Abstract
Background
We evaluated the effect of erenumab, a fully human monoclonal antibody that inhibits the canonical calcitonin gene-related peptide receptor, on migraine-related disability, impact, and health-related quality of life among patients with episodic migraine.
Methods
Patients enrolled in a phase 3, 6-month, double-blind, placebo-controlled study of once-monthly erenumab 70 and 140 mg for migraine prevention (STRIVE) used an eDiary during the baseline and double-blind treatment phases to complete validated, specific questionnaires, including the modified (monthly) Migraine Disability Assessment Questionnaire; Headache Impact Test; and Migraine-Specific Quality of Life Questionnaire-role function-restrictive (MSQ-RFR), -role function-preventive (MSQ-RFP), and -emotional function (MSQ-EF).
Results
A total of 955 patients were randomized to receive erenumab 70 mg (n = 317), erenumab 140 mg (n = 319), or placebo (n = 319). Erenumab versus placebo resulted in significantly greater improvements in all patient-reported outcomes; changes from baseline were numerically higher with 140 mg erenumab. Improvements occurred rapidly and were maintained over 6 months of treatment. Between-group differences from placebo over months 4–6 for the 70- and 140-mg dose groups were, respectively, −2.1 and −2.8 for modified (monthly) Migraine Disability Assessment Questionnaire, −2.1 and −2.3 for Headache Impact Test, 5.1 and 6.5 for MSQ-RFR, 4.2 and 5.4 for MSQ-RFP, and 5.2 and 6.7 for MSQ-EF (p < 0.001 for all). Erenumab also significantly reduced the proportion of patients with severe and very severe migraine-related disability and increased the proportion of patients with clinically meaningful improvements in migraine-related impact and health-related quality of life.
Conclusion
Erenumab reduced migraine disability and impact and improved patients’ health-related quality of life, reinforcing its role as a promising new therapy for migraine prevention.
Keywords
Introduction
Migraine can result in severe disability and may impose a substantial burden on patients and families (1,2). Disruption to social and leisure activities and physical and emotional functioning, as well as reduction in health-related quality of life (HRQoL), may occur both during and between migraine attacks (3,4). Migraine-related disability can result in loss of productivity while at work (presenteeism) as well as absence from work (absenteeism) (5,6), both of which may result in a substantial economic burden to the individual, employers, and society (5,7). This burden is present in both episodic migraine (fewer than 15 headache days per month) and chronic migraine (at least 15 headache days per month, with at least eight migraine days per month).
Guidelines for the management of people with migraine provide recommendations for pharmacologic intervention (acute and preventive medications) as well as behavioral and physical treatments for migraine (8). Expert consensus recommends that preventive pharmacologic treatment should be offered or considered depending on the number of headache days per month and level of headache-related disability (9). The key goals of preventive migraine therapy are twofold: to reduce attack frequency and severity, and to improve function and reduce disability.
Preventive pharmacologic therapy has been shown to reduce migraine attacks and headache days, and improve migraine-related disability, impact, and HRQoL. Improvements in migraine-related disability and impact (Migraine Disability Assessment Questionnaire [MIDAS] and Headache Impact Test [HIT-6™]) were observed in an open-label study of preventive therapy with topiramate (10), and improvements in migraine-related impact (HIT-6) and HRQoL (Migraine-Specific Quality of Life Questionnaire [MSQ]) were observed in placebo-controlled studies of preventive therapy with onabotulinumtoxinA (11,12).
Erenumab (in the US, erenumab-aooe) is a fully human monoclonal antibody that selectively binds to and inhibits the canonical calcitonin gene-related peptide (CGRP) receptor (13). In STRIVE, a pivotal, phase 3, placebo-controlled clinical trial in patients with episodic migraine, preventive treatment with once-monthly (QM) 70 or 140 mg erenumab significantly reduced the number of monthly migraine days and monthly acute migraine-specific medication days compared with placebo (14). In addition, the odds of having at least a 50% reduction in mean migraine days were approximately 2.1 and 2.8 times greater with erenumab (70 and 140 mg, respectively; both p < 0.001) versus placebo (14). Erenumab also led to significant improvements in patients’ physical functioning, as measured by the Migraine Physical Function Impact Diary (MPFID) (14), a patient-reported outcome tool that was developed to evaluate the benefit of migraine interventions on the average daily impact of migraine on patients’ physical impairment and everyday activities (15,16).
Here, we extend the assessment of patient-reported outcomes from the 6-month double-blind treatment phase of the STRIVE study by evaluating the effect of erenumab on migraine-related disability, impact, and HRQoL among patients with episodic migraine using MIDAS, HIT-6™, and MSQ, respectively.
Methods
Study design and patient population
Details of the design of this phase 3, multicenter, randomized, double-blind, placebo-controlled study (ClinicalTrials.gov, NCT02456740) have been published (14). Migraine was defined in accordance with the International Classification of Headache Disorders-3 beta version (ICHD-3 beta) (17), the same definition as the current, final ICHD 3rd edition (18). Eligible patients were 18–65 years old with a history of migraine (based on medical records and/or self-reported) for at least 12 months before screening and episodic migraine (4–14 migraine days per month, on average) during the 3 months before screening and during the 4-week baseline phase of the study. Patients were excluded if they had no therapeutic response to more than two migraine-preventive treatment categories (i.e. no benefit in frequency, severity, or duration of migraine attacks after at least 6 weeks of treatment at the recommended dose based on the investigator’s assessment), as documented in patient records or reported by the patient.
At the end of the 4-week baseline phase, 955 eligible patients from sites in Canada, Austria, Belgium, Czech Republic, Finland, Germany, Poland, Slovakia, Sweden, the United Kingdom, Turkey, the Netherlands, and the United States were randomized 1:1:1 to receive monthly subcutaneous injections of erenumab 70 mg, erenumab 140 mg, or placebo for 6 months (double-blind phase). Randomization was stratified according to region (North America versus other) and use of migraine-preventive medication (current use, previous use only, or no previous or current use). Patients in each double-blind treatment group were then rerandomized 1:1 to receive erenumab 70 or 140 mg for 7 months (active treatment phase, dose blinded). Here, we report patient-reported outcomes from the double-blind treatment phase of the STRIVE study.
The trial protocol was approved by an independent ethics committee at each trial site, and all patients provided written informed consent before the start of any study-related procedures.
Outcome measures
During the baseline and double-blind treatment phases of the study, patients recorded information about their migraine and nonmigraine headaches using a handheld electronic diary (eDiary). Patients also used their eDiary to complete a number of validated, self-administered, specific questionnaires, including the MPFID (results previously published (14)), MIDAS, HIT-6™, and MSQ version 2.1. Each questionnaire was translated for patients in non–English-speaking countries.
MIDAS
MIDAS is a five-item questionnaire that was developed to measure migraine-related disability over the previous 3 months (19). In this study, we utilized the modified (monthly) MIDAS (mMIDAS), which has a 4-week recall period to reduce recall bias and improve the accuracy of the results (20,21). mMIDAS assesses absenteeism (complete disability) and presenteeism (reduced participation) in several domains, including work, school, family, social, and leisure activities. The mMIDAS score is the sum of missed days due to a migraine from paid work, school, housework, and nonwork (family, social, and leisure) activities; and days at paid work or housework where productivity was reduced by at least half.
To facilitate the interpretability of the results, a post hoc analysis was performed in which the mMIDAS was converted into an every-3-month assessment of disability, in line with the original instrument: the converted mMIDAS score over months 4, 5, and 6 of the double-blind phase of the study was calculated by taking the average of the available postbaseline assessments and multiplying the average by 3 (if at least one mMIDAS was present at months 4, 5, and 6). The converted total scores were categorized based on the scoring system for the 3-month MIDAS, with scores of 0–5 representing minimal or infrequent disability (grade 1); 6–10, mild or infrequent disability (grade 2); 11–20, moderate disability (grade 3); and ≥21, severe disability (grade 4).
The between-group minimally important difference (defined as the smallest difference in scores between groups that patients perceive as beneficial or important) for MIDAS (3-month recall period) is a 5-point decrease (−5) (22).
Analyses of MIDAS included change from baseline in mMIDAS (total, absenteeism, and presenteeism) scores; converted, 3-month MIDAS scores; and the percentage of patients who had 3-month MIDAS total scores of ≥21 (severe disability) or ≥41 (very severe disability) (23).
HIT-6™
HIT-6™ (4-week recall period) is a six-item tool that assesses the impact of headache, including the frequency of headache pain severity, headaches limiting daily activity, wanting to lie down when headache is experienced, feeling too tired to work or do daily activities because of headache, feeling “fed up” or irritated because of headache, and headaches limiting ability to concentrate or work on daily activities (24–26). Response categories are “never”, “rarely”, “sometimes”, “very often”, and “always”, which are assigned 6, 8, 10, 11, and 13 points, respectively, and then summed to produce a total HIT-6™ score ranging from 36 to 78. HIT-6™ scores are then categorized into four grades, with a score of ≤49 representing little or no impact; 50–55, some impact; 56–59, substantial impact; and 60–78, severe impact due to headache (24,26). The between-group minimally important difference for HIT-6™ is a 1.5-point decrease (−1.5) (27).
Analyses of HIT-6™ include change from baseline in HIT-6™ total scores and the percentage of patients who achieved a ≥5-point reduction (improvement) from baseline in HIT-6™ score.
MSQ
MSQ (4-week recall period) measures three dimensions: how migraine attacks limit daily social and work-related activities (role function-restrictive, RFR [seven items]) and how they prevent these activities (role function-preventive, RFP [four items]) as well as the emotions associated with migraine attacks (emotional function, EF [three items]) (28,29). Patients respond to each item using a six-point scale: “none of the time”, “a little bit of the time”, “some of the time”, “a good bit of the time”, “most of the time”, and “all of the time”, which are assigned scores of 1–6, respectively. Scores are then rescaled from 0 to 100, with higher scores indicating better migraine-specific HRQoL. The between-group minimally important differences for MSQ domains are 3.2-, 4.6-, and 7.5-point increases for MSQ-RFR, MSQ-RFP, and MSQ-EF, respectively (30).
Analyses of MSQ included change from baseline in MSQ scores (MSQ-RFR, MSQ-RFP, and MSQ-EF), percentage of patients who achieved a ≥5-point clinically meaningful increase (improvement) from baseline in MSQ-RFR and MSQ-RFP scores (post hoc analysis), and percentage of patients who achieved a ≥8 point, clinically meaningful increase (improvement) from baseline in MSQ-EF scores (post hoc analysis).
Statistical analysis
Patient-reported outcome data were analyzed for all randomized patients who received at least one dose of assigned treatment and had at least one change from baseline in outcome measurement with the instrument of interest during the double-blind treatment phase of the study. Data were analyzed using SAS 9.4 (Cary, NC, USA).
For monthly continuous endpoints, changes from baseline values were calculated for each monthly time point. Least-squares means at each monthly time point and the mean over months 4–6 were estimated from a generalized linear mixed-effects model that included treatment group, baseline values, stratification factors, scheduled visits, and the interaction between treatment and scheduled visits without imputation for missing data. For the 3-month MIDAS total score, an analysis of covariance model that included treatment, stratification factors, and baseline value as covariates was used to estimate the least-squares means, with missing data imputed using the last observation carried forward method. For dichotomous endpoints, odds ratios were estimated from a stratified Cochran-Mantel-Haenszel test, with missing data imputed as nonresponse. p-values comparing each erenumab dose group with placebo are descriptive and are not adjusted for multiplicity for the analyses of these exploratory endpoints. The patient-reported outcomes are from exploratory endpoints; therefore, the reported p-values are descriptive and not adjusted for multiplicity. Statistical significance was determined by comparing descriptive p-values with nominal significance level at p ≤ 0.05.
Results
Patient disposition and demographics
A total of 955 eligible patients were randomized to receive erenumab 70 mg (n = 317), erenumab 140 mg (n = 319), or placebo (n = 319); 858 (89.8%) patients completed the 6-month double-blind treatment phase of the study, among whom 96% completed the patient-reported outcomes at each time point. The clinical trial sites are listed in Supplementary Table 1. Details regarding the demographics and baseline characteristics of the randomized patients have been published (14). In brief, most patients were women (85%), with a mean age of 41 years and mean duration of illness of 20 years. Almost 40% of patients had previously failed one or more preventive treatments for migraine due to insufficient efficacy and/or tolerability, and the mean monthly migraine days at baseline was 8.3. Patient demographics and clinical characteristics were balanced between the treatment groups.
Disability due to migraine: MIDAS
Baseline migraine-related disability and headache-impact scores.

Note: Patient-reported outcomes were analyzed for all randomized patients who received ≥1 dose of assigned treatment and had ≥1 change from baseline in monthly migraine days during the double-blind treatment phase of the study.
HIT-6™: Headache Impact Test; MIDAS: Migraine Disability Assessment Questionnaire; MSQ: Migraine-Specific Quality of Life Questionnaire; SD: standard deviation.
Erenumab treatment versus placebo resulted in a greater reduction (i.e. improvement) in migraine-related disability. Separation between the erenumab and placebo groups occurred as early as month 1, and reductions in scores remained consistently greater for erenumab throughout the 6 months of treatment (Figure 1(a),(b)). The between-group differences at 70 and 140 mg versus placebo over months 4–6 were, respectively, −2.1 (p< 0.001) and −2.8 (p< 0.001) for overall mMIDAS, −1.0 (p = 0.003) and −1.6 (p < 0.001) for absenteeism, and −1.1 (p < 0.001) and −1.3 (p < 0.001) for presenteeism (Table 2).
Change in monthly MIDAS and 3-month MIDAS: total days lost to disability. Efficacy analysis set. Data are presented as LSM (SE). The shaded area represents values averaged over months 4–6. (a) Change from baseline in monthly MIDAS. *p < 0.001 and †p < 0.05 for the erenumab dose groups versus placebo, not adjusted for multiplicity. (b) Change from baseline in 3-month MIDAS averaged over months 4–6. *p < 0.001 for erenumab 140 mg versus placebo; ‡p = 0.002 for erenumab 70 mg versus placebo, not adjusted for multiplicity. Change from baseline in migraine-related disability, impact, and HRQoL scores averaged over months 4–6. Note: The 3-month MIDAS scores were obtained by averaging the monthly mMIDAS scores over months 4–6 and multiplying by 3 (if at least one monthly mMIDAS was present). Last observation carried forward was used to impute missing every-3-month assessments. Between-group difference is the difference between erenumab 70 mg or 140 mg versus placebo. p-values are nominal and are not corrected for multiplicity. Common ORs, 95% CIs of the ORs, and p-values are obtained from a CMH test, stratified by region and prior/current treatment with migraine prophylactic medication. The MID for mMIDAS has not been established. The MID for 3-month MIDAS is a 5-point decrease (−5). The MID for HIT-6™ is a 1.5-point decrease (−1.5). The MIDs for MSQ-RFR, MSQ-RFP, and MSQ-EF are 3.2-, 4.6-, and 7.5-point increases, respectively. BL: baseline; CI: confidence interval; CMH: Cochran-Mantel-Haenszel; HIT-6™: Headache Impact Test; LSM: least-squares mean; MID: between-group minimally important difference; mMIDAS: modified (monthly) Migraine Disability Assessment Questionnaire; MSQ: Migraine-Specific Quality of Life Questionnaire (EF: emotional function; RFP: role function-preventive; RFR: role function-restrictive); OR: odds ratio; SE: standard error.

After converting the mMIDAS total scores to 3-month MIDAS total scores, the between-group differences at 70 and 140 mg versus placebo over months 4–6 were, respectively, −5.8 (p = 0.002) and −9.0 (p < 0.001), both of which exceeded the between-group minimally important difference for MIDAS (−5.0) (Table 2).
Percentage of patients with severe or very severe migraine-related disability over months 4–6.
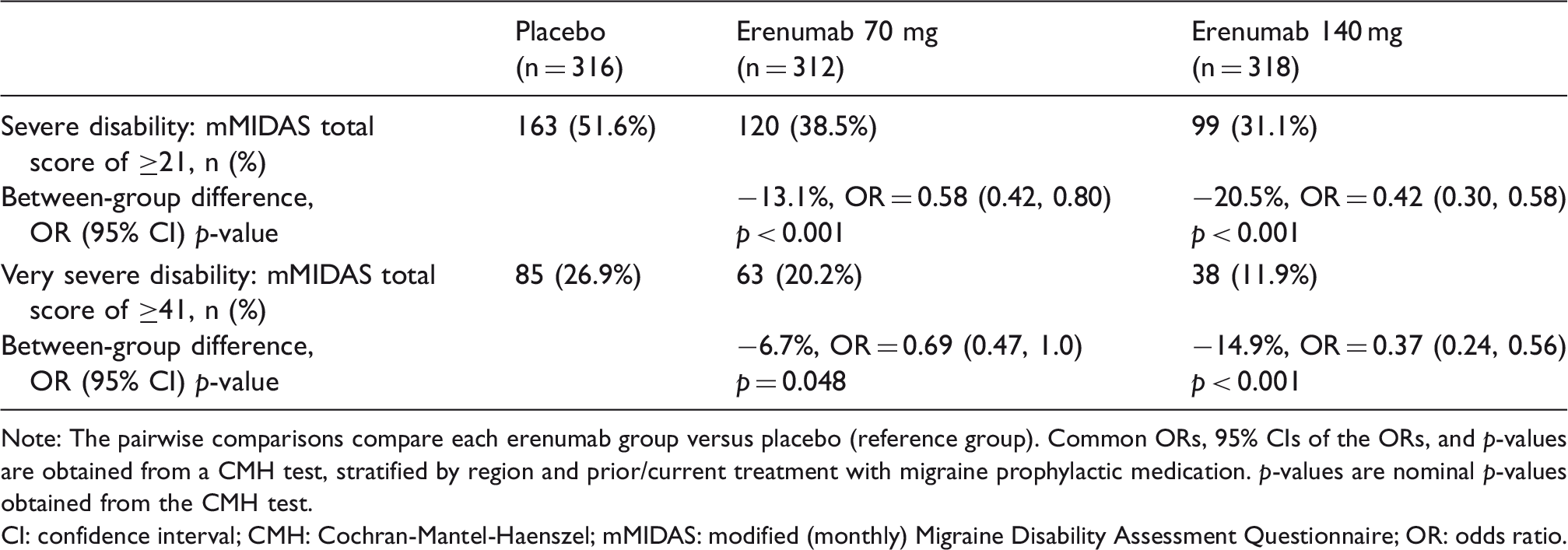
Note: The pairwise comparisons compare each erenumab group versus placebo (reference group). Common ORs, 95% CIs of the ORs, and p-values are obtained from a CMH test, stratified by region and prior/current treatment with migraine prophylactic medication. p-values are nominal p-values obtained from the CMH test.
CI: confidence interval; CMH: Cochran-Mantel-Haenszel; mMIDAS: modified (monthly) Migraine Disability Assessment Questionnaire; OR: odds ratio.
Impact of headache: HIT-6™
Baseline HIT-6™ scores were similar across treatment groups (Table 1), with scores of 59–60 representing substantial to severe migraine impact. Erenumab treatment resulted in greater reductions (i.e. improvement) in HIT-6™ total scores compared with placebo. Like MIDAS, separation between the erenumab and placebo groups occurred early, with greater reductions in the erenumab groups observed throughout the 6 months of treatment (Figure 2). The between-group differences at 70 and 140 mg versus placebo over months 4–6 were, respectively, −2.1 (p < 0.001) and −2.3 (p < 0.001), both of which exceeded the between-group minimally important difference for HIT-6™ (−1.5) (Table 2).
Change from baseline in HIT-6™ total scores. Efficacy analysis set. Data are presented as LSM (SE). The shaded area represents values averaged over months 4–6. *p ≤ 0.001 and †p = 0.031 for corresponding erenumab dose group versus placebo, not adjusted for multiplicity.
The proportion of patients who achieved a clinically meaningful ≥5-point reduction (improvement) from baseline in HIT-6™ scores over months 4–6 after receiving 70 mg, 140 mg, or placebo was 56.4%, 49.7%, and 39.9%, respectively (Table 2).
Effect of headache on HRQoL: MSQ
Baseline scores for each MSQ domain were similar among treatment groups, with patients reporting moderate levels of HRQoL, reflecting migraine-related limitations in daily social and work-related activities (MSQ-RFR) (Table 1).
Both doses of erenumab resulted in greater improvement in each MSQ domain over months 4–6 compared with placebo, signifying overall improvement in patients’ HRQoL (Table 2). Again, improvement in HRQoL occurred early, and mean changes in all MSQ domains were consistently greater with erenumab versus placebo and with 140 mg versus 70 mg erenumab at all monthly visits (Figure 3(a)–(c)). The between-group differences at 70 and 140 mg versus placebo over months 4–6 were, respectively, 5.1 (p < 0.001) and 6.5 (p < 0.001) for MSQ-RFR (both exceeded the minimally important difference of 3.2 for this domain); 4.2 (p < 0.001) and 5.4 (p < 0.001) for MSQ-RFP (140 mg exceeded the minimally important difference of 4.6); and 5.2 (p < 0.001) and 6.7 (p < 0.001) for MSQ-EF (neither exceeded the minimally important difference of 7.5) (Table 2).
Change from baseline in MSQ scores. Efficacy analysis set. Data are presented as LSM (SE). (a) MSQ-RFR. (b) MSQ-RFP. (c) MSQ-EF. The shaded area represents values averaged over months 4–6. *p < 0.001 and †p ≤ 0.005 for corresponding erenumab dose group versus placebo, not adjusted for multiplicity.
A higher proportion of patients treated with erenumab 70 or 140 mg versus placebo achieved a ≥5-point increase (improvement) from baseline in MSQ-RFR and MSQ-RFP and a ≥8-point increase (improvement) from baseline in MSQ-EF, indicating clinically meaningful improvements in HRQoL (31) (Table 2).
Discussion
One of the main goals of preventive migraine therapy is to improve function and reduce disability (8). In this phase 3 trial, treatment with erenumab versus placebo was associated with significant reductions in the effects of migraine on daily activities, as measured using the MPFID (14). In addition, treatment with erenumab was associated with improvements in numerous patient-reported outcomes, including measures of migraine disability, impact, and HRQoL.
At study baseline, patients had moderate disability (as measured by mMIDAS), substantial to severe impact (as measured by HIT-6™), and moderate ratings of HRQoL (as measured by the MSQ); in particular, limitations in their daily social and work-related activities. During the study, all patient-reported outcome measures improved with both doses of erenumab versus placebo, as evidenced by statistically significant reductions in migraine-related disability, headache-related impact, and improvements in HRQoL. These benefits occurred rapidly and were apparent from the first assessment, one month after treatment initiation with both doses of erenumab compared with placebo. Benefits were maintained through the six months of double-blind treatment.
Two complementary approaches were used to assess the clinical meaningfulness of the statistically significant differences in patient-reported outcome scores after treatment with erenumab versus placebo: (a) comparison with established between-group minimally important differences (when available) and (b) assessment of specific improvement thresholds. Placebo-adjusted between-group differences for both doses of erenumab over months 4–6 exceeded the between-group minimally important differences for 3-month MIDAS (−5 points), HIT-6™ (−1.5 points), and MSQ-RFR (+3.2 points), confirming the relevance of these changes in the clinic. In addition, treatment with erenumab resulted in a higher proportion of patients achieving a ≥5-point reduction from baseline in HIT-6™ scores, a ≥5-point increase from baseline in MSQ-RFR and MSQ-RFP, and a ≥8-point increase from baseline in MSQ-EF. Taken together, these data support clinically meaningful improvements in measures of disability, impact, and HRQoL among patients receiving erenumab.
Improvements from baseline were numerically greater among patients who received 140 mg versus 70 mg of erenumab. Although STRIVE was not designed to test differences between the two doses, the numerically greater improvements in patient-reported outcomes observed with 140 mg versus 70 mg erenumab are consistent with the previously reported efficacy outcomes, including reduction in monthly migraine days, reduction in monthly acute migraine-specific medication days, and increase in the proportion of patients achieving a ≥50% reduction in monthly migraine days, compared with placebo (14).
The following study limitations should be considered. mMIDAS, HIT-6™, and MSQ are all prone to recall bias because they are monthly assessments; however, this is in part balanced by the previously published results of the MPFID (14), a daily assessment utilized in the STRIVE study. With regards to the mMIDAS, the monthly format and scoring system are not yet validated and may underestimate the patient’s actual burden. It should also be considered that our data refer to patients selected in the context of a clinical trial and should be confirmed and further enriched by real-world data.
Together with previously reported data on the MPFID (14), which evaluated the average daily impact of migraine on patients’ physical impairment and everyday activities, and data on improvements of patient-reported outcomes in patients with chronic migraine treated with erenumab (32), the results of these analyses demonstrate that preventive treatment with 70 or 140 mg erenumab results in statistically significant and clinically meaningful improvements in measures of migraine-related disability, impact, and HRQoL using instruments commonly employed in migraine-prevention trials. These patient-reported outcomes data from the STRIVE pivotal study are consistent with and complementary to the results on standard efficacy measures (14). Overall, these results demonstrate the benefit of erenumab in reducing the burden of migraine and improving the HRQoL of patients and confirm its role as a promising new therapy for the prevention of episodic migraine.
Supplemental Material
Supplemental material for Migraine-related disability, impact, and health-related quality of life among patients with episodic migraine receiving preventive treatment with erenumab
Supplemental material for Migraine-related disability, impact, and health-related quality of life among patients with episodic migraine receiving preventive treatment with erenumab by Dawn C Buse, Richard B Lipton, Yngve Hallström, Uwe Reuter, Stewart J Tepper, Feng Zhang, Sandhya Sapra, Hernan Picard, Daniel D Mikol and Robert A Lenz in Cephalalgia
Footnotes
Clinical implications
Disability, impact on daily life activities, and overall HRQoL are very important aspects to assess in patients with migraine, together with measures of migraine frequency and severity.
In the STRIVE pivotal study, erenumab 70 and 140 mg resulted in statistically significant and clinically meaningful, rapid improvement in all patient-reported outcomes (as measured by monthly MIDAS, HIT-6™, and the MSQ), with sustained benefits over six months of treatment.
These patient-reported outcomes data complement the results on standard efficacy measures, supporting the benefit of erenumab in reducing the burden of migraine and improving the HRQoL of patients, and confirming its role as a promising new therapy for the prevention of episodic migraine.
Acknowledgements
Kathryn Boorer, PhD, of KB Scientific Communications, LLC, provided medical writing support on behalf of Amgen Inc. Previous presentations: The results of this analysis were presented in part at the American Headache Society, 59th Annual Meeting, Boston, MA; 8–11 June 2017.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DCB has received research grants from Allergan, Amgen, Avanir, and Promius; received honoraria and consulting fees from Alder, Amgen, Eli Lilly, Promius, and Teva; and is on the editorial board of Current Pain and Headache Reports. RBL has received research support from the National Institutes of Health (NIH): 2PO1 AG003949 (Program Director), 5U10 NS077308 (PI), 1RO1 AG042595 (Investigator), RO1 NS082432 (Investigator), K23 NS09610 (Mentor), and K23 AG049466 (Mentor); received support from the Migraine Research Foundation and the National Headache Foundation; is on the editorial board of Neurology and a senior advisor for Headache; has reviewed for National Institute on Aging (NIA) and National Institute of Neurological Disorders and Stroke (NINDS); holds stock options in eNeura Therapeutics; holds stock in Biohaven; serves as a consultant to, advisory board member for, or reports honoraria from American Academy of Neurology, Alder, Allergan, American Headache Society, Amgen, Autonomic Technologies, Avanir, Biohaven, BioVision, Boston Scientific, CoLucid, Dr. Reddy’s, electroCore, Eli Lilly, eNeura Therapeutics, GlaxoSmithKline, Merck, Pernix, Pfizer, Supernus, Teva, Trigemina, Vector, and Vedanta; and received royalties from Wolff’s Headache, 8th edition (2009), Oxford University Press, Wiley, and Informa. YH is a consultant for Amgen and Novartis. UR is a consultant for Allergan, Amgen, Autonomic Technologies, electroCore, Eli Lilly, Novartis, and Teva; and serves on speakers’ bureaus for Allergan, Amgen, Eli Lilly, Novartis, and Teva. SJT has received research grants from Alder, Allergan, Amgen, Autonomic Technologies, Avanir, Dr. Reddy’s, electroCore, eNeura, Scion NeuroStim, Teva, and Zosano; is a consultant for Acorda, Alder, Allergan, Amgen, Autonomic Technologies, Avanir, BioVision, Charleston Labs, DeepBench, Dr. Reddy’s, electroCore, Eli Lilly, eNeura, GLG, Guidepoint Global, Kimberly-Clark, Neurolief, Novartis, Pfizer, Scion NeuroStim, Supernus, Teva, and Zosano; holds stock and stock options in ATI; is on advisory boards for Acorda, Alder, Allergan, Amgen, Autonomic Technologies, Avanir, Dr. Reddy’s, electroCore, eNeura, Kimberly-Clark, Pernix, Pfizer, Scion NeuroStim, Slingshot Insights, Supernus, Teva, and Zosano; is on the board of directors of the American Headache Society; has received royalties from Springer; and holds stock options in Autonomic Technologies. FZ, SS, HP, DDM, and RAL are employees of and hold stock in Amgen.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Amgen Inc. and Novartis (ClinicalTrials.gov, NCT02456740).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
