Abstract
Introduction
It has not been established if migraine headache and migraine aura share common pathophysiological mechanisms. Sildenafil, a phosphodiesterase-5 inhibitor, causes cGMP accumulation and provokes migraine-like headache in patients with migraine without aura. We investigated if sildenafil induced aura and migraine-like headache in patients with migraine with aura.
Methods
In a randomized, double-blinded, placebo-controlled crossover study, 16 patients with migraine with aura (of whom 11 patients exclusively had attacks of migraine with aura) received 100 mg sildenafil or placebo on two separate days. The development, duration, and characteristics of aura and headache were recorded using a questionnaire. The primary outcome was the incidence of migraine aura.
Results
Aura symptoms were induced in three patients (19%) after sildenafil and none after placebo (P < 0.001). After administration of sildenafil, 12 patients (75%) developed headache compared with two patients (12.5%) after placebo (Fisher’s exact test, P < 0.001). The headache in nine patients (56%) after sildenafil and one patient (6%) after placebo fulfilled the criteria for migraine-like attacks (Fisher’s exact test, P = 0.002). All patients, who fulfilled the criteria for migraine-like attacks, reported that the attack mimicked the headache phase during their usual migraine attacks.
Discussion
Sildenafil have a moderate migraine headache-inducing and a modest aura-inducing effect in patients with migraine with aura, even in those who exclusively experienced attacks of migraine with aura in their spontaneous attacks. These findings suggest that accumulation of cGMP by PDE5-inhibition do not play any significant role in the initiation of migraine aura and refute the hypothesis of sildenafil being a tool for pharmacological provocation of this phenomenon. These findings further support dissociation between the aura and the headache phase.
Introduction
During the past three decades, human migraine models have been developed to elucidate the initiating mechanisms of migraine. Studies of experimental migraine induction have revealed important signaling pathways involved in the initiation of migraine headache, including those mediated by the second messenger molecules cyclic adenosine monophosphate (cAMP) and cyclic guanosine monophosphate (cGMP) (1–3). However, several outstanding questions remain to be answered: Do migraine aura and migraine headache share common pathophysiological mechanisms? Can cortical spreading depression (CSD), a slowly propagating wave of strong neuronal and glial depolarization presumed to be the underlying pathophysiological mechanism of migraine aura (4–7), be induced experimentally in human? While a great deal of effort has been spent in the development of a safe and effective human model of migraine aura, such a model has not been established yet, although recently it has been suggested that levcromakalim, an ATP-sensitive potassium channel opener, may be a novel aura-inducing substance in humans (8–11). Given the unpredictable and short-lasting nature of migraine aura, the development of a human migraine aura model is warranted to fully understand the role of aura in migraine pathophysiology.
Sildenafil, an inhibitor of cGMP-degrading phosphodiesterase 5 (PDE5), used for pulmonary arterial hypertension and erectile dysfunction, induces headache in healthy individuals and migraine-like headache in patients with migraine without aura (1,12). Interestingly, in a case report, the selective PDE5-inhibitor, tadalafil, consistently induced typical visual aura without headache 72 hours after administration in a patient with no history of migraine or significant visual disturbances (13). It is of great interest to investigate if cGMP accumulation can induce migraine-like headache, and, more importantly, migraine aura in patients with migraine with aura (MA) to disclose a possible common pathway of the aura and migraine headache initiation and to determine if such a pathway involves the signaling molecules known to be important in the induction of migraine-like headache.
In the present randomized, double-blinded, placebo-controlled crossover study, we investigated if sildenafil induced aura and migraine-like headache in patients with MA.
Methods
Participants
In total 16 individuals diagnosed with MA according to the International Headache Society (IHS) Criteria (14) were included in the study. Participants were recruited through specific patient recruitment websites and among the hospital staff from March 2016 to June 2019. Exclusion criteria were: age <18 and >50 years; weight <60 and >95 kg; any other type of headache, except tension-type headache ≤4 days per month; attack frequency of MA less than 2 and more than 12 every year; history of cardio- or cerebrovascular diseases; hypertension (systolic blood pressure >140 mmHg or diastolic pressure >90 mmHg); hypotension (systolic blood pressure <90 mmHg or diastolic pressure <50 mmHg); any daily intake of medication, including prophylactic migraine treatment, except oral contraceptives; pregnancy and lactation. A full medical history, physical examination, electrocardiography, and vital signs (heart rate and blood pressure) were obtained on the day of recruitment. All participants provided written informed consent before inclusion.
This study was approved by the Ethics Committee of Copenhagen (H-15008491), the Danish Data Protection Agency, and the Danish Medicines Agency and conducted in accordance with the Declaration of Helsinki of 1964, as revised in 2008. In addition, this study was registered at ClinicalTrials.gov (NCT02795351).
Design and experimental protocol
In this randomized, double-blinded placebo-controlled crossover study, patients suffering from MA were randomly assigned to receive 100 mg sildenafil or placebo (lactose, potato starch, gelatin, magnesium stearate, talcum) in an opaque gelatine capsule on 2 experimental days separated by at least 5 days. The Capital Region Hospital Pharmacy performed the balanced randomization, in which half of the participants received sildenafil and the other half placebo on the first day and crossed over on the second day.
On both study days, the participants arrived at the clinic at 8 am, free from headache and fasting for at least 8 h. Intake of medication, except oral contraceptives was not allowed 12 h before the start of the study. The experiment was postponed if participants experienced headache on the study day before drug administration or had suffered from a migraine attack within 72 h prior to the start of the study. All measurements were performed in a quiet room with a temperature of 22–24°C. After 5 minutes of rest, blood pressure and heart rate were measured, and an electrocardiogram was performed. Participants then received either 100 mg sildenafil or placebo; aura and headache intensity and characteristics were recorded every 15 min until 120 min after drug administration. Subsequently, a second measurement of blood pressure, heart rate, and an electrocardiogram was performed.
Aura, headache and migraine-like attack criteria
The development, duration, and characteristics of visual, sensory, or speech disturbances were recorded using a validated diagnostic aura diary (15). Headache intensity was scored on a verbal scale from 0 to 10; a score of 1 represented a very mild headache (including a feeling of pressing or throbbing), 5 a headache of moderate intensity, and 10 the worst headache imaginable (16). Headache characteristics and accompanying symptoms were also recorded as well as premonitory symptoms (defined as symptoms preceding and forewarning of a migraine attack by 2–48 h, occurring before the aura in MA and before the onset of pain in migraine without aura) (14), adverse events, and if the reported headache mimicked their spontaneous migraine attacks. After discharge from the hospital, participants continued these recordings by a self-administered questionnaire every hour until 14 h after drug administration or until bedtime. Participants could treat headache with their usual migraine treatment or with over-the-counter pain medication. Because few patients completed the questionnaire for the 14th hour, we restricted follow-up to 13 h.
Experimentally induced migraine by pharmacological intervention cannot fulfill strict IHS criteria (2,17). Therefore, we defined a migraine-like attack, induced 0–14 h after administration of sildenafil, if one of the two following criteria was met:
Headache fulfilling criteria C and D for migraine without aura according to the IHS criteria: C. Headache has at least two of the following characteristics: unilateral location; pulsating quality; moderate to severe pain intensity (equal to ≥4 on the verbal scale); aggravation by cough (in-hospital period) or causing avoidance of routine physical activity (out-hospital period). D. During headache, at least one of the following associated symptoms: nausea and/or vomiting; photophobia and phonophobia.
Headache described as mimicking usual migraine attack and treated with acute migraine medication.
Statistical analysis
All values are presented as means with standard deviation unless otherwise stated. Sample size was chosen based on the results of previous similar headache induction studies in migraine patients (1,2,17–20). The primary outcome was incidence of aura. Secondary outcomes were incidence of migraine-like attacks, headache intensity, and incidence of headache. The incidence of aura, migraine-like attacks, and headache after sildenafil and placebo was compared using Fisher’s exact test. Area under the curve, calculated according to the trapezium rule (21), was applied as a summary measure to analyze the difference between treatments. Differences in area under the curve for headache intensity were tested with the Wilcoxon signed rank test. All statistical analyses were performed with SAS statistical software (SAS 9.4, SAS Institute, Cary, North Carolina, USA). A two-sided p-value <0.05 was considered statistically significant.
Data availability
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
Results
In total, 16 patients (14 women, two men; mean age 26 [range 19–42]) were included in the study (Table 1). All patients completed both study days. In their spontaneous attacks, 11 patients experienced aura symptoms preceding every headache phase of migraine, and five patients experienced both MA and migraine without aura (MO) attacks. The frequency of spontaneous MA attacks ranged from 2 to 12 per year; all patients usually experienced visual aura. In addition, four patients also experienced sensory aura and two patients had symptoms of aphasia.
Baseline characteristics of study population.

NSAID, non-steroidal anti-inflammatory drugs; –, no treatment.
Aura
Patient 3 reported visual aura with left-sided flickering light spots 1 hour after intake of sildenafil. The aura was accompanied by headache which fulfilled the criteria for migraine-like attacks. The aura symptoms lasted for 6 hours, and the patient described these symptoms as being similar to those of a spontaneous MA attack. The day after sildenafil intake, outside the study observation period, the patient experienced a left-sided blind spot, which lasted for 14 days and was deemed unrelated to sildenafil intake.
Patient 4 reported visual aura with bilateral flickering light spots 3 hours after sildenafil intake. The symptoms lasted approximately 15 min. The aura was accompanied by headache which fulfilled the criteria for migraine-like attacks. Apart from the duration of the period between termination of the aura phase and onset of headache (usually 30 min), the patient described these symptoms as being similar to those of a spontaneous MA attack.
Patient 13 reported visual aura with right-sided flickering light spots 2.5 hours after sildenafil intake. The symptoms lasted for approximately 5 hours. Headache was reported 10 min after onset of the aura symptoms and fulfilled the criteria for migraine-like attacks. Apart from the duration of aura symptoms (usually less than one hour), the patient described these symptoms as being similar to those of a spontaneous MA attack.
Headache, migraine-like attacks, and systolic blood pressure
The clinical characteristics of headache and associated symptoms during the 13 h observation period after administration of sildenafil and placebo are shown in Table 2. Figure 1 displays the headache intensity during the 13 h observation period after administration of sildenafil and placebo. In total, 12 patients (75%) experienced headache after intake of sildenafil compared with two patients (12.5%) after placebo (Fisher’s exact test, P < 0.001). The headache in nine patients (56.3%) after sildenafil and one patient (6.3%) after placebo fulfilled the criteria for migraine-like attacks (Fisher’s exact test, P = 0.002).
Clinical characteristics of headache and associated symptoms during the 13 h observation period in patients with MA after sildenafil and placebo.

+, present; –, not present or not applicable.aLocalization/quality (throb = throbbing; pres=pressing)/intensity/aggravation (by cough during in-hospital phase or by movement during out-hospital phase).
bNausea/photophobia/phonofobia.

Individual (thin lines) and median (thick line) headache intensity during the 13 h observation period in patients with MA after sildenafil and placebo.
After sildenafil administration, the median time: i) to fulfilment of criteria for migraine-like attacks was 5 h (range 2–6), ii) for the headache to mimic a spontaneous migraine headache according to patients was 3 h (range 1–6), and iii) to peak headache was 4.5 h (range 3–7).
The area under the curve for headache intensity during the observation period was significantly larger after sildenafil compared with placebo (Wilcoxon signed rank test, P < 0.0001). Likewise, the median peak headache intensity was significantly higher after sildenafil (5, range 0–8.5) compared with placebo (0, range 0–6) (Wilcoxon signed rank test, P = 0.0003).
During the observation period, four patients experienced neck stiffness after sildenafil compared with one patient after placebo. Four patients also reported unusual tiredness after sildenafil compared with two patients after placebo. These symptoms did not fulfill the criteria for a premonitory symptom in any of the patients (i.e. these either were not followed by a migraine attack or did not precede a migraine attack by at least 2 h). No patients reported adverse events such as palpitations, flushing, or dizziness on either study days.
On the sildenafil day, six patients took their usual medication to treat their headache. Three patients responded well to their usual migraine treatment. The three patients, who did not respond well to their usual migraine treatment, reported that during spontaneous migraine attacks medication only occasionally relieved their headache. On the placebo day, one patient took usual medication to treat the headache and responded well.
Systolic blood pressure during the in-hospital observation period was not significantly different between sildenafil and placebo (Wilcoxon signed rank test, P > 0.05) (Figure 2).
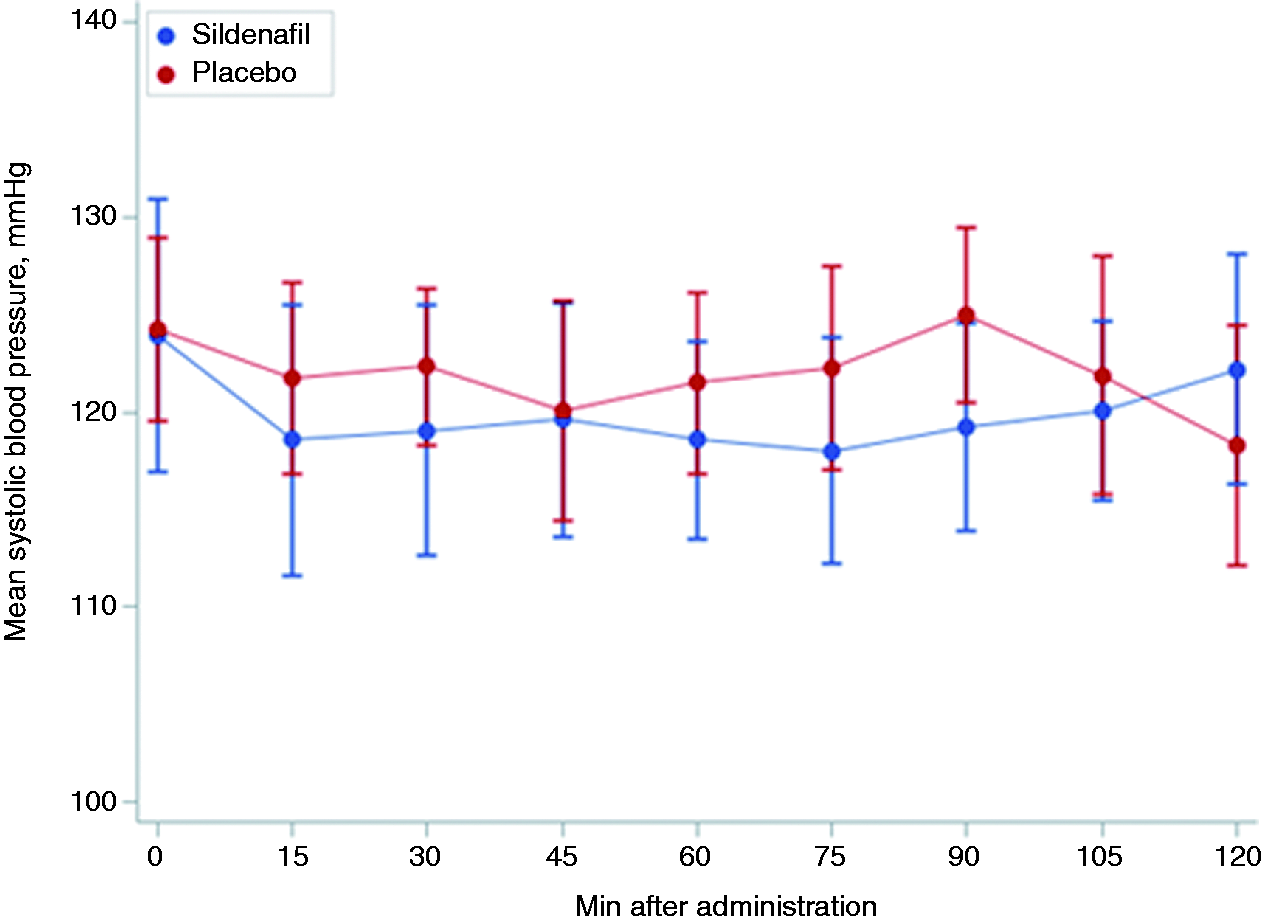
Systolic blood pressure during the two-hour in-hospital observation period in patients with MA after sildenafil and placebo.
Discussion
In this randomized, double-blinded, placebo-controlled crossover study, we investigated the aura- and headache-inducing effects of the PDE5-inhibitor, sildenafil, in 16 patients with MA. The present study yielded the following findings: First, sildenafil induced migraine aura in less than 20% of patients. Second, sildenafil induced migraine-like headache in more than half of the patients.
Signaling pathways involving the second messenger molecules, cAMP and cGMP, are important for the initiation of migraine headache, and the trigeminovascular system may include common relay points such as in the trigeminal ganglion (22). In experimental provocation studies, compounds increasing the concentration of cGMP (nitric oxide donors and sildenafil) (1,3) and cAMP (calcitonin gene-related peptide, pituitary adenylate cyclase-activating polypeptides, and cilostazol) (2,17,18) have consistently induced migraine-like headache in patients with migraine without aura. Figure 3 The effects of some these compounds have also been investigated in patients with MA. In two studies, glyceryl trinitrate administration triggered migraine-like headache, but not migraine aura, in patients with MA (23,24). In another study, calcitonin gene-related peptide infusion appeared to induce both migraine aura and migraine-like headache, but with very modest effects (28% and 56% of patients, respectively) (19). More recently, the selective PDE3-inhibitor, cilostazol also failed to trigger migraine aura, despite inducing migraine-like headache in 75% of the patients (20). The aura- and headache-inducing effects of sildenafil in patients with MA have not been investigated until now, and the present study is therefore the first to investigate these effects of a PDE5-inhibitor in patients with MA. Among 16 patients with MA, of whom the majority exclusively experienced MA attacks, sildenafil induced possible migraine aura in three patients, corresponding to less than 20% of all patients. Although these patients reported that the aura symptoms provoked by sildenafil intake were overall similar to those experienced during their spontaneous MA attacks, the duration of symptoms was substantially longer in two of the patients (5 and 7 hours, respectively). Interestingly, in a case series of 17 otherwise healthy men, intake of 100 mg sildenafil was associated with visual disturbances (including photophobia, disrupted color perception, impaired visual acuity, and deficiencies in stereopsis) that persisted for more than 24 hours but resolved within 21 days (25). As sildenafil also inhibits PDE6, in addition to PDE5, these symptoms were likely caused by inhibition of PDE6, which is expressed specifically in rod and cone photoreceptor cells in the retina, as well as hemodynamic changes in the ophthalmic arteries in response to sildenafil (25,26). Although the long duration of the possible migraine aura in the present study may raise the possibility that these visual disturbances may simply be a manifestation of PDE6-inhibition rather than migraine aura, we believe that this is unlikely to be the case given the different symptomatology. Even if the reported symptoms in the present study reflect “true” migraine aura, the finding that less than 20% of patients with MA developed migraine aura after sildenafil intake suggest that accumulation of cGMP by PDE5-inhibition do not play any significant role in the initiation of migraine aura and refute the hypothesis of sildenafil being a tool for pharmacological provocation of this phenomenon.

Experimental provocation of migraine headache and migraine aura in patients with migraine with aura.
Despite the modest aura-inducing effect of sildenafil, this PDE5-inhibitor provoked migraine-like attacks, mimicking the headache phase during their spontaneous migraine attacks, in slightly more than half of patients with MA. These findings are important, from two perspectives. First, the headache-inducing effect of sildenafil seems to be less pronounced in patients with MA than in those with migraine without aura (56% versus 83%), and sildenafil also appears to be less effective in provoking migraine-like headache than other previously investigated compounds in patients with MA, including glyceryl trinitrate, cilostazol, and levcromakalim (an ATP-sensitive potassium channel opener) (11,20,23). While this finding may be due to differences in thresholds or definitions for migraine headache induction and characteristics in enrolled patients across the studies, it can be speculated that PDE5-inhibition is of lesser importance in the induction of migraine headache in patients with MA, which may relate to different key signaling mechanisms involved, than in those of migraine without aura. This finding supports the concept that MA and migraine without aura may inherently be different headache entities. Nevertheless, this is speculation only and therefore warrants further study. Second, the finding that sildenafil to some extent induces migraine-like headache, and to a lesser degree aura, in patients with MA, even in those suffering exclusively from migraine attacks with aura, suggests that PDE5-inihibition and intracellular accumulation of cGMP may activate pathways relevant for the initiation of migraine headache downstream to the cascade of migraine aura initiation and propagation. On the other hand, this finding also lends support to the notion that migraine aura and migraine headache are triggered by distinct mechanisms in patients with MA.
Although the signaling pathways involving the second messenger molecules cAMP and cGMP are important in the migraine headache pathophysiology, the totality of evidence suggests that these pathways play little to no role in the induction of migraine aura (19,20,23,24). Several methods for an experimental provocation of migraine aura have been attempted, including visual stimulation, physical activity, and induction by other pharmaceutical compounds, with rather disappointing results (8–10). What, then, triggers migraine aura? CSD is considered the underlying pathophysiological mechanism of migraine aura, and this electrophysiological phenomenon can be induced by cortical pinprick injury or application of potassium on the brain surface in animal models (4–7). In this regard, the findings of a recent randomized cross-over study investigating the aura-inducing potential of levcromakalim, an ATP-sensitive potassium channel opener, in 17 patients with MA are of great interest (11). In this study, levcromakalim induced migraine-like attacks in 82% and, more importantly, migraine-like attacks with aura in 59% of patients (11). Although levcromakalim, based on these findings, appears to be a novel aura-inducing substance in humans, the mechanisms by which this compound exerts its migraine and aura-inducing effects are not fully elucidated, and further studies are needed to confirm these promising data and to elucidate the pathophysiological mechanisms.
Limitations
The findings of this study should be viewed in the context of a number of limitations. The majority of studies on experimental provocation of migraine aura included patients who exclusively had attacks of MA. In our study of 16 patients with MA, 11 patients exclusively had attacks of MA. Thus, five patients experienced migraine attacks both with and without aura, though the majority of these primarily were migraine attacks with aura. Ideally, patients should exclusively experience attacks of MA in order to maximize the possibility of aura provocation and to assess the relation between migraine aura and migraine headache. However, restricting our study population to patients who exclusively had attacks of migraine with aura yielded similar findings as the main analyses. As in other migraine-provoking studies, the majority of our study population in our study comprised women, and as per the exclusion criteria, patients were otherwise healthy and had no cardiovascular risk factors. Thus, the generalizability of the model for men and patients with concomitant medical conditions or cardiovascular risk factors may be limited. In addition, a direct comparison between migraine provocation studies may not be likely given the differences in thresholds or definitions for migraine headache induction and characteristics in enrolled patients across the studies. Finally, the propensity or susceptibility to aura, even in an experimental setting, may change due to the hormonal state of the individual (in women), recent sleep patterns, medications, diet, and a variety of other factors.
Conclusions
Sildenafil induced migraine-like headache in more than half of the patients with MA, but not migraine aura. These findings suggest that PDE5-inhibition and cGMP accumulation do not play any significant role in the initiation of migraine aura and support a dissociation between the aura and headache phase in patients with MA.
Article highlights
In a randomized, double-blinded, placebo-controlled crossover study including 16 patients with migraine with aura, sildenafil, a phosphodiesterase-5 inhibitor, induced aura symptoms in three patients (19%) versus none after placebo. Sildenafil induced migraine-like headache in nine patients (56%) compared with one patient (6%) after placebo.
Footnotes
Abbreviations
cAMP: Cyclic adenosine monophosphate
cGMP: Cyclic guanosine monophosphate
CSD: Cortical spreading depression
PDE: Phosphodiesterase
MA: Migraine with aura
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by The Novo Nordisk Foundation Grant number NNF14OC0011637 and NNF18OC0031840.
