Abstract
Improved health-related quality of life (HRQOL) and reduced activity limitations are prime objectives of migraine therapy, but no data on the effect of preventive treatments on these outcomes are available. We monitored changes in HRQOL using the Short Form 36 (SF-36) and headache-related activity limitations using the Migraine Disability Assessment Score (MIDAS) in 141 consecutive migraine without aura patients on prophylaxis. A total of 102 patients completed the 3-month study. Mean (SD) number of headache days/month dropped from 8.0 (3.7) to 5.0 (2.3) (P <0.001). Mean monthly consumption of acute drugs reduced from 7.4 (3.5) to 4.4 (3.1) (P <0.001). MIDAS total score reduced (improved) significantly. All SF-36 scale scores increased (improved), most significantly. This first study to assess prospectively the impact of prophylaxis on HRQOL and daily activities in a large series indicates that migraine prophylaxis has the potential to reduce the global burden of migraine on individuals and society.
Keywords
Introduction
Migraine without aura is characterized by recurrent headache attacks. The pain is moderate to severe, of variable duration (up to several days) and can be associated with other symptoms, such as phonophobia, photophobia, nausea and vomiting. Prodromes such as somnolence and mood changes may also be present (1, 2) and the condition is present for long periods in patients’ lives. Migraine therefore has a major impact on sufferers’ lives. Several studies have shown that migraineurs are impaired in daily activities (3–5) and that their health-related quality of life (HRQOL) is generally poor compared both with the general population and with sufferers from other chronic disorders such as hypertension, diabetes and myocardial infarction (4, 6, 7). The high prevalence of migraine, particularly during the age of greatest productivity and responsibility (8), explains why the condition affects society as a whole and has considerable economic impact, particularly in terms of indirect costs (9–11).
Improvement in HRQOL and reduction in activity limitations (or functional disability) are prime objectives of migraine therapy (12, 13). While several studies have shown that appropriate acute treatments may improve these outcomes (14–21), little evidence is available on the effects of preventive treatments on the impact of migraine.
The aim of this study was to use standard validated instruments to monitor changes in HRQOL and headache-related activity limitations over time, in migraine without aura patients prescribed prophylactic therapy.
Materials and methods
Patients
Participants were recruited from among those attending 11 headache centres in the Region of Lombardy, northern Italy, between June 2002 and January 2003. Inclusion criteria were diagnosis of migraine without aura according to International Headache Society criteria (1988) (22), and presence of at least one of the following: at least four headache attacks per month or at least two attacks per month lasting 3 days; migraine significantly interfering with patient’s daily activities despite acute treatment; and contraindication to, failure of, or adverse effects from acute treatments. These eligibility criteria were drawn from most recent preventive treatment guidelines (12, 13, 23).
Study design and procedures
We carried out a prospective open longitudinal multicentre study. At the first visit (V1), after complete history, neurological examination and verification of eligibility, patients were informed of the aim and design of the study and gave written informed consent to participate. Responses to currently used acute treatments were also evaluated and modified where appropriate. Each patient was then given a diary card with instructions to report the dates of migraine episodes and all acute medications taken to abort attacks during the successive 2-month run-in period, during which no preventive treatment was given. An appointment was fixed for a return visit in 2 months.
At the second visit (V2) the patient completed the Migraine Disability Assessment Score (MIDAS) questionnaire (24) and the Short Form 36 (SF-36) survey (25). After verification that the patient still conformed to the eligibility criteria (by inspection of the diary card), prophylaxis was started. The prophylactic drug was chosen from those included in the most recent guidelines (12) and tailored to each patient’s characteristics. No changes in acute treatment were allowed during this phase of the study. Patients were instructed to continue compiling the diary card. An appointment was fixed for the third visit (V3) 3 months later. During the third visit, the diary card was again reviewed and the MIDAS and SF-36 forms were completed a second time.
Outcome measures
Headache frequency during run-in and prophylaxis, as number of days with headache per month, and acute medication consumption during the same periods, as number of medications used per month, were determined from the diary cards.
Headache-related activity limitations were assessed from MIDAS. MIDAS consists of five questions investigating the influence of headache on three activity domains over the preceding 3 months: questions 1 and 2 investigate paid work, enquiring about number of days off work due to headache and number of days where productivity was reduced by half or more; questions 3 and 4 ask the same questions about household work; question 5 enquires about missed days of recreational, social and family activities (24). The MIDAS total score is obtained by summing the scores (number of days affected) of these five questions.
HRQOL was determined using the SF-36 (25). The SF-36 is the most commonly used generic HRQOL questionnaire. It consists of 36 items grouped into eight domains: Physical Functioning (PF)—extent to which health limits physical activities; Physical Role Limitation (RP)—extent to which physical health interferes with work or other daily activities; Bodily Pain (BP)—effect of pain on work, inside and outside the home; General Health (GH)—personal evaluation of health, and resistance to illness; Vitality (VT)—personal assessment of level of energy and ‘pep’; Social Functioning (SF)—extent to which physical health or emotional problems interfere with work or other daily activities; Role Limitation Emotional (RE)—extent to which emotional problems interfere with normal social activities; and Mental Health (MH)—general mental health, including depression, anxiety, and behavioural-emotional control. The validated Italian versions of MIDAS (26) and of SF-36 were used (27).
Data analysis
The significance of differences between the completer and non-completer groups was tested using Fisher’s exact test. Changes in the outcome measures between V2 and V3 were compared using the Wilcoxon signed test or paired Student’s t-test. The Stata Statistical Software, release 7.0 (Stata Corporation, College Station, TX, USA), was used.
Results
Between June 2002 and January 2003, 141 consecutive patients conforming to the eligibility criteria were recruited. Seven patients did not present for the second visit and another withdrew during run-in as she became pregnant. Thus 133 patients presented at the second visit. In 14 of these patients the inclusion criteria no longer held: in 10 there had been a clinical improvement during run-in, presumably due to a prescribed change [from non-steroidal anti-inflammatory drugs (NSAIDs) to triptans] in acute treatment at V1, resulting in a reduction in headache frequency, impact or both. Thus, 119 patients were prescribed prophylaxis.
The drugs prescribed were: flunarizine (42 patients); β-blockers, propranolol or metoprolol (23 patients); antidepressants, amitriptyline or selective serotonin reuptake inhibitors (SSRIs) (22 patients); antiepileptic drugs, valproate or topiramate (eight patients), and other drugs (pizotifen, NSAIDs) (seven patients).
Seventeen of the 119 patients who began the prophylaxis dropped out. Three withdrew consent during the first month for personal reasons; eight did not present at the third visit, and six withdrew during the first 2 months due to adverse events (one for depression and two for increase in body weight in those given flunarizine; two for fatigue and hypotension in those on β-blockers, one for nausea and vomiting in a patient on valproate). A total of 102 patients therefore completed the study.
Mean (SD) number of days with headache/month dropped from 8.0 (3.7) at V2 to 5.0 (2.3) at V3 (paired Student’s t-test, P < 0.001). Mean monthly consumption of acute drugs reduced from 7.4 (3.5) at V2 to 4.4 (3.1) at V3 (paired t-test, P < 0.001). Median (interquartile range) MIDAS total score was 25.5 (11–40) at V2 and 8.0 (2–23) at V3 (Wilcoxon signed test, P < 0.0001). Median changes in all individual MIDAS scores also indicated a significant improvement in activity limitations, as reported in Table 2.
Changes in MIDAS total score and individual question scores in 102 migraine without aura patients after 3 months of prophylaxis

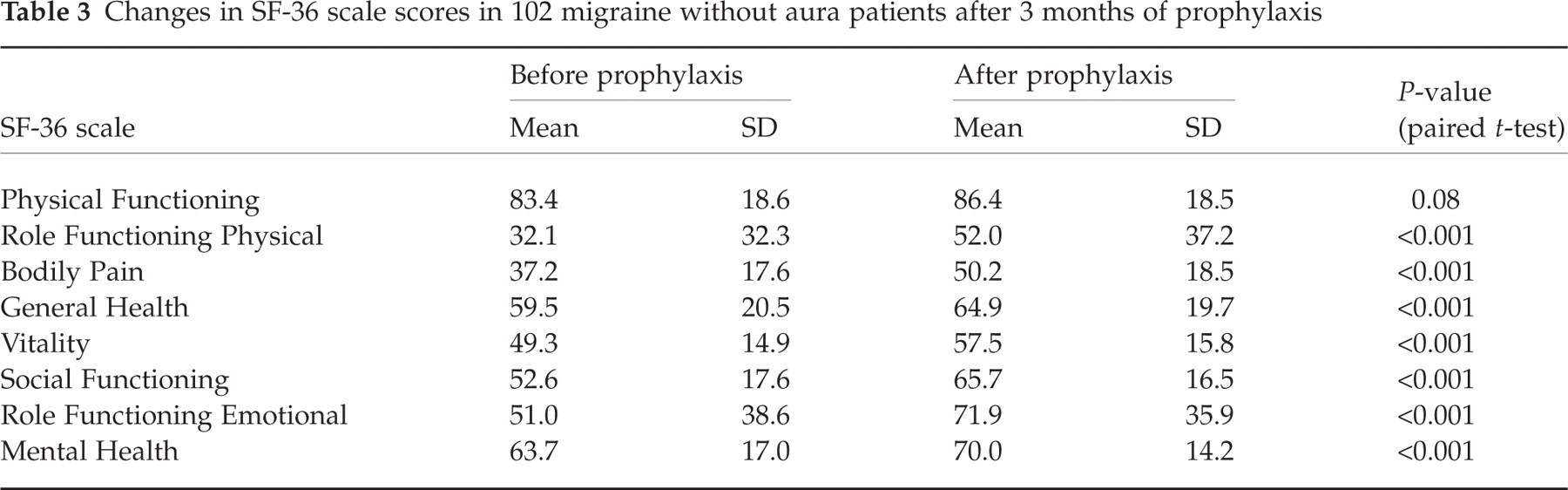
All SF-36 scale scores increased after prophylaxis. Differences were highly significant for all except Physical Functioning (Table 3).
Changes in SF-36 scale scores in 102 migraine without aura patients after 3 months of prophylaxis
Discussion
Over the last decade attention has focused on characterizing and quantifying the personal and social impact of migraine. Standardized instruments to assess HRQOL and activity limitations have been used in clinical series and in population studies. These studies have demonstrated that migraine impairs work productivity, limits daily activities in family and social spheres, and that migraineurs have poor HRQOL (3–11).
Recent guidelines on the treatment of migraine stipulate that recovery of normal functioning and improvement in HRQOL should be among the primary objectives of therapy (12, 13).
Changes in the functional consequences of migraine, in patients receiving appropriate acute treatment, have been evaluated by several studies. In patients taking triptans, the impact of migraine on work and non-work activities reduced, and HRQOL improved (14–21). However, much less is known about the effect of preventive treatments on the impact of migraine. A recent study assessed whether preventive medication reduced resource consumption related to migraine management (28), finding that prophylaxis was effective in reducing use of other medications, number of visits to the physician and emergency departments, and number of neuroimaging examinations.
There are few data on the recently formulated therapy objectives (changes in HRQOL and headache-related activity limitations) over time in migraine without aura patients prescribed prophylactic therapy (29, 30). The present study is the first to assess prospectively the effects of different prophylactic treatments on HRQOL and ability to perform daily activities using both a generic questionnaire and a migraine-specific tool in a large series. We found significant reductions in headache frequency and symptomatic drug consumption as a result of prophylaxis. We also found a marked reduction in MIDAS total score, indicating significant improvements in ability to function in different daily activities, with a decrease in number of days on which work and non-work activities were missed or significantly impaired. Improvements in HRQOL as assessed by the SF-36 were also marked, with significant increases in scores of scales exploring: extent to which physical health interfered with roles and activities in social, work and personal areas; general sense of well-being; and behavioural-emotional control. The lack of a significant improvement in the Physical Functioning scale requires comment. This scale assesses functions requiring moderate physical effort such as lifting a weight, walking and going up and down stairs; and these activities are little compromised in migraine sufferers, as illustrated by a high baseline (V2) score, close to that of the general population.
Dahlof in 1989 (29) used a questionnaire (Minor Symptom Evaluation, MSE) to assess appreciation of treatment in a sample of Swedish migraineurs after prophylaxis with propranolol or flunarizine in a double-blind, parallel-group study. Three domains (contentment, vitality and sleep) derived from the 15 questions were analysed, but no significant improvement was found during the 5-month follow-up.
Improvement in HRQOL has been recently reported in an analysis of data pooled from three randomized, double-blind, placebo-controlled studies on topiramate prophylaxis in migraine (30). All three domains (role restriction, role prevention and role emotional) of the migraine-specific quality-of-life questionnaire (MSQ) improved significantly after treatment, in line with our own findings.
In our study, the participating clinicians were free to prescribe the prophylactic medication they considered most appropriate; the study is therefore inappropriate for comparing different prophylactics. Nevertheless, we performed an ad hoc analysis of changes in MIDAS and SF-36 in the three largest medication groups (flunarizine, β-blockers and antidepressants). We found that MIDAS scores were significantly lower (indicating improvement in activity limitations) after prophylaxis in all three groups. For SF-36, the score improvements in patients on flunarizine and antidepressants were closely similar to those of the entire completer group, while for patients on β-blockers, improvements were not significant for Bodily Pain, General Health, Vitality, or Role Functioning Emotional (data not presented). This finding suggests it may be worth designing specific studies to determine whether prophylactics differ significantly in their ability to improve migraine-related functional compromise and quality of life.
A confounding effect of acute treatments can be excluded because the patients used the same symptomatic drugs during the 3-month prophylaxis period as they did in the run-in period.
As in all clinical studies in migraine, placebo effects may have played a role. Placebo effects can be large in studies that monitor pain (31, 32). Furthermore, expectations arising from recruitment to a trial may have contributed to reducing the emotional and functional impact of migraine. However, placebo effects could not be estimated in this open uncontrolled trial.
Of the patients who were given prophylaxis, 14% did not complete the course (six for side-effects, 11 for reasons that are unclear). This is rather a high proportion; however, the characteristics of non-completers did not to differ greatly from those of completers (Table 1). Note, however, that non-completers did use triptans more often than completers and the efficacy of these drugs in reducing the intensity and duration of attacks may have induced patients to stop taking the prophylactic.
Characteristics of 119 patients started on migraine prophylaxis

AED, Antiepileptic drugs.
Significantly superior in completers vs. non-completers, Fisher’s exact test, P < 0.05.
Significantly superior in non-completers vs. completers, Fisher’s exact test, P < 0.04.
No significant difference, Fisher’s exact test, P = 0.6.
The presence of other chronic disorders was not systematically assessed. Migraineurs often have comorbidities, particularly psychiatric conditions (33), which may influence the aspects we measured. However, none of our patients received specific treatments for other disorders during the study period. It is possible that antidepressive compounds could have improved coincidental depression, with consequent influence on migraine-related impact. However, the improvements in disability and HRQOL scores in patients receiving antidepressants were similar to those in the entire completer group.
These limitations, and particularly the fact that no placebo run-in period or control group were used, mean that the improvements found in this study have to be interpreted cautiously. Nevertheless, they suggest that preventive treatments for migraine can significantly improve ability to function and perceived quality of life. Thus, prophylaxis can not only influence the classical endpoints of migraine treatment (headache frequency and analgesic consumption) but also reduce the global burden of migraine on individuals and on society. A corollary of these findings is that both MIDAS—a migraine specific instrument—and the generic SF-36 appear sensitive to clinical changes following prophylaxis. However, controlled studies are required to corroborate these conclusions.
Footnotes
Acknowledgements
The following members of the Lombardy Headache Project Group (Gruppo Progetto Cefalee Lombardia) participated in this study: P. Bernardoni, Neurology Service, Pieve di Coriano, Italy; D. D’Amico and S. Usai, C. Besta National Neurological Institute, Milan, Italy; R. De Marco and T. Partziguian, Neurological Department, Ospedali Riuniti, Bergamo, Italy; E. Ferrante, Neurological Department, Niguarda Cà Granda Hospital, Milan, Italy; F. Frediani, Neurological Department, Policlinico S. Pietro, Ponte S. Pietro, Italy; N. Massetto and M. Secchi, Neurological Department, S. Paolo Hospital, Milan, Italy; S. Misceo, Neurological Department, Predabissi Hospital, Vizzolo Predabissi, Italy; F. Moschiano, Neurological Department, L. Mandic Hospital, Merate, Italy; R. Rao, Neurological Department, Spedali Civili, Brescia, Italy; P. Santoro and E. Agostoni, Neurological Department, S. Gerardo Hospital, Monza, Italy; C. Tonini and A. Coppola, Neurological Department, G. Salvini Hospital, Garbagnate Milanese, Italy.
