Abstract
Background
A pharmacological class effect was initially proposed for monoclonal antibodies against the calcitonin gene related peptide pathway. However, preliminary evidence shows that switching patients who were non-responding to one monoclonal antibody to another could provide some benefit. Herein, we assess treatment response to an anti-calcitonin gene related peptide/receptor monoclonal antibody in patients who have failed to respond to anti-calcitonin gene related peptide/ligand monoclonal antibodies calcitonin gene related peptide/ligand monoclonal antibodies and vice versa. In addition, we select non-responders to the first anti- monoclonal antibody by three or five more stringent variables.
Methods
Retrospective cohort study including outpatients treated consecutively with two anti-calcitonin gene related peptide monoclonal antibodies. Ineffectiveness to the first monoclonal antibody was assessed using three (MIDAS score, monthly headache days, and analgesic monthly days) variables or five (monthly headache days, MIDAS score, analgesic monthly days, analgesic monthly number and HIT-6 score) variables in the same cohort of patients. The primary endpoints were the absolute change from baseline in monthly headache days, response rate, and persistence in medication overuse at three months of treatment with the second anti-CGRP mAb.
Results
In patients selected by three variables, a sustained reduction in monthly headache days, analgesic monthly days, MIDAS and HIT-6 scores was observed at month-3 of treatment with the second monoclonal antibody. Ten (45.4%) patients achieved at least a ≥30% response rate. No difference was reported switching anti-CGRP mAb against ligand or receptor. In the patient subgroup selected by five variables, only HIT-6 was reduced from baseline at month-3. However, a trend toward a reduction in monthly headache days, analgesic monthly days, and MIDAS score was observed at month-3.
Conclusions
Switching anti-calcitonin gene related peptide monoclonal antibodies in selected patients might be an option to achieve or improve clinical benefit. More studies are required to establish the effectiveness of switching these treatments.
Introduction
Three monoclonal antibodies against the calcitonin gene related protein (CGRP) pathway (anti-CGRP mAbs) are currently available in Europe. One (erenumab) is directed against the CGRP receptor (R) (anti-CGRP/R mAb) and two (galcanezumab and fremanezumab) against the ligand (L) (anti-CGRP/L mAbs). Eptinezumab, another anti-CGRP/L mAb, has been approved recently. A pharmacological class effect was initially proposed for anti-CGRP mAb, as randomized clinical trials (RCTs) and real-world studies demonstrated similar efficacy and tolerability of all these new medicines in chronic (CM) and episodic migraine (EM), regardless of medication overuse (MO) and various previous treatment failures, including onabotulinumtoxinA (1).
However, a proportion (15–30%) of patients treated with anti-CGRP mAbs have discontinued treatment due to lack of efficacy, defined according to different variables (1). In these patients, switching between anti-CGRP mAbs could be an option, considering the target (receptor vs. ligand), structural and pharmacokinetic differences among mAbs (2,3). An initial observational study (4), and some case series (5–9), have suggested that switching between anti-CGRP mAbs might be a successful option in a portion of migraine patients who previously failed one of them (2,4). No RCTs, designed to assess the effect of the anti-CGRP mAb switch in non-responders, have been performed so far. The recent European Headache Federation (EHF) guidelines stated that evidence on the potential benefits of switch is insufficient, but it may be an option (10). Therefore, data on responses to anti-CGRP mAb switch are urgently needed.
Ineffectiveness of anti-CGRP mAbs in migraine needs to be carefully evaluated (11,12), particularly in patients with severe migraine, characterized by medication overuse (MO), several failures with preventive treatments, and elevated burden of disease. Although the most common parameters assessed to define effectiveness are ≥30% (usually in CM) or ≥50% (in EM/CM) reduction in MMDs/MHDs, the therapeutic benefit of anti-CGRP mAbs is not easily recapitulated by a single parameter (12,13). Anti-CGRP mAbs have been shown to ameliorate analgesic monthly number (AMN), analgesic monthly days (AMDs), pain intensity, quality of life, and disability (14,15), with or without decreasing the overall number of monthly headache days (MHDs). However, the most valuable parameter(s), and relative cut-off, to evaluate treatment response remains undetermined (14).
Herein, we assess treatment response to an anti-CGRP/R mAb in patients who have failed to respond to anti-CGRP/L mAbs and vice versa. In addition, we evaluate results by selecting non-responders by three or five more stringent migraine-related variables.
Materials and methods
Patient features and variables collected
We performed a retrospective analysis of prospectively collected data, including all consecutive out-patients treated subsequentially with two anti-CGRP mAbs (i.e., erenumab, galcanezumab, or fremanezumab), at the Headache Center of the Careggi University Hospital.
All patients signed informed consent and started treatment with an anti-CGRP mAbs (anti-ligand or receptor) after discontinuation for ineffectiveness only (see below) with another anti-CGRP mAb. If the first treatment was an anti-CGRP/L mAb the second was an anti-CGRP/R and vice versa. Therefore, no switches were performed between galcanezumab and fremanezumab. Reimbursement policies in some countries (including Italy) did not allow the switch among mAbs in case of ineffectiveness, insufficient response, or non-tolerance. In particular, ineffectiveness is defined by the Italian Medicines Agency (AIFA) as a <50% reduction in MIDAS (Migraine Disability Assessment) score at month-3 and month-6 of treatment (16). In accordance with these rules, all switches in this study were performed with no costs for the Italian national health service or patients. The study was reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines and was approved as part of the Registro Italiano Cefalee (RICe) study by the local Ethics committee (Studio RICe, 14591_oss).
Study participants were patients older than 18 years with EM or CM according to ICHD-3 (17) criteria, with or without MO, who started a preventive therapy with an anti-CGRP mAb (erenumab 70–140 mg monthly; galcanezumab 240 mg first dose and 120 mg monthly; or fremanezumab 225 mg monthly). Before the first anti-CGRP mAb, all patients reported failure for lack of efficacy (no meaningful improvement in the frequency of headaches after the administration of drugs for ≥3 months at appropriate dose) with at least three preventive treatments, including onabotulinumtoxinA.
During the treatment phases, patients completed a paper headache diary recording MHDs and acute medication use (AMN, and AMDs per month). A headache day was defined as any day on which a patient recorded any type of headache and response rates as a reduction in MHDs ≥30%, ≥50% ≥75%, or 100%.
Furthermore, patients compiled the Headache Impact Test (HIT-6) questionnaire monthly and the MIDAS questionnaire quarterly. Adverse events (AEs) were reported during both treatments. Demographics, migraine characteristics (presence of aura, disease duration, and CM onset), previous failures of drug classes, including beta-blockers, tricyclic antidepressants, antiseizure medications, onabotulinumtoxinA, and current concomitant preventive and acute symptomatic treatments, were collected.
Study design and patient selection
In accordance with a previous report (4), the study consisted of two observational phases of four months. Each phase started with a baseline period of one month, defined as baseline-1, before starting the three-month period of treatment with the one anti-CGRP mAb, and baseline-2, before starting the second three-month period with another anti-CGRP mAb. A non-observational phase of at least six months (ranging from six to 15 months) occurred between the last administration of the first anti-CGRP mAb and the first administration of the second anti-CGRP mAb (Figure 1). For this study, a month was defined as 30 days. To select non-responsive patients to the first anti-CGRP mAb, ineffectiveness was defined a priori combining three different variables sequentially, starting with a <50% reduction in MIDAS score, then a <30% reduction in MHDs, and finally a <50% reduction in AMDs. These cut-offs were chosen because MIDAS < 50% is required by AIFA to continue the prescription program, MHD < 30% is considered below the minimum clinically meaningful response in CM patients (12), and AMD < 50%, as a third parameter, to recapitulate analgesics use.

Study design (a) and flowchart of patients (b).
As we obtained unexpected results showing significant differences in parameters (HIT-6 and total number of analgesics [AMN]) not included in the a priori criteria used for patient selection, and in the absolute value of MIDAS score at month-3 with the first anti-CGRP mAb, a subgroup analysis to select patients with additional, more stringent, variables was performed. These criteria, selected a posteriori, included two additional variables and stricter cut-offs to define ineffectiveness: MHD < 30%, MIDAS < 30%, AMD < 30%, AMN < 30% and HIT-6 < 30%.
Endpoints and analysis
The primary endpoints were the absolute change from baseline in MHDs, response rates (≥30%, ≥50% ≥75%, and 100% reduction in MHDs), and persistence in MO at month-3 in the second cohort compared to baseline-2. The secondary endpoint was the absolute change from baseline of the overall number of AMN and AMDs, as well as MIDAS and HIT-6 scores at the same timepoint. The same analysis was performed in the overall population selected with three variables, and the subgroup cohort selected with five variables. Finally, we compared the group of patients who switched from anti-CGRP/L mAbs to the anti-CGRP/R mAb and vice versa. Only patients with three complete months of data with both treatments were included in the study. Patients who discontinued treatments for adverse events, non-compliance with administration time, or lost at follow-up were excluded (Figure 1).
Statistical Analysis
This is a retrospective analysis, and the sample size was not based on any statistical consideration. However, the sample size is in line with previous studies (4,5,7) and with the stringent criteria adopted. Due to the study design and aims, no direct comparison was made between the first and the second anti-CGRP mAb. Demographic and baseline characteristics were summarized descriptively, namely mean ± standard deviation [SD] or median interquartile range [IQR] for continuous variables and number (percentage) for categorical data. Normality assumption was assessed using the Shapiro-Wilk test. Considering the non-normally distributed data, a Wilcoxon signed-rank test was calculated to study effectiveness variables pre-post changes in quantitative variables. An exact McNemar's test was run for categorical dependent variables. A Mann-Whitney U test was run to determine the difference between the two independent groups with continuous variables. Following the observation that only five and four patients had missing data for migraine and chronicization duration, respectively, no imputation was done for missing data. No other missing data were present. A p-value <0.05 was considered significant for all variables. Bonferroni correction was applied for multiple comparisons. All data were analyzed using SPSS software version 26.0 (IBM Corp. SPSS Statistics, Armonk, NY, USA) and graphs designed using GraphPad Prism version 9.00 (La Jolla, USA).
Results
Cohort characteristics
Overall, 457 patients were treated with at least one anti-CGRP mAb between December 2019 and July 2022. Among them, 31 were subsequently treated with two different anti-CGRP mAbs. Nine were excluded according to the inclusion criteria based on the three variables used for initial patient enrollment and reasons are reported in Figure 1. Of the remaining 22 patients, all were female (100%), 19 (86.4%) with CM and three (13.6%) with EM. The mean age was 49.6 ± 11.8 years at baseline-1. The mean duration of migraine was 33.9 ± 14.5 years. A history of aura was reported in two patients (9.1%) and 18 (81.8%) had MO at baseline. Fifteen (68.2%) patients had daily headache at baseline and no other headaches. All demographic and clinical features (Table 1), as well as concomitant and prior treatments at baseline, are reported in Table 2. Baseline features, prior and concomitant treatments, and details on the switches of the subgroup (n = 11) selected with five variables are reported in online Supplementary Tables S1 and S2.
Patients demographic and clinical features at baselines.

IQR, interquartile range; SD, standard deviation. Percentages are expressed on column total. §Only patients with chronic migraine. #Calculated on 17 and 15 patients, respectively.
Concomitant and prior preventive treatments.

SD, standard deviation; SSRI, selective serotonin reuptake inhibitors; SNRI, serotonin-norepinephrine reuptake inhibitor. Percentages are expressed on column total.
First anti-CGRP monoclonal antibody
Eleven (50.0%) patients received erenumab, whereas eight (36.4%) and three (13.6%) patients received galcanezumab or fremanezumab as first anti-CGRP mAb, respectively (Table 1). No significant differences were reported from baseline-1 to month-3 in MHDs (median [IQR], −1.5 [4.2], p = 0.18) and AMDs (−2.0 [5.2], p = 0.20), with slight change compared to baseline (Table 3). However, an unexpected difference was reported in the absolute values for AMN (−12.5 [24.2], p = 0.042), MIDAS score (−15.5 [55.2], p = 0.015), and HIT-6 score (−2.5 [8.0], p = 0.018) at month-3 (Table 3). Furthermore, significant reductions were detected at month-1 for MHDs, which, however, were lost at month-2 and month-3 (Table 3). A reduction was also observed for AMDs at month-1 and month-2, but not at month-3 (Table 3). Notably, despite the decrease in the number of analgesics at month-3 (p = 0.042), the number of MO patients (according to ICHD-3 criteria) did not change (p = 1.00). Although they did not reach the significance level at month-3, decreases in MHDs or AMDs at month-1 or month-2 could indicate a clinically meaningful response to the anti-CGRP mAb.
Changes in migraine-related variables during first anti-CGRP mAb treatment in the overall cohort.

The changes are calculated using the Wilcoxon signed-rank test and expressed as median reduction [IQR]. All p values and reductions are vs baseline. P values are corrected for multiple comparisons if appropriate and significative values are in bold. IQR, interquartile range; mAbs, monoclonal antibodies.
As a potential initial response was not detected by combining MHDs, MIDAS, and AMDs and related cut-offs used, we re-selected patients at month-3 with five variables and more stringent cut-offs (all <30%), as reported in the methods section. No significant outcomes were shown at any time point, except for AMDs at month-1, an effect lost at month-2 and month-3 (online Supplementary Table S3). No differences in MO were reported. As expected, the absolute reduction effect in all variables was diminished compared to patients selected with three variables. Only two non-severe AEs and no serious AEs were reported. In particular, one patient had somnolence and another patient had scalp paresthesia.
Second anti-CGRP monoclonal antibody
All patients who received galcanezumab (n = 8) and fremanezumab (n = 3) as first treatment switched to erenumab 140 mg, whereas patients treated with erenumab (n = 11) all switched to galcanezumab (240 mg loading dose then 120 mg monthly). Therefore, 11 patients switched from anti-CGRP/L abs to the anti-CGRP/R mAb, and 11 patients vice versa. All patients started the second anti-CGRP mAb after at least six months from the last administration of the first anti-CGRP mAb.
In the cohort selected with three variables (n = 22), MHDs, AMDs, and MIDAS and HIT-6 scores showed a sustained reduction from baseline-2 at month-1, month-2, and month-3 (Figure 2A; Table 4). In particular, at month-3, the primary outcomes, MHDs and AMDs, were reduced by 2.5 (13.0) (p = 0.003) and 6.0 (11.0) (p = 0.003), respectively (Figure 2A; Table 4). One patient achieved a ≥75% response rate at month-3, whereas three (13.6%) and six (27.3%) patients achieved ≥30% and ≥50% response rates, respectively (Figure 3A; online Supplementary Table S5). Overall, 10 (45.4%) patients achieved at least a ≥30% response rate. No difference (p = 0.65) was reported between daily vs. non-daily headache patients in achieving at least a 30% response. AMNs did not change from baseline-2 to month-3. However, it should be noted that the absolute number of acute medications, which at baseline-1 was 52.2 (75), dropped to 32.5 (44) at baseline-2. This difference may suggest some effectiveness of the first anti-CGRP mAb that was maintained over the >6 months of observation without treatment. On the other hand, no difference was reported for AMDs and MO patients. No differences were reported in the comparison between anti-CGRP/L mAbs and anti-CGRP/R mAb switches for all outcomes at month-3 (MHDs p = 0.38, AMDs p = 0.84, AMN p = 0.57, MIDAS p = 0.87, HIT-6 p = 0.23) and other months of treatment.
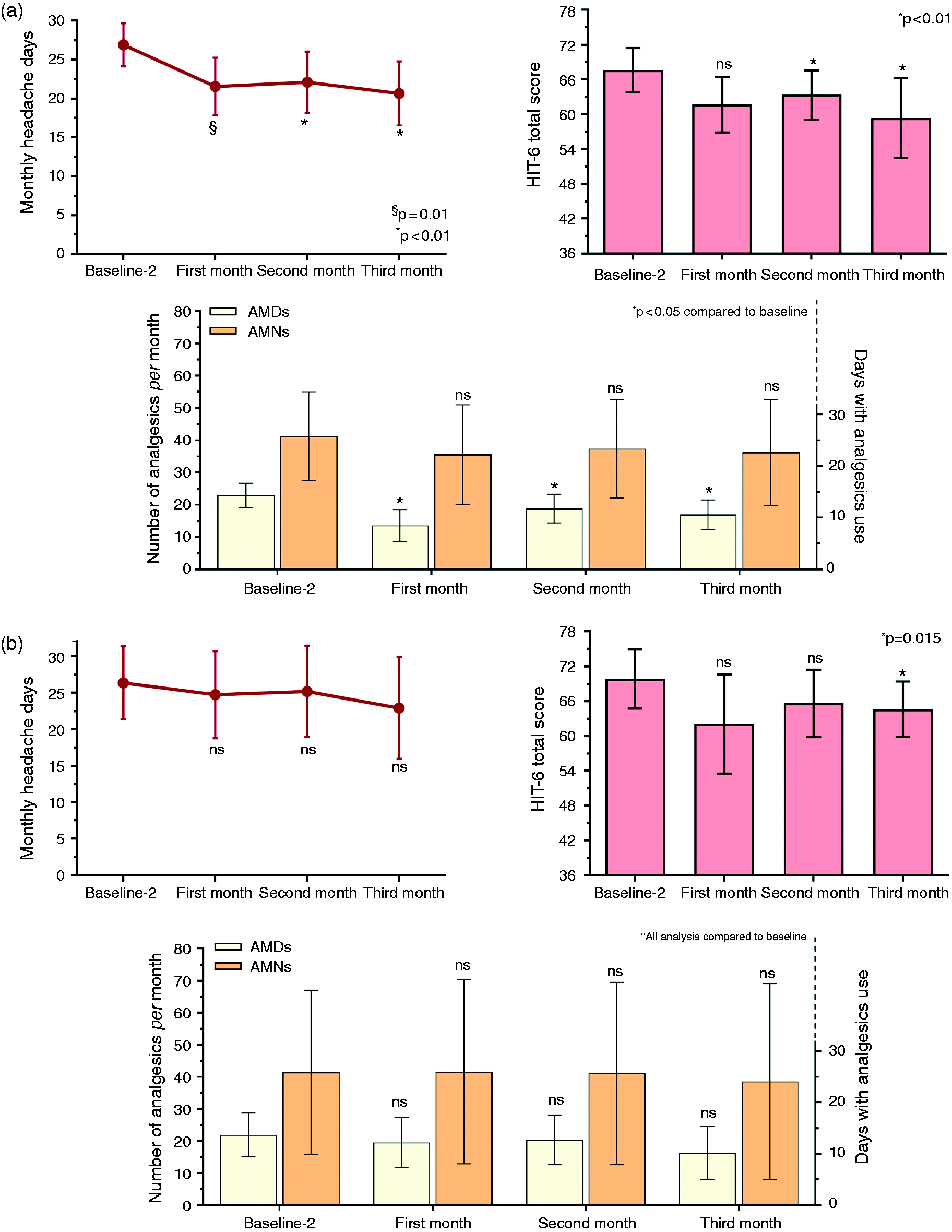
Number of monthly headache days, number of analgesics per month and days with at least one analgesic use per month and HIT-6 score in the second anti-CGRP mAbs cohort selected with three (a) or five (b) variables. AMDs, days with at least one analgesic; AMN, absolute number of analgesics; Ns, not significative. Baseline-2 specifically refers to the baseline of the second mAb treatment.
Changes in migraine-related variables during second anti-CGRP mAb treatment in the overall cohort.

The changes are calculated using the Wilcoxon signed-rank test and expressed as median reduction [IQR]. All p values and reductions are vs baseline. P values are corrected for multiple comparisons if appropriate and significative values are in bold. *Primary and secondary endpoints.
IQR, interquartile range; mAbs, monoclonal antibodies.

Response rates in the second anti-CGRP mAbs cohort selected with three (a) or five (b) variables. Percentages are calculated on the total number of patients per follow-up.
Different results were obtained in the subgroup of patients selected by using 5 variables (n = 11). Only HIT-6 score at month-1, month-2, and month-3 was reduced compared with baseline-2 (4.0 [5.0], p = 0.015) (Figure 2B; online Supplementary Table S4). Although not significantly different, a trend to a reduction was found for MHDs (1.0 [3.0] p = 0.081), AMDs (4.0 [10.0], p = 0.054), and MIDAS score (7.0 [55.0], p = 0.051) at month-3 (Figure 2B; online supplementary table S4). Similarly, at month-3 of treatment with the first anti-CGRP mAb, no difference in the number of patients with MO was reported (p = 1.00), two patients (18.2%) achieved a ≥50% response rate at month-3, and 1 achieved a ≥30% response rate (Figure 3B; online Supplemental Table S5). No difference (p = 0.15) was reported between daily vs. non-daily headache patients in achieving at least a 30% response. No AEs were reported during the treatment with the second mAb.
Discussion
This real-world study suggests a potential clinical benefit of switching anti-CGRP mAbs in a selected cohort of patients with drug-resistant migraine. The study, however, underlines the importance of the choice of outcomes to assess effectiveness. Patients, selected with three parameters (MIDAS, MHDs, and AMDs), showed a significant clinical benefit when treated with the second anti-CGRP mAb, as 45.4% of patients achieved at least a ≥30% response rate (with one patient achieving a ≥75% response rate) at month-3. On the other hand, patients selected with five parameters (MHDs, MIDAS, AMDs, AMN, and HIT-6) showed only a significant reduction in HIT-6 score at month-3, whereas other outcomes showed only a trend to improvement. While a placebo effect is limited in a population with several previous treatment failures (18), and even less likely after failure with another anti-CGRP mAb, the absence of a control group cannot exclude this bias. Although the number of acute medications was significantly decreased, the second mAb failed to reduce the number of MO patients in both the three- and five-variable cohorts. This contradictory result may be due to the exceedingly high number of acute medications reported at baseline-2, and the absolute number of medications used per month that remained above the limit used to define MO. No differences were shown between patients that switched from the anti-CGRP/L mAb to anti-CGRP/R mAbs or vice versa in the cohort selected with three variables. Considering the small sample size of the five-variables cohort, a separate analysis between anti-ligand and receptor mAbs was not performed. No severe or clinically important adverse events have been reported, confirming anti-CGRP mAb tolerability.
A first study (4) reported that the switch from erenumab to galcanezumab or fremanezumab provided clinical benefit (MHD response rate ≥30%) in 32% of the patients three months after the beginning of the second treatment. Interestingly, the subgroup of patients with daily headache showed a response inferior to the second mAbs as compared to non-daily headache patients. It should be underlined that in the previous study (3) non-responder patients were selected only by MHDs, whereas in the present study three or five criteria were used for patient selection. Other retrospective small case series (5–9) included patients that switched for several reasons, including adverse events, costs, or other reasons, and the criteria for ineffectiveness were not defined (9), or just MHDs were considered (5). In some cases, patients had a partial response to the first drug (5,7) or switched immediately to the second mAb with no wash out period (5). A possible carryover effect may challenge the interpretation of the results with the second mAb. All these studies reported some benefit in switching anti-CGRP mAbs, regardless of the target, ligand or receptor. However, biases, mainly related to partial response to the first anti-CGRP mAb, cannot be excluded. Furthermore, only MHD reduction was considered to select non-responder patients. According to discontinuation studies (19–23), a rapid deterioration in migraine starts from the first month of discontinuation but just after three half-lives it is unlikely that the mAbs maintain a protective action.
Our results support the proposal to switch to a second anti-CGRP mAb when the first anti-CGRP mAb achieved insufficient response in our selected patients, who failed three or more preventive treatments, and for whom the first anti-CGRP mAb and further therapeutic options were limited or absent. In addition, our results show that the identification of responders to anti-CGRP mAbs could depend on the type and number of the variables used to select patients. The inclusion of patient reported outcomes, which evaluate the impact on quality of life, disability, improved efficacy of analgesics, and patient preference should be, in principle, superior to one single variable to define responders vs. not responders. In addition, the choice of appropriate time points for the evaluation and decision of treatment continuation seems to have a major impact on patient treatment and costs optimization. It cannot be excluded that additional benefit is achieved after three months of treatment, as real-world studies have shown further improvement at six months (19,24). If, in general, six months seem better than three months to assess effectiveness, but the present patients did not report any effectiveness of the three or five variables at three months, a delayed onset of efficacy may be unlikely. A switch may be performed at any time point during long-term treatment, considering clinical and patient pragmatic treatment goals.
We did not find any differences in response rate between daily vs. non-daily headache patients as reported in a previous study (4) where patients with daily headache did not respond to the switch. However, the small number of non-daily headache patients (30%) at baselines precluded an appropriate stratification of daily vs. non-daily headache in our study. This population deserves further careful investigation in future studies, both with switch of anti-CGRP mAbs and conventional long-term real-world studies with preventive treatments. Ideally, a prospective double blind, crossover study, controlled with placebo (or continuing the same anti-CGRP in a blind manner), RCTs, or well-designed large observational clinical studies, may provide fundamental information for clinical practice and regulatory policies.
The are several strengths to our study. For example, it is the first study reporting the switch from anti-CGRP/L mAbs to the anti-CGRP/R mAb, enrolling patients with ineffectiveness defined using three or five variables, and the second study to investigate only patients selected because of ineffectiveness to the first anti-CGRP mAb. Careful selection of non-responders to the first anti-CGRP mAb should limit misinterpretation on the results of the second anti-CGRP mAb. Our cohort consisted of CM and EM patients who failed at least three previous preventive treatments, and most of them with MO at baseline. We performed different analyses on acute medication use and included disability questionnaires. There are some limitations to our study. For example, the retrospective design, although with prospectively collected data, and the small sample size due to the stringent criteria used to define ineffectiveness and country-specific regulations. In addition, although a longer course of disease is usually a negative predictor for treatment response (25), the non-homogenous time elapsed between the first and second anti-CGRP mAb might be a confounding factor. Another limitation is the use of only MHDs instead of distinguishing MHDs from MMDs. Some patients received concomitant preventive therapy (regardless of clinical indication) during anti-CGRP mAbs treatments. However, as doses remained stable before and after baseline-1 and -2, their impact on the results is unlikely. Finally, due to the study design and the selection bias to enroll patients who failed the first anti-CGRP mAb, results do not allow any conclusion of superiority between the various anti-CGRP mAbs.
Conclusions
As has been reported for triptans (26,27), increasing evidence and clinical practice suggest that a class effect may not be the case for anti-CGRP mAbs. The pathophysiological mechanism due to differential response to anti-CGRP mAbs is still unknown and the therapeutic effect of agents targeting the CGRP pathway may involve distinct mechanisms of action (3). Switching anti-CGRP mAbs in selected patients might be an option to improve or achieve clinical benefit as assessed by several clinical and patient-reported outcomes. Further studies, accurately selecting patients with no-response to the first anti-CGRP mAb, are necessary to establish the effectiveness of switching anti-CGRP mAbs.
Clinical implications
A pharmacological class effect was initially proposed for monoclonal antibodies against the CGRP pathway. Preliminary evidence shows that switching patients who were non-responding to one monoclonal antibody to another could provide some benefit Switching anti-CGRP mAbs in selected patients might be an option to improve or achieve clinical benefit as assessed by several clinical and patient-reported outcomes.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231160519 - Supplemental material for Switching anti-CGRP(R) monoclonal antibodies in multi-assessed non-responder patients and implications for ineffectiveness criteria: A retrospective cohort study
Supplemental material, sj-pdf-1-cep-10.1177_03331024231160519 for Switching anti-CGRP(R) monoclonal antibodies in multi-assessed non-responder patients and implications for ineffectiveness criteria: A retrospective cohort study by Luigi Francesco Iannone, Andrea Burgalassi, Giulia Vigani, Giorgio Tabasso, Francesco De Cesaris, Alberto Chiarugi and Pierangelo Geppetti in Cephalalgia
Footnotes
Acknowledgment
The Società Italiana per lo Studio delle Cefalee (SISC) is acknowledged for the Registro Italiano delle Cefalee (RICe).
Authors’ contributions
PG, LFI, AC and FDC designed the study; LFI, AB, GV, GT, AC, FDC, and PG performed the research; LFI, PG, AC, and FDC analyzed and interpreted the data. LFI and PG wrote the paper. AC, AB, GV, GT, and FDC revised the draft manuscript. All authors critically reviewed the manuscript, agreed to be fully accountable for ensuring the integrity and accuracy of the work, and read and approved the final manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PG: received personal fees from Allergan, Eli Lilly, Novartis, Amgen, TEVA; Grants from Amgen, TEVA, Eli-Lilly, Allergan, Chiesi; Scientific Advisory Board, Endosome Therapeutics; Founding scientist of FloNext srl, Spinoff of the University of Florence. FDC received personal fees from TEVA, Eli Lilly, Novartis. LFI received personal fees from Eli-Lilly. Other authors have no conflicting interests.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
