Abstract
Background
Previous studies have reported various levels of association between migraine and preeclampsia. However, there are no published data on migraine and its association with preeclampsia in African countries, including Sudan.
Methods
A case–control study was conducted at Rabak Maternity Hospital in White Nile State, Central Sudan. The cases were pregnant women with preeclampsia, while the controls were healthy pregnant women. All participants were interviewed using questionnaire. The adjusted odds ratio (AOR) and a 95% confidence interval (CI) were calculated in a multivariate regression analysis.
Results
Of 148 pregnant women with preeclampsia, 96 (64.9%) women had mild preeclampsia and 52 (35.1%) women had severe preeclampsia. Of the 148 study participants with preeclampsia, 57 (38.5%) had a history of migraine and 19/296 (6.4%) women in control group had a history of migraine (p < 0.001). Pregnant women with a history of migraine have higher odds of preeclampsia than pregnant women without a history of migraine (AOR = 9.01, 95% CI = 4.81–16.86). A history of preeclampsia, being overweight and obesity were associated with preeclampsia.
Conclusion
Our findings are consistent with the findings of previous studies on the association between migraine and preeclampsia. More studies are needed on this topic.
Introduction
Preeclampsia is the occurrence of hypertension and one or more organ dysfunction (1). Preeclampsia is one of the leading global health problems, with a worldwide prevalence of 2–8% of pregnant or parturient women, which is higher in countries with less resources. Preeclampsia can lead to several adverse maternal and perinatal outcomes, and it is the leading and primary cause of both maternal and perinatal morbidity and mortality (2). Despite advances in various sciences, the exact cause and pathophysiology of preeclampsia are not yet fully understood. There are several sociodemographic, reproductive, obstetric, clinical, nutritional, biochemical, infectious, and genetic factors associated with preeclampsia (3). Sudan is the third largest country in Africa, and preeclampsia or eclampsia is responsible for 4.2% of hospital admissions of pregnant women in Sudan, with preeclampsia/eclampsia being one of the leading causes of the country’s high maternal and perinatal morbidity and mortality rates (4).
Women—especially during their reproductive years—are more susceptible to migraines, and it has been estimated that approximately 20.0% of women of reproductive age have migraines (5). Pregnant women who have migraines are at higher risk of adverse pregnancy and neonatal outcomes, such as preterm birth (6–8), low birthweight deliveries (8,9), increased rate of cesarean deliveries (8), increased rate of neonatal intensive care unit admissions, and newborn respiratory distress syndrome (10). Previous studies have reported different levels of association between migraine, hypertensive disorders of pregnancy (6,7,11–13), and preeclampsia (6,8,9,14–17). Furthermore, most—if not all—of these studies were conducted in high-income countries, and there are no published data on migraine and its association with preeclampsia in Africa. In Africa (including Sudan), several factors, such as malnutrition, malaria, bacterial, and viral infections, are associated with preeclampsia (18–20).
It has recently been reported that 7.5% of Sudanese medical students have migraines, and the prevalence of migraine is higher among female medical students (21).
Thus, several risk factors for preeclampsia may differ between different populations and in different settings. Therefore, it is necessary to investigate the risk factors for preeclampsia in different populations. Investigating the risk factors for preeclampsia will yield critical data with which clinicians, researchers, and health planners can identify preventive and screening tools used in antenatal care (ANC) that need to be modified to address the risk factors—including a history of migraine. This study was conducted at Rabak Maternity Hospital in White Nile State, Central Sudan, to investigate the association between a history of migraine and preeclampsia.
Methods
Design
A non-matched case–control study was conducted at Rabak Maternity Hospital, White Nile State, Central Sudan, from February to December 2021. Rabak maternity hospital is a tertiary hospital located in Rabak, one of the largest and most prominent cities in Sudan. Rabak is 362 m above sea level, and it lies between the geographical coordinates 13°9′0″N and 32°44′0″E (22). The Strengthening the Reporting of Observational studies in Epidemiology (STROBE) Statement standard checklists were followed (23).
The cases were women with a singleton pregnancy who had preeclampsia. As recently recognized (1), preeclampsia is defined as the “occurrence of systolic blood pressure equal to or more than 140 mmHg and/or diastolic blood pressure equal to or more than 90 mmHg on two occasions at least four hours apart in previously normotensive women with one of the following new‐onset conditions at or after 20 weeks of gestation: proteinuria (≥30 mg/mol protein or creatinine ratio; ≥300 mg/24 hour or ≥2+ dipstick), maternal organ dysfunction (e.g., acute kidney injury, liver involvement, neurological complications, hematologic complications [platelets count <150 000/μL], disseminated intravascular coagulation, and hemolysis], and uteroplacental dysfunction (fetal growth restriction, abnormal umbilical artery Doppler waveform analysis, or stillbirth)”. The controls were healthy pregnant women who attended the labor ward of the hospital. Women with multiple pregnancies and systematic diseases (diabetes mellites, epilepsy, hypertension, and thyroid disease) were excluded from the cases and the controls.
After signing a written informed consent form, all women in both the case and control groups were interviewed using a questionnaire that included questions designed to capture sociodemographic, clinical, and reproductive data (age, parity, level of education, history of miscarriage, and ANC level) of women. The questionnaires were administered by two female medical officers. A history of maternal migraine was queried with the question: Have you ever been told by a doctor that you have a migraine headache? and the responses were recorded as yes or no. The measured weights and heights of the sampled pregnant women were used to compute their body mass index (BMI), which was expressed as weight divided by the square of the height (kg/m2) and the BMI was categorized as underweight (<18.5 kg/m2), normal weight (18.5–24.9 kg/m2), overweight (25.0–29.9 (kg/m2) and obese (≥30.0 kg/m2) (24).
Calculation of sample size
Sample sizes of 148 cases and 296 controls (control to case ratio of 2:1) were calculated per the case–control study formula (25). We assumed that 10.0% of pregnant women in the control group and 20.0% of the cases had a history of migraine. Our assumption is based on recent findings of a prevalence of migraine among female medical students in Khartoum, Sudan (21). The sample sizes were calculated assuming a two-sided test, a 5% significance level, a 95% confidence interval, and 80% power (26).
Ethics
This study received ethical clearance from the ethical committee of the Faculty of Medicine, University of El Imam El Mahdi, Kosti, Sudan (No 06/2021). The study participants signed an informed consent form before participating in the study.
All research methods employed in this study were performed in accordance with the Declaration of Helsinki.
Statistics
The data were analyzed using the Statistical Package for the Social Sciences (SPSS) for Windows, version 22 (IBM, Armonk, NY, USA). The data were checked for normality using the Shapiro–Wilk test. Median (interquartile range [IQR]), frequency, and percentage were used to describe the characteristics of the participants. A chi-square test was used to compare proportions between the women with preeclampsia and the controls. Univariate analyses were performed with preeclampsia as the dependent variable, while maternal age, parity, education, history of miscarriage, ANC level, history of migraine, and BMI were the independent variables. Subsequently, a multivariable analysis was performed if the p-value was less than 0.2 in the univariate analysis. The adjusted odds ratios (AORs) and 95% confidence interval (CI) were also computed; a p-value of less than 0.05 was considered statistically significant.
Results
One hundred and forty -eight pregnant women were the cases of preeclampsia. Of 148 women with preeclampsia, 96 (64.9%) were mild and 52 (35.1%) were severe preeclampsia. Fifteen (10.1%) women with preeclampsia had early onset preeclampsia (gestational age: <34 weeks). Age and BMI were significantly higher among the women with preeclampsia than among the controls. There was no difference in parity, level of education, blood group, hemoglobin level, and gender of the newborn between the women with preeclampsia (n = 148) and the pregnant women in the control group (n = 296). The number of pregnant women with a history of migraine was higher among the women with preeclampsia than in the control group. While 57 (38.5%) of the 148 women with preeclampsia had a history of migraine, only 19 (6.4%) of the pregnant women in the control group had a history of migraine (p < 0.001). Compared to the controls, a higher number of women with preeclampsia had a prior history of preeclampsia, (33/148 [22.3%] versus 2/296 [0.7%], P < 0.001), were overweight (47/148 [31.8%] versus 58/296 [19.6%], P = 0.001), and were obese (37/148 [25.0%] versus 35/296 [11.8%], P < 0.001), (Table 1).
Univariate analysis of sociodemographic and reproductive variables among women with preeclampsia and controls at Rabak Maternity Hospital, Sudan, in 2021.
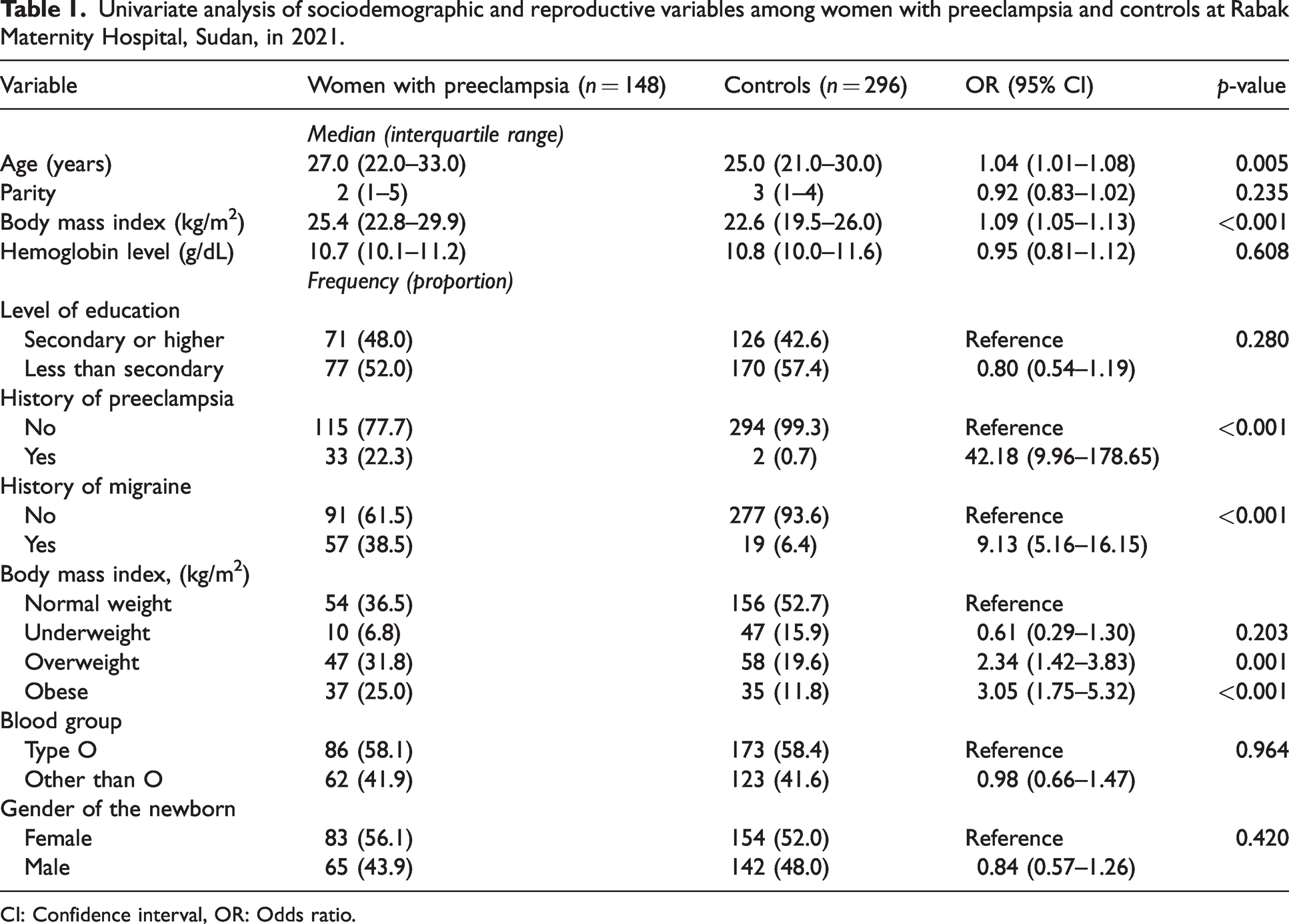
CI: Confidence interval, OR: Odds ratio.
An adjusted multivariate logistic regression analysis revealed that pregnant women with a history of migraine have higher odds of preeclampsia than those who did not have a history of migraine (AOR = 9.01, 95% CI = 4.81–16.86, p < 0.001). Furthermore, pregnant women with a history of preeclampsia have higher odds of preeclampsia recurrence (AOR = 44.88, 95% CI = 9.70–204.03, p = <0.001). Overweight women (AOR = 2.54, 95% CI = 1.41–4.58, p = 0.002) and obese women (AOR = 3.95, 95% CI = 2.07–7.51, p < 0.001) have higher odds of preeclampsia (Table 2).
Multivariate analysis of sociodemographic and reproductive variables among women with preeclampsia and controls at Rabak Maternity Hospital, Sudan, in 2021.
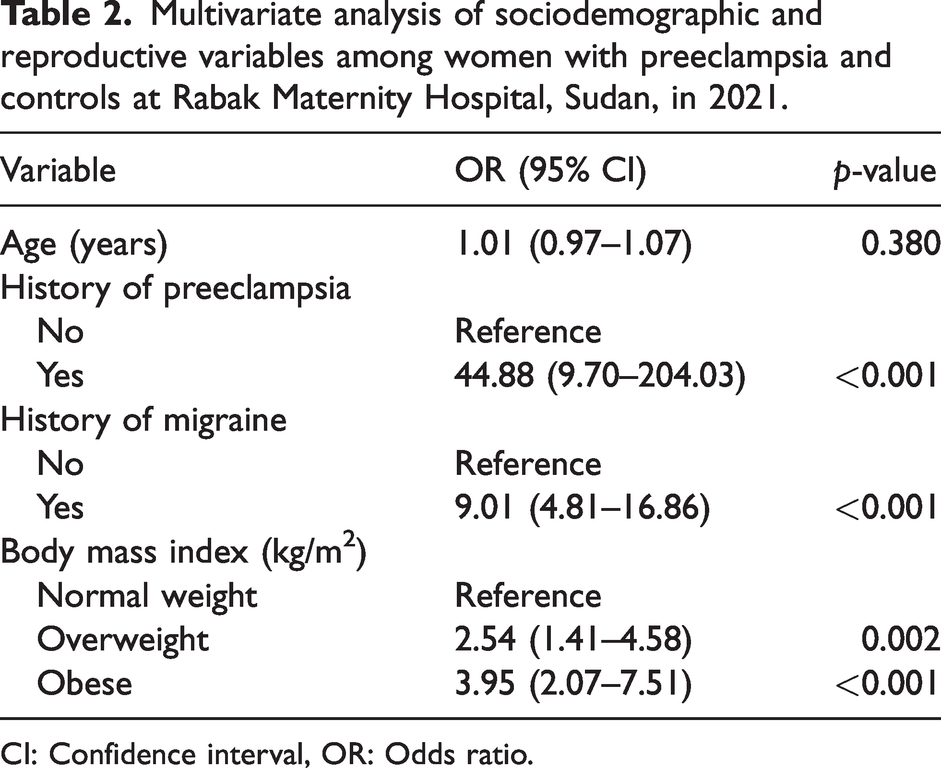
CI: Confidence interval, OR: Odds ratio.
Discussion
The primary finding of this study is that pregnant women with a history of migraine have 9.02 times higher odds of preeclampsia than those who do not have a history of migraine. Our results are consistent with the results of several previous studies on the same subject, which reported varied levels of association between migraine and preeclampsia (6,8,9,11,14–17). It has recently been demonstrated that pre-pregnancy migraine or migraine during early pregnancy was associated with an 81% increased risk of preeclampsia (relative risk = 1.19). Marcoux et al., in a case–control study of Canadian women (15), reported that pregnant women with a history of migraines had a 2.4 times higher risk of preeclampsia than their peers who did not have migraines. Adeney et al. (14), in a case–control study (244 women with preeclampsia and 470 normotensive women) conducted in the Pacific Northwest of the USA, observed that pregnant women with a history of diagnosed migraine had a 1.8 times higher risk of preeclampsia than women in the control group who did not have history of migraine. Furthermore, Sanchez et al., in a study on Peruvian women (cases: 339 women with preeclampsia, and controls: 337 normotensive women), found that women who had a history of having had any headaches before or during pregnancy had a 2.4 times increased risk for preeclampsia than those who did not have such a history. They also observed that women who had migraines before pregnancy had a 3.5 times increased risk of preeclampsia than those who did not, and women who had migraines during pregnancy had a four times higher risk of preeclampsia (16). Previously, Facchinetti et al. (17), in a study involving a large prospective cohort of 702 Italian women with singleton pregnancies, reported that migraine was associated with preeclampsia. In another large cohort study (19,694 participants), women who had pre-pregnancy migraine were found to have a 60.0% higher risk of preeclampsia (relative risk = 1.40) than those who did not have pre-pregnancy migraine (6). A recent systematic review that included 14 studies revealed that pregnant women who had migraines had a 2.07 times higher risk of having preeclampsia than pregnant women who did not have migraine (9). Chen et al., in a three-year nationwide population-based database study in Taiwan, found that pregnant women who had migraines had a 1.34 times higher risk of preeclampsia than pregnant women who did not have migraine (8). As mentioned earlier, migraine is associated with an elevated risk of hypertensive disorders of pregnancy (6,7,11–13).
Our findings should be compared cautiously with the results of other studies because different methods were used to assess migraine itself, and differences in sociodemographic factors must be considered. As mentioned earlier, the exact relationship between migraine and preeclampsia is not yet fully understood. It is conceivable that triggering factors, such as cytokines, inflammatory biomarkers, cardiometabolic factors, and thrombogenic susceptibility, are overlapping factors for migraine and preeclampsia (27–29).
Primarily, we aimed to assess the association between migraine and preeclampsia; however, our findings indicate that women with a history of preeclampsia, being overweight and obesity were associated with preeclampsia. These factors have been extensively studied previously, and a discussion of these factors is beyond the scope of this study.
Limitations
Due to facility constraints, the diagnosis of migraine in this study was determined based on a simple history and not on a proper diagnosis of migraine, which should be based on the International Classification of Headache Disorders (30). This point (diagnosing migraine by history) could have a bias-effect on our results. Our study was a non-matched case-control study which has its limitations effects of not matching for age and other variables. Several factors such as malnutrition, malaria, bacterial, and viral infections (18–20) are linked to preeclampsia in Sudan; these factors have not been ruled out.
Conclusion
Our findings are consistent with the findings of previous studies on the association between migraine and preeclampsia. Nonetheless, more longitudinal, prospective cohort studies are needed to assess the association between migraine and the risk of preeclampsia.
Clinical implications
Higher number of women with preeclampsia had a history of migraine. Pregnant women with a history of migraine have higher odds of preeclampsia. Pregnant women with a history of preeclampsia have higher odds of preeclampsia. Preventive measures (aspirin, micronutrients, and close follow-up) for preeclampsia may be considered for pregnant women with a history of migraine.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
