Abstract
Introduction
Identifying female-specific risk markers for cerebrovascular disease is becoming increasingly important. Both migraine and preeclampsia have been associated with higher incidence of brain white matter lesions (WML) and stroke. We assessed the association between WML and migraine among formerly (pre)eclamptic women.
Methods
A total of 118 women (76 formerly (pre)eclamptic and 42 control women) were screened for migraine and WML presence. Independent effects of migraine and (pre)eclampsia on WML were assessed.
Results
Migraine prevalence did not differ between the (pre)eclamptic (26/76; 34%); and control group (10/42; 24%), p = 0.17. Age-adjusted regression analysis failed to show a significant independent effect of migraine (OR 1.14; 95% CI 0.47–2.76; p = 0.77) on WML presence, and showed a non-significant effect of (pre)eclampsia (OR 2.30; 95% CI 0.90–5.83; p = 0.08).
Conclusion
Migraine prevalence was not found to be an independent risk factor for WML prevalence in formerly (pre)eclamptic women. Since this study had a small sample size, larger prospective studies are needed to examine female-specific risk factors for WML and its consequences.
Introduction
Identifying female-specific risk markers for the development of cardiovascular and cerebrovascular disease is becoming increasingly important. If it is possible to identify at-risk women at a relatively young age, these women may benefit from early lifestyle interventions. Two conditions that may be important risk markers are hypertensive disorders of pregnancy such as (pre)eclampsia, and migraine. Migraine is a primary headache syndrome, considered a chronic disabling condition causing individual suffering and impaired quality of life. The lifetime prevalence of migraine is higher in females (33%) than in males (13%), and has an age-dependent peak during childbearing years (1). Previous studies have found a relationship between migraine and brain white matter lesions (WML) and subclinical infarctions in later life (2). Cortical spreading depression, a cortical wave of neuronal and glial depolarization that is thought to be the biological substrate of migraine aura, may cause disruption of the blood-brain barrier by activating brain matrix metalloproteinases (3). In preclinical studies, this disruption is suggested to result in transient local tissue damage and WML (3). Similarly, women with a history of early-onset preeclampsia or eclampsia (tonic-clonic seizures in a woman with preeclampsia) demonstrate a high prevalence of WML (4,5). Several reports state that women who suffer from migraine are at higher risk to develop hypertensive disorders during pregnancy compared to non-migraineurs, with odds ratios (ORs) ranging between 1.1 and 3.7 (6). Diagnostic criteria for preeclampsia or migraine, however, frequently have not been strictly applied in these cohorts and information on structural brain pathology is lacking.
In this study we therefore assessed the association between the presence of brain WML and migraine among a group of relatively young women who suffered from (pre)eclampsia, using strict diagnostic criteria.
Methods
Participants
Formerly eclamptic, preeclamptic and control women who had normotensive pregnancies, who were previously enrolled as part of a follow-up study assessing long-term cerebrovascular consequences of (pre)eclampsia, received a new invitation (4,5). Preeclampsia and eclampsia were strictly defined according to the criteria of the International Society for the Study of Hypertension in Pregnancy (7). Women with a diagnosis of eclampsia between 1988 and 2005 were identified from the electronic databases of the University Medical Center Groningen (UMCG) and matched for age and year of index pregnancy with a formerly preeclamptic woman. Additional formerly eclamptic women were recruited through collaboration with the VU University Medical Center Amsterdam and Isala Clinics Zwolle. Parous women in the control group were recruited through either the department’s electronic delivery database or among hospital employees and their family members. Medical records were reviewed for accuracy of diagnosis and to extract clinical and demographic characteristics. Exclusion criteria were: a history of epilepsy, demyelinating disorders, intracranial infections, or any cranial neurosurgical procedure. For all women, elapsed time since the index pregnancy had to be ≥12 months.
All (n = 212) women who participated in the aforementioned studies (4,5) were invited by regular mail or email to participate in the migraine study now reported. They were asked to access the Leiden University Migraine Neuro-Analysis (LUMINA) website (https://www.lumc.nl/org/hoofdpijn-onderzoek/migraine/onderzoek) and complete the Web-based questionnaire enabling accurate migraine diagnosis based on the International Classification of Headache Disorders, third edition beta (ICHD-III-beta) criteria of the International Headache Society (8,9). Neuroimaging studies were performed between July 2005 and January 2012 and LUMINA questionnaires were filled out between July 2012 and July 2013. This project was approved by both the UMCG and Leiden University Medical Center (LUMC) Institutional Review Boards. All women provided written informed consent.
Migraine assessment
The LUMINA questionnaire is a validated, self-reported, Web-based questionnaire to diagnose migraine headache and aura symptoms. This questionnaire is a part of the LUMINA project (8). Participants were asked to complete a migraine screening questionnaire that had been validated previously (1). Participants who screened positive were sent a validated Web-based extended migraine diagnostic questionnaire (8) based on the ICHD-III-beta criteria. In addition to questions that were necessary for accurate migraine diagnose and subtype, the extended questionnaire also included items pertaining to demographic factors, acute and prophylactic headache medication use, and migraine attack frequency.
Cerebral magnetic resonance imaging (MRI) protocol
MRI imaging took place on average 6 ( ±5.3) years following the index pregnancy.
All studies were performed on a 3-T MRI system (Philips Intera; Philips Medical Systems, Best, the Netherlands) at the Neuro-Imaging Center of the School for Behavioural and Cognitive Neurosciences in Groningen (4,5). An experienced neuroradiologist, blinded to patient category, rated the presence and number of subcortical/periventricular WML and other structural brain abnormalities. WML were defined as hyperintense on fluid-attenuated inversion recovery (FLAIR), proton density-weighted and T2-weighted images, and not hypointense on T1-weighted images (5).
Physical parameters
Following MRI and a period of rest, blood pressure was measured manually using an aneroid sphygmomanometer. Patients with a systolic blood pressure of ≥140 mmHg, diastolic blood pressure ≥90 mmHg, and/or currently using antihypertensive medication for their high blood pressure were designated as currently hypertensive. One individual in the (pre)eclampsia group used antihypertensive medication as migraine prophylaxis.
Statistics
Statistical analysis was performed using IBM SPSS Statistics for Windows version 20 (IBM Corp., Chicago IL, USA). An a priori sample size calculation was not performed because the participants of this study had already been included at an earlier stage and hence the maximum sample size was fixed. Demographic data were compared using a chi-square test for categorical data, one-way analysis of variance (ANOVA) for normally distributed data or Kruskal-Wallis test for non-parametric data. The presence of migraine according to the LUMINA questionnaire was compared between patient groups (eclampsia and preeclampsia combined) versus controls using a one-sided Fisher’s exact test. The presence of WML in women with and without migraine was tested similarly. Binary logistic regression analyses were used to study the independent effect of (pre)eclampsia and migraine on WML presence, corrected for age.
Results
General characteristics
Participant characteristics.

Results are expressed as mean (SD), median (minimum–maximum) or number (percentage). BMI: body mass index (missing data in respectively n = 8, 15 and 16); EGA: estimated gestational age; y: years; wk: weeks. an = 40.
Prevalence of migraine in (pre)eclampsia groups
Overall migraine prevalence was 30.5% (36/118) and did not significantly differ between (pre)eclamptic women (26/76; 34%) and controls (10/42; 24%) p = 0.17. Of the 36 women who screened positive for migraine, 24 completed the extended survey. Of these, six of 17 (35%) (pre)eclamptic and one of seven (14%) control women had migraine with aura (non-significant). In 14/17 (82%) (pre)eclamptic women initial onset of migraine was before pregnancy, as was the case in six of seven (86%) control women (non-significant). Average age at migraine onset was 19 ( ± 8.4) years in (pre)eclamptic women 20 (±11.4) years in controls (non-significant).
Prevalence of WML and relationship with migraine
Overall, WML were predominantly found in the frontal areas. Subcortical WML were found in 28/76 (37%) of (pre)eclamptic women and 11/42 (26%) of controls (Figure 1). Periventricular WML were found in four preeclamptic women, cortical infarctions in one eclamptic and one preeclamptic woman, and a lacunar infarction in one control. The age-adjusted regression analysis failed to show a significant independent effect of migraine (OR 1.14; 95% CI 0.47–2.76; p = 0.77) on WML presence, and showed a non-significant effect of (pre)eclampsia (OR 2.30; 95% CI 0.90–5.83; p = 0.08).
Presence of WML in migraineurs and non-migraineurs.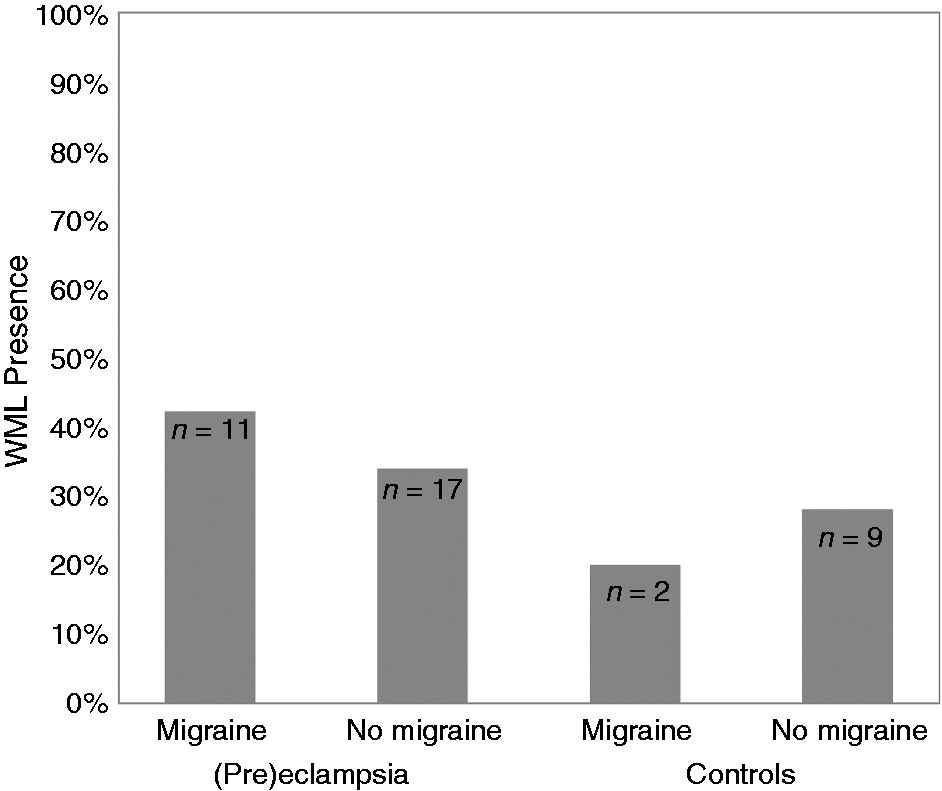
Discussion
Migraine prevalence did not differ between formerly (pre)eclamptic women and women with normotensive pregnancies. A history of (pre)eclampsia showed a non-significant effect as a determinant of WML presence, independent of migraine.
Several studies have reported a higher risk of developing preeclampsia in migraineurs; others failed to find such a relationship (6). Such studies used various definitions for migraine and preeclampsia and were often performed during or shortly after pregnancy. We found a migraine prevalence of 33–36% among formerly (pre)eclamptic women with an average age of 43, which is higher than the prevalence 20–28% reported during the childbearing years (20–50 years) (1). Previous studies, including those by our own group, demonstrated that a history of migraine or a history of (preterm) (pre)eclampsia were associated with a higher prevalence of brain WML (2,4,5). Although the significance of WML in these conditions has not been elucidated yet, such WML have been associated with cognitive impairment in midlife adults, and with stroke and dementia in the elderly (10). WML are suggested to be of vascular origin. Both migraine as well as preeclampsia involve abnormal vascular reactivity, endothelial dysfunction, and increased platelet function in combination with a higher cardiovascular risk profile (11,12). Women with migraine may have a predisposition to poor compensation in response to vascular stressors such as pregnancy, and hence an increased likelihood to develop preeclampsia. Indeed, decreased arterial diameters and compliance in combination with increased blood pressure have been reported in migraine (13). The current study was not able to show a difference in migraine prevalence between formerly (pre)eclamptic women and women with normotensive pregnancies. In addition, the effect of a history of (pre)eclampsia as a determinant of WML presence was non-significant, while our previous studies clearly demonstrated such as effect in a larger cohort (4,5).
This study describes the prevalence of migraine in formerly (pre)eclamptic women and controls, and examines the relationship with WML, using strict diagnostic standardized and validated criteria for both migraine and (pre)eclampsia. This study knows some methodological limitations. Most important, study groups were relatively small and included relatively young women. Around 55% of the women who participated in our previous neuroimaging studies participated in the current study, which may have given rise to selection bias. Non-responder analysis, however, revealed no significant differences. As the cross-sectional study design hampers conclusions on cause-effect relationships, future longitudinal studies with longer follow-up may have the potential to assess the interaction between migraine and (pre)eclampsia with regard to WML. Interestingly, in our study in most women the onset of migraine preceded the development of (pre)eclampsia.
In conclusion, in this study migraine prevalence was not found to be an independent risk factor for the presence of WML in formerly (pre)eclamptic women and women with normotensive pregnancies. Because of the cross-sectional nature of our study and the limited number of participants, no definite conclusion on cause-effect relationships can be drawn. It is, however, of the utmost importance to study female-specific risk factors for cerebrovascular disease, taking migraine into account. Our study shows that larger, long-term, prospective studies are needed.
Article highlights
In this relatively small study, migraine was not found to be an independent risk factor for white matter lesion (WML) prevalence in formerly (pre)eclamptic women. Large, long-term, prospective studies are warranted to examine female-specific risk factors for cerebrovascular disease.
Footnotes
Funding
This work was supported by ZonMW (90700235), the Netherlands organization for health research and development.
Conflicts of interest
Dr G.M. Terwindt reports support for conference visits from Menarini, and independent support from NWO. Dr W.P.J. van Oosterhout reports support for conference visits from Menarini and Allergan. The other authors have nothing to declare.
Acknowledgments
This project was approved by both the UMCG (reference number METC 2004.216) and LUMC (reference number P07.079 C2. Addendum) Institutional Review Boards (Medisch Ethische Toetsingscommissie). All women signed informed consent.
