Abstract
Abstract
Background
Patients diagnosed with New Daily Persistent Headache and Persistent Post-Traumatic Headache belong to a heterogeneous group of primary and secondary headache disorders, with the common clinical feature that these conditions start abruptly, continue unabated, and are refractory to conventional migraine preventive treatments.
Objective
This is a real-world, medium-term audit to explore whether erenumab improves quality of life in a pooled group of 82 abrupt-onset, unremitting and treatment refractory patients, where the diagnosis is new daily persistent headache and persistent post-traumatic headache in the majority of cases.
Methods
Eighty-two patients were treated with erenumab every 28 days over a two to three-year period, beginning in December 2018. These patients were “longstanding chronic” and refractory with a median of eight (IQR 4–12) prior failed migraine preventive treatments and median duration of disease of seven (IQR 3–11) years. The starting dose of erenumab was 70 mg in 79% of cases and 140 mg in the remaining patients (individuals with a BMI of more than 30). All patients were asked to complete three migraine specific Quality of Life questionnaires or Patient Reported Outcome Measures before starting treatment and typically at 3–12 intervals until the end of June 2021 or cessation of treatment. The Patient Reported Outcome Measures included: Headache Impact Test-6, Migraine Associated Disability Assessment test and Migraine-Specific Quality-of-Life Questionnaire. Patients generally only stayed on treatment after 6–12 months if there was deemed to be an improvement of at least 30% and there were no significant side effects. The longest treated cases have quality of life data for 30 months after starting erenumab.
Results
Of the 82 patients, 29 (35%) had improvement in Quality of Life scores, with no significant side effects, and wished to stay on treatment. Fifty-three patients (65%) stopped treatment during the first 6–25 months due to lack of efficacy and/or patient reported side effects (n = 33 and n = 17, respectively) or a combination of both, pregnancy planning (n = 2), and lost to follow up (n = 1).
Conclusion
Significant improvements in Quality of Life scores were recorded by one-third of patients over a period of 11–30 months, with a 35% persistence after a median of 26 months of treatment. This contrasts with our recently published, treatment resistant, chronic migraine cohort where the persistence with erenumab treatment was almost 55% after a median time of 25 months.
Keywords
Introduction
Abrupt onset headache disorders which are treatment refractory, constant from the beginning and persisting for many years, are challenging cases which are not infrequently encountered in specialist headache clinics (1). This heterogenous grouping includes patients diagnosed with ‘primary’ New Daily Persistent Headache or NDPH (formally classified in ICHD-3 as a primary headache disorder) (2) and Persistent Post-Traumatic Headache or PPTH (classified in ICHD-3 as a secondary headache disorder) (2), and various other rarer secondary headache conditions. Overall, these patients represent a minority of cases seen in headache clinics, but they are invariably disabled and the diagnosis is often complicated by the fact that many may display other neurological, predominantly migraine associated symptoms, including nausea, disequilibrium, movement sensitivity, photophobia and phonophobia (3,4). In effect, they very commonly have a migraine phenotype clinically.
‘Primary’ NDPH is essentially a clinical description of a headache disorder, with a clearly documented abrupt onset, that continues without relief for more than three months (typically many years in our clinical practice and in this cohort). Primary NDPH can be sub-classified into NDPH with a Migraine Phenotype (MP) or a Tension-Type Phenotype (TTP) (2). Additionally, primary NDPH may be sub-classified as spontaneous (where there is no obvious associated antecedent or preceding trigger factor) or triggered. The list of generally recognised triggers for primary NDPH include: a single stressful or traumatic life event, a viral or flu-like illness (presenting as an upper respiratory tract infection), and extracranial surgery. Other possible triggers include hormone manipulation with progesterone, exposure to chemical substances (such as hydrocarbons), withdrawal from or exposure to certain prescribed medications (such as antibiotics or selective serotonin reuptake inhibitors (SSRIs)) and human papillomavirus (HPV) vaccination (5).
As noted above and as suggested by the name, primary NDPH is formally classified as a primary headache condition, but this assignment is not without controversy. Some experts propose that NDPH should be divided into a primary or secondary headache disorder (6). To add to the classification and diagnostic difficulties, many ‘primary NDPH’ cases are clinically indistinguishable from patients who are deemed to have a secondary abrupt onset persistent headache disorder, such as constant daily unremitting headache from (typically minor) direct head trauma, blast and whiplash injuries (PPTH) (6). In addition, these primary NDPH patients may be clinically very similar to patients with chronic migraine (CM) who have no completely normal or crystal-clear days, and are therefore in a continuous migraine state. For this report, we have grouped all of our ‘abrupt onset unremitting’ or ‘daily persistent from the beginning’ headache patients together, based on the fact that they all have ongoing intractable daily headache and associated migraine symptoms for at least one year (and typically several years). As is the situation worldwide, the vast majority of these patients struggle to improve with conventional headache or migraine preventive treatments and are therefore described as medically refractory or treatment intractable (7).
Erenumab is a fully-human anti-calcitonin gene-related peptide (CGRP) monoclonal antibody that binds to the canonical CGRP receptor (CLR/RAMP1) to prevent CGRP from docking (8). It was licensed for the prevention of migraine by the US food and drug administration (FDA) in May 2018 and the european medicines agency (EMA) in August 2018 (9). To date, there is limited real-world evidence for the use of CGRP monoclonal antibodies in primary NDPH or secondary disorders which clinically mimic this condition (10–12).
Methods
Aim
The aim of this audit was to evaluate whether treatment with erenumab improved quality of life, by using Quality of Life (QoL) questionnaires or Patient Reported Outcome Measures (PROMs), in a heterogenous cohort of 82 patients with an intractable daily headache disorder and a migraine phenotype. We hypothesised that erenumab may work in a proportion of these patients as they have all displayed some migraine symptoms, typically manifesting clinically manifesting clinically with exacerbations of their daily headache disorder.
Study design
This is a retrospective, open-label, observational audit (survey) in a real-world setting. It mirrors the design of our recently published report on erenumab in 177 resistant chronic migraine patients (13). The vast majority of patients included in this report were diagnosed with primary NDPH or a secondary abrupt onset persistent headache condition (typically PPTH). All but one of the patients have had continuous symptoms for two years or more and have responded inadequately or have failed multiple conventional migraine preventive therapies. This is the primary analysis of this data and the a priori assumption was that at least 20–25% of patients would show a subjective response to treatment in this very refractory cohort. The cohort is a convenience sample from patients seen in our clinics from December 2018 to October 2019. No sample size calculation was conducted. QoL questionnaires were filled in by the patients as part of our routine clinical practice just before starting treatment (December 2018 to October 2019 in 80 of the cases) and then periodically, in practice on average every 3–12 months, to the end of June 2021. Erenumab was kindly made available free of charge to our patients by Novartis through a free Managed Access Program (MAP) in Ireland from November 2018 until the first half of 2022. Erenumab is now available in Ireland through the national public healthcare system since November 2021.
Setting
This audit was conducted in three Specialist Headache/Migraine clinics in Ireland. The clinics are supervised by Headache/Migraine specialists (neurology consultants) and exclusively see patients suffering with headache. These centres receive referrals from primary care practitioners, other Irish non-headache neurologists and non-neurologist medical/surgical hospital consultants from all over Ireland. They are considered to be designated national headache clinics.
Inclusion criteria
Eighty-two patients older than 18 years with an abrupt onset and ‘continuous from the beginning’ clinical phenotype, mainly diagnosed with primary NDPH (ICHD-3, code 4.10) and secondary PPTH (ICHD-3, codes 5.2.1, 5.2.2, 5.4, and 5.6) according to the ICHD-3 criteria, (2) were included in the study (Figure 1). Patients are deemed to have a secondary condition if there were clear provoking or precipitating factors. This included patients with (typically relatively minor) direct head trauma, whiplash or blast injury (all falling within PPTH diagnostic classification) (2), brain haemorrhage, arterial dissection, abnormal cerebrospinal Fluid (CSF) volume, etc. Three experienced headache neurologists (authors RL, PJG and MHR) had reviewed all the patients clinically and made the diagnosis over the preceding 5–10 years. All patient charts were reviewed again by two of the authors (AB and MHR) in 2021/2022 to ensure diagnostic certainty.

Clinical phenotype of patients with abrupt onset continuing and unremitting headache conditions.
For this report, a migraine phenotype (MP) was clinically defined as the presence of at least two of the following associated migraine features, which were typically asked about during the history taking in the context of clinical exacerbations: throbbing/pounding moderate to severe pain, aggravation or worsening of symptoms with movement or routine physical activity, photophobia, phonophobia, nausea, vomiting or osmophobia. We only included patients in this audit who had a migrainous phenotype using these criteria.
Exclusion criteria
Abrupt onset headache patients were excluded from the audit if they were prescribed erenumab and had evolving chronic daily migraine immediately prior to the onset of their condition or had an NDPH-tension type headache (TTH) phenotype. In addition, abrupt onset headache patients in our clinics are excluded from being prescribed erenumab if they are pregnant or considering pregnancy in the next 6–9 months, have a latex allergy, or have had a recent cardiovascular event (such as stroke, transient ischaemic attack, myocardial infarction, etc.) in the last 6–12 months.
Data collection
Patients received erenumab 70 mg/140 mg subcutaneous injection every four weeks. Our usual practice in 2018/2019 was to start at a dose of 70 mg for the first three to four months, and then increase the dose to 140 mg if there was inadequate response and no significant side effects. A minority of patients (21%) started on 140 mg from the outset if they had a significantly raised BMI (above 30). Patients were asked to complete three PROM or QoL questionnaires: Headache Impact Test (HIT-6), Migraine Disability Assessment (MIDAS) and Migraine-Specific Quality of Life (MSQ). These questionnaires were completed before starting treatment, and typically at 3–12-month intervals, for up to 30 months in some cases. Patients were routinely reviewed twice a year, unless there were unusual circumstances such as possible significant side effects (patients reviewed more frequently) or clinics were postponed/cancelled due to COVID-19 (patients reviewed less frequently). Moreover, some consultations were virtual given the pandemic. As a general rule, the decision regarding whether to continue with treatment was based on at least a 30% overall subjective improvement in severity of headache and associated neurological (migraine) symptoms.
Ethics
Ethics approval was not required for this audit, as completion of headache and/or migraine QoL questionnaires or PROMs by patients is a routine part of our clinical practice. No additional patient data or information was collected, and specific tests/investigations were not performed. Patients were informed that anonymised data would be collected, and verbal informed consent was obtained from all patients. This audit was registered in the Dublin hospitals where patients were seen.
Statistical analysis
The data was analysed using baseline (time-zero) patient QoL questionnaire outcomes and then compared to scores at various time periods after starting treatment (1–6 months, 7–12 months, 13–18 months, 19–24 months and 25–30 months). Data was reported as observed and no imputation was carried out for missing data. From analysing the raw scores and reviewing histograms, we concluded that the data is unlikely to be normally distributed, and therefore median and interquartile ranges (IQR) for HIT-6, MIDAS, and MSQ scores were used for displaying the results. Calculations were carried out using RStudio 1.4.1106 and R 4.0.5 for Windows. Confidence intervals were set at 95% when comparing medians and a significance level of 5% was used for two-tailed statistical analysis. Wilcoxon Signed Rank tests were used to compare the median scores across times. Mann-Whitney U tests were carried out to verify that excluded patients (i.e. those who did not record a score at both times being tested) did not systematically impact the tested median scores.
Results
Patient groups and demographics
Eighty-two patients with abrupt onset, continuous daily from the beginning, and intractable headache disorders were included. All but one patient had constant daily symptoms for at least two years at the time of starting erenumab. Sixty-five percent of patients were female (n = 54) and 35% male (n = 28). Thirty three percent of patients (n = 27) had no previous premorbid history of headache, while 67% (n = 55) had a previous history of episodic migraine or infrequent headache. The median (IQR) length of disease prior to starting treatment with erenumab was seven years (IQR 3–11). A median (IQR) of eight conventional headache/migraine preventive therapies (IQR 4–12) were tried before receiving erenumab. Approximately 76% of patients (n = 62) had failed or only responded partially to at least two cycles of onabotulinumtoxin A (PREEMPT Protocol) before receiving erenumab. Demographics, diagnosis and summary of clinical details of the cohort are presented in Table 1. More detailed clinical information is presented in Online Appendix A. Almost 65% of patients (n = 53) were clinically classified as having primary NDPH, while approximately 35% (n = 29) had a variety of secondary abrupt onset intractable headache disorders (Figure 1).
Demographics and clinical details of patients with abrupt onset headache conditions.

Outcome of treatment with erenumab
Seventy-nine percent of patients were started on the 70 mg dose of erenumab (n = 64/82) whereas 21% (n = 18/82) were started on erenumab 140 mg (mainly patients with a raised BMI) every four weeks. Approximately 35% of patients (n = 29) reported a subjective improvement in severity of headache and migraine symptoms following treatment with erenumab, and therefore remained on treatment at 13–18 months and beyond (median duration of treatment was 26 months). Figure 2 shows the response to erenumab according to the underlying clinical phenotype. Relatively similar percentages of patients with primary NDPH (34%, n = 18/53) and secondary cases (38%, n = 11/29) remained on erenumab at 18–30 months. Approximately 65% of patients (n = 53/82) discontinued erenumab during the first 6–25 months of treatment, with the majority in the first 6–12 months. The main reasons for cessation of treatment included: lack of efficacy (n = 33), inadequate treatment response and side effects (n = 17) (with the major reasons listed in Table 2 and Online Appendix A) or pregnancy planning (n = 2) (Figure 3). One patient was lost to follow up. Among patients with secondary headache conditions who benefited from erenumab, nine had PPTH, one electrocution, and one patient had trigeminal herpes zoster.

Response to erenumab according to the clinical phenotype.
Median HIT-6 score at baseline and follow up in months. Median difference in HIT-6 compared to baseline and percentage change in HIT-6 compared to baseline. Proportion of patients contributing to the data.

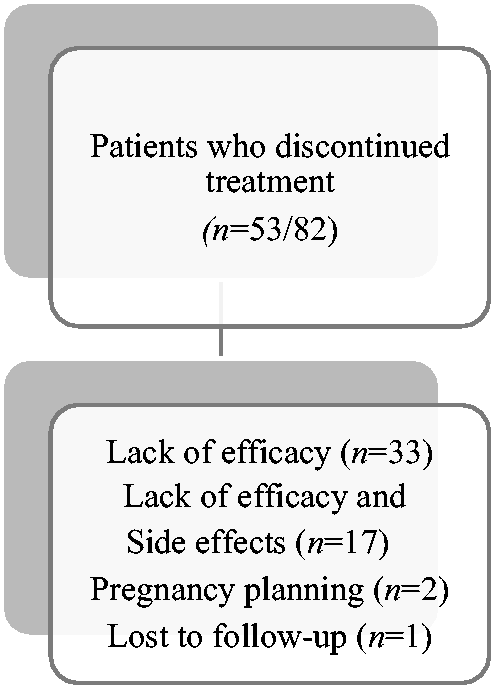
Main reason of discontinuation of treatment with erenumab.
Overall, approximately 79% (23/29) of patients who reported a benefit with erenumab after 18–30 months of treatment had a history of episodic migraine or infrequent headache. In contrast, 60% (32/53) of patients who reported no improvement (non-responders) had a prior history of migraine or episodic headache.
QOL data analysis
We used three quality of life measures (HIT-6, MSQ and MIDAS) to assess whether there was perceived improvement with treatment. Medians, change in median scores and proportions of patients contributing to completed questionnaires are shown in Tables 2–4. The progressions of measure medians are shown in Figures 4–9. Three questionnaire records were removed from analysis due to not having recorded time-zero scores; two of these were for MIDAS and one was for MSQ. Descriptive statistics of length of treatment and a summary of treatment length by patient group and time are shown in Tables 5–6 and Figure 10. All comparisons determined by the Wilcoxon Signed Rank test were statistically significant with P-values of <0.01, apart from the MSQ time 13–18 and time 25–30 scores which were statistically significant but with P-values of 0.017 and 0.032, respectively. All exclusions were tested using Mann-Whitney U tests and were not statistically significant. For HIT-6, self-reported scores showed a sustained reduction compared to baseline in those patients who remained on treatment with erenumab indicating subjective improvement in quality of life. Median reduction in HIT-6 over the first year was between 4.25 and 5 points but increased to an 11-point reduction in the second year, indicating a clinically significant improvement. There was a sustained reduction at 25–30 months, but at a slightly lower value of 9 points. Similarly for MIDAS, patients reported overall improvement in scores at 13–18 months compared to baseline (127 points). The median MIDAS score reduced at 19–24 months and 25–30 months (by 123 and 151 points, respectively). The MSQ median scores also reduced by 14 points after 12 months. There was a further reduction in the MSQ score at 13–18 and 19–24 months (by 13 and 15.5 points, respectively), but remained at 12 points of reduction at 25–30 months. Overall, our results showed an improvement in quality-of-life outcomes that was sustained up to 18–30 months.
Median MIDAS score at baseline and follow up in months. Median difference in MIDAS compared to baseline and percentage change in MIDAS compared to baseline. Proportion of patients contributing to the data.

Median MSQ score at baseline and follow up in months. Median difference in MSQ compared to baseline and percentage in MSQ compared to baseline. Proportion of patients contributing to the data.


Median HIT-6 score (y-axis) at baseline and up to 30 months of follow-up (x-axis). 95% confidence interval of median shown by vertical bars. m0106, m0712, m1318, m1924, and m2530 represent the treatment periods: 1–6 months, 7–12 months, 13–18 months, 19–24 months, and 25–30 months, respectively.
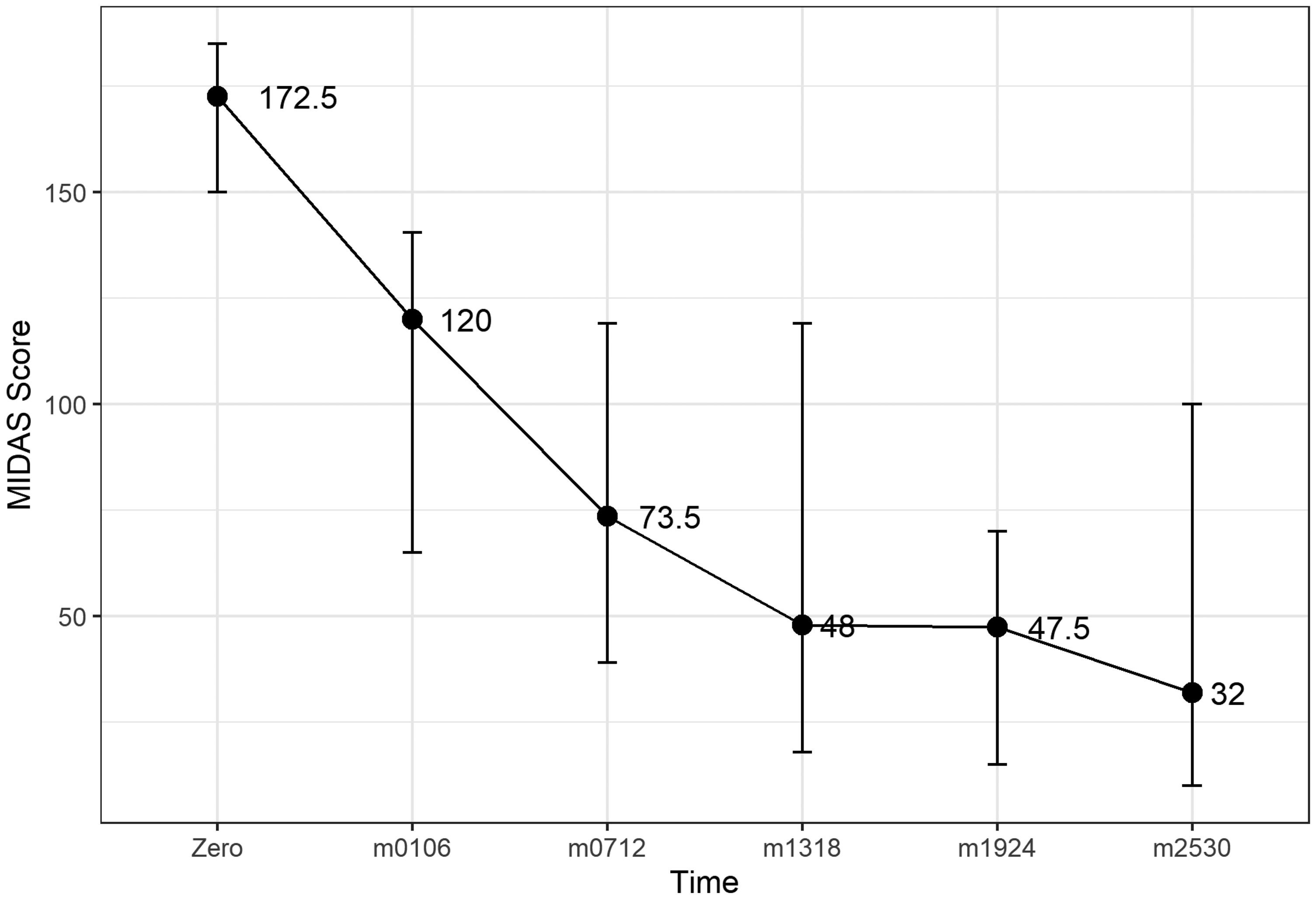
Median MIDAS score at baseline (y-axis) and up to 30 months of follow-up (x-axis). 95% confidence interval of median shown by vertical bars. m0106, m0712, m1318, m1924, and m2530 represent the treatment periods: 1–6 months, 7–12 months, 13–18 months, 19–24 months, and 25–30 months, respectively.

Median MSQ score at baseline (y-axis) and up to 30 months of follow-up. 95% confidence interval of median shown by vertical bars. m0106, m0712, m1318, m1924, and m2530 represent the treatment periods: 1–6 months, 7–12 months, 13–18 months, 19–24 months, and 25–30 months, respectively.
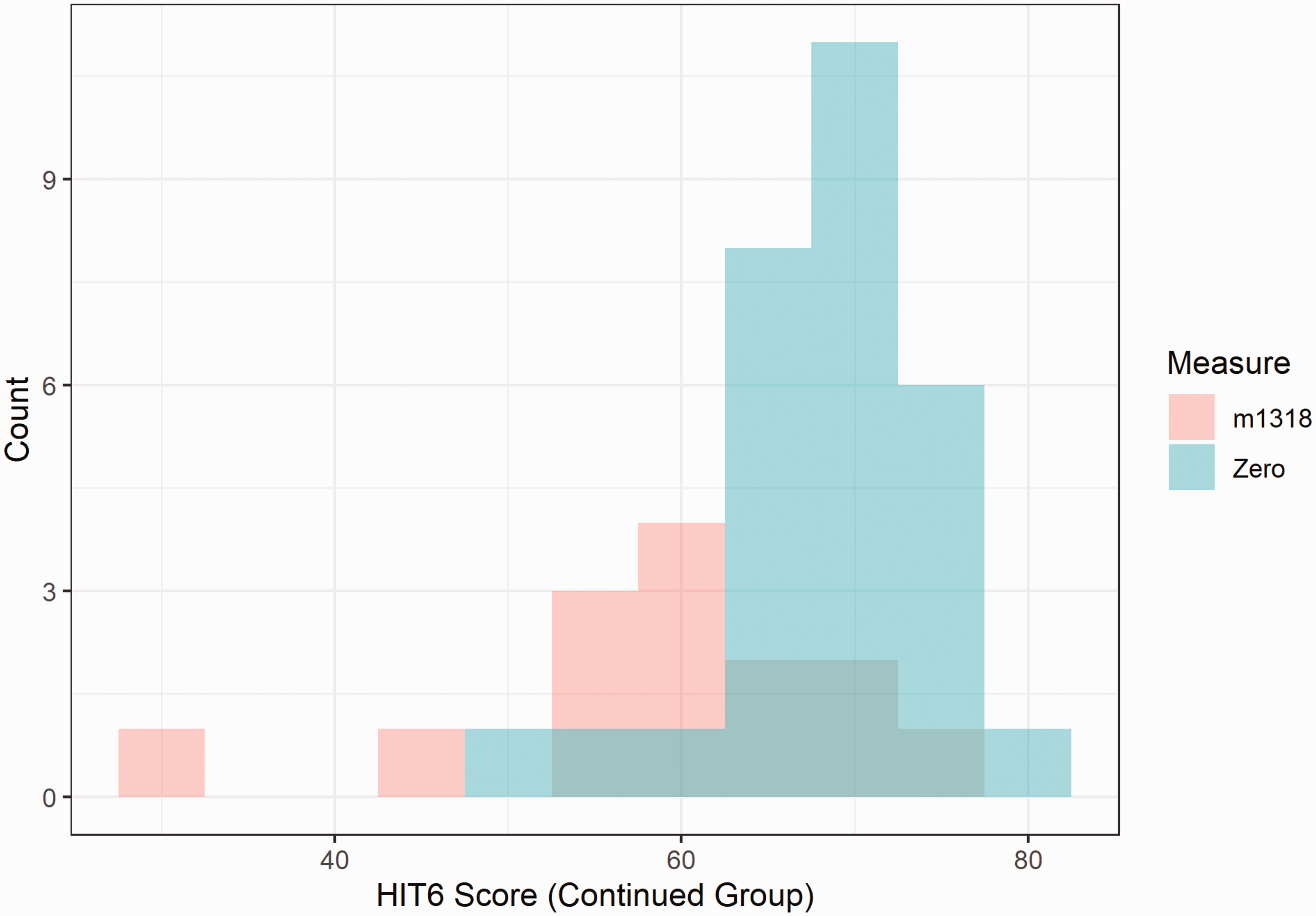
Histogram comparing time-zero and m1318 (treatment period months 13–18). Distribution of HIT-6 scores for patients who remained on treatment beyond June 2021.

Histogram comparing time-zero and m1318 (treatment period months 13–18). Distribution of MIDAS scores for patients who remained on treatment beyond June 2021.

Histogram comparing time-zero and m1318 (treatment period months 13–18). Distribution of MSQ scores for patients who remained on treatment beyond June 2021.
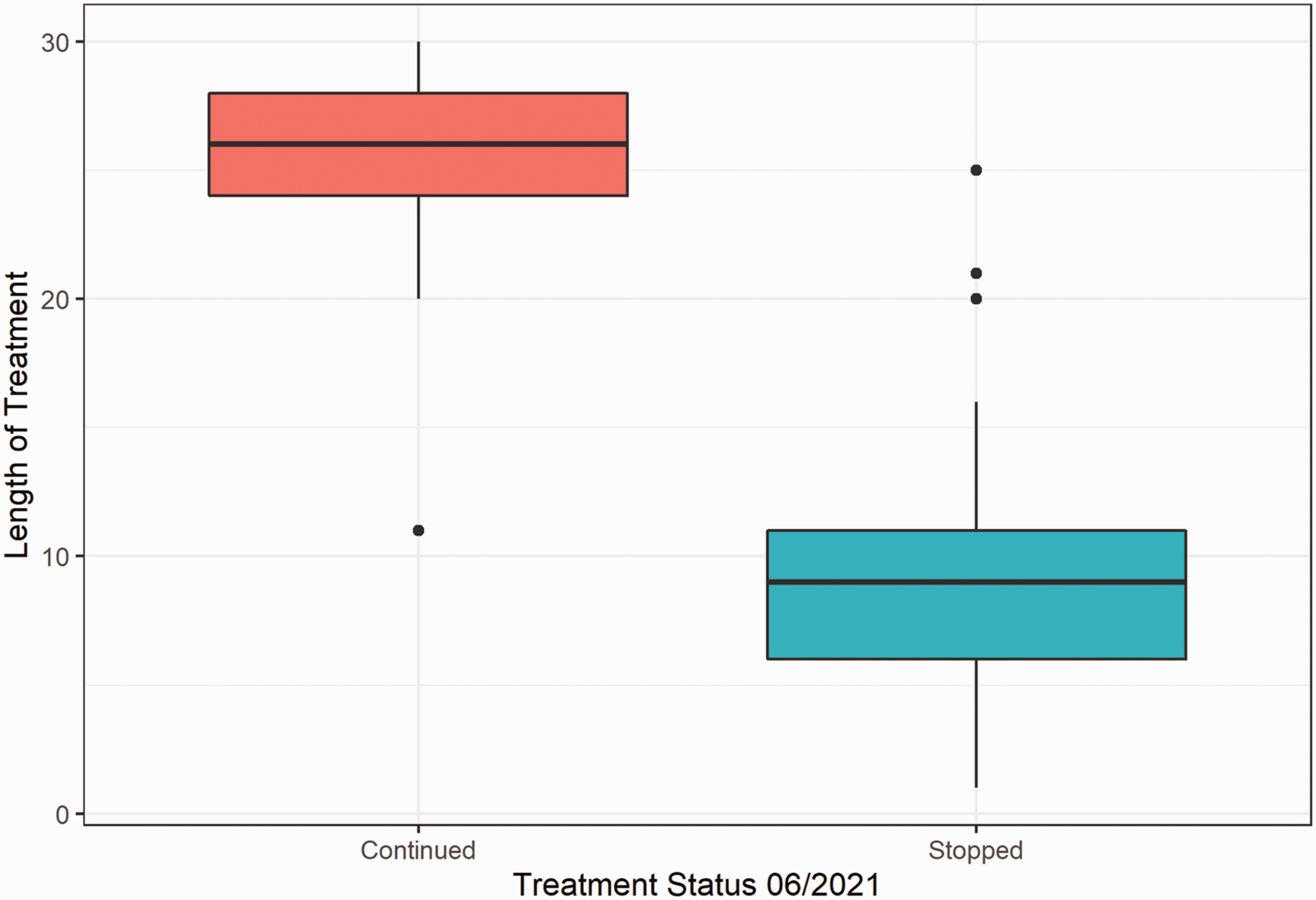
Boxplot comparing length of treatment between patients who stopped treatment vs patients who continued treatment.
Descriptive statistics of the length of treatment per treatment group.

Number of patients per treatment group (Continued or Stopped by June 2021) per time period. Group Zero was determined to be patients who stopped treatment between months 1 and 5, as these were outside normal follow-up. Individual group totals and cumulative totals also included.

Discussion
The classification of patients presenting with an NDPH clinical phenotype remains somewhat controversial. Historically, NDPH has been designated as a primary headache disorder. However, patients with ‘primary NDPH’ are often clinically indistinguishable from those with secondary NDPH, in which case the condition is precipitated by various clearly identified factors including trauma (typically mild direct head trauma, whiplash and blast injuries), intracranial vascular accidents and cranial surgery. It is important to reiterate that the overriding clinical feature, which is common to all of these abrupt onset, daily persistent headache patients is that they are generally refractory to conventional headache and migraine preventive treatments, especially if the condition has been present for at least two years (as is the case in 81 of our 82 patients). It is also important to note that a number of important and more sinister clinical conditions can cause persistent daily headache from onset and should be excluded. For example, cerebral venous sinus thrombosis (CVST), CSF leak, intracranial neoplasms, chronic meningitis, and chronic subdural haematoma can all present with a new daily persistent headache phenotype and are mimics of the more benign condition (7).
Several authors have therefore proposed a more clinically orientated view of abrupt onset headache with an NDPH phenotype. They have suggested that the clinical syndrome is described, rather than labelling the patient with a diagnosis, and importantly, have advised investigation of these patients comprehensively (6,7). Flu-like illness, extracranial surgery and stressful life events have all been frequently reported as being associated with or preceding primary NDPH (2), but it is not clear that there is a causative link, unlike the clearer correlation with trauma or cranial surgery in secondary abrupt onset persistent headache conditions.
In a small proportion of cases, primary NDPH and secondary abrupt onset headache disorders such as PPTH can spontaneously remit after one to two years. However, in general, this group of patients remain symptomatic for many years and can be very challenging for clinicians to manage. They are often regarded by many experts as the most treatment refractory of all headache disorders (14). Unfortunately, patients with these conditions routinely fail multiple conventional headache/migraine therapies. There are a few isolated reports that some cases remit while on preventive medication with topiramate (15) or gabapentin (15). Treatment with onabotulinumtoxin A has been shown to reduce the more severe headache frequency and overall headache severity in a small retrospective study of 19 patients with NDPH (14). In our cohort, three quarters of patients failed treatment completely or had an inadequate response to (typically only 30–40% subjective improvement) onabotulinumtoxin A prior to starting erenumab.
The development of the anti-CGRP monoclonal antibodies represents a new era for the treatment of episodic and chronic migraine (8). However, there is currently limited supporting evidence for the use of CGRP antagonists such as erenumab in primary NDPH or secondary clinical correlates (with or without a chronic migraine phenotype) (10,11,16).
The exact pathogenic mechanism(s) of NDPH is unknown. In addition, the question as to why many NDPH patients exhibit a migraine phenotype clinically is unclear. What is apparent is that there appears to be a clinical correlation between these two entities in a majority of these abrupt onset, daily from beginning, unremitting patients. Therefore, we believe that it is reasonable to postulate that some patients may respond to migraine specific preventive treatments, including anti-CGRP monoclonal antibodies (such as erenumab, fremanezumab and galcanezumab). One proposed link between these two conditions is the relationship between elevated pro-inflammatory cytokines (including TNF-alpha) and increased CGRP levels, which in turn may result in persistent headache. In a small study of 20 cases with NDPH, 19 patients were found to have high levels of TNF-alpha in CSF (17). It has been shown previously that TNF-alpha increases CGRP release in animal models (18,19). While reducing excess CGRP released as a consequence of elevated TNF-alpha may be one possibility, the paucity of evidence for a role of inflammation by anything less than a rather expansive definition (20) leaves much more to be done to understand NDPH.
The only data for the use of CGRP monoclonal antibodies in primary NDPH comes from a retrospective study on a paediatric population by Greene et al., who investigated 112 adolescents with refractory headaches treated with either erenumab, fremanezumab or galcanezumab (Table 7) (16). Among these 112 adolescents, a total of 18 had abrupt onset unremitting headache (12 had refractory primary NDPH and six had PPTH or a secondary NDPH phenotype) and the remainder had treatment resistant chronic migraine. The CGRP monoclonal antibody used for these 18 abrupt onset patients is not specified. Furthermore, the treatment outcome for the subgroups of primary NDPH and PPTH (18 patients in total) were not presented separately in this paper (16). It is noteworthy that in the above-mentioned study of resistant headache with predominantly chronic migraine patients (84%, n = 94/112), over two-thirds of the cases benefited from treatment with CGRP monoclonal antibodies, which is more consistent with adult studies in relatively non-refractory migraine (and slightly higher than our findings in treatment resistant CM).
PPTH is one of the most common causes of a secondary NDPH phenotype and is generally not responsive to treatment with conventional headache/migraine therapies. A 12-week open label study of erenumab treatment in 100 adults with PPTH following mild traumatic injury showed some reduction of moderate and severe headache, according to Ashina and colleagues (11). In addition, VanderEnde et al, reported a small case series of five women with PPTH who had a 51% reduction in headache intensity following treatment with erenumab (10). In contrast, a recently reported randomised, placebo-controlled trial failed to demonstrate an effect of fremanezumab 675 mg s/c monthly in adults with PPTH. It is noted that there were some complexities in interpreting the results of this study, including power issues with regard to recruitment and endophenotyping being an unresolved issue (12). A summary of previous studies using CGRP monoclonal antibodies in primary NDPH and secondary abrupt onset headache conditions (including PPTH) is provided in Table 7. Although it is difficult to directly compare these prior studies with our current report, it is clear from this table that there is very little published about this population of refractory patients in a medium-term, real-world setting. This is especially the case for primary NDPH.
Published reports of CGRP monoclonal antibody treatment of ‘primary’ NDPH, PPTH or other abrupt onset headache conditions.

NDPH: New Daily Persistent Headache; PPTH: Persistent Post-Traumatic Headache; PROM: Patient Reported Outcome Measures.
Our results indicate that only approximately one-third of our pooled patients with refractory ‘primary’ NDPH and secondary abrupt onset ‘constant from the beginning’ headache conditions respond to treatment with erenumab and remained on treatment for two years or more. Our findings demonstrate significant improvements in the quality of life of this 35% of very refractory cases, which for each patient is very meaningful as they often suffer without significant improvement for many years. However, it should be noted that the magnitude of this improvement is relatively modest when compared to our standard cohort of resistant CM patients treated with erenumab (13). For comparison purposes, approximately 55% of our resistant chronic migraine patients respond to erenumab (median treatment time of 25 months). In addition, it should be pointed out that there are also a small (but significant) proportion of ‘super-responders’ in our resistant CM patients, a feature which is not seen in the pooled refractory primary NDPH and secondary abrupt onset headache group reported here (13). It is important to point out specifically that none of our 82 abrupt onset patients described in this report have managed to achieve significant periods with ‘crystal clear days’ and they therefore have had several years of daily unremitting symptoms. In contrast, we do see prolonged periods of normality returning for a proportion of previously resistant and daily CM cases.
Primary NDPH and secondary abrupt onset persistent patients often record a significantly lower quality of life than episodic migraine, chronic migraine or healthy controls. This is clearly shown in the QoL scores for our intractable group of 82 patients (even when compared to our treatment-resistant longstanding CM patients). We therefore believe that it is clinically very worthwhile to treat these refractory ‘abrupt onset and daily from the beginning’ patients with a 9–12 month trial of a CGRP mAb, especially if there is a prior personal history of definite migraine or episodic/infrequent moderate to severe headache (probable migraine). QoL improved significantly (albeit modestly) in one-third of the cases in our small cohort, but this should be regarded as a (cautiously optimistic) positive outcome for patients with abrupt onset headache disorders, as they are typically such a treatment refractory group of headache conditions.
Limitations
First, this was an open label, retrospective audit, and it is therefore possible that there may be a significant placebo effect as we did not have a control group for comparison. This is coupled with potential for spontaneous improvement over the two-year period since treatment was started in 2018/2019. Arguing against a placebo effect, one would have expected the benefit of treatment to subside gradually after one to two years, and this does not appear to be the case in the 35% of responders. Secondly, the QoL questionnaires we used (HIT-6, MIDAS, MSQ) have been mainly developed for migraine. To our knowledge, there are no QoL questionnaires specifically developed for primary NDPH or secondary abrupt onset headache conditions which mimic NDPH. However, we considered that these QoL questionnaires will provide generally reliable data for patients with primary NDPH or secondary abrupt onset headache disorders as the phenotype in these cases is often indistinguishable from the clinical picture seen with our refractory chronic migraine patients; daily headache and associated neurological (migraine) symptoms with no completely normal (symptom free) or crystal-clear days. Thirdly, it should be noted that diagnosing primary NDPH and secondary abrupt onset headache disorders (such as PPTH) is clinically challenging, and therefore misdiagnosis cannot be completely excluded in a very small proportion of patients. However, we believe that the clinical diagnosis is correct in all patients included in this audit. Finally, another limitation is that we describe a very small group of intractable, abrupt onset daily from the beginning, unremitting patients. Our findings need to be replicated in much larger studies, taking care to stratify the patients by the presence or absence of an associated personal migraine biology or a strong family history in at least two first degree relatives.
Conclusions
Erenumab appears to have improved the quality of life of slightly more than one-third of our very refractory and very chronic migraine phenotype, primary NDPH and secondary abrupt onset unremitting headache patients (mainly PPTH with a migraine phenotype). This subgroup of patients has remained on treatment for two years or more, and generally the treatment is well tolerated. We therefore propose that erenumab is a treatment option for this group of intractable patients, especially in cases where there is a personal history of migraine or episodic moderate/severe headache (probable migraine) prior to the onset of their daily disorder.
Clinical implications
Erenumab showed improvement in quality-of-life measures in one-third of patients with intractable primary NDPH and secondary headache conditions with a similar clinical phenotype. Almost 80% of the patients who responded to erenumab had a prior history of episodic migraine or probable episodic migraine.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231182126 - Supplemental material for Two-year, real-world erenumab persistence and quality of life data in 82 pooled patients with abrupt onset, unremitting, treatment refractory headache and a migraine phenotype: New daily persistent headache or persistent post-traumatic headache in the majority of cases
Supplemental material, sj-pdf-1-cep-10.1177_03331024231182126 for Two-year, real-world erenumab persistence and quality of life data in 82 pooled patients with abrupt onset, unremitting, treatment refractory headache and a migraine phenotype: New daily persistent headache or persistent post-traumatic headache in the majority of cases by Alina Buture, Esther M. Tomkins, Arif Shukralla, Emma Troy, Katie Conaty, Esther Macken, Roisin Lonergan, Jane Melling, Niamh Long, Kieran Birrane, Eamonn Shaikh, Peter J. Goadsby and Martin H. Ruttledge in Cephalalgia
Footnotes
Acknowledgements
We would like to thank our neurology administration teams for their support during this audit. We thank Novartis for the opportunity to treat our more chronic migraine patients through their Managed Access Program (MAP). We would also like to thank our patients for completing the questionnaires.
Authors’ contributions
Concept and design: MHR, EMT; Data acquisition: MHR, AB, EMT, ET, ES, JM, NL, RL; Clinical diagnosis and review of charts/patients: PJG, AB, EMT, ES, RL, MHR; Data analysis: PJG, AS, KB, MHR; Interpretation of data: All authors; Drafting of the manuscript: AB, PJG, ET, KB, MHR; Critical revision of the manuscript for important intellectual content: All authors; Supervision: MHR.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AB: has received personal fees and/or honoraria from Novartis and Eli Lilly. EMT: received personal fees and/or honoraria from Allegran/Abbvie, Novartis, Pfizer, Teva, and Lundbeck. AS, ET, KC, EM, RL, JM and NL have no conflict to declare. ES: has received honoraria from Teva. KB: has received personal fees from Novartis. PJG: reports, over the last 36 months, a grant from Celgene, and personal fees from Aeon Biopharma, Allergan/Abbvie, Amgen, CoolTech LLC, Dr Reddys, Eli-Lilly, Epalex, Impel Neuropharma, Lundbeck, Novartis, Pfizer, Praxis, Sanofi, Satsuma, Shiratronics, Teva Pharmaceuticals, and Tremeau, and personal fees for advice through Gerson Lehrman Group, Guidepoint, SAI Med Partners, Vector Metric, and fees for educational materials from CME Outfitters, and publishing royalties or fees from Massachusetts Medical Society, Oxford University Press, UptoDate and Wolters Kluwer, and for medicolegal advice in headache, and a patent magnetic stimulation for headache (No. WO2016090333 A1) assigned to eNeura without fee. MHR: has received personal fees and/or honoraria from Allegran/Abbvie, Grunenthal, Eli-Lilly, Novartis, Teva, Lundbeck and MSD.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The statistical analysis for this manuscript was made possible and supported by an educational grant from Novartis. There was no other funding for this project.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
