Abstract
Background
The discovery of calcitonin gene-related peptide (CGRP) as a key player in migraine pathophysiology has revolutionized the approach to preventive treatment. Atogepant, an oral small-molecule CGRP receptor antagonist, has shown promising efficacy in randomized controlled trials (RCTs) for both episodic and chronic migraine. However, real-world evidence, particularly in individuals with chronic migraine and multiple preventive treatment failures, remains limited. This study is aimed to evaluate the effectiveness, safety, and tolerability of daily atogepant 60 mg in a homogeneous cohort of resistant individuals with chronic migraine over a 24-week period to extend the short-term observation assessed in previous real-world studies.
Methods
In the present real-world, prospective, monocentric study, a total of 100 participants (93% female; mean ± SD, age 43 ± 11 years) with chronic migraine with at least three previous treatment failures without medication overuse headache were consecutively recruited and received atogepant 60 mg daily for six months. All participants had failed a median of six previous preventive treatments, including CGRP-monoclonal antibodies (mAbs) (68%) and onabotulinumtoxin-A (BoNT-A) (14%). Primary outcomes included change in monthly migraine days (MMDs) and greater than 50% responder rate at 12 and 24 weeks. Secondary outcomes included changes in monthly headache days (MHDs), acute medication intake (MAMI), headache impact (Headache Impact Test (HIT-6)), anxiety and depression (Hospital Anxiety and Depression Scale (HADS)) and patient satisfaction (Patient's Global Impression of Change (PGIC)), change in MMDs, demographic and clinical features associated with greater than 50% responder rate, as well as effectiveness in individuals with previous CGRP-mAbs failure. Treatment-emergent adverse events (TEAEs) were also recorded.
Results
At weeks 12 and 24, MMDs were reduced by 5.6 and 7.1 days from baseline, respectively (p < 0.001), while 45% and 53% of participants achieved a ≥ 50% reduction in MMDs. Significant improvements were also seen in MHDs (−8.1 days), MAMI (−5.1 days) and HIT-6 scores (−6.2 points). Conversion from chronic to episodic migraine occurred in 60% of participants. PGIC results showed that 69% of participants reported feeling “much” or “very much” better. Logistic regression identified higher socioeconomic status (odds ratio = 2.87) as a positive predictor and previous CGRP-mAb failure (odds ratio = 0.38) as a negative predictor of treatment response. Nevertheless, among individuals with more than one CGRP-mAb failure, 47% achieved a ≥50% reduction in MMDs. TEAEs were reported by 53% of participants, with constipation (28%) and fatigue (16%) being the most common.
Conclusions
Atogepant 60 mg daily demonstrated meaningful clinical benefit and good tolerability in real-world individuals with treatment-resistant chronic migraine over a 24-week period. These findings extend data from RCTs and real-world studies limited to 12-week period of observation, supporting atogepant as an effective option even in individuals with prior CGRP-mAb failure.
This is a visual representation of the abstract.
Introduction
The therapeutic approach to chronic migraine has traditionally relied on preventive treatment by using non-specific oral medications, the so-called “repositioning drugs” that, until a few years ago, were the only available pharmacological options (1-2). The identification of the pivotal role played by calcitonin gene-related peptide (CGRP) in migraine pathophysiology has, in recent years, revolutionized the therapeutic landscape of this disabling neurological disorder (3,4). By targeting the CGRP pathway, either the ligand or its receptor, through monoclonal antibodies (CGRP-mAbs), remarkable clinical outcomes have been achieved in terms of efficacy and tolerability, as demonstrated in both randomized controlled trials (RCTs) and real-world studies (5,6). More recently, the therapeutic arsenal available to clinicians has been further expanded with the introduction of gepants—small-molecules which antagonize CGRP receptors, capable of preventing the meningeal vasodilation and neurogenic inflammation believed to underlie migraine attacks (7). Among the new-generation gepants, both atogepant and rimegepant have been approved for the preventive treatment of migraine; however, only atogepant has demonstrated efficacy in individuals with two to four prior preventive treatment failures. Based on their efficacy, safety and tolerability, multiple expert consensus statements have recommended CGRP-targeting therapies as first-line options for migraine prevention (8,9), even though current insurance coverage in most countries does not support their use as a first-line treatment. Atogepant, administered orally at daily doses of 10 to 60 mg, received approval from the US Food and Drug Administration (FDA) in 2021 for the preventive treatment of episodic migraine, based on robust data from a phase 2/3 trial, the pivotal phase 3 ADVANCE trial and a 52-week open-label extension study (10-12). Based on these foregoing, atogepant became the first oral gepant approved by the European Medicines Agency for the prevention of both episodic and chronic migraine. However, evidence from real-world clinical practice remains limited, restricting our understanding of the drug's performance in routine care settings across broader and more heterogeneous patient populations. Such data are essential to enhance the generalizability of RCTs findings and to achieve a more pragmatic approach to migraine management.
To date, only two real-world studies have evaluated the effectiveness, safety and tolerability of atogepant 60 mg in individuals with migraine and multiple prior preventive treatment failures (13,14). Nonetheless, both investigations were constrained by a short observation period (i.e. 12 weeks) and by the heterogeneity of their cohorts, which included individuals with high-frequency episodic migraine (8–14 migraine days per month), as well as those with chronic migraine.
The present real-world study therefore aims to evaluate the effectiveness, safety and tolerability of atogepant as a preventive treatment in a homogeneous cohort of individuals with chronic migraine and multiple previous preventive treatment failures over a 24-week observation period.
Methods
Study design and participants
This is an observational, prospective, non-randomized study designed to evaluate the efficacy and safety of daily atogepant 60 mg as a preventive treatment in individuals with resistant chronic migraine and a history of more than three prior preventive treatment failures. One hundred and eleven participants aged 18 years or older with a diagnosis of chronic migraine without medication overuse headache were consecutively recruited from the migraine population being referred to the Headache Center at NeuroClinic Norway. According to the International Classification of Headache Disorders, 3rd edition (ICHD-3), chronic migraine was defined as a headache occurring on 15 or more days per month for more than three months, with migraine features present on at least eight of those days each month (15). A semi-structured clinical interview, performed by a neurologist specialized in headache disorders (Dr C. Sundal), was used to establish the diagnosis. Participants were eligible for inclusion if they had failed at least three prior preventive treatments due to lack of efficacy or intolerable side effects, with no upper limit on the number of previous treatment failures. Treatment failure was defined as “lack of efficacy” in terms of no meaningful improvement in the frequency of headaches (<50% reduction in MMDs) after the administration of standard of care treatments (the so-called repositioning drugs) for more than weeks at the appropriate dose, and/or two cycles (24 weeks) of BoNT-A and/or at least three months of subcutaneous CGRP-mAbs according to the European Headache Federation criteria (16). Tolerability failure was defined as documented discontinuation due to adverse events at any time of the treatment. All participants received daily atogepant 60 mg and were followed-up for six months.
Other oral migraine preventive medications (alone or in combination) with a stable dose for at least three months were allowed concomitantly to atogepant administration.
Collected variables
At baseline (T0) data aimed at assessing demographic data such as (i) gender, (ii) age, (iii) body mass index (BMI) and (iv) socioeconomic status (i.e. low, medium and high), as well as clinical information of migraine history such as (a) familiarity for migraine, (b) age at migraine onset, (c) age at migraine diagnosis, (d) age at migraine chronification onset, (e) presence of aura phenomenon, (f) comorbidities (e.g. depression, anxiety, fibromyalgia, attention deficit hyperactivity disorder (ADHD), endometriosis and cardiovascular diseases) and (g) previous treatment failures among topiramate, amitriptyline, venlafaxine, candesartan or lisinopril, beta-blockers, flunarizine, BoNT-A and CGRP-mAbs. Participants’ comorbidities, such as anxiety and depression, were considered based on prior clinical diagnoses. Cardiovascular diseases were defined as previous diagnosis of angina, myocardial infarction or heart failure, as established by a cardiologist. Furthermore, at baseline (T0) and at the end of the third (T1) and the sixth month (T2) of atogepant preventive treatment, the following parameters were collected (i) headache frequency, considering both MMDs and monthly headache days (MHDs), (ii) average headache pain intensity experienced during the attacks (i.e., mild, moderate and severe), (iii) monthly days with acute medication intake (MAMI), (iv) migraine related impact by Headache Impact Test (HIT-6) (17-18), and (vi) concomitant migraine preventive treatment. Participants self-reported pain intensity based on their own perception, categorizing each headache attack as mild, moderate, or severe, in the absence of a standardized scale. The Patient's Global Impression of Change (PGIC) (19) was administered at the end of the third (T1) and of the sixth month (T2) of atogepant preventive treatment. All participants were instructed to fill in an electronic diary (https://nordicbraintech.com/products/braintwin). A “headache day” was defined as any day on which the patient reported a headache, including those with tension-type features. A “migraine day” was defined as any day with a headache exhibiting migraine characteristics or responding to triptan administration. During the six months of atogepant preventive treatment, all treatment emergent adverse events (TEAEs) were recorded and used as safety outcome. The dataset was complete, and no missing data were observed during data collection or analysis.
The protocol was reviewed and approved by the Ethical Committee of Norway, REK #604900. Each patient gave a free, informed consent for participation in the study and the analysis and publication of the protocol data.
Primary outcomes
The co-primary effectiveness outcomes of the present real-word observation (20) were:
Change in MMDs from baseline to week 12 and to week 24. Percentage of MMDs “responders” (participants who achieved at least 50% reduction from their individual baseline in MMDs) after 12 and 24 weeks of preventive treatment with atogepant 60 mg.
Secondary outcomes
Secondary outcomes included the evaluation from baseline to week 12 and to week 24 of:
Change in MHDs. Change in MAMI. Changes in migraine related impact (evaluated by HIT-6). Percentage of MHDs “responders” (participants who achieved at least 50% reduction from their individual baseline in MHDs) after 12 and 24 weeks of preventive treatment with atogepant 60 mg. Percentage of MMDs super-responders (participants who achieved at least 75% reduction from their individual baseline in MMDs). Percentage of participants converting from chronic migraine to episodic migraine. Changes in number of days with mild, moderate and severe headache. Changes in anxious and depressive symptoms (evaluated by HADS). Patient satisfaction (investigated by PGIC questionnaire). Baseline clinical variables associated with MMDs 50% responder rate among demographic data (age, gender, BMI, socioeconomic status) and clinical parameters of disease severity such as presence of aura phenomenon, previous CGRP-mAbs failure. Percentage of TEAEs.
Among participants with chronic migraine and previous CGRP-mAbs failure we explored:
xii. Reduction in MMDs, MHDs and MAMI. xiii. Percentage of responders (participants who achieved at least 50% reduction from their individual baseline in MMDs).
Among participants with chronic migraine and previous BoNT-A failure, we explored:
xiv. Reduction in MMDs. xv. Percentage of responders (participants who achieved at least 50% reduction from their individual baseline in MMDs).
Statistical analysis
The sample size was calculated by means of G*Power software (version 3.1.9.7) developed by the University of Dusseldorf. Specifically, an aggregated effect size (Cohen's d) of 0.41 was calculated by combining data from the three placebo-controlled trials (10-12) using a weighted average based on the inverse variance of each individual effect size estimate. A sample size of at least 80 participants was then estimated to achieve a power of 95% and a significance level of 5% (two-sided) for detecting an effect size of 0.41 between paired variables (i.e. changes in MMDs) assuming dependent samples and a normal distribution of the statistical parameter in the populations studied.
Normality of the distribution of continuous variables was assessed using the Shapiro–Wilk test prior to applying parametric or non-parametric statistical analyses. Continuous variables with a normal distribution are reported as the mean ± SD, while continuous variable without a normal distribution are reporter as the median ± interquartile (IQR) range. Rates and categorical values are reported as subjects-counts and percentage. A paired t-test was used for comparisons of normally distributed continuous variables, whereas the Wilcoxon signed-rank test was applied for non-normally distributed data. The McNemar test for proportions of paired samples was applied to compare categorical outcomes measured at different time points. Hypothesis testing was one-tailed. p < 0.05 was considered statistically significant. Bonferroni correction for multiple comparisons was applied. Only subjects with complete information regarding the primary outcome (MMDs) were included in the analysis. Finally, a binary logistic regression analysis (forced entry method) was conducted to ascertain whether baseline demographic variables (age, gender, BMI and socioeconomic status) and parameters of disease severity (presence of aura and prior CGRP-mAbs failure) were associated with treatment response, defined as a >50% reduction in MMDs at week 24. All variables were entered simultaneously into the model. The analysis was performed only for patients with complete follow-up data at week 24. All statistics were performed using STATA, version 16 (StataCorp) and SPSS, version 20 (IBM Corp.).
Results
The whole population consisted of 111 participants with chronic migraine. Eleven subjects (10%) dropped out of the treatment with atogepant during the first 12 weeks of treatment due to lack of effectiveness (seven participants), pregnancy (two participants) or poor tolerability (two participants discontinued atogepant for severe fatigue) (Figure 1). Only individuals for whom the MMDs were fully available at baseline, week 12 and week 24 were included in the analysis of effectiveness. Therefore, one-hundred individuals were included in the final analysis (93% female, aged 43 ± 11 years, range 24–75 years). The average time since migraine onset was 25.87 ± 13.51 years, while the average time years with chronic migraine was 31.05 ± 10.64 years. Participants had tested, before starting atogepant, at least three preventive treatments, including BoNT-A (in 14% of participants) and CGRP-mAbs (median of 1 ± 2) in the majority of them (68% of participants). Demographic and baseline headache characteristics of participants included in the study are reported in Table 1.

Flow chart showing patients’ enrollment. CM = chronic migraine.
Demographic and clinical features of the 100 participants who completed the 24-week follow-up.

BMI = body mass index; CGRP = calcitonin gene-related peptide; F = female; HIT-6 = Headache Impact Test-6; HADS = Hospital Anxiety and Depression Scale; IQR = interquartile range; M = male; mAbs = monoclonal antibodies; MAMI = monthly days with acute medication intake; MHDs = monthly headache days; MMDs = monthly migraine days; n = number; NSAID = non-steriodal anti-inflammatory drug.
Primary outcomes
Compared to the baseline, MMDs decreased at week 12 and week 24 by 5.6 days (SD = 5.9; p < 0.001) and 7.1 days (SD = 5.4; p < 0.001), respectively (Figure 1). The percentages of participants reporting a reduction of at least 50% of MMDs at week 12 and week 24 were 45% and 53%, respectively (Figure 2).

Monthly migraine days (MMDs) variation from baseline to 12 and 24 weeks of atogepant 60 mg treatment in (i) participants with chronic migraine as a whole cohort (red line), (ii) participants with chronic migraine with previous failure of calcitonin gene-related peptide (CGRP)-monoclonal antibodies (mAbs) (blue line) and (iii) participants with chronic migraine without previous failure of CGRP-mAbs (green line). *p < 0.001.
Secondary outcomes
Compared to the baseline, at week 12 and week 24 (Table 2):
MHDs decreased by 6.3 days (SD = 6.9; p < 0.001) and 8.1 days (SD = 6.7; p < 0.001), respectively; MAMI decreased by 4.7 days (SD = 2.7; p < 0.001) and 5.1 days (SD = 2.6; p < 0.001), respectively. HIT-6 decreased from 63.6 (SD = 3.35) to 59.1 (SD = 6.62; p < 0.001) and 57.4 (SD = 6.68; p < 0.001) points, respectively.
Change in clinical outcomes from baseline to weeks 12 and week 24 in the 100 participants who completed the 24-week follow-up.

Values are the mean ± SD or ± interquartile range (IQR) or number (%).
CM = chronic migraine; EM = episodic migraine; HADS = Hospital Anxiety and Depression Scale; HIT-6 = Headache Impact Test-6; MAMI = monthly days with acute medication intake; MHDs = monthly headache days; MMDs = monthly migraine days; n = number.
*Statistically significant (in the comparison with baseline).
Furthermore, the percentages of participants reporting at week 12 and week 24:
Reduction of at least 50% of MHDs were 30% and 38%, respectively. Reduction of at least 75% of MMDs were 21% and 20%, respectively. Reversion from chronic migraine to episodic migraine were 57% and the 60% of participants, respectively. Contrariwise, no changes were observed in symptoms of anxiety over the 24 weeks of treatment with atogepant.
Comparing baseline demographic and clinical features in the 50% responder and non-responder groups (Table 3), responders showed a reduced HIT-6 score as well as a higher socioeconomic status. Logistic regression showed that socioeconomic status, treated as an ordinal variable (low = 1, middle = 2, high = 3) and failure of at least one previous preventive treatment with CGRP-targeted therapy (treated as a dicotomic variable where previous failure = 1 and no previous failure = 0) were significantly associated with treatment response. The odds of being a responder increased by a factor of 2.87 for each one-level increase in socioeconomic status (odds ratio (OR) = 2.87, 95% confidence interval = 1.32–6.23, p = 0.008) suggesting a higher likehood of being a responder to atogepant preventive treatment in people with higher socioeconomic status. Moreover, previous failure to CGRP-mAbs was negatively associated with response to the current treatment with a significantly lower likehood of being a responder to atogepant preventive treatment in people with previous CGRP-mAbs failure (OR = 0.38, 95% confidence interval = 0.15–0.99, p = 0.049). Conversely, logistic regression analysis did not reveal any significant association between age, gender, BMI, or presence of aura and the likelihood of being a responder, defined as achieving a >50% reduction in MMDs compared to baseline (Table 4).
Comparison of baseline features between 50%-responders and non-responders.
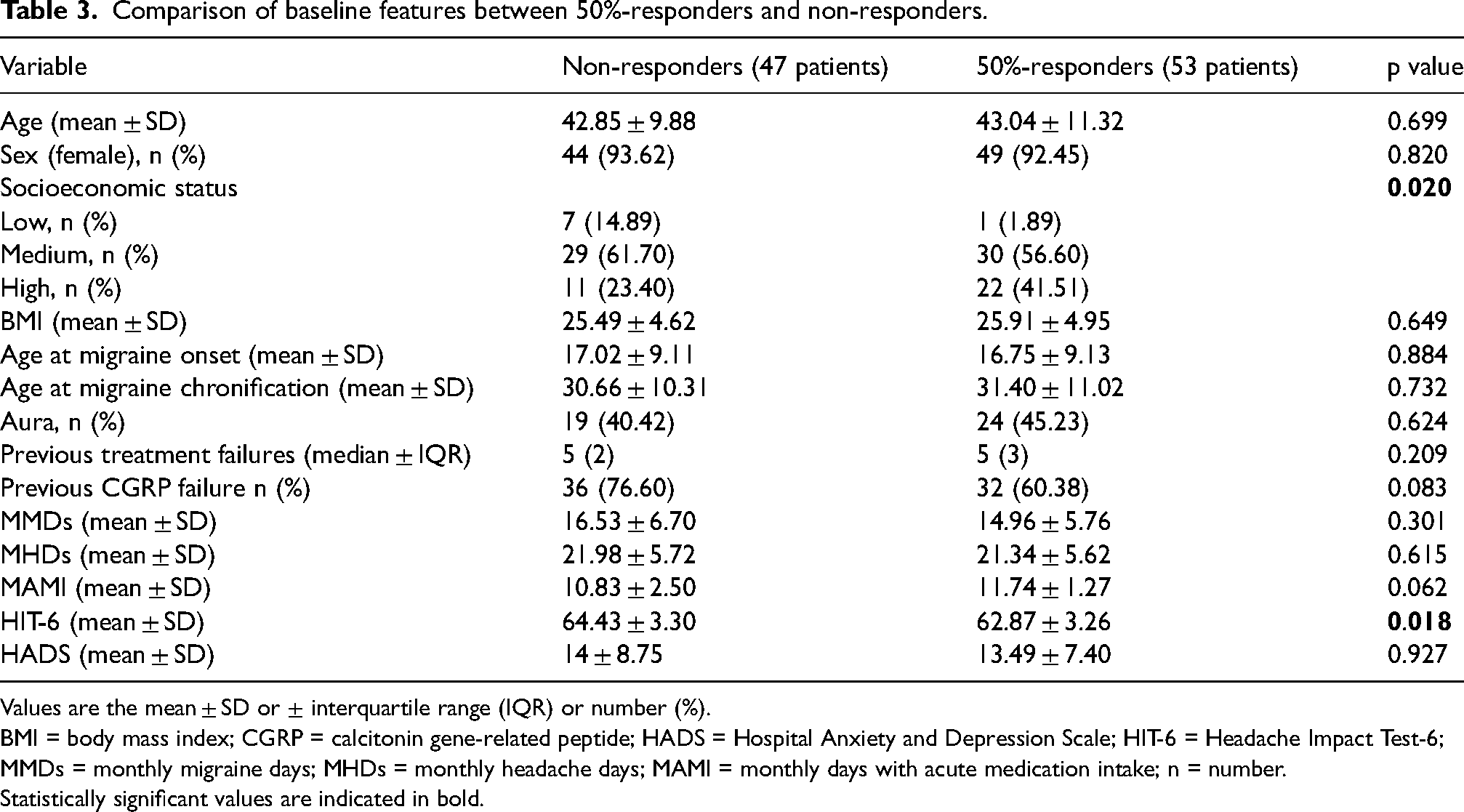
Values are the mean ± SD or ± interquartile range (IQR) or number (%).
BMI = body mass index; CGRP = calcitonin gene-related peptide; HADS = Hospital Anxiety and Depression Scale; HIT-6 = Headache Impact Test-6; MMDs = monthly migraine days; MHDs = monthly headache days; MAMI = monthly days with acute medication intake; n = number.
Statistically significant values are indicated in bold.
Logistic regression analyses to assess association between demographic and clinical parameters with a 50% responder rate.

BMI = body mass index; CGRP = calcitonin gene-related peptide; SE = standard error; χ2 (2) = 43.89, p < 0.059, pseudo r2 = 0.08.
Statistically significant values are indicated in bold.
Specifically considering participants with previous preventive treatment failure with at least one CGRP-mAb, compared to the baseline to week 12 and week 24:
MMDs reductions were of 4.9 days (SD = 6.3; p < 0.001) and 6.7 days (SD = 5.6; p < 0.001), respectively. MHDs reduction were by 6.1 days (SD = 6.9; p < 0.001) and 7.6 days (SD = 7.1; p < 0.001), respectively. MAMI decreased by 4.5 (SD = 3.1; p < 0.001) and 4.9 (SD = 2.8; p < 0.001), respectively. The percentages of participants with previous preventive treatment failure to at least one CGRP-mAb reporting a reduction of at least 50% of MMDs at week 12 and week 24 were 42.7% and 47.1%, respectively (Figure 2).
Furthermore, among the 14 patients with prior BoNT-A failure, MMDs were reduced by 9.4 days from baseline (p < 0.001), and 57% achieved a ≥50% reduction in MMDs at week 24.
Patients’ global impression of their clinical condition following atogepant preventive treatment, assessed by PGIC at 24-week of treatment, revealed that 69% of the whole cohort felt “much better” or “very much better”, while 22% of participants had a worth noting improvement. Only 9% of participants reported no significant changes compared to the baseline (Figure 3).

Changes in the percentage of mild, moderate and severe headache from baseline to week 12 and 24 of treatment with daily atogepant 60 mg. CGRP = calcitonin gene-related peptide; mAbs = monoclonal antibodies; MMD = monthly migraine days.
Sixty-three participants (63%) reported TEAEs related to atogepant at week 12. The most common TEAEs were constipation (40%), fatigue (25%), nausea (12%), stomach pain (7%), hair loss (4%) and loss of appetite (6%). Contrariwise, fifty-three participants (53%) reported TEAEs related to atogepant after week 24. The most common TEAEs were constipation (28%), fatigue (16%), nausea (7%), stomach pain (5%), hair loss (2%) and loss of appetite (2%).
No significant changes in BMI were observed at weeks 12 and 24 compared to baseline (Figure 4). Specifically, mean BMI was 25.7 at baseline, 25.6 at week 12 (p = 0.931) and 25.6 at week 24 (p = 0.894).

Patient Global Impression of Change (PGIC) at 24 weeks of treatment with daily atogepant 60 mg. No individuals reported “No change/condition has got worse” or “Almost the same/hardly any change at all” (options not included in the graphic).
Discussion
The present is the first real-world observation reporting atogepant effectiveness and tolerability in a cohort of participants with chronic migraine with multiple previous preventive treatments failure, observed over a 24-week period.
The recent introduction of CGRP-targeted therapies (i.e., CGRP-mAbs and gepants) marked a major advancement in the preventive treatment of migraine (5). Among these, atogepant has been shown to be an effective, safe, and well-tolerated therapeutic option, supported by robust evidence from randomized, double-blind, placebo-controlled trials in individuals with chronic migraine as well as with episodic migraine with prior failures to preventive treatments. Specifically, in the PROGRESS trial, 778 individuals with chronic migraine were enrolled, showing over 12 weeks, a mean MMDs reduction from baseline of −6.9 days in the 60 mg once-daily group compared to −5.1 in the placebo group (11). Besides, the efficacy of atogepant in individuals with previous failure to two-four classes of “standard of care” oral preventive treatments was explored in the ELEVATE study, where individuals with episodic migraine, followed over 12 weeks, showed a significantly greater reduction in MMDs if treated with atogepant 60 mg once daily compared to the individuals treated with placebo (−4.2 vs. −1.9) (12). In both studies, constipation was the most common TEAEs, reported in 10% of participants receiving atogepant versus 3% in the placebo group.
Very recently, two Italian real-world studies (the STAR and GIANT studies) investigated the effectiveness of atogepant 60 mg, over a 12-week period, in cohorts with both individuals with high frequency episodic migraine and chronic migraine (13,14). Along with a favourable safety profile, atogepant significantly reduced MMDs as well as migraine-related functional impact and disability in both populations, irrespective of prior preventive treatment failures, including CGRP-mAbs.
Another retrospective cohort study evaluated the effectiveness of a three-month atogepant treatment in individuals with episodic and chronic migraine who had previously discontinued CGRP-mAbs due to lack or loss of efficacy (21). Among 44 participants, primarily with chronic migraine, atogepant resulted in a ≥50% reduction in MHDs in 18.2% of participants. Significant improvements were also observed in moderate-to-severe headache days, acute medication use and HIT-6.
In the present study, we observed, after 12 weeks, a reduction of 5.6 MMDs compared to baseline, which is less than that observed in the PROGRESS study (i.e. 6.9 MMDs) and a lower reduction compared to observations from other real-world studies. The above-mentioned difference may be explained by the fact that our patient cohort exclusively consisted of individuals with treatment-resistant chronic migraine. Indeed, while in the PROGRESS study the number of previous treatment failures was not specified, individuals enrolled in other real world observations showed a median of four previous preventive treatment failures at baseline. Similarly, while previous CGRP-mAbs failure represented an exclusion criterion in the PROGRESS study, the percentage of individuals with previous CGRP-mAbs failure in real world investigations ranged from 41.5% in the GIANT study to 53% in the STAR study. Our cohort of participants showed a median of six prior preventive treatment failures (IQR 2) and, notably, 68% had also failed at least one treatment with CGRP-mAbs. Intriguingly, at 24 weeks, MMDs decreased by 7.1 compared to the baseline, suggesting that longer periods of treatment could be necessary to capture the therapeutic benefit in individuals with such a severe treatment-resistant chronic migraine (22).
In our cohort, the percentage of participants reporting a reduction of at least 50% of MMDs after the third month of treatment was 45%. This is slightly better than data emerging from the PROGRESS study (43% with atogepant 30 mg twice a day and 41% with 60 mg once a day), while it was substantially in line with data emerging from the real world STAR study (46.4%). Interestingly, the percentage of participants reporting a reduction of at least 50% of MMDs after 24 weeks increased to 53% (Figure 5). Along with the reduction of MMDs, at week 24 of atogepant 60 mg treatment, the 60% of participants converted from chronic to episodic migraine: specifically, 33% of participants converted to high frequency episodic migraine and 27% to low frequency episodic migraine.

Percentage of participants (whole cohort, individuals with previous failure of calcitonin gene-related peptide (CGRP)-monoclonal antibodies (mAbs) and individuals without previous failure of CGRP-mAbs) reaching the reduction of >50% in monthly migraine days at 12 and 24 weeks of atogepant 60 mg treatment.
Twenty percent of participants were classified as super-responders, defined as those reporting a >75% reduction in MMDs compared to baseline. This result appears slightly lower than that reported in previous real-world investigations, such as the STAR study (31.1%) and the GIANT study (36.6%).
Beyond the improvement of migraine attack frequency, a significant reduction in the number of attacks of moderate or severe intensity as well as in MAMI were observed.
A substantial improvement was observed in migraine-related impact on daily living activities over the 24 weeks of atogepant 60 mg treatment as our participants showed a HIT-6 reduction of more than 6 points, considered a clinically significant difference in HIT-6 score, indicating not only a statistically meaningful, but also a clinically perceivable improvement in headache-related disability (23).
Logistic regression analysis identified the history of CGRP-mAbs failure and socioeconomic status as key predictors of clinical response to atogepant. More specifically, individuals without previous CGRP-mAbs failures and with high socioeconomic status showed higher odds of achieving ≥50% reduction of MMDs. Contrariwise, differently from previous investigations, BMI did not emerge as a predictor of response to atogepant, nor did it show any significant variation over the three-month treatment period. Higher socioeconomic status may act as a positive predictive factor for treatment response in individuals with migraine due to a combination of structural and behavioural advantages. Although socioeconomic status is not a biological determinant, it profoundly shapes the environment in which treatment occurs. Indeed, individuals with higher socioeconomic status typically have improved access to healthcare services, greater health literacy, and, arguably, fewer barriers to initiating and adhering to prescribed treatments (24-25). On the other hand, previous failure CGRP-mAbs may represent a negative predictor of response to atogepant. Since both CGRP-mAbs and gepants act on the same pathway, although with different mechanisms and effects, that would suggest that a lack of efficacy with one CGRP-modulating treatment could reflect a sort of “resistance” to CGRP inhibition as a preventive strategy in certain individuals. Interestingly despite the negative predictor role of previous failure CGRP-mAbs on response to atogepant, we found a significant percentage of responders (>50% reduction in MMD compared with baseline) in 42.6% and 47% of participants with previous CGRP-mAbs failure at weeks 12 and 24, respectively. The efficacy of atogepant in individuals with chronic migraine and prior failure to CGRP-mAbs suggests that, in certain individuals, treatment failure may result from insufficient and/or inadequate blockade of the pathway (26).
Indeed, several molecular mechanisms may underlie the superior inhibition of CGRP signalling by atogepant compared to CGRP-mAbs. Unlike ligand-binding CGRP-mAb, which are degraded upon internalization, atogepant co-internalizes with the receptor and continues inhibiting signalling within endosomes, potentially enhancing therapeutic efficacy (27). Additionally, atogepant antagonizes α-CGRP-induced β-arrestin recruitment and receptor internalization, indicating broader intracellular effects. It also binds amylin and adrenomedullin receptors, both implicated in migraine pathophysiology (28,29). Moreover, preclinical data show that atogepant attenuates responses of C-fibers and Aδ-fibers following cortical spreading depolarization (30–32). Finally, pharmacokinetically, atogepant differs from mAbs by being orally administered, having a shorter half-life and smaller size. About the latter, approximately 1.4% crosses the blood–brain barrier, although the relevance of its central effects remains uncertain (33).
In the present study, Atogepant demonstrated a favourable tolerability profile, with no serious adverse events reported. The most commonly observed TEAEs were constipation, fatigue, and nausea. Both the nature and frequency of adverse events are consistent with findings from other real-world evidence studies and with recent data from the US FDA Adverse Event Reporting System (FAERS) database (14,34). In contrast to the FAERS data, no unexpected adverse events were identified during the 24-week treatment period with atogepant.
Interestingly, the incidence of TEAEs declined over the course of treatment, suggesting that TEAEs may be more frequent during the initial phases with a tendency to reduce over time, potentially enhancing long-term adherence. As a proof of the concept, the discontinuation rate was low (10%), and only two participants discontinued treatment due to adverse events, both referring to fatigue. Notably, although previous research has shown that individuals with migraine naïve to CGRP-targeting therapies strongly prefer treatments without nausea or constipation, only a few participants discontinued atogepant due to fatigue, and none discontinued treatment due to nausea or constipation (35).
A notable strength of this study is the homogeneity of the participants’ sample, which consisted exclusively of individuals with chronic migraine without medication overuse headache, thereby reducing clinical variability and enhancing the interpretability of treatment effects within this specific population, with a consequent consistent generalizability of the findings in similar migraine populations. Furthermore, the present observation is focused on hard-to-treat individuals, giving substantial insight on the treatment of a specific patients’ population characterized by several previous preventive treatment failures. Finally, the longer observation period, compared to the real-word data available to date, is crucial for capturing the full therapeutic potential of atogepant, particularly in real-world settings, as it allows for a more accurate assessment of sustained effectiveness, treatment adherence, and clinical responses.
On the other hand, the study is not exempt from some limitations. Primarily, as a non-randomized, open-label design, it lacked both a placebo and an active comparator arm, requiring cautious interpretation of the results, as uncontrolled variables may have influenced the observed outcomes.
Nevertheless, open-label studies do not necessarily overestimate treatment effectiveness, particularly when the efficacy and safety profiles of the intervention, such as atogepant in chronic migraine, are already well-established (36,37).
In conclusion, the present observations support the effectiveness, safety, and tolerability of daily atogepant 60 mg as preventive treatment, during a six-month period of observation, in individuals with hard-to-treat migraine phenotypes such as chronic migraine with multiple previous treatment failures. Specifically, the present findings build upon evidence from randomized controlled trials by demonstrating significant reductions in pain intensity and migraine related impact, meaningful benefits in individuals with previous failure of CGRP-mAbs. Further large-scale studies are needed to confirm these results and to more clearly define the role of atogepant within individualized migraine management strategies.
Key findings
After 24 weeks of treatment with atogepant 60 mg/day, participants showed a mean reduction of MMDs, with 53% achieving a ≥50% reduction.
After 24 weeks, sixty percent of participants converted from chronic to episodic migraine, with significant reductions in both headache days and acute medication use.
Among individuals with prior failure to CGRP monoclonal antibodies, 47% still achieved a ≥50% reduction in MMDs after 24 weeks.
Atogepant showed a favourable tolerability profile, with no serious adverse events and a low discontinuation rate (10%), while the most common TEAEs (constipation, fatigue and nausea) were more frequent in the early phases and tended to decline over time.
Footnotes
Declaration of conflicting interests
AR: has received speaker honoraria from Allergan, Lilly, AbbVie, Pfizer, Novartis and Teva and serves as an associate editor of Frontiers in Neurology (Headache Medicine and Facial Pain section). MS: has received speaker honoraria from Novartis, Pfizer, AbbVie, Teva and Lilly.
IF: Grants/honorarium/Consulting fees: Allergan, Amgen, Eli Lilly, J and J, Merck, Purdue, Teva, Aralez, Novartis, Lundbeck. DS: None. AA: None. AHA: has received personal fees for lectures/advisory boards: Novartis, Abbvie, TEVA, Roche, Lundbeck, Pfizer, Boehringer Ingelheim. Non-personal research grants from several sources, including KlinBeForsk, EU, Boehringer Ingelheim, Medtronic, BMS. EC: has received honoraria from Novartis, Chiesi, Lundbeck, MedScape, Lilly; his salary has been partially funded by Río Hortega grant Acción Estratégica en Salud 2017–2020 from Instituto de Salud Carlos III (CM20/00217) and Juan Rodés fellowship, Subprograma Estatal de Incorporación de la Acción Estratégica en Salud 2023 (JR23/00065). He is a junior editor for Cephalalgia. PPR: has received, in the last three years, honoraria as a consultant and speaker from AbbVie, Amgen, Dr Reddy's, Eli Lilly, Lundbeck, Medscape, Novartis, Organon, Pfizer and Teva Pharmaceuticals. Her research group has received research grants from AbbVie, AGAUR, EraNet Neuron, FEDER RIS3CAT, Instituto Investigación Carlos III, MICINN, Novartis, and Teva Pharmaceuticals, and has received funding for clinical trials from AbbVie, Amgen, Biohaven, Eli Lilly, Lundbeck, Novartis, Pfizer and Teva Pharmaceuticals. She is the Honorary Secretary of the International Headache Society, is on the editorial board of Revista de Neurologia, is an associate editor for Cephalalgia, Headache, Neurologia and Frontiers of Neurology, and is an advisor of the Scientific Committee of the Editorial Board of The Journal of Headache and Pain. She is a member of the Clinical Trials Guidelines Committee and Scientific Committee of the International Headache Society. She has edited the Guidelines for the Diagnosis and Treatment of Headache of the Spanish Neurological Society. She is the founder of ![]() . ET: has received personal fees for lectures/advisory boards: Novartis, Eli Lilly, Abbvie, TEVA, Roche, Lundbeck, Pfizer, Biogen, Organon. Consultant for and owner of stocks and IP in Man & Science. Stocks and IP in Nordic Brain Tech. Stocks in Keimon Medical. Non-personal research grants from several sources, including Norwegian Research Council, KlinBeForsk, EU. Commissioned research (non-personal): Lundbeck. CS: has received honorarium for lectures/advisory boards: Novartis, Abbvie, TEVA and Vigil NeuroScience.
. ET: has received personal fees for lectures/advisory boards: Novartis, Eli Lilly, Abbvie, TEVA, Roche, Lundbeck, Pfizer, Biogen, Organon. Consultant for and owner of stocks and IP in Man & Science. Stocks and IP in Nordic Brain Tech. Stocks in Keimon Medical. Non-personal research grants from several sources, including Norwegian Research Council, KlinBeForsk, EU. Commissioned research (non-personal): Lundbeck. CS: has received honorarium for lectures/advisory boards: Novartis, Abbvie, TEVA and Vigil NeuroScience.
Ethical statement
Each patient gave a free, informed consent for participation in the study and the analysis and publication of the protocol data.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
