Abstract
Background
The King-Devick test is a timed rapid number naming task that involves complex cerebral functions. The objective of this pilot exploratory study is to determine whether there is a difference in the King-Devick test during a migraine attack compared to the interictal phase.
Methods
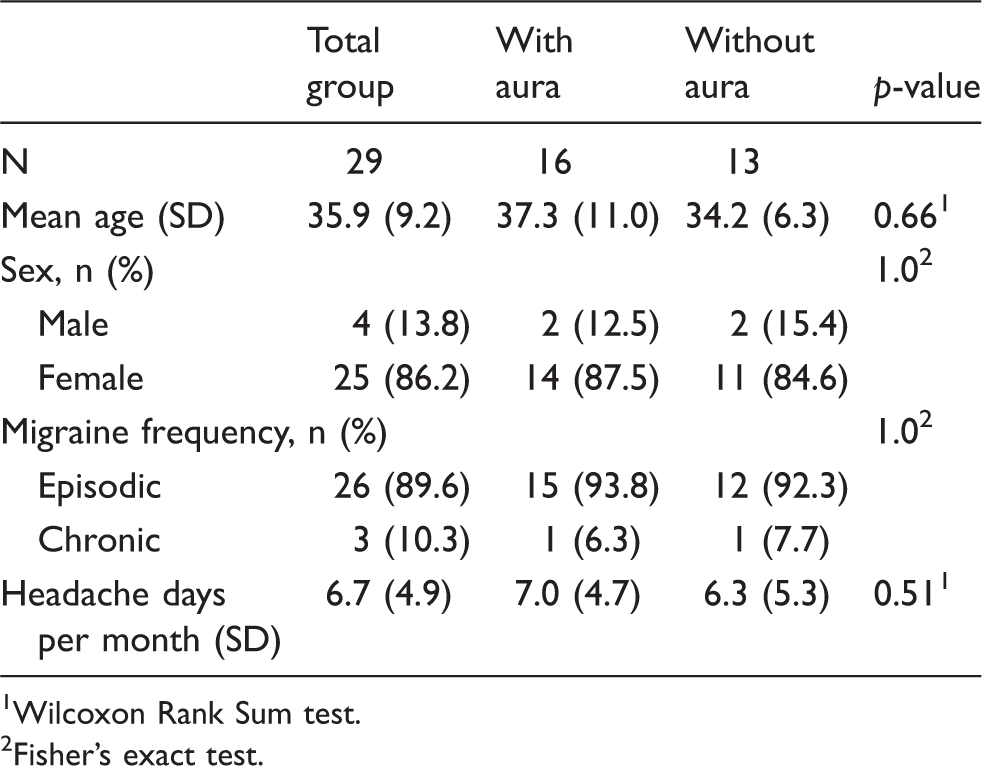
We evaluated 29 adult subjects with migraine with aura or migraine without aura. For each participant, we performed King-Devick tests during migraine attacks and interictal phases. Subjects served as their own controls.
Results
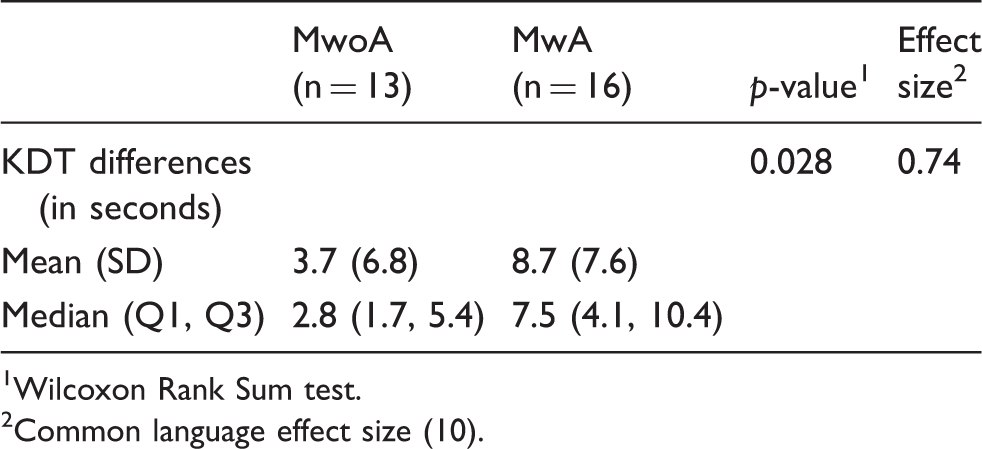
The King-Devick test was slower during the migraine attack compared to the interictal baseline (median 4.6 sec slower, p < 0.001). The slowing of the King-Devick test during migraine attack was more prominent in those with migraine with aura compared to subjects with migraine without aura (median 7.5 vs. 2.8 sec, p = 0.028).
Conclusions
This exploratory, observational study shows changes in the King-Devick test during migraine compared to the interictal phase. Future studies are required to determine if the King-Devick test may be used as a rapid and simple tool to objectively characterize migraine-associated disability.
Introduction
Migraine is accompanied by a myriad of symptoms such as sensory sensitivities (photophobia, phonophobia), visual impairment and cognitive dysfunction, which reflects altered central nervous system functioning. Past studies have demonstrated alterations in central nervous system functioning with impaired neuropsychological performances (1). However, the studies were done comparing migraine patients to healthy controls; therefore, it was unclear how migraine attacks affect cerebral function compared to one’s interictal phase. Additionally, the neurocognitive exams could take a long time to perform. It would be beneficial to have a simple, fast and objective testing modality that could be applied to quantify altered task-performing ability and characterize migraine-related dysfunction and disability during attacks.
The King-Devick test (KDT) is a rapid number naming test performed on an iPad or tablet that requires intact vision, saccadic eye movements, language, concentration and attention. Participants are asked to read the numbers on the test cards from left to right as quickly as possible without making errors (Figure 1). The time required to complete each test card and the number of errors are recorded. The KDT was originally designed to assess reading difficulties in children. It has since been applied to characterize cerebral function in different neurological disorders. KDT is now a validated sideline concussion screening tool (2), and it has also been shown to be sensitive to hypoxia (3) and sleep deprivation (4), as well as the progression of neurological diseases such as multiple sclerosis (5) and Parkinson’s disease (6).
The King-Devick test. Subjects were asked to read the numbers on the cards from the left to the right as quickly as they could, without making any errors. This requires intact saccadic eye movement, vision, language, concentration and attention.
The objective of this exploratory, observational study is to determine whether KDT: a) Demonstrates a difference during a migraine attack compared to the interictal phase, b) demonstrates between-group differences among patients with migraine with aura (MwA) and migraine without aura (MwoA).
Methods
This study was approved by the institutional review board at Mayo Clinic in August 2016. All subjects provided signed written informed consent prior to enrollment. The enrollment and data collection took place from September 2016 to November 2017.
Subjects
Demographic and disease characteristics of patients with migraine with aura and migraine without aura.
Wilcoxon Rank Sum test.
Fisher’s exact test.
Methods
For each participant, we planned to obtain KDT data during a migraine attack, and between migraine attacks (interictal phase). The interictal phase was defined by a duration of at least 48 hours since the termination of the last migraine attack, and without experiencing any headache or premonitory symptoms. We asked our study participants to contact us to complete KDT tests during a migraine attack and an interictal phase. The KDT tests were conducted by the same examiner (C, C-C). To avoid the learning effect, participants underwent testing in random sequence; testing was performed either first during the interictal phase then later during the migraine attack phase, or vice versa.
During the first visit with a subject, we confirmed the diagnosis of migraine and collected their migraine characteristics before proceeding with the KDT. Migraine characteristics included headache frequency, whether they had aura and the types of aura, the types of acute headache medications, the frequency of acute medication use, and whether they were using preventive medications and the types of preventive medications. During their migraine attack we collected information on the time of headache onset, whether they had taken acute headache medications, the headache pain level on visual analog scale (from 0–10), and symptoms they were experiencing other than headache, such as aura symptoms, photophobia, phonophobia, blurry vision, nausea, allodynia, neck stiffness, and cognitive dysfunction (including mental fogginess, mental slowing, and difficulty with concentration).
For the KDT, participants were asked to read numbers on tests cards on an iPad from left to right without error as quickly as possible. The amount of time and the number of errors made were recorded. At least two trials were performed during the interictal and acute migraine phases. The shortest time for which they could read without making any errors was used as their score. The iPad was held by the participants approximately 40 cm from their eyes. The iPad display was set at maximum brightness for each test and the ambient light setting was similar among clinic rooms where tests were performed. Collected data was kept in a Redcap database for analysis.
Statistical analysis
As a pilot exploratory study, power analysis was not utilized. All paired comparisons were performed using the Wilcoxon Signed Rank test, while the Wilcoxon Rank Sum test was used to perform all non-paired comparisons between groups. Correlation between numerical variables was quantified using Spearman’s correlation. All analyses were performed using SAS v9.4 (SAS Institute; Cary, NC). All hypothesis tests performed were two-sided.
Results
KDT comparing migraine attack vs. interictal
The mean differences in the KDT performances during a migraine attack vs. interictal baseline of the group (n = 29).

Wilcoxon Signed Rank test.
Migraine attack vs. interictal baseline in migraine without aura vs. migraine with aura
Differences in KDT performance during a migraine attack compared to the interictal baseline for migraine without aura versus migraine with aura.
Wilcoxon Rank Sum test.
Common language effect size (10).
The effect of pain level on change in KDT scores
The average pain level during migraine attacks at the time of testing was 3 on an 11-point visual analog scale (VAS) among all subjects (ranged 1 to 7). There was no correlation between pain level and the change in KDT scores.
Effect of non-headache symptoms during migraine attack on the change in KDT scores
Analyses of the effect of non-headache symptoms during migraine attack showed no difference between the KDT scores and the presence of symptoms including photophobia or cognitive deficits during migraine attack. We did not observe enough events of the following symptoms to evaluate their respective effects on KDT scores: Vision changes, language deficits, nausea, neck pain, and allodynia.
Effect of headache frequency and the time of migraine onset on the KDT scores
KDT tests were performed between 1–144 hours (mean 19 hours) after headache onset, and our subjects’ headache frequency ranged from 1 day per month to 20 days (mean 6.7 days) per month. We did not find any correlation between the headache frequency, or the timing between the headache onset to test time, of the KDT slowing during migraine attack compared to their interictal baseline (KDT Spearman correlation = 0.16, p = 0.40, and Spearman correlation = −0.33, p = 0.08, respectively).
Preventive medications
Median KDT was 4.0 seconds slower during migraine attack compared to the interictal baseline in subjects taking preventive medications (n = 9; including six subjects on topiramate, one on amitriptyline, one on magnesium, two on onabotulinumtoxinA, one on propranolol, and one on paroxetine). For subjects not on preventive medications, the KDT was 5.0 seconds slower (n = 20) compared to the interictal baseline. There was no between-group difference (p = 0.81).
Discussion
The KDT is a brief and simple test of rapid number naming. It requires a number of complex central nervous system functions including intact vision, saccadic eye movements, attention, processing, reading and speech skills. Our exploratory pilot study showed that the KDT was slower during a migraine attack compared to the interictal baseline (median of differences 4.6 seconds). Between MwA and MwoA, the effect of migraine attack on change in KDT performance is more prominent in those with MwA compared to MwoA (median 7.5 vs. 2.8 seconds slower during migraine attack compared to baseline, p = 0.028). Of note, none of the testing was performed while subjects were experiencing visual aura. A previous functional MRI study reported that patients with MwA had an altered resting-state visual network connectivity compared to those with MwoA and healthy controls during the interictal phase (7), which could be related to more slowing during the KDT testing.
Migraine is starting to be recognized as more than just a headache disorder and therefore having ways of assessing migraine beside pain levels is important. We did not find any correlation between the change in KDT scores and the pain level during tests. This requires further evaluation, but may suggest that changes in the KDT are capturing components of migraine-associated disability beyond pain associated disability. In regard to the effect of non-headache symptoms during testing on the change in KDT scores, there was no difference in KDT scores depending on the presence of photophobia or cognitive deficits. Ten of our subjects reported cognitive deficits during migraine attacks when testing was performed. For those subjects, their KDT score was 8.5 seconds (median) slower than their interictal baseline. For those that did not perceive cognitive deficits while testing, the KDT was 4.0 seconds (median) slower than their interictal baseline. This suggests possible cerebral function changes even when there was no subjective cognitive dysfunction reported. Many reports of migraine-associated disability rely on subjective patient report. Having an objective test that can capture dysfunction regardless of subjective patient report may be useful. One subject reported language difficulties during migraine while testing, and the KDT score was 22.1 seconds slower compared to the interictal baseline. While this observation was interesting, the number of subjects was too small to make any conclusion.
Since migraine typically presents with photophobia and pain, it might be a straightforward assumption that performance on visual tasks, such as the KDT, might be poorer during a migraine attack compared to the interictal phase. Currently, there are limited ways to objectively measure the dysfunction and disability experienced by patients during a migraine attack, especially measures that are brief and simple. To establish the practical utility of KDT in migraine, further studies are required to test whether the KDT may be used as an objective and quantitative measure of migraine disability, which could provide clinicians and researchers with another indicator of disease severity and treatment efficacy in addition to the Visual Analog Scale for headache pain during the migraine attack phase.
Furthermore, examining whether there is a difference in the KDT during other migraine phases (premonitory and postdrome) or leading up to these phases, may allow us to further understand migraine pathophysiology. Additionally, future studies are warranted to duplicate our current findings, and see whether these findings are unique to migraine by comparing KDT scores among different headache disorders (for example tension-type headache).
We chose to use the iPad version to perform KDT due to benefits of electronic testing including ease of storing test history and consistency in timing with automatic timing. A previous study comparing the paper and electronic version of KDT showed high reliability using the same platform and incompatibility across platforms (8). Migraine is often accompanied by photophobia and difficulty using screens, which might make use of an iPad test more difficult for patients. Given that the majority of people use screens in their everyday lives, we believe using the iPad version of the KDT is a reasonable testing format.
The results of this exploratory, observational study are promising but future studies will be needed. The small sample size is a limitation of this study. It is possible that the results of this study could be from selection bias in a small group of subjects.
Although the KDT testing takes no more than a few minutes, the version of the KDT used for this study required a trained test administrator. From a clinical standpoint, the requirement of a test administrator makes the testing more difficult and might limit the clinical utility. A new version of the KDT has been developed which uses voice-recording software so the testing can be individually administered. A study evaluating the individually administered KDT is pending, and if it shows similar changes to those seen in the current study would have more clinical utility given the ease of use and portable nature. Additionally, the individually administered KDT would allow for more recording opportunities and therefore the ability to potentially capture test results in the premonitory and postdrome phase.
Previous concussion studies reported learning effects for the KDT. For athletes without a head trauma, the post-fight KDT score was 1.9 seconds faster (9). To avoid learning effects in our study, we chose to administer the tests in random sequences. For eight of the 29 subjects, data during migraine attack was collected prior to interictal testing. For 21 subjects, interictal testing was performed first. As the majority of the subjects underwent interictal testing first, the slowing during migraine attack would go against a practice effect.
Overall, this is an exploratory study to determine whether there would be differences between the KDT during a migraine and the interictal phase and differences between MwA and MwoA. These potential differences may serve as an objective assessment of the migraine attack phase versus the interictal state. Performance of the KDT requires intact saccadic eye movements, vision, language, concentration and attention. Our study does not allow us to determine whether a deficit among one, some or all of these required skills explains the changes in KDT performance. Future research should confirm these findings in a larger sample, evaluate the KDT during the premonitory and postdrome phase, and compare KDT results among different headache disorders.
Conclusion
The KDT, an objective quantitative measure of rapid number naming that requires intact saccadic eye movements, vision, language, concentration, and attention, showed slowing during migraine attacks compared to the interictal baseline. Furthermore, the slowing of the KDT score was more prominent for those that had MwA compared to those with MwoA. Future studies are required to see if the KDT could be an effective tool to objectively characterize migraine-associated disability and its clinical utility.
Abbreviations
KDT: King-Devick test; SD: standard deviation; MwA: migraine with aura; MwoA: migraine without aura.
Clinical implications
The KDT score was slower during the migraine attack compared to the interictal baseline, and the slowing of KDT during migraine attack was more prominent in those with MwA compared to subjects with MwoA. The KDT may be a rapid and simple tool to objectively characterize migraine-associated disability.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C-CC has nothing to disclose; AJS has received compensation from serving on advisory boards and/or consulting within the past 5 years for Alder, Amgen, Eli Lilly & Company, eNeura, and Novartis. AJS has also received funding for travel and speaking from the American Academy of Neurology and American Headache Society. MRB and MAG have nothing to disclose; JVdP reports speaking fees for Amgen and Novartis, and consulting for Healint and Teva. She has received a research grant from Amgen.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
