Abstract
Background
Migraine is associated with hypertensive disorders of pregnancy through common pathophysiological features. This study evaluates the association between migraine diagnosis and treatment, and risk of hypertensive disorders of pregnancy.
Methods
We conducted a prospective cohort study in the Clinical Practice Research Datalink GOLD, a large longitudinal database of patient records in the UK. We analyzed data from liveborn or stillborn singleton deliveries from 1993–2020 with at least 24 months of medical history and no history of cardiovascular disease (n = 1,049,839). We ascertained migraine through diagnosis or prescription codes before 20 weeks of gestation and hypertensive disorders of pregnancy through diagnosis codes between 20 weeks of pregnancy and delivery. We used log-binomial regression models to estimate the risk ratio and 95% confidence intervals, comparing risk of hypertensive disorders of pregnancy among individuals with migraine to those without migraine, adjusting for confounders.
Conclusions
A history of migraine prior to pregnancy was associated with an increased risk of hypertensive disorders of pregnancy (RR = 1.17, 95% CI: 1.09–1.26). The greatest risk was among those with pre-pregnancy migraine that persisted into the first trimester (RR = 1.84, 95% CI: 1.35–2.50). Use of migraine medication was associated with a higher risk of hypertensive disorders of pregnancy compared to non-migraineurs (RR = 1.50, 95% CI: 1.15–1.97). Results from this study indicate that migraine is a potential risk factor for hypertensive disorders of pregnancy.
Introduction
Migraine is two to three times more common in females than males, and most burdensome in females of reproductive age (1,2). The prevalence of migraine among females of reproductive age in the United Kingdom (UK) is approximately 20% (1). Hypertensive disorders of pregnancy (HDOP) include gestational hypertension and preeclampsia/eclampsia. In the UK, gestational hypertension occurs in an estimated 6–7% of pregnancies, with 15–26% of these cases progressing to preeclampsia (3). HDOP are associated with fetal death, preterm birth, and neonatal intensive care admission, and may be a marker for future cardiovascular disease (4,5).
Migraine may be associated with HDOP through common pathophysiological features such as increased platelet activity, endothelial dysfunction, and high vasoactive sensitivity (5–8). Genomic analysis has identified genetic variants common to migraine susceptibility and vascular and smooth muscle function (4,9). Several clinic-based case-control studies found approximately two to four times the risk of HDOP among those with migraine, compared with non-migraineurs (10–13), although use of hospital controls made these studies vulnerable to selection bias. A prospective cohort study of women at antenatal clinics in Italy found that migraineurs had a greater risk of HDOP than non-migraineurs (OR: 2.85, 95% confidence interval 1.40–5.81) (14).
Recent population-based cohort studies report more modest associations, finding a 30–60% increased risk of HDOP among individuals with migraine, although exposure and outcome definitions varied (4,6,8,15,16). A 2019 meta-analysis reported a pooled odds ratio of 1.94 (95% confidence interval: 1.37–2.76) for the adjusted association between migraine and preeclampsia, although only five studies were included in this estimate (17). A multicenter cohort study found that pregnant people who experienced worsening of migraine during pregnancy had a 13-fold higher risk of hypertensive disorders than those whose migraine improved. While they were unable to differentiate between migraine subtypes, the authors postulated that the lack of metabolic and vascular adaptations in individuals for whom migraine persists in pregnancy contributes to an increased HDOP risk (14).
While migraine medication use may be an indication of severity, particularly with prescription medication, safety concerns and the desire to reduce medication use complicate the study of migraine medication use during pregnancy (18). One prospective cohort study found a positive association between use of prescription medication for migraine and preeclampsia, compared with migraineurs who did not use medication (OR = 1.44, 95% CI: 1.17–1.76) (19). A second prospective cohort study reported an OR of 1.54 (95% CI: 0.6–4.1) for the association between use of triptans and preeclampsia, compared with migraineurs who did not take triptans and an OR of 1.33 (95% CI: 0.7–2.5) when comparing triptan users to a non-migraine comparison cohort, although estimates were imprecise (20).
In the present study, we examined the association between migraine diagnosis and treatment, and risk of HDOP, using a large, population-based cohort with prospectively collected data. We focused on critical windows of exposure from pre-pregnancy through the 20th week of pregnancy, to determine if migraine that persisted through pregnancy and migraine that required prescription treatment were associated with an increased risk of HDOP.
Methods
Study population
We conducted a prospective cohort study in the Clinical Practice Research Datalink (CPRD) GOLD from 1993–2020. The CPRD is a large longitudinal database of de-identified patient records from hundreds of primary care practices in the UK. The CPRD data used in this study contained information from January 1993 through December 2020 and is updated on an ongoing basis. Data in the CPRD include patient characteristics, clinical diagnoses, laboratory results, hospitalizations, prescribed medications, and other patient data. The CPRD is described in detail at cprd.com.
The UK has universal health care coverage and requires that general practitioners act as the gatekeeper to manage access to healthcare, including pre-conception, antenatal, and postpartum care. As such, records from primary care include comprehensive information on service utilization from pregnant people, including diagnoses and prescriptions from consulting (specialty) providers, and hospitalizations. Validation studies of the CPRD have found high accuracy and completeness of the data (21,22).
For the present study, we selected all liveborn or stillborn deliveries between 1993 and 2020 with at least 24 months of medical history prior to the delivery date. The study population contained 1,093,431 deliveries to 768,991 patients over the 27-year period.
Assessment of exposure
We ascertained exposure status through the presence of at least one diagnosis code for migraine or prescription for migraine-specific medication (e.g., triptans, calcitonin gene-related peptide receptor (CGRP) antagonists) at any time in the pre-pregnancy medical history or during pregnancy. We used the date of the clinical encounter resulting in the diagnosis or prescription to identify the timing relative to pregnancy. We defined the start of pregnancy as 280 days prior to the delivery date and examined exposure during multiple time periods before and during pregnancy, including pre-pregnancy, ever during pregnancy, 1–12 weeks of pregnancy, and 13–20 weeks of pregnancy.
Assessment of outcome
We ascertained HDOP through the presence of diagnosis codes (e.g., hypertension complicating pregnancy, mild, moderate, or severe preeclampsia, eclampsia, toxaemia) between 20 weeks of pregnancy and delivery. To further examine the spectrum of HDOP severity, we also separately analyzed the component outcomes of gestational hypertension and preeclampsia or eclampsia.
Assessment of covariates
We ascertained covariate information through the medical record, similarly to exposure and outcome. Covariates included patient age at pregnancy start, last recorded pre-pregnancy BMI (body mass index), last recorded pre-pregnancy smoking status, mood disorders (pre-pregnancy and before 20 weeks of pregnancy), and history of diabetes and asthma. A count of previous delivery codes in the medical record was used to identify primiparity and multiparity. Pre-pregnancy BMI was missing for 36% of deliveries, and pre-pregnancy smoking status was unknown for 14% of deliveries. All other covariates were available for all patients.
Exclusions
From the 1,093,431 deliveries in the study population, we excluded 5485 multifetal pregnancies, 9634 deliveries to individuals with a prior diagnosis of hypertensive disorders of pregnancy, and 28,302 deliveries to individuals with a prior diagnosis of cardiovascular disease or hypertension. If an individual without a history of such conditions was diagnosed with HDOP during pregnancy, or hypertension or cardiovascular disease after delivery, their subsequent pregnancies were excluded.
Finally, we excluded 171 deliveries from four practices where there were no individuals with migraine who delivered during the study period, due to data quality concerns with those practices. The final analytic sample included 1,049,839 deliveries,
Statistical analysis
We reviewed anonymized, individual CPRD patient records from a random sample of 364 records from all exposure and outcome levels to confirm that exposure and outcome data were correctly coded in the population. We then calculated exposure prevalence during all time periods of interest and examined the distribution of exposure, covariates, and outcome in the study population. To generate values for missing BMI (36% missing) and unknown smoking status (14%), we used multiple imputation by fully conditional specification methods (23). We created twenty imputed datasets and statistically combined coefficients and standard errors across datasets. We then examined the distribution of covariates by the presence or absence of migraine codes prior to pregnancy using descriptive statistics. All statistical analyses were conducted using SAS version 9.4 (SAS Institute, Cary NC).
We used log-binomial regression models to estimate the risk ratio (RR) and 95% confidence intervals (CI) comparing risk of HDOP among individuals with migraine to those without migraine, adjusting for confounders. We repeated this analysis examining exposure at multiple time points (pre-pregnancy, first 20 weeks of pregnancy, first 12 weeks of pregnancy, weeks 13–20 of pregnancy). We also examined preeclampsia/eclampsia and gestational hypertension separately as the outcomes of interest. Deliveries were the unit of analysis for this study. We used robust standard errors to account for non-independence of outcomes among patients who had more than one eligible delivery in the sample.
We selected confounders based on a literature review and adjusted for confounders via multivariable modeling. In the final models we adjusted for: age at pregnancy start (continuous), calendar year at pregnancy start (five-year increments), last recorded body mass index prior to pregnancy start (kg/m2, <18.5, 18.5–24.9, 25–29.9, 30–34.9, ≥35), last recorded smoking status before pregnancy (smoker, former smoker, non-smoker), parity (parous/nulliparous), presence of diagnosis codes for asthma before pregnancy, presence of diagnosis codes for diabetes before pregnancy, and presence of diagnosis codes for mood disorders before 20 weeks of pregnancy.
We stratified by primiparity, defined as first delivery in the medical record, to determine if the association between migraine and HDOP is stronger in first pregnancies, that have a higher underlying risk of HDOP (24–26). We also stratified by the time between the last pre-pregnancy migraine diagnosis or prescription and the estimated pregnancy start date (within 90 days of pregnancy start, >90 days to 1 year prior to pregnancy, or greater than 1 year prior to pregnancy). We conducted this analysis to assess the extent to which individuals with migraine closer to the estimated start of pregnancy had a higher risk of HDOP than those who had not received a recent diagnosis or prescription for migraine.
To evaluate the hypothesis that persistent migraine is associated with a higher risk of HDOP than migraine that resolved in the first trimester, we examined patterns of migraine diagnoses or prescription timing in the pre-pregnancy period, first trimester, and 13–20 weeks of pregnancy. Due to small numbers of individuals with some patterns, final models examined the relationship between six of the patterns and HDOP: 1) never, 2) pre-pregnancy migraine only, 3) first trimester migraine with pre-pregnancy migraine, 4) first trimester migraine without pre-pregnancy migraine, 5) 13–20th week migraine with first trimester migraine, and 6) 13–20th week migraines without first trimester migraine.
We classified individuals with migraine codes during pregnancy as treated with migraine-specific medications (treated), treated with medication that is not specific to migraine (possibly treated), or untreated. Migraine-specific treatments included medications that are only indicated for treatment of migraine, such as triptans, ergotamine, and CGRP antagonists, whereas non-migraine specific medications included medications with migraine treatment or prophylaxis among their indications, such as analgesics, beta-blockers, and anti-depressants (online Supplemental Table 1). If an individual received a prescription for both migraine-specific and non-migraine specific treatment, they were classified as treated. Therefore, treated, possibly treated, and untreated were mutually exclusive categories.
Results
Descriptive results
Of the 1,049,397 deliveries, there were 6345 cases of hypertensive disorders of pregnancy (0.6%), including 4308 cases of preeclampsia or eclampsia (0.4%) and 2741 cases of gestational hypertension (0.3%). Twenty percent of participants had a diagnosis and/or prescription code for migraine at any point in their medical record (n = 209,321). The prevalence of migraine prior to pregnancy was 12% (n = 127,295). Only 1% of deliveries had a migraine code during pregnancy (n = 12,246). Prevalence of migraine was similar in the first trimester (n = 6,891, 0.7%) and weeks 13–20 of pregnancy (n = 7,662, 0.8%).
Deliveries to individuals with pre-pregnancy migraine were slightly more likely to be first deliveries than deliveries to individuals without pre-pregnancy migraine (64% vs. 57%). Deliveries to individuals with pre-pregnancy migraine were also slightly more likely to have a pre-pregnancy history of asthma (23% vs. 16%), and substantially more likely to have a pre-pregnancy history of mood disorders (40% vs. 25%) (Table 1).
Pre-Pregnancy characteristics by migraine status* of 1,049,837 deliveries between 1993 and 2020 in CPRD Gold.

*Migraine status determined by presence of a migraine diagnosis or migraine-specific medication code prior to delivery.
Pre-pregnancy migraine and HDOP
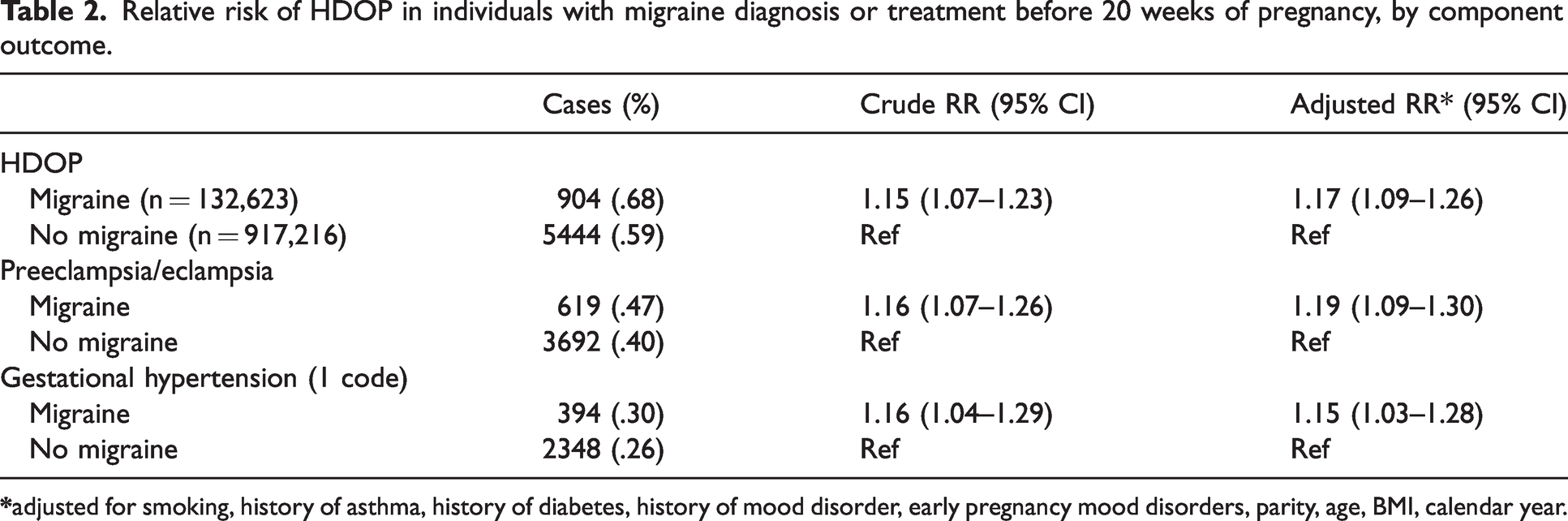
Migraine at any time before 20 weeks of pregnancy (pre-pregnancy or during pregnancy) was associated with a slightly increased risk of HDOP overall (0.68% of migraineurs and 0.59% of non-migraineurs; RR = 1.17, 95% CI: 1.09–1.26), and among nulliparous individuals (first delivery in medical record) (RR = 1.19, 95% CI: 1.10–1.30), as well as parous individuals (RR = 1.11, 95% CI: 0.96–1.28). Estimates were similar for preeclampsia/eclampsia (0.47% of migraineurs and 0.40% of non-migraineurs; RR = 1.19, 95% CI: 1.09–1.30) and gestational hypertension (0.30% of migraineurs and 0.26% of non-migraineurs; RR = 1.15, 95% CI: 1.03–1.28), despite preeclampsia/eclampsia being more severe outcomes (Table 2).
Relative risk of HDOP in individuals with migraine diagnosis or treatment before 20 weeks of pregnancy, by component outcome.
First trimester migraine and HDOP
While pre-pregnancy migraine overall was associated with an increased risk of HDOP (0.99% of migraineurs and 0.60% of non-migraineurs), the highest risk was among deliveries with first trimester migraine, regardless of whether migraine was also present later in pregnancy. Migraine during the first trimester of pregnancy (regardless of migraine status during other time periods) was associated with a 1.58-fold greater risk of HDOP (95% CI: 1.21–2.01) (Table 3). Results for the component outcomes were similar: the RR for preeclampsia/eclampsia was 1.55 (95% CI: 1.16–2.08) and for gestational hypertension was 1.74 (95% CI: 1.24–2.45).
Relative risk of HDOP in individuals with migraine diagnosis or treatment in the first trimester of pregnancy, by component outcome.

Migraine occurrence patterns and HDOP
Figure 1 shows the possible patterns of migraine occurrence (diagnosis or treatment) from pre-pregnancy to the 20th week of pregnancy. Pre-pregnancy migraine which persisted into pregnancy was uncommon. When migraine did persist into early pregnancy, it continued into the second trimester in about 25% of deliveries. A small percentage of deliveries (5328; 0.6%) without a pre-pregnancy history of migraine had codes for migraine during the first 20 weeks of pregnancy.

Distribution of migraine prescription or diagnosis codes throughout pregnancy, by time periods of interest.
Table 4 shows the association between different migraine occurrence patterns and HDOP, with no migraine during pre-pregnancy or pregnancy as the reference group. There was a small increase in risk of HDOP among deliveries with pre-pregnancy migraine only (0.66% vs. 0.59%; RR = 1.14, 95% CI: 1.04–1.23). There was an elevated risk of HDOP among individuals with first trimester migraine overall (0.95% vs. 0.69%; RR = 1.56, 95% CI: 1.19–2.05). However, this association was limited to those with pre-pregnancy and first trimester migraine; we found no meaningful association among those with new onset first trimester migraine who did not have pre-pregnancy migraine (RR = 1.04, 95% CI: 0.59–1.82).
Relative Risk of HDOP by migraine timing.

The majority of migraineurs (80%) received a migraine diagnosis or prescription more than one year before the estimated start of pregnancy, while 13% had a code >90 days to one year before pregnancy, and 7% had a code within 90 days before pregnancy. The RR for the association between first trimester migraine and HDOP among individuals with a migraine occurrence within three months before pregnancy was 1.58 (95% CI: 1.00–2.52). This association was similar among individuals with less recent last pre-pregnancy migraine codes: RRs among individuals with migraine codes >90 days to one year, and greater than one year before pregnancy were 1.52 (95% CI: 0.86–2.52), and 1.46 (95% CI: 0.85–2.53), respectively (Table 5).
Relative risk of HDOP and first trimester migraine, by date of last pre-pregnancy migraine relative to pregnancy start.

Second trimester migraine and HDOP
The risk of HDOP was also elevated among those with migraine in the 13–20th week of pregnancy, compared to those with no migraine (RR = 1.32, 95% CI: 1.03–1.69) (Table 5). When deliveries with migraine in the 13–20th week of pregnancy were stratified by the presence or absence of migraine in the first trimester, the association between migraine and HDOP was much greater among those with migraine in both trimesters (RR = 1.86, 95% CI: 1.13–3.08), compared to those whose first migraine code was in the 13–20th week of pregnancy (RR = 1.22, 95% CI: 0.92–1.63).
Migraine treatment and HDOP
Overall, most individuals with migraine were either treated (57%) or possibly treated (15%) (Figure 2). While treatment or possible treatment was markedly lower during pregnancy than in pre-pregnancy (51% vs. 72%), treatment or possible treatment was substantially more common in the first trimester (68%) than in the 13–20th weeks of pregnancy (37%). While untreated migraine was associated with a modestly increased risk of HDOP (0.78% vs. 0.60%; RR = 1.28, 95% CI: 0.98–1.67), migraine that was treated or possibly treated during pregnancy was associated with greater increases in the risk of HDOP (RR = 1.50, 95% CI: 1.15–1.97 and RR = 1.56, 95% CI: 0.91–2.69, respectively) (Table 6). Use of migraine-specific treatment was not associated with a greater increase in HDOP risk than use of other treatment.

Distribution of migraine treatment among individuals with migraine, by timing of migraine diagnosis or prescription.
Relative risk of HDOP by migraine treatment codes during pregnancy.

**Received a prescription specifically for migraine treatment or prophylaxis.
***Received a prescription for a medication often, but not exclusively, used for migraine treatment or prophylaxis.
Discussion
In this population-based prospective cohort study of deliveries in the United Kingdom CPRD GOLD from 1993–2020, we found that the greatest risk of HDOP in relation to migraine occurred among individuals with pre-pregnancy migraine that persisted into the first trimester of pregnancy. This risk elevation occurred regardless of whether they continued to experience migraine after their first trimester but did not occur among those with new onset migraine in the first trimester. A history of migraine prior to pregnancy overall was associated with a slightly increased risk of HDOP. Individuals who used medication for their migraine had a slightly higher risk than those who did not. Findings were consistent when we examined HDOP as a composite outcome, and when we analyzed preeclampsia and gestational hypertension separately.
The present study, which used prospectively-collected data from patient records, may help clarify the extent to which migraine is associated with the risk of HDOP. While previous literature has consistently identified migraine as a risk factor for preeclampsia, the magnitude of this association has varied greatly across studies (12,15,16,27). Migraine attacks tend to decrease during pregnancy (28) however prior studies have also found that individuals who continue to experience migraine in pregnancy despite maternal adaptations may be at greater risk of HDOP than those for whom migraine improves (5,27).
Our study did not find an increased risk among those with migraine later in pregnancy, beyond the risk experienced by those with first trimester migraine. This could be an artifact of misclassification, as this period was only seven weeks long. Individuals may have experienced migraine, but not seen a physician, particularly if they were hesitant to treat their migraine with prescription medication during pregnancy. A study of migraine medication use in pregnancy found that discontinuation of medication was common (29).
An important limitation of this study is that we did not have information on the date of medication use during pregnancy. We acknowledge that a small subset of the medications under study (8%) are contraindicated among women of reproductive age or during pregnancy (i.e., topiramate, sodium valproate) due to potential teratogenicity. If migraine medication use increased the risk of miscarriage (i.e., pregnancy loss before 20 weeks of gestation), we would expect that conditioning on ongoing pregnancy (≥20 weeks of gestation) in our study would have attenuated the association between migraine medication use and risk of HDOP.
Use of diagnosis and medication codes to ascertain migraine status likely resulted in a more specific and less sensitive measure than self-report (30). Indeed the prevalence of migraine in our cohort and other medical registry studies was appreciably lower than estimates from previous studies using self-reported migraine (4,18,19,31–34). It is likely that migraine was under-ascertained because less severe migraine may not be discussed with a healthcare provider or captured in the medical record. Research on medical consultation for migraine in the United Kingdom found that only a third of migraineurs had seen a doctor for their headaches within one year (35). The relatively small number of individuals experiencing migraine in pregnancy limited precision in some stratified analyses.
As this was a prospective cohort study, we would expect exposure misclassification to be non-differential and result in an underestimation of the effect estimate. Therefore, it is unlikely that non-differential misclassification would explain our positive findings. However, it is also possible that migraine severity is an effect measure modifier, and less severe migraine does not increase the risk of HDOP, limiting the generalizability of our findings. In our study, 96% of individuals with pre-pregnancy migraine did not have migraine diagnosis or prescription codes in the first trimester of pregnancy. This may be due to inconsistent coding or the desire to reduce medication use during pregnancy; however, individuals were likely unaware of their pregnancy status for several weeks in their first trimester, and we did observe a marked decrease in medication use later in pregnancy.
Outcome misclassification was also likely in this study, particularly for gestational hypertension, which is a less severe outcome and therefore may be less reliably coded than preeclampsia or eclampsia (36). Less severe HDOP is less likely to be included in the delivery hospital discharge letter sent to the general practitioner or less likely to be coded in the primary care record by the general practitioner, and therefore under-ascertained in our study. The prevalence of hypertensive disorders of pregnancy in this cohort was substantially lower than previous estimates, suggesting under-ascertainment of outcome (3). Low sensitivity and high specificity of outcome measurement likely biased our effect estimates towards the null. The magnitude of bias would increase with the degree to which HDOP was under-ascertained, unless there was perfect specificity of the outcome, in which case the risk ratios would be unbiased. In summary misclassification due to low sensitivity and high specificity of both exposure and outcome likely led to bias towards the null. Our findings may underestimate the true association between migraine and HDOP.
Another potential source of bias in this study was unmeasured or residual confounding, as we were limited to covariate information available in the medical records. Factors related to seeking care for migraine and unmeasured migraine triggers such as alcohol, physical activity, and psychological stress may vary in pregnancy and relate to the risk of HDOP, resulting in unmeasured confounding and either an under- or over- estimate of the measure of association.
We observed that having a code for migraine or migraine treatment in the medical record was associated with a small increase in risk of HDOP, and that the risk of HDOP is highest among those with pre-pregnancy migraine that persists in the first trimester of pregnancy. These and previous findings suggest that individuals who experience migraine in pre-pregnancy and during pregnancy may be at increased risk of HDOP and therefore may benefit from increased monitoring during pregnancy.
Results from this study will help clinicians better understand migraines as a potential risk factor for HDOP, leading to appropriate education and monitoring, which will potentially improve the health of parent and baby. Individuals with a history of migraine who become pregnant need as much information as possible on the expected course of their condition and its impacts on their reproductive health. As migraine is a relatively common chronic condition, increased attention to this at-risk population has high public health relevance. In addition, individuals who experience HDOP are more likely to be diagnosed with hypertension later in life (4,5), and increased understanding of HDOP may lead to improved vigilance in preventing future health complications.
Public health relevance
Individuals who experience migraine in pre-pregnancy and during pregnancy may be at increased risk of HDOP and therefore may benefit from increased monitoring during pregnancy. As individuals who experience HDOP are more likely to be diagnosed with hypertension later in life, increased understanding of HDOP may also lead to improved vigilance in preventing future health complications.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231161746 - Supplemental material for Migraine and risk of hypertensive disorders of pregnancy: A population-based cohort study
Supplemental material, sj-pdf-1-cep-10.1177_03331024231161746 for Migraine and risk of hypertensive disorders of pregnancy: A population-based cohort study by Holly M Crowe, Amelia K Wesselink, Elizabeth E Hatch, Lauren A Wise and Susan S Jick in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and patient consent
CPRD data is obtained under license from the UK Medicines and Healthcare products Regulatory Agency. The study protocol was approved by the Independent Scientific Advisory Committee for Medicines and Healthcare products Regulatory Agency database research (protocol 21_000709). The interpretation and conclusions contained in this study are those of the authors alone. All data accessed were de-identified and do not require patient consent for use.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported through funds provided by the National Institute of Child Health and Human Development, National Institutes of Health (R01-HD086742, R21-HD072326). HC is supported by the National Research Service Award (T32 HD 104612).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
