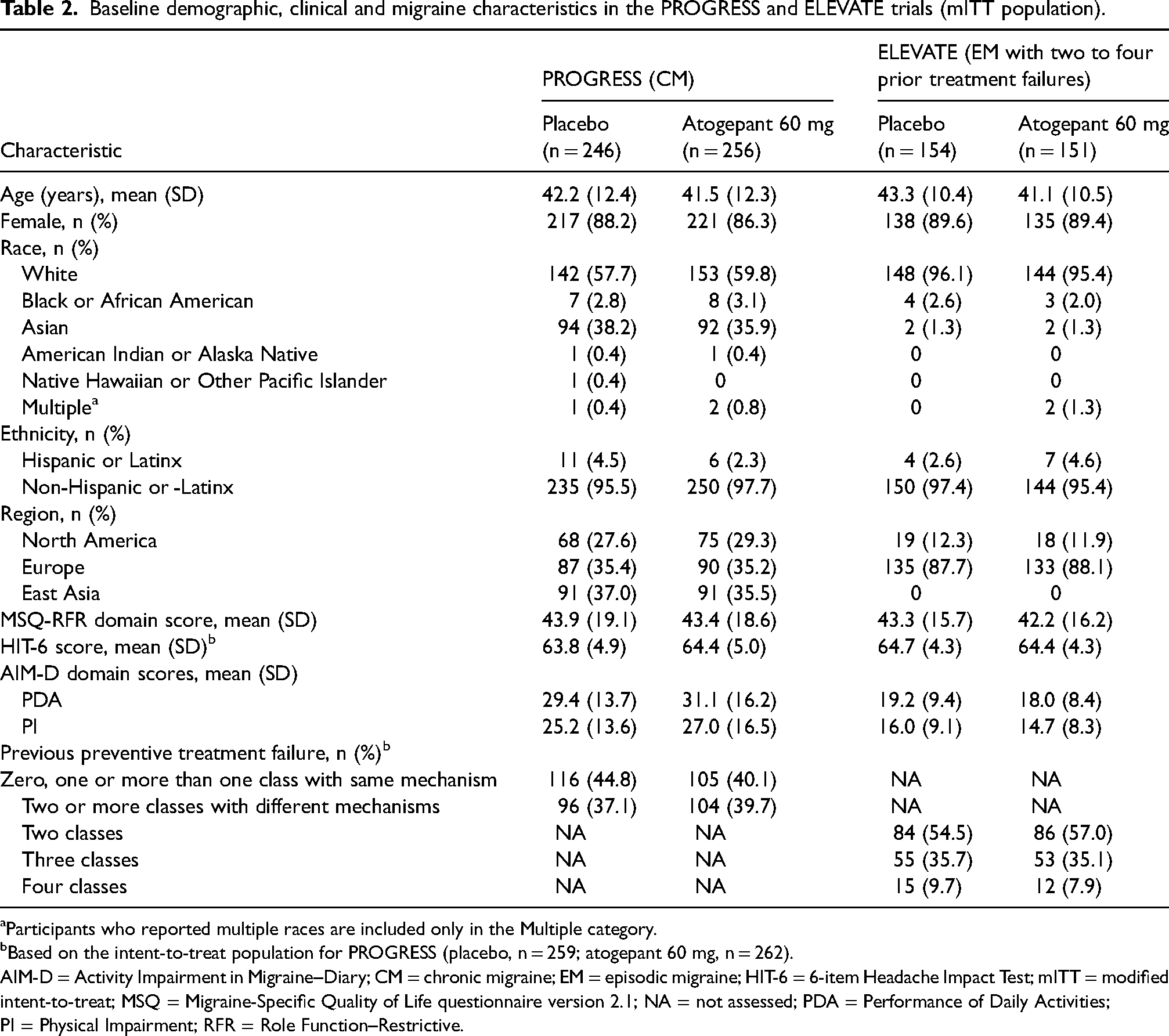
Participants who reported multiple races are included only in the Multiple category.
Based on the intent-to-treat population for PROGRESS (placebo, n = 259; atogepant 60 mg, n = 262).
AIM-D = Activity Impairment in Migraine–Diary; CM = chronic migraine; EM = episodic migraine; HIT-6 = 6-item Headache Impact Test; mITT = modified intent-to-treat; MSQ = Migraine-Specific Quality of Life questionnaire version 2.1; NA = not assessed; PDA = Performance of Daily Activities; PI = Physical Impairment; RFR = Role Function–Restrictive.
In ELEVATE, 315 participants were randomized; 305 had evaluable follow-up data and were included in the mITT population (placebo, n = 154; atogepant 60 mg once-daily, n = 151). The mean age was in the range 41.1–43.3 years and 89.4%–89.6% of participants were female (Table 2). The majority of participants in ELEVATE were previously failed by two classes of preventive treatment (placebo, 54.5%; atogepant, 57.0%).
Within all three trials, baseline scores for the PROMs were generally similar between the placebo and atogepant groups (Tables 1 and 2).
Patient-reported outcomes
Migraine-Specific Quality of Life questionnaire v2.1
In all three trials, improvements from baseline in MSQ-RFR domain scores were greater at weeks 4, 8 and 12 for participants receiving atogepant 60 mg once-daily than with placebo (all comparisons p < 0.05) (Figure 1). The least squares mean difference (LSMD) (95% confidence interval (CI)) in change from baseline MSQ-RFR domain score for atogepant compared with placebo at week 12 was 12.0 (95% CI = 6.0–18.0; p < 0.0001) among participants with LFEM in ADVANCE, 9.9 (95% CI = 3.4–16.4; p = 0.0028) among participants with HFEM in ADVANCE, 6.2 (95% CI = 2.5–9.8; alpha-controlled, p = 0.0009) in PROGRESS and 17.7 (95% CI = 13.1–22.3; alpha-controlled, p < 0.0001) in ELEVATE. MSQ-RFP and MSQ-EF domain scores also improved to a greater degree with atogepant than placebo from baseline to weeks 4, 8 and 12 (all p < 0.05) in ADVANCE (both LFEM and HFEM), ROGRESS, and ELEVATE (Figures 2 and 3).
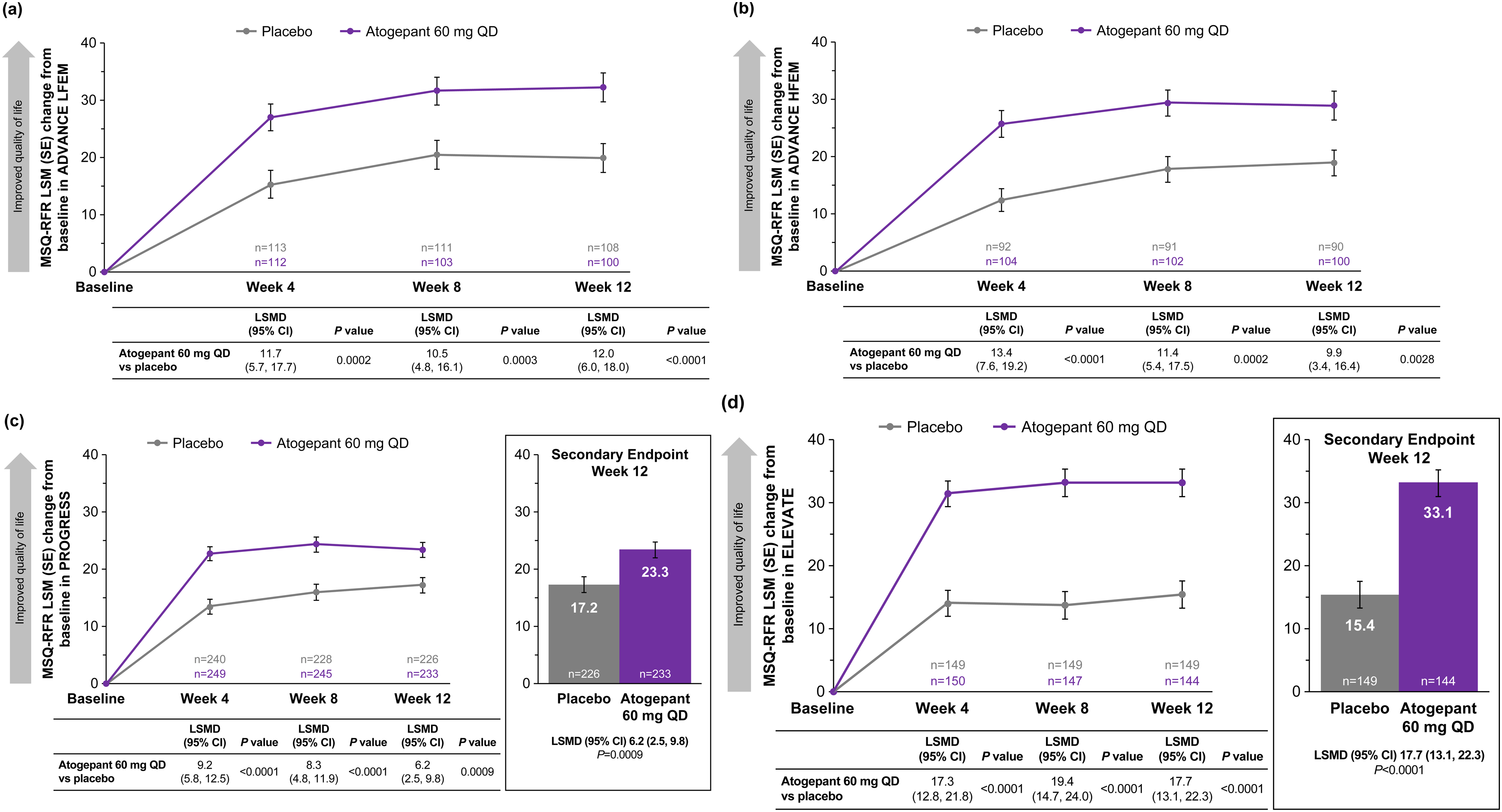
Changes from baseline in MSQ-RFR domain scores (mITT population) in (a) ADVANCE LFEM, (b) ADVANCE HFEM, (c) PROGRESS (CM) and (d) ELEVATE (EM with two to four prior treatment failures). All p values are nominal except for week 12 in PROGRESS and ELEVATE. The LSMDs exceeded the previously reported between-group MID of 3.2 points (31). CM = chronic migraine; EM = episodic migraine; HFEM = high-frequency episodic migraine; LFEM = low-frequency episodic migraine; LSM = least squares mean; LSMD = least squares mean difference; mITT = modified intent-to-treat; MSQ = Migraine-Specific Quality of Life questionnaire version 2.1; QD = once-daily; QoL = quality of life; RFR = Role Function–Restrictive domain.
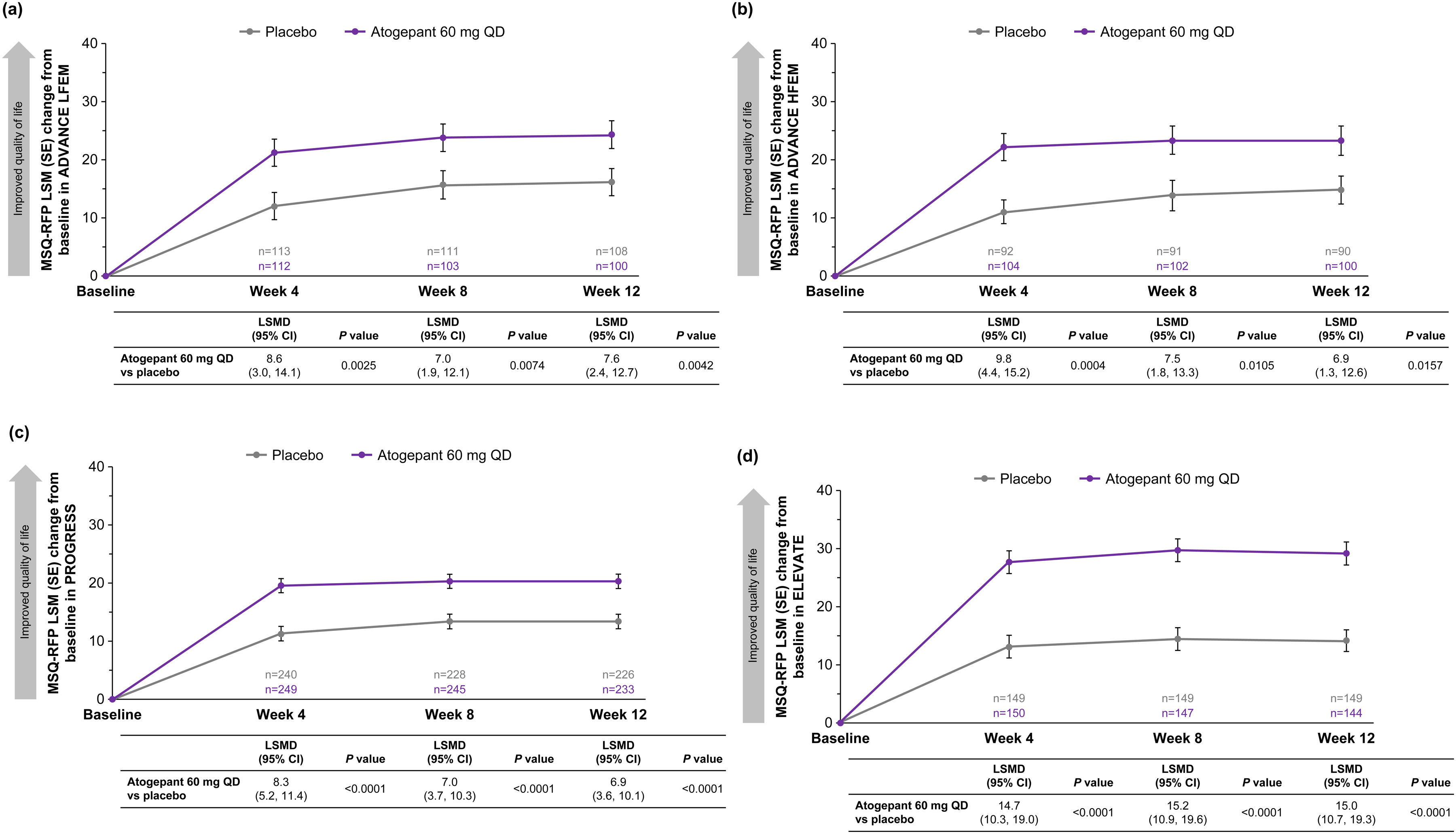
Changes from baseline in MSQ-RFP domain scores (mITT population) in (a) ADVANCE LFEM, (b) ADVANCE HFEM, (c) PROGRESS (CM) and (d) ELEVATE (EM with two to four prior treatment failures). All p values are nominal. The LSMDs exceeded the previously reported between-group MID of 4.6 points (31). CM = chronic migraine; EM = episodic migraine; HFEM = high-frequency episodic migraine; LFEM = low-frequency episodic migraine; LSM = least squares mean; LSMD = least squares mean difference; mITT = modified intent-to-treat; MSQ = Migraine-Specific Quality of Life questionnaire version 2.1; QD = once-daily; RFP = Role Function–Preventive domain.
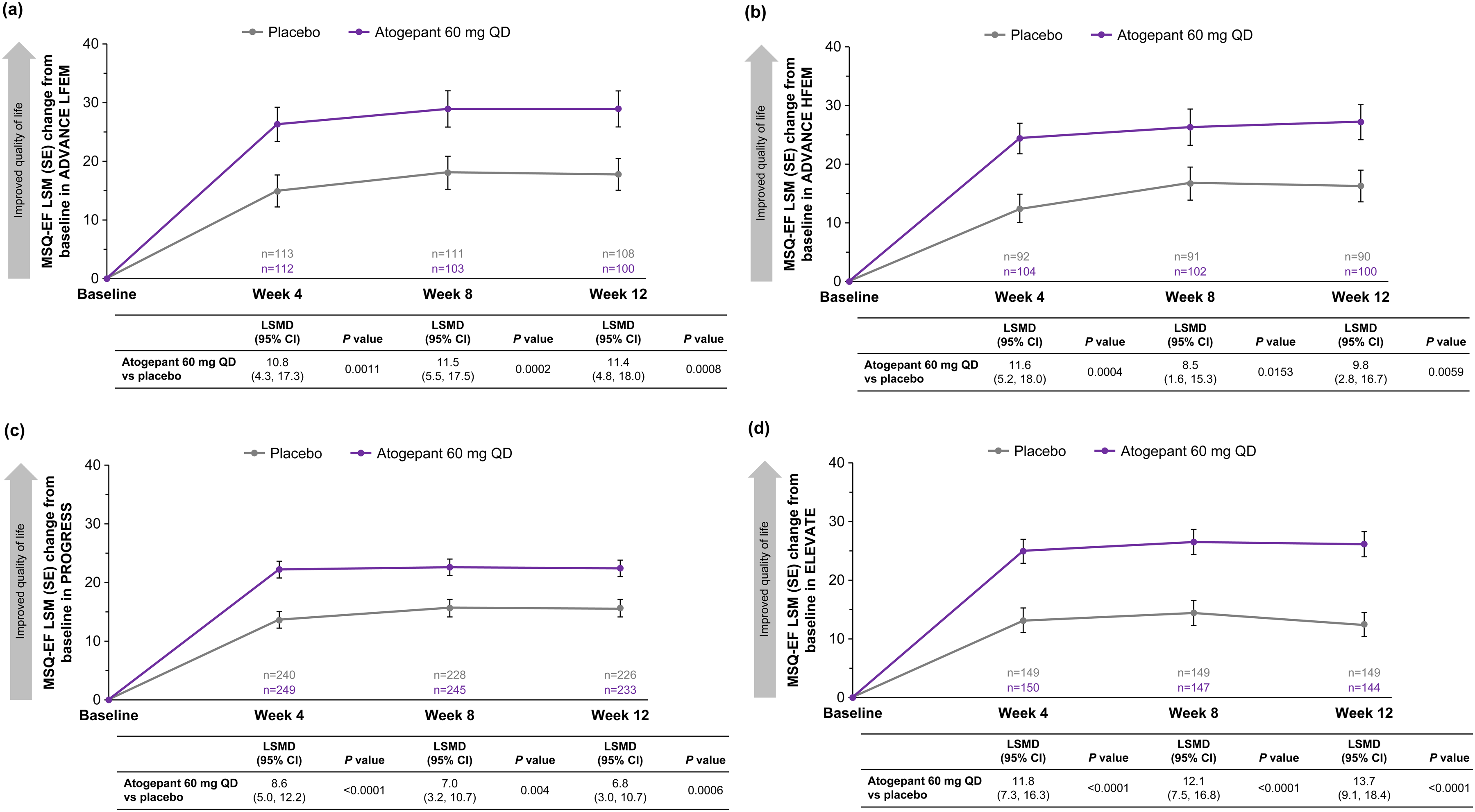
Changes from baseline in MSQ-EF domain scores (mITT population) in (a) ADVANCE LFEM, (b) ADVANCE HFEM, (c) PROGRESS (CM) and (d) ELEVATE (EM with two to four prior treatment failures). All p values are nominal. The LSMDs exceeded the previously reported between-group MID of 7.5 points, except for weeks 8 and 12 in the PROGRESS trial (31). CM = chronic migraine; EF = Emotional Function domain; EM = episodic migraine; HFEM = high-frequency episodic migraine; LFEM = low-frequency episodic migraine; LSM = least squares mean; LSMD = least squares mean difference; mITT = modified intent-to-treat; MSQ = Migraine-Specific Quality of Life questionnaire version 2.1; QD = once-daily.
Headache Impact Test–6
In all three trials, atogepant 60 mg once-daily demonstrated improvement in HIT-6 scores compared with placebo at weeks 4, 8 and 12 (all p < 0.01) (Figure 4). At week 12, LSMD (95% CI) in change from baseline in HIT-6 total score for atogepant in relation to placebo was −4.7 (95% CI = −6.7 to −2.7; p < 0.0001) among participants with LFEM (ADVANCE), −3.4 (95% CI = −5.5 to −1.2; p = 0.002) among participants with HFEM (ADVANCE), −2.8 (95% CI = −4.1 to −1.4; alpha-controlled, p < 0.0001) in PROGRESS and −6.5 (95% CI = −8.3 to −4.7; alpha-controlled, p < 0.0001) for ELEVATE. These LSMDs exceeded the previously reported between-group MIDs of −1.5 for EM and −2.3 for CM (32,33).
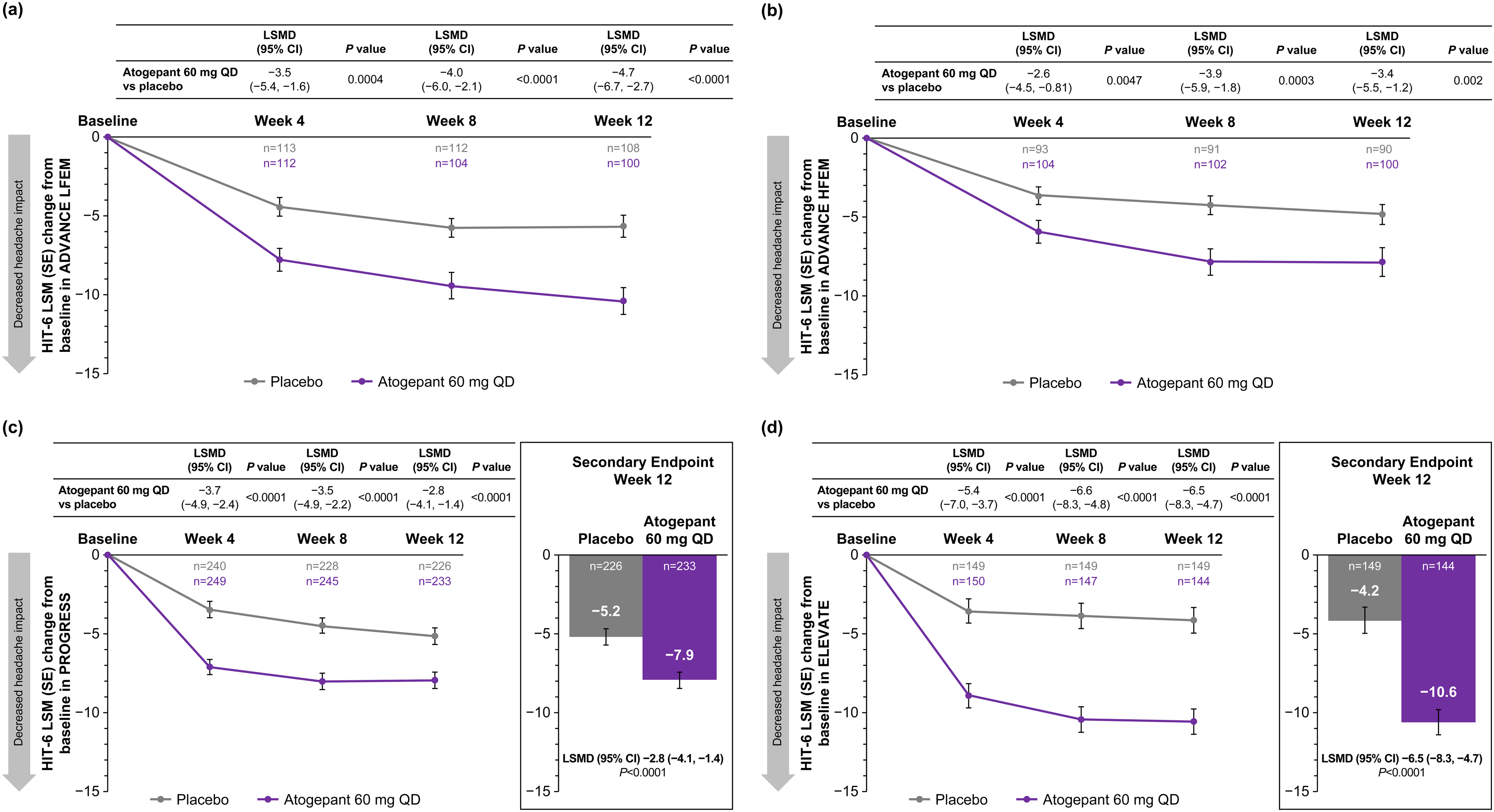
Changes from baseline in HIT-6 total scores (mITT population) in (a) ADVANCE LFEM, (b) ADVANCE HFEM, (c) PROGRESS (CM) and (d) ELEVATE (EM with two to four prior treatment failures). All p values are nominal except for week 12 in PROGRESS (secondary endpoint for the EU and Canada) and week 12 in ELEVATE (secondary endpoint for the EU). Minimally important differences are −1.5 for EM and −2.3 for CM (32,33). CM = chronic migraine; EM = episodic migraine; EU = European Union; HFEM = high-frequency episodic migraine; HIT-6 = 6-item Headache Impact Test; LFEM = low-frequency episodic migraine; LSM = least squares mean; LSMD = least squares mean difference; mITT = modified intent-to-treat; QD = once-daily.
Activity Impairment in Migraine–Diary
In all three trials, improvements from baseline in AIM-D PDA domain score were greater across the 12-week treatment period for participants receiving atogepant 60 mg once-daily compared with placebo (all p < 0.01) (Figure 5). Across weeks 1–12, the LSMD (95% CI) for change from baseline in AIM-D PDA for atogepant vs. placebo was −2.3 (95% CI = −3.9 to −0.7; p = 0.0060) among participants with LFEM (ADVANCE), −4.5 (95% CI = −6.9 to −2.2; p = 0.0002) among participants with HFEM (ADVANCE), −3.4 (95% CI = −5.2 to −1.5; alpha-controlled, p = 0.0005) in PROGRESS and −4.7 (−6.4 to −3.1; alpha-controlled, p < 0.0001) in ELEVATE. AIM-D PDA domain score improvements also were greater for atogepant vs. placebo during weeks 1–4, 5–8 and 9–12 (all p < 0.05), with the exception of weeks 5–8 among participants with LFEM in ADVANCE (p = 0.0616) and weeks 9–12 in PROGRESS (p = 0.669). Similarly, change from baseline in AIM-D PI domain scores were greater in all three trials for participants receiving atogepant compared with placebo across most time periods evaluated (all p < 0.005), with the exception of weeks 5–8 and 9–12 among participants with LFEM in ADVANCE (p = 0.2243 and p = 0.0948, respectively) and weeks 9–12 in PROGRESS (p = 0.1738) (Figure 6).
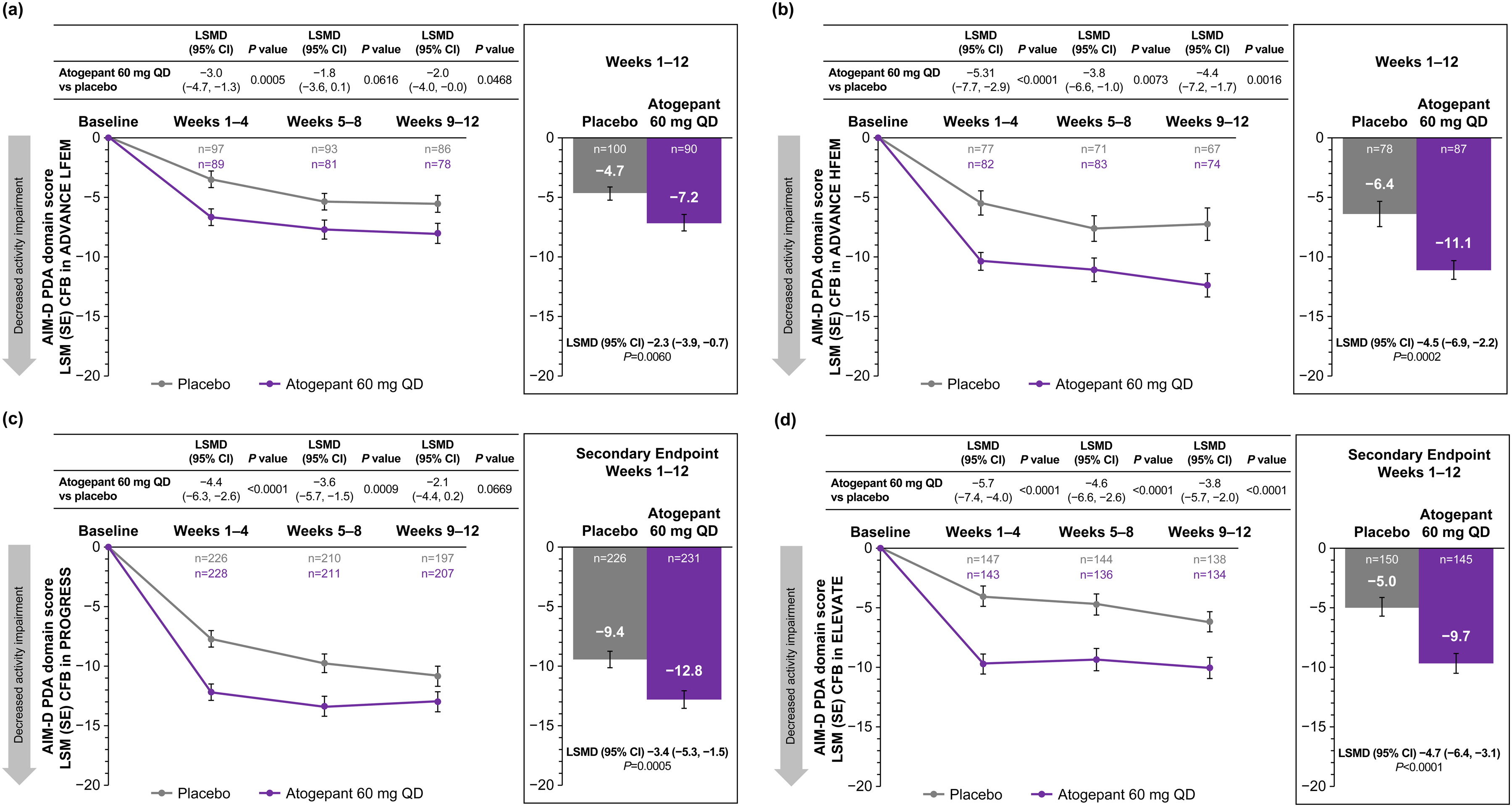
Changes from baseline in AIM-D PDA scores (mITT population) in (a) ADVANCE LFEM, (b) ADVANCE HFEM, (c) PROGRESS (CM) and (d) ELEVATE (EM with two to four prior treatment failures). All p values are nominal except for weeks 1–12 in PROGRESS (secondary endpoint for all regions except for the EU and Canada) and weeks 1–12 in ELEVATE (secondary endpoint for the US only). AIM-D = Activity Impairment in Migraine–Diary; CFB = change from baseline; CM = chronic migraine; EM = episodic migraine; EU = European Union; HFEM = high-frequency episodic migraine; LFEM = low-frequency episodic migraine; LSMD = least squares mean difference; mITT = modified intent-to-treat; PDA = Performance of Daily Activities; QD = once-daily.
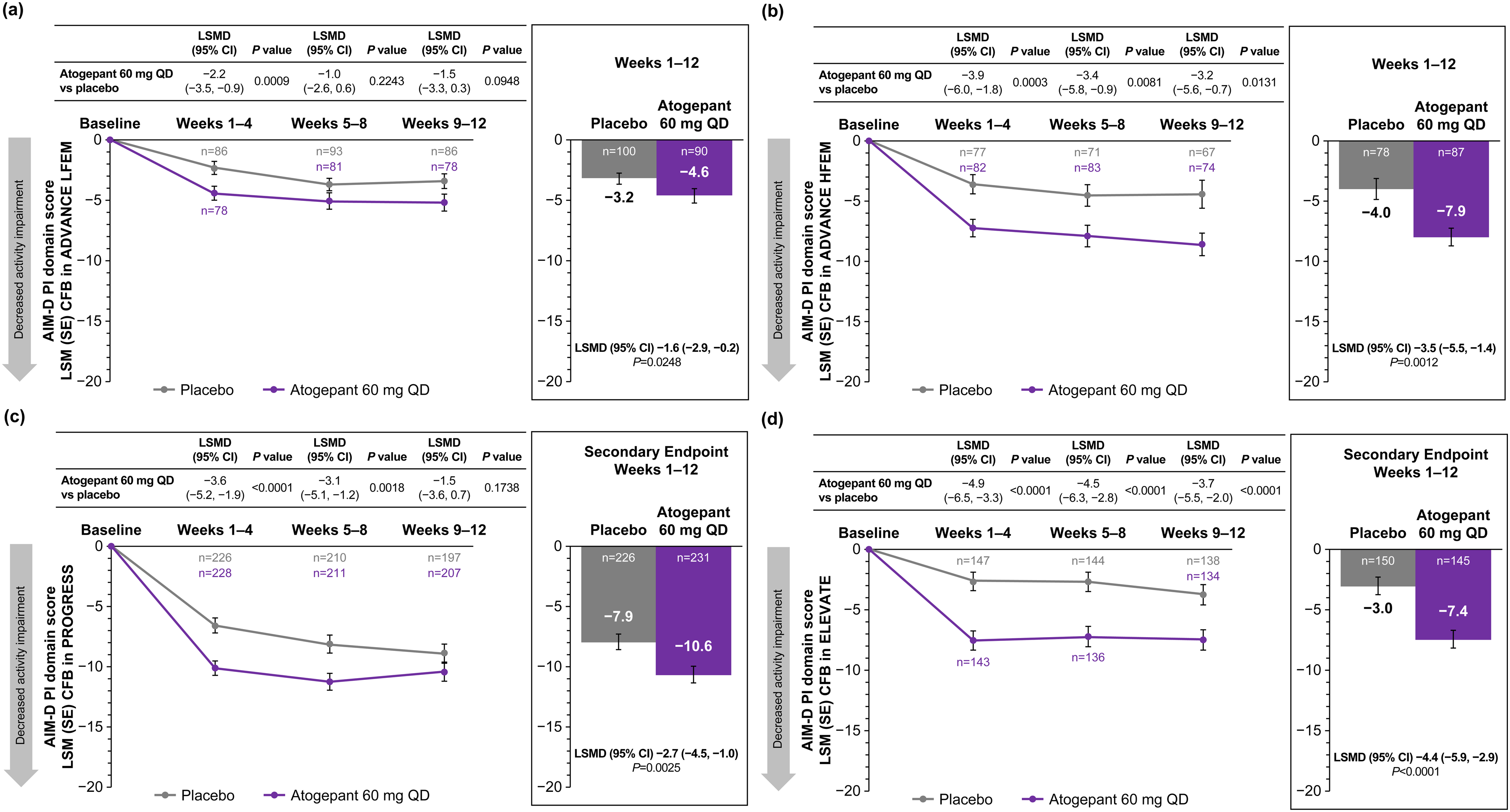
Changes from baseline in AIM-D PI scores (mITT population) in (a) ADVANCE LFEM, (b) ADVANCE HFEM, (c) PROGRESS (CM) and (d) ELEVATE (EM with two to four prior treatment failures). All p values are nominal except for weeks 1–12 in PROGRESS (secondary endpoint for all regions except for the EU and Canada) and weeks 1–12 in ELEVATE (secondary endpoint for the US only). AIM-D = Activity Impairment in Migraine–Diary; CFB = change from baseline; CM = chronic migraine; EM = episodic migraine; HFEM = high-frequency episodic migraine; LFEM = low-frequency episodic migraine; LSMD = least squares mean difference; mITT = modified intent-to-treat; PI = Physical Impairment; QD = once-daily.
Responder analyses
MSQv2.1 responder rates
In ADVANCE, proportions of responders based on MIDs at week 12 were higher for atogepant vs. placebo for all three MSQv2.1 domains among participants with LFEM (MSQ-RFR: 80.0% vs. 61.1%; odds ratio (OR) = 2.64 (95% CI = 1.4–5.1); p = 0.0038; MSQ-RFP: 76.0% vs. 61.1%; OR = 2.19 (95% CI = 1.0–4.7); p = 0.0433; MSQ-EF: 68.0% vs. 55.6%; OR = 1.97 (95% CI = 1.0–3.8); p = 0.0389) but were not different among those with HFEM (p > 0.05 for all) (Table 3). In PROGRESS, higher proportions of participants who received atogepant compared with placebo met responder criteria at week 12 for MSQ-RFP (68.7% vs. 57.5%; OR = 1.69 (95% CI = 1.1–2.6); p = 0.0141) and MSQ-EF (63.1% vs. 50.4%; OR = 1.87 (95% CI = 1.2–2.9); p = 0.0044), but not MSQ-RFR (p = 0.0791) (Table 4). In ELEVATE, higher proportions of participants who received atogepant compared with placebo were responders for all three MSQv2.1 domains at week 12 (MSQ-RFR: 81.3% vs. 51.0%; OR = 4.22 (95% CI = 2.5–7.2); p < 0.0001; MSQ-RFP: 76.4% vs. 58.4%; OR = 2.64 (95% CI = 1.5–4.6); p = 0.0007; MSQ-EF: 72.2% vs. 45.6%; OR = 3.60) (95% CI = 2.1–6.3); p < 0.0001) (Table 4).
Summary of MSQv2.1 and HIT-6 responder rates at week 12 in the ADVANCE trial by headache frequency (mITT population).
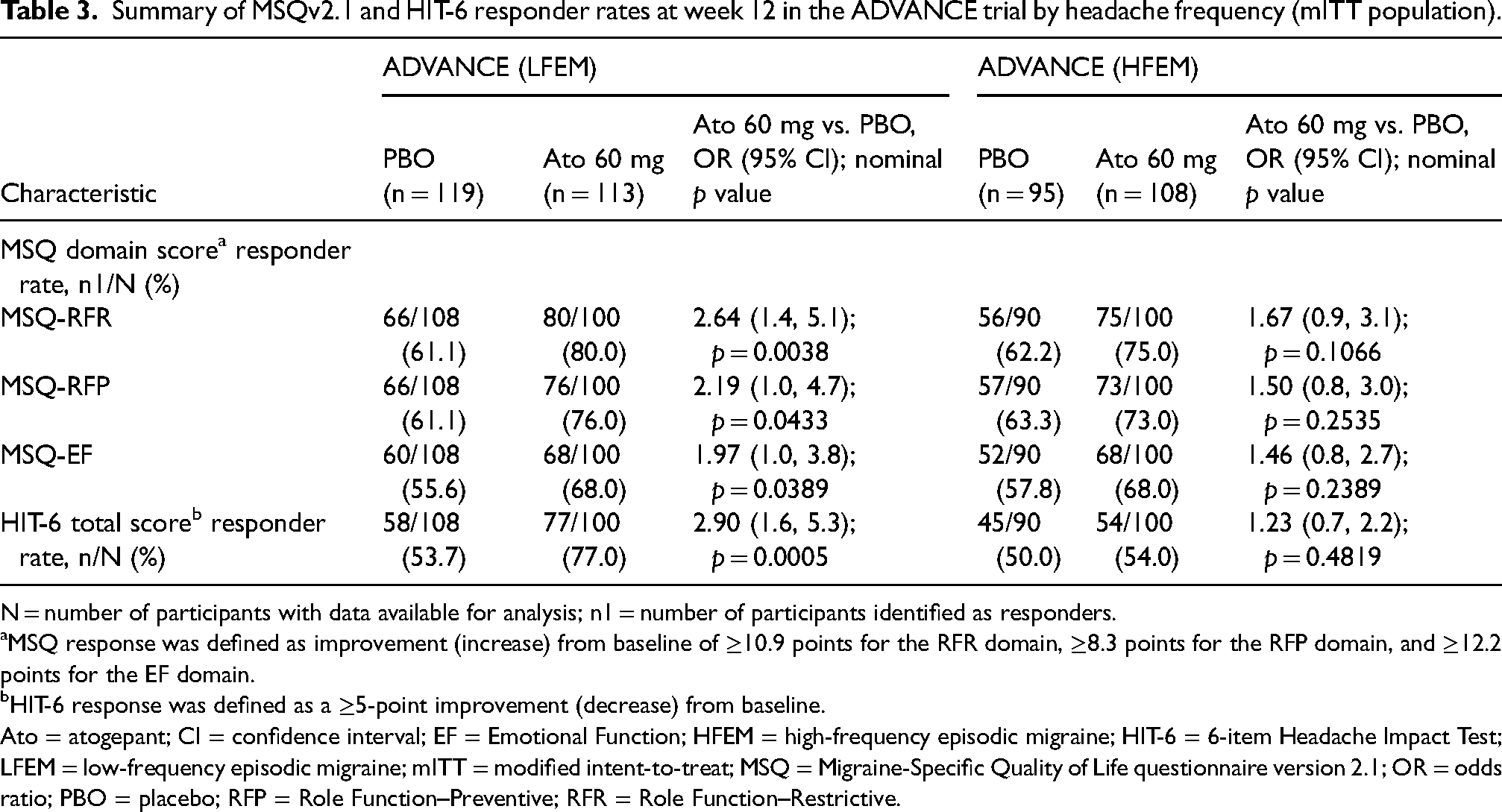
N = number of participants with data available for analysis; n1 = number of participants identified as responders.
MSQ response was defined as improvement (increase) from baseline of ≥10.9 points for the RFR domain, ≥8.3 points for the RFP domain, and ≥12.2 points for the EF domain.
HIT-6 response was defined as a ≥5-point improvement (decrease) from baseline.
Ato = atogepant; CI = confidence interval; EF = Emotional Function; HFEM = high-frequency episodic migraine; HIT-6 = 6-item Headache Impact Test; LFEM = low-frequency episodic migraine; mITT = modified intent-to-treat; MSQ = Migraine-Specific Quality of Life questionnaire version 2.1; OR = odds ratio; PBO = placebo; RFP = Role Function–Preventive; RFR = Role Function–Restrictive.
Summary of MSQv2.1 and HIT-6 responder rates at week 12 in the PROGRESS and ELEVATE trials (mITT population).
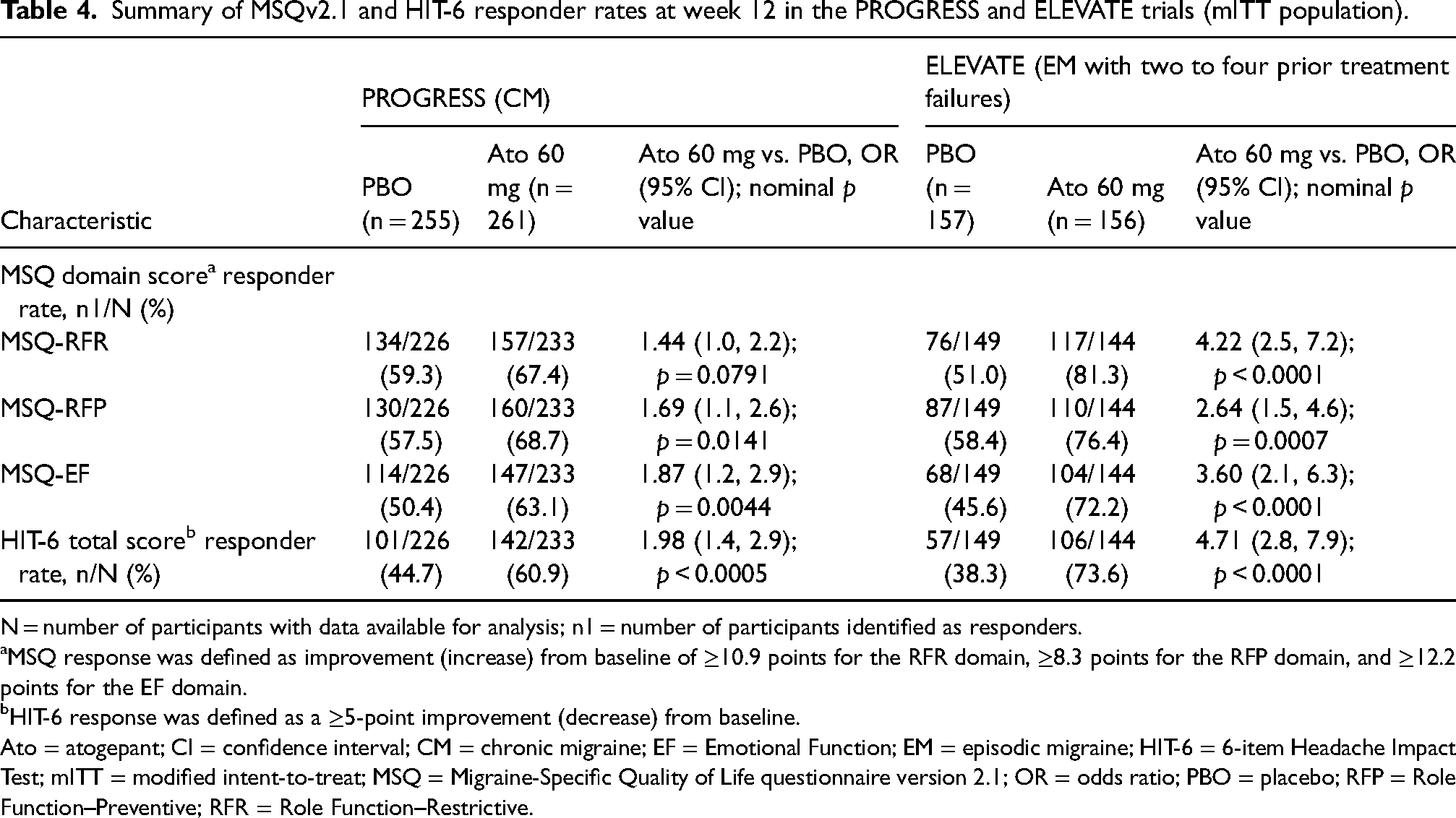
N = number of participants with data available for analysis; n1 = number of participants identified as responders.
MSQ response was defined as improvement (increase) from baseline of ≥10.9 points for the RFR domain, ≥8.3 points for the RFP domain, and ≥12.2 points for the EF domain.
HIT-6 response was defined as a ≥5-point improvement (decrease) from baseline.
Ato = atogepant; CI = confidence interval; CM = chronic migraine; EF = Emotional Function; EM = episodic migraine; HIT-6 = 6-item Headache Impact Test; mITT = modified intent-to-treat; MSQ = Migraine-Specific Quality of Life questionnaire version 2.1; OR = odds ratio; PBO = placebo; RFP = Role Function–Preventive; RFR = Role Function–Restrictive.
HIT-6 responder rates
In ADVANCE, the HIT-6 responder rate based on MIDs at week 12 was higher for atogepant vs. placebo among participants with LFEM (77.0% vs. 53.7%; OR = 2.9 (95% CI = 1.6–5.3); p = 0.0005) but not among those with HFEM (p = 0.4819) (Table 3). Greater proportions of atogepant- vs. placebo-treated participants were considered HIT-6 responders at week 12 in both PROGRESS (60.9% vs. 44.7%, OR = 1.98 (95% CI = 1.4–2.9), p = 0.0005) and ELEVATE (73.6% vs. 38.3%, OR = 4.71 (95% CI = 2.8–7.9), p < 0.0001) (Table 4).
Safety
Detailed safety outcomes for all three trials have been previously reported (16,18,19). Treatment-emergent adverse events occurring in ≥2% of participants (safety population) in ADVANCE, PROGRESS and ELEVATE for atogepant 60 mg once-daily vs. placebo included constipation (6.9% vs. 0.5%, 10.0% vs. 3.1%, and 10.3% vs. 2.5%, respectively) and nausea (6.1% vs. 1.8%, 9.6% vs. 3.5% and 7.1% vs. 3.2%, respectively). Safety outcomes were consistent with the known safety profile of atogepant in all three trials (see supplementary material, Table S2).
Discussion
In these analyses of ADVANCE, PROGRESS and ELEVATE trial data, atogepant 60 mg once-daily showed greater improvements from baseline during 12 weeks of treatment in PROMs assessing QoL (MSQv2.1), daily functioning (AIM-D) and migraine-related impairment (HIT-6) across the spectrum of people with migraine. Together, the trials presented here include not only a broad EM population (LFEM and HFEM), but also a CM population and a population of patients with EM previously failed by two to four classes of oral preventive treatment, representing individuals with substantial unmet treatment needs. Participants who received atogepant consistently showed improvements from baseline compared with placebo in all three MSQv2.1 domain scores at week 12, HIT-6 total score at week 12, and AIM-D PDA and PI domain scores across the 12-week treatment period. In PROGRESS and ELEVATE, improvements on these PROMs in the OTHE population (see supplementary material, Table S3) were similar to those presented for the mITT population.
Improvements in the same PROMs assessed here have been previously reported after atogepant treatment for broad EM populations in ADVANCE (27) and a long-term safety trial (28). In the long-term trial, significant improvements in all three MSQv2.1 domain scores, HIT-6 total score and both AIM-D domain scores for atogepant-treated participants with EM were sustained throughout 52 weeks (28). The present analysis expands on earlier findings by demonstrating that atogepant also improved QoL, headache-related impairment and daily functioning among people with EM failed by prior preventive treatment, as well as those with HFEM, who are at risk of transformation to CM in the absence of effective treatment (34). These PROM data are an important complement to the efficacy and safety data underpinning the American Headache Society's endorsement of atogepant and other CGRP-targeted agents as first-line preventive migraine treatments (35) and reflect the rapid improvement in well-being for patients who respond to atogepant.
These analyses demonstrated that MSQv2.1 and HIT-6 responder rates favored atogepant in the overall population, but did not reach statistical significance for atogepant vs. placebo among those with HFEM. A likely explanation for these findings is the fact that LFEM/HFEM subgroup analyses were not adequately powered in the ADVANCE trial. In addition, QoL improvement with atogepant vs. placebo was numerically greatest in participants with EM not responding to 2–4 prior preventive treatments (ELEVATE), followed by LFEM/HFEM (ADVANCE) and then CM (PROGRESS). For example, the change from baseline in MSQ-RFR score at 12 weeks for atogepant vs. placebo was 17.7 in ELEVATE, 12.0 and 9.9 among participants with LFEM and HFEM (ADVANCE), respectively, and 6.2 in PROGRESS. It is possible that differences in QoL improvements for atogepant users relative to placebo would narrow between EM and CM over an observation period >12 weeks. Nevertheless, these findings both highlight the robust treatment response observed among participants who had previously been failed by multiple preventive treatments and corroborate evidence that individuals with lower headache frequencies respond favorably to preventive migraine treatment (36). Treating migraine early in the disease course, when headaches are less frequent, may therefore provide an opportunity to improve outcomes and prevent disease progression (34,36).
The relevance of PROMs is of growing interest and has been highlighted in collaborative research that situates PROMs as the future of migraine trials (37). An international collaborative of migraine patients and specialists developed a core outcome set for migraine (COSMIG) noting that preventive trials should include PROMs, including migraine-specific measures of pain and QoL (38). In addition, migraine frequency and PROMs may not always be strongly correlated. For instance, a randomized trial of mindfulness-based stress reduction among participants with migraine demonstrated no improvement in migraine frequency, but showed improvements in disability, QoL and depression (39), corroborating the value of PROMs and elevating the importance of patient-centric data that reflect daily and social well-being in migraine trials. To further examine the relationship among PROMs, migraine frequency and treatment response history across atogepant trials (5,6,8), a pooled analysis of data from patients with LFEM, HFEM, CM, and varying numbers of prior treatment failures is being considered.
The strengths of these analyses include using rigorously validated PROMs in a large, varied group of people with migraine that reflects the populations encountered in clinical practice. Use of the MSQv2.1 and HIT-6 is recommended by the National Institute of Neurological Disorders and Stroke, and the AIM-D is a novel measure for assessing activity impairment (a migraine impact of great importance to patients) that was developed in alignment with guidance from the US Food and Drug Administration (25,40). A potential limitation of these analyses is that the treatment period for each trial was only 12 weeks. Because PROMs may require longer periods to show improvement, studies with longer durations are being conducted. In addition, the four-week recall periods of both the MSQv2.1 and HIT-6 may introduce the possibility of recall bias relative to a daily assessment scale such as AIM-D.
Conclusions
Among individuals experiencing LFEM, HFEM and CM, and those with EM previously failed by two to four classes of oral preventive treatment, atogepant 60 mg once-daily consistently demonstrated meaningful improvements in PROMs after 12 weeks that reflect improved daily functioning and QoL, and reduced migraine-related physical impairment.
Atogepant 60 mg once-daily improved quality of life, daily functioning, and headache-related impairment among participants with migraine, including those with low- or high-frequency EM, CM or multiple preventive treatment failures These results demonstrate the value of preventive treatment with atogepant for reducing migraine-attributed impacts on multiple aspects of daily life, health and well-being across the spectrum of people with migraine
Supplemental Material
sj-docx-1-cep-10.1177_03331024241300305 - Supplemental material for Effect of preventive treatment with atogepant on quality of life, daily functioning, and headache impact across the spectrum of migraine: Findings from three double-blind, randomized, phase 3 trials
Supplemental material, sj-docx-1-cep-10.1177_03331024241300305 for Effect of preventive treatment with atogepant on quality of life, daily functioning, and headache impact across the spectrum of migraine: Findings from three double-blind, randomized, phase 3 trials by Christopher Gottschalk, Pranav Gandhi, Patricia Pozo-Rosich, Suzanne Christie, Cristina Tassorelli, Jonathan Stokes, Yingyi Liu, Lei Luo, Krisztian Nagy, Joel M. Trugman and Richard B. Lipton in Cephalalgia
Footnotes
Acknowledgments
We thank all the participants of these studies and the clinical personnel who managed the studies. Allergan (now AbbVie) funded this study and contributed to the study design, the collection, analysis and interpretation of data, as well as the review and approval of the final version for publication. All authors had access to relevant data and participated in the drafting, review and approval of this publication. No honoraria or payments were made for authorship. Medical writing support was provided to the authors by Peloton Advantage, LLC, an OPEN Health company, and was funded by AbbVie.
Declaration of conflicting interests
Christopher Gottschalk, MD, FAHS, has received personal fees as a consultant or member of an advisory board from AbbVie, Lundbeck, Pfizer, Spherix Global Insights, and Theranica; and is a trustee of the Headache Cooperative of the Northeast (HCNE); and does not hold any stock or options. Patricia Pozo-Rosich, MD, PhD, has received, in the last 3 years, personal fees for advisory boards and speaker panels from AbbVie, Amgen, Dr. Reddy’s, Eli Lilly, Lundbeck, Medscape, Novartis, Pfizer, and Teva, and for serving on the scientific advisory board of Lilly Foundation Spain and Gadea Science Foundation; is the principal investigator for clinical trials sponsored by AbbVie, Amgen, Biohaven, Eli Lilly, Lundbeck, Novartis, Pfizer, and Teva; has received (with her group) grants from AbbVie, ERANet Neuron, Instituto de Salud Carlos III, International Headache Society, Novartis, FEDER RIS3CAT, and Teva; serves as an associate editor for Cephalalgia, Headache, Neurologia, The Journal of Headache and Pain, and Revista de Neurologia; is the Honorary Secretary of the International Headache Society; and does not hold any stock or options. Suzanne Christie, MD, has received compensation for consulting and/or honoraria from Allergan (now AbbVie), Lilly, Novartis, and Teva; has participated on advisory boards for AbbVie, Lundbeck, and Miravo; and has received research support from Novartis. Cristina Tassorelli, MD, PhD, has participated in advisory boards for AbbVie, Dompé, Eli Lilly, Ipsen, Lundbeck, Pfizer, and Teva; has lectured at symposia and is a principal investigator or collaborator in clinical trials sponsored by AbbVie, Eli Lilly, Ipsen, Lundbeck, Pfizer, and Teva; has received research grants from the European Commission, Italian Ministry of Health, Italian Ministry of University, Migraine Research Foundation, and Italian Multiple Sclerosis Foundation; and serves as an associate editor for Cephalalgia. Jonathan Stokes, MBA; Krisztian Nagy, MD; Joel M. Trugman, MD; Lei Luo, MPH, BS; Yingyi Liu, PhD; and Pranav Gandhi, PhD, are employees of AbbVie and may hold AbbVie stock. Richard B. Lipton, MD, has received research support from the National Headache Foundation, the National Institutes of Health, and the US Food and Drug Administration; serves as consultant for, advisory board member of, or has received honoraria or research support from AbbVie/Allergan, Amgen, Biohaven, Dr. Reddy’s Laboratories (Promius), electroCore, Eli Lilly, GlaxoSmithKline, Lundbeck, Merck, Novartis, Teva, Vector, and Vedanta Research; receives royalties from Wolff’s Headache, 8th edition (Oxford University Press, 2009), and Informa; and holds stock/options in Axon, Biohaven, CoolTech, and Manistee.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and publication of this article: This study was sponsored and funded by Allergan (now AbbVie). Medical writing support was provided to the authors by Peloton Advantage, LLC, an OPEN Health company, and was funded by AbbVie.
Author contributions
All authors were responsible for the study design. LL and YL were responsible for collection and assembly of data. LL and YL were responsible for data analysis. All authors were responsible for data interpretation. All authors were responsible for manuscript preparation. All authors were responsible for manuscript review and revisions. All authors approved the final version of the manuscript submitted for publication.
Data availability
AbbVie is committed to responsible data sharing regarding the clinical trials we sponsor. This includes access to anonymized, individual and trial-level data (analysis data sets), as well as other information (e.g. protocols, clinical study reports or analysis plans), as long as the trials are not part of an ongoing or planned regulatory submission. This includes requests for clinical trial data for unlicensed products and indications. These clinical trial data can be requested by any qualified researchers who engage in rigorous, independent, scientific research, and will be provided following review and approval of a research proposal, Statistical Analysis Plan (SAP), and execution of a Data Sharing Agreement (DSA). Data requests can be submitted at any time after approval in the USA and Europe and after acceptance of this manuscript for publication. The data will be accessible for 12 months, with possible extensions considered. For more information on the process or to submit a request, see: ![]() .
.
Ethical statement
These trials complied with the International Council on Harmonisation Good Clinical Practice Guideline and the Declaration of Helsinki. Investigators obtained approval from an independent ethics committee or local institutional review board. Participants provided written informed consent prior to study initiation.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
