Abstract
Abstract
Background
The connection between migraine aura and headache is poorly understood. Some patients experience migraine aura without headache, and patients with migraine aura with headache commonly experience milder headaches with age. The distance between the cerebral cortex and the overlying dura mater has been hypothesized to influence development of headache following aura. We tested this hypothesis by comparing approximated distances between visual cortical areas and overlying dura mater between female patients with migraine aura without headache and female patients with migraine aura with headache.
Methods
Twelve cases with migraine aura without headache and 45 age-matched controls with migraine aura with headache underwent 3.0 T MRI. We calculated average distances between the occipital lobes, between the calcarine sulci, and between the skull and visual areas V1, V2 and V3a. We also measured volumes of corticospinal fluid between the occipital lobes, between the calcarine sulci, and overlying visual areas V2 and V3a. We investigated the relationship between headache status, distances and corticospinal fluid volumes using conditional logistic regression.
Results
Distances between the occipital lobes, calcarine sulci and between the skull and V1, V2 and V3a did not differ between patients with migraine aura with headache and patients with migraine aura without headache. We found no differences in corticospinal fluid volumes between groups.
Conclusion
We found no indication for a connection between visual migraine aura and headache based on cortico-cortical, cortex-to-skull distances, or corticospinal fluid volumes overlying visual cortical areas. Longitudinal studies with imaging sequences optimized for measuring the cortico-dural distance and a larger sample of patients are needed to further investigate the hypothesis.
Introduction
Approximately 30% of patients with migraine experience migraine with aura (MA), i.e., transient, fully-reversible neurological deficits, most frequently visual symptoms, which may or may not be accompanied by headache (1,2). Patients with MA frequently report episodes of migraine without aura (MO) as well (3). How aura and headache are connected is unknown, but based on animal models it is hypothesized that cortical spreading depression (CSD), the electrophysiological substrate of migraine aura, may directly trigger headache by activating trigeminal nociceptive fibers in the dura mater through the release of noxious substances including potassium and nitric oxide (4,5). A recent positron emission tomography study suggested that inflammation occurs in meninges and calvarial bone overlying visual cortical areas following migraine with visual aura, supporting the notion of CSD-induced meningeal nociception (6).
In clinical experience, patients who initially report migraine aura with headache (MAWH) may with increasing age experience milder headaches accompanying aura and attacks of migraine aura without headache (MAWOH) (7). Theoretically, this clinical course could be explained by age-related atrophy of the brain, in which the distance between the cortical surface and the dura mater increases. This could in turn reduce the propensity for CSD to activate dural trigeminal nociceptive fibers due to the increased distance. To investigate this hypothesis, we included patients with migraine with aura who never experience headache in relation to their aura and age-matched patients who always experience aura-related headache. Based on magnetic resonance images (MRI) acquired at 3.0 tesla field strength, we approximated distances between cortical visual areas and overlying skull as a proxy for cortico-dural distance. We hypothesized that patients with MAWOH have greater cortico-dural distances compared to age-matched patients with MAWH. We additionally measured volumes of corticospinal fluid (CSF) overlying visual cortical areas as secondary analyses.
Methods
We based our analyses on data from the population-based Women with Migraine with Aura Neuroimaging (WOMAN) study, of which details and imaging data are previously published (8–10). In brief, female twins born from 1931 to 1982 were recruited using the Danish Twin Registry (11). Subjects received a baseline questionnaire on health and lifestyle issues between February 2011 and April 2014. Eligible responders to a questionnaire screening for migraine (12) were telephone interviewed by physicians who classified each participant’s headache disorders according to the International Classification of Headache Disorders, 3rd edition beta version (13). Detailed eligibility criteria have been presented previously (8). Eligible subjects were invited to participate in the MRI part of the study at a single center in Copenhagen, Denmark.
As part of the clinical interview, participants with migraine with aura reported whether they had 1) aura always followed by headache, 2) aura sometimes followed by headache, or 3) aura never followed by headache. For the present study, we investigated participants reporting migraine aura attacks never followed by headache (cases) and compared these to age-matched participants reporting aura always followed by headache (controls). No comparisons within twin pairs were carried out and zygosity of the twins was not taken into account.
MRI
All participants underwent whole-brain MRI using the same 3.0 T MRI scanner (Siemens Magnetom Verio) equipped with a 32 channel receive head coil. Structural imaging included a T1-multi-echo 3D FLASH sequence which was acquired to enable quantitative estimation of whole-brain T1 (14) with TR = 18.7 ms, TE = 2.20, 4.70, 7.20, 9.70, 12.20 and 14.70 ms, flip angle 20°, FOV = 256 mm, 176 sagittal slices. However, as the segmentation procedures applied are optimized for sequences with regular T1-weighting an image with contrast equivalent to a typical T1-weighted sequence was produced by averaging across the 6 echoes in the T1-multi-echo sequence. T2-weighted images were obtained using a 3D turbo spin echo sequence with the following parameters: TR = 3200 ms, TE = 409 ms, FOV = 256 mm, 176 sagittal slices. All images were assessed by a consultant radiologist with experience in neuroradiology, blinded to headache diagnoses and clinical data.
Data analysis
The analyses were done using a combination of the FreeSurfer (15) image analysis suite v.6.0.0 (http://surfer.nmr.mgh.harvard.edu) and the complete head anatomy reconstruction (CHARM) method (16).
For the primary analysis, we calculated distance-based measures between the two hemispheres (left and right) for the pericalcarine and occipital cortical regions, and between the inner skull surface and each of the visual areas V1, V2, and V3a. The distances were calculated based on the automated segmentation and surface reconstruction output from CHARM and FreeSurfer respectively. The distances were calculated automatically using an in-house Python script, which measured the minimum distance from each node on the FreeSurfer generated to the closest point on the skull volume generated by CHARM (Figure 1). For the cortico-cortical distances, the minimum distance from each node on the FreeSurfer pial surface within the cortical region-of-interest (ROI) in one hemisphere to all nodes in the same ROI on the other hemisphere was calculated, and the same process was repeated on the other hemisphere, i.e., we used a symmetric distance, and finally a histogram of the node-wise minimum distances was constructed. From the histogram we extracted the fifth percentile value as an estimate of a robust minimum distance between the cortical areas. For the cortex-to-skull distances, the minimum distance from each node on the FreeSurfer pial surface within the visual area ROI to the inner skull surface was calculated, and the same histogram analysis was repeated to estimate a robust minimum distance.

Example of distance measurement between cortical surface (outlined in blue) and the skull (yellow). (a) Overview. Nodes (white dots) were automatically defined using the FreeSurfer map and (b) For each cortical node the distance was measured to all nodes in the skull (grey lines), and the smallest distance measured was extracted (in this case the green line).
For the secondary analyses, the CSF volumes between the two occipital lobes, between the calcarine sulci and overlying cortical areas V2 and V3a were determined. The cortical CSF volumes within the occipital area were extracted by first masking the CHARM CSF segmentation with a region-of-interest limited in the superior-inferior and left-right directions by the extent of the occipital cortex, defined by the FreeSurfer cortical labels cuneus, lateraloccipital, lingual, and pericalcarine, and in the posterior-anterior direction by the superior and inferior sagittal sinuses extracted from the CHARM segmentation. To ensure that the volume masks included only the CSF between the occipital lobes, and not in the ulci, the masks were manually corrected by one author (AVT), based on T2-weighted images, using as boundaries the superior sagittal sinus, the straight sinus, the parieto-occipital sulcus and the inferior borders of the occipital lobes (Figure 2). As a quality control of the manual corrections, five randomly selected samples were evaluated by another author (OP).

Midline sagittal section of MRI T2-map. Cerebrospinal fluid between the occipital lobes (“midline CSF”) is marked in red.
OP inspected the five random subjects independently and subsequently OP and AVT had a joint meeting to discuss the manual segmentations by AVT. OP and AVT agreed that no further corrections were needed. Both AVT and OP were blinded to the case/control-status of the subjects.
The CSF volume between the calcarine sulci was derived by restricting the midline volume to the FreeSurfer pericalcarine label. Additionally, the volumes of the CSF directly overlying visual areas V2 (from FreeSurfer’s Brodmann area map) and V3a (from the PALS-B112 atlas) bilaterally was automatically extracted from the CHARM cortical CSF segmentation by summing the volume of those CSF voxels that overlapped with dilated cortical masks of the V2 and V3a areas. The cortical masks were created by mapping the surface labels to voxels using FreeSurfer’s mri_label2vol method, and the masks were further dilated three times using a fully-connected (26 neighbors) kernel.
Statistics
Statistics were performed using R version 4.1.0 (©The R Foundation for Statistical Computing). Baseline characteristics were compared between cases and controls using unpaired t-tests for continuous variables and chi-squared tests for categorical variables. p-values <0.05 were considered significant.
For the primary analysis we fitted conditional logistic regression models for each of our distances of interest (calcarine-calcarine, occipital-occipital, V1-skull, V2-skull and V3a-skull) using case status as dependent variable and volumes of interest as independent variables. We further included as independent variables age, total intracranial volume and total CSF volume as these were presumed a priori to correlate positively with the distances of interest. For the secondary analyses we fitted similar conditional logistic regression models for each of our volumes of interest (CSF in the midline between the calcarine sulci, CSF in the midline between the occipital lobes, CSF overlying V2, and CSF overlying V3a) while controlling for the same variables as in our primary analyses. We further calculated CSF volumes of interest normalized to intracranial volume and compared these volumes between cases and controls using unpaired t-tests. For regression models and for t-tests comparing CSF volumes, we did not correct for multiple comparisons due to the exploratory nature of the study, and a p-value < 0.05 was considered significant.
Results
The original dataset included MRI data from 172 twins with migraine with aura, 34 co-twins and 139 control subjects. All subjects were female. Twelve scans were excluded due to technical issues resulting in 333 available scans (166 twins with MA, 30 co-twins and 137 controls). Twelve of these participants had exclusively MAWOH and were included in the present study. We further aimed to include four participants with MAWH as controls for each MAWOH case. We matched MAWH to MAWOH participants according to age by five-year strata and were able to include 45 age-matched MAWH participants. Three twin pairs were included, one pair with a twin with MAWOH and a co-twin with MAWH and two pairs where both twins had MAWH.
Baseline characteristics of patients with MAWOH vs MAWH are provided in Table 1. MAWOH participants had a shorter disease duration (17.8 vs 28.1 years) but otherwise the two groups did not differ with regard to age, headache, aura characteristics or lifestyle factors.
Baseline characteristics of study participants.

Baseline characteristics of study subjects. Values are means ± SDs for continuous variables and n/n for categorical variables. *Active diagnosis is defined as occurrence of at least one episode within the last 12 months. MAWH, Migraine aura with headache; MAWOH, Migraine aura without headache.
The measured distances are presented in Table 2. Boxplots of distances are shown in Figure 3(a) to (e). For our primary analyses, we found no statistically significant distance differences for any of the cortical regions or between cortex and skull between cases with MAWOH and their controls with MAWH. None of the co-variates (CSF volumes of interest, age, total intracranial volume, and total CSF-volume) significantly influenced odds ratios of case status.
Distances between the two hemispheres and between the inner skull surface and the visual areas.
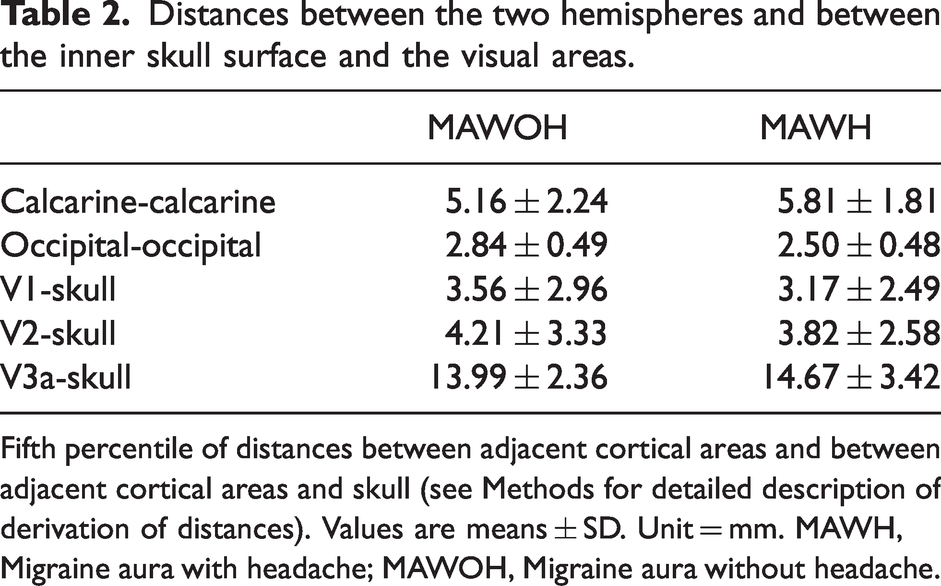
Fifth percentile of distances between adjacent cortical areas and between adjacent cortical areas and skull (see Methods for detailed description of derivation of distances). Values are means ± SD. Unit = mm. MAWH, Migraine aura with headache; MAWOH, Migraine aura without headache.

Boxplots of distribution of fifth percentile distances between calcarine sulci (a), occipital lobes (b), visual area V1 and skull (c), visual area V2 and skull (d), and visual area V3a and skull (e) in patients with migraine aura without headache (MAWOH) and patients with migraine aura with headache (MAWH).
Secondary analyses – distances
As our distances of interest are hypothesized to correlate with age, we performed simple linear regression on our distances as a function of age in our subset, and in none of the models was age significantly associated with the distance of interest (p-values: Calcarine-calcarine: 0.40; occipital-occipital: 0.17; V1-skull: 0.40; V2-skull: 0.34; V3a-skull: 0.86). Pearson’s correlation coefficients between age and distances of interest were: Calcarine-calcarine: 0.11; occipital-occipital: 0.19; V1-skull: 0.11; V2-skull: 0.13; V3a-skull: 0.02.
Secondary analyses – volumes
The measured volumes are presented in Table 3. Boxplots of CSF volumes between the calcarine sulci, between the occipital lobes, overlying V2, and overlying V3a are additionally shown in Figure 4(a) to (d). None of the volumes or co-variates were significantly associated with case status.
Volumes of intracranial CSF (total and overlying specific areas) and brain.

Intracranial CSF volumes. V2, V3a: CSF volumes overlying cortical areas V2 and V3a, respectively. Values are means ± SD. Unit = mm3. CSF, Cerebrospinal fluid; MAWH, Migraine aura with headache; MAWOH, Migraine aura without headache.; TIV, Total intracranial volume; TBV, Total brain volume.

Boxplots of distribution of cerebrospinal fluid (CSF) volumes in the midline between the occipital lobes (a), overlying visual area V2 (b), overlying visual area V3a (c), and between the calcarine sulci (d) in patients with migraine aura without headache (MAWOH) and patients with migraine aura with headache (MAWH).
We performed unpaired t-tests comparing CSF volumes of interest between cases and controls. The groups did not differ significantly for any volume.
Post-hoc analyses
As the investigated models did not predict the investigated CSF volumes, we carried out a range of post hoc analyses to test whether our results might in part be explained by insufficient power. As age would be expected to correlate with total brain volume, we decided to investigate if this correlation was present in the entire WOMAN dataset of 333 available scans and in our dataset of 57 subjects regardless of aura status. We performed simple linear regression on these variables. Age was significantly correlated with total brain volume for both the full dataset and our subset (p-values 7.2 * 10−4 and 0.0046 respectively). We additionally performed simple linear regression on our volumes of interest as a function of age in our subset, and in none of these cases was age significantly associated with the volumes of interest (p-values 0.96, 0.64, 0.46 and 0.86 for midline, V2, V3a and calcarine sulci, respectively). Pearson’s correlation coefficients between age and volumes of interest were −0.0066, −0.063, −0.10 and 0.02 for midline, V2, V3a and calcarine sulci, respectively.
As we found disease duration to differ between cases and controls (mean 17.8 years and 28.1 years, respectively, see Table 1), we repeated our regression analyses adding disease duration as an independent variable. For all analyses, disease duration was associated with case status, while our measured volumes and distances were not associated with case status (data not shown).
Discussion
We measured cortico-cortical and cortex-to-skull distances for pre-defined early cortical visual areas and hypothesized that patients with migraine with visual aura but no subsequent headache would have greater distances compared to patients with migraine with visual aura accompanied by headache. We did not find any association between distances and headache status when controlling for other intracranial volumes, and thus our findings do not corroborate the hypothesis. Neither were CSF volumes overlying pre-defined cortical visual areas associated with case status. Controlling for disease duration did not affect these findings. While there may be no true association between the distances and volumes in question and headache status, it is possible that our failure to demonstrate an association relies on insufficient power.
The trigeminovascular system is known to play a central role in migraine pathophysiology (17). Trigeminal projections innervate several intracranial structures including the dura mater and dural vessels as well as pial vessels and possibly the pia mater itself to a smaller extent (18). Mechanical stimulation of dural blood vessels and large cerebral vessels has been shown to cause a sensation of deep, throbbing pain accompanied by nausea, not unlike the typical pain experienced by migraine patients during attacks (19). Additionally, mechanical stimulation of the tentorium cerebelli and other structures adjacent to occipital cortex may lead to pain referred to the frontal and periorbital regions, which are frequently involved in migraine attacks.
It has been shown in animal models that CSD can activate nociceptive fibers in the dura mater. CSD involves the release of many substances, including potassium ions, adenosine triphosphate, glutamate, nitrogen oxide, and arachidonic acid metabolites. These substances may activate trigeminal nerve endings, likely in a concentration dependent manner. As the concentration of a diffusing solute falls off by a factor 1/radius, the distance from the cortex to the trigeminal fibers embedded in the dura is possibly of relevance in the sequential hypothesis of migraine development. As most migraineurs with aura experience visual aura, visual areas and adjacent structures are relevant to investigate further in the future. We note that all but one out of 57 subjects in our study reported visual aura.
The sequential hypothesis of migraine initiation, in which CSD leads to migraine headache, does not account for migraine attacks in which headache precedes or immediately accompanies aura (1). It is possible that the wave of CSD in these attacks originates in non-eloquent areas, initiates mechanisms that downstream lead to headache, and then spreads to eloquent areas such as visual or somatosensory cortex. However, a non-sequential model should also be considered, in which some unknown factor leads to both aura and headache (20).
Apart from direct activation of trigeminal nociceptive fibers by CSD, an association between migraine with visual aura and inflammation in meninges and bone overlying the occipital lobes has been recently reported (6). This inflammation might be relevant to headache pathogenesis in sensitizing trigeminal fibers and increasing susceptibility to headache. More studies in this area are required.
While CSD may activate trigeminal nociceptive fibers peripherally, there is also evidence to suggest that CSD can activate second-order trigeminal neurons via central mechanisms (21). If CSD does indeed trigger nociception via central mechanisms, we would not expect the size of the subarachnoid space to influence this.
Strengths and weaknesses
A main strength of this study was the inclusion of clinically well-characterized and well-matched patients with migraine. The migraine diagnosis was established through telephone interviews with physicians based on internationally acknowledged criteria and all subjects were scanned using the same MRI scanner at a single center.
A significant shortcoming of our study is the sample size of 12 cases, which was a natural limitation of the data set used. We suggest that future studies include more patients to increase power.
While all subjects had a history of migraine with aura, we note that only 8/12 cases and 25/45 controls reported active migraine with aura (i.e., at least one aura episode within the last 12 months). Subjects with inactive migraine might not be pertinent to the analyses. Additionally, we did not have any information about natural evolution of aura in each subject. Even young patients with migraine with aura may never experience headache in relation to aura symptoms, and underlying pathophysiology in these patients may be different from patients with MAWH as well as patients progressing from MAWH to MAWOH.
Our data included three pairs of twins of whom two were concordant for migraine type (MAWH) and one was discordant. Although observations in a pair of twins cannot formally be considered independent, we considered that this would have negligible effects on the statistical analyses.
In conclusion, we found no differences in cortico-cortical distances, cortex-to-skull distances, or CSF volumes overlying visual cortical areas between patients with migraine aura with and without headache. We suggest that future studies are needed, ideally with longitudinally acquired imaging data in patients with migraine with aura, and with imaging protocols optimized for measuring the cortico-dural distance. Such studies should include detailed aura- and headache phenotyping to accurately assess the clinical relation between migraine aura and headache.
Article highlights
The connection between migraine aura and headache is incompletely understood. The distance between the cortical surface and the dura mater has been implicated in this connection. Using MRI in cases with migraine aura without headache and controls with migraine aura with headache we were unable to corroborate the role of the subarachnoid space in migraine pathophysiology.
Footnotes
Ethics approval and patient consent
The WOMAN study was approved by the Ethics Committee of the Region of Southern Denmark and the Danish Data Protection Agency. Written informed consent was obtained from all participants prior to any study procedures.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AVT, DG, KOK, KM and AT report no disclosures. OP is supported by the Lundbeck foundation (R360-2021-395). MA reports personal fees from AbbVie, Amgen, Eli Lilly, Lundbeck, Novartis, Pfizer and Teva. MA participated in clinical trials as the principal investigator for AbbVie, Amgen, Eli Lilly, Lundbeck, Novartis and Teva. MA received a research grant from Lundbeck Foundation, Novo Nordisk Foundation, and Novartis. MA was supported by the Lundbeck Foundation Professor Grant (R310‑2018–3711). MA has no ownership interest and does not own stocks of any pharmaceutical company. MA serves as associate editor of Cephalalgia, associate editor of the Journal of Headache and Pain, and associate editor of Brain.
HRS has received honoraria as speaker from Sanofi Genzyme, Denmark, Lundbeck AS, Denmark, and Novartis, Denmark, as consultant from Sanofi Genzyme, Denmark, Lophora, Denmark, and Lundbeck AS, Denmark, and as editor-in-chief (Neuroimage Clinical) and senior editor (NeuroImage) from Elsevier Publishers, Amsterdam, The Netherlands. He has received royalties as book editor from Springer Publishers, Stuttgart, Germany and from Gyldendal Publishers, Copenhagen, Denmark.
AH has received honoraria for lecturing and/or writing from Allergan, Novartis, Teva, Lundbeck, and Eli Lilly. He serves as Assistant Editor of Headache.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: HRS holds a five-year professorship in precision medicine at the Faculty of Health Sciences and Medicine, University of Copenhagen which is sponsored by the Lundbeck Foundation (Grant Nr. R186-2015-2138).
