Abstract
Background
Understanding characteristics and reasons associated with using calcitonin gene-related peptide monoclonal antibodies (CGRP mAb) for migraine prevention may help clinicians individualize treatment plans and achieve better patient outcomes.
Methods
We analyzed 2019–2020 cohort data of OVERCOME (US), a population-based survey among adults with migraine. Eligible participants were categorized based on current CGRP mAb usage (“NEVER” and “EVER” users [“Continued”, “Switched” and “Discontinued”]). Machine learning techniques followed by logistic regression were used to examine, among 60 sociodemographic, clinical, migraine-related- and migraine treatment utilization characteristics, those associated with CGRP mAb use status.
Results
Of 39,113 participants, 25.6% had ever used migraine preventive medication(s) and 5.0% used CGRP mAbs (of which 46.1% Continued, 14.7% Switched and 39.3% Discontinued). Top factors associated with higher odds of CGRP mAb EVER vs. NEVER use were currently using recommended acute medication (odds ratio (OR) = 2.43; 95% confidence interval (CI) = 2.09–2.82) and contraindications for triptan use (OR = 2.32; 95% CI = 2.06–2.60). Continuing use vs. switching or discontinuing was most associated with current use of botulinum toxin for migraine (OR = 2.21; 95% CI = 1.42–3.44 and OR = 4.53; 95% CI = 3.14–6.55, respectively).
Conclusions
At the time of survey, CGRP-targeted mAbs remain underutilized for migraine and multiple characteristics are associated with CGRP mAb use patterns.
This is a visual representation of the abstract.
Introduction
Migraine is a highly prevalent neurological disease that is associated with substantial individual and socioeconomic burden (1). Recent advances in migraine treatment provide potential acute and preventive pharmacologic options for more effective care (2,3). The first monoclonal antibody (mAb) targeting calcitonin gene-related peptide (CGRP) or its receptor was approved in 2018 by the United States (US) Food and Drug Administration as a preventive treatment for migraine, with subsequent approval of other CGRP-targeted preventive medications (3–5). CGRP mAbs, which are mechanism-based drugs, are generally well-tolerated and effective in improving patient outcomes, including reducing migraine attack/headache frequency and severity (4,6–9) and are considered a first-line option for the prevention of migraine by the American Headache Society (AHS) and the European Headache Federation (7,9). Despite the potential benefits of CGRP mAbs, only a small percentage of eligible people with migraine are actually receiving these treatments. Findings from an observational claims study showed that only 5.3% of the people with migraine used CGRP mAbs in 2020 (10). Examining factors associated with CGRP mAb use may help clinicians better individualize preventive treatment plans for their patients. The aim of the current study was to characterize CGRP mAb utilization in more detail and examine the factors and patient-reported reasons associated with CGRP mAb use.
Methods
Study design and population
OVERCOME (US) is a prospective, longitudinal, multicohort, web-based survey conducted annually from 2018 to 2020 in an adult sample of the US population (representative of geographic region, age, race and sex); this was the first study of its kind to be administered after the introduction of CGRP mAbs in clinical practice (11,12). The Sterling Institutional Review Board (IRB ID #6425-001) approved this non-interventional survey study. Participation was voluntary and all respondents provided electronic informed consent to take part in the survey.
Respondents were considered to have active migraine if they reported having had ≥1 headache(s) in the previous 12 months and met the International Classification of Headache Disorders, 3rd edition (ICHD-3) criteria (13) for migraine assessed via the validated American Migraine Study/American Migraine Prevalence and Prevention migraine diagnostic questionnaire (14,15). Those who met the criteria for active migraine then completed the full migraine survey. A more comprehensive description of the OVERCOME (US) study design and population has been previously reported (11,12,16).
The current analysis combined baseline migraine cohorts from 2019 and 2020 of the OVERCOME (US) study and included people with migraine who indicated that they had ever taken any migraine preventive medication (including traditional oral medications in the classes of anti-depressant, anti-seizure, cardiovascular and other medications, botulinum toxins, and CGRP-targeted mAbs). Eligible participants were retrospectively categorized into two groups: those who had used a CGRP mAb preventive medication for migraine at any point in time (labeled as “EVER” users) and those who had never used a CGRP mAb preventive medication (labeled as “NEVER” users). The EVER users were then stratified into three mutually exclusive subgroups: “Continued” and currently taking the initial CGRP mAb (never switched or discontinued); “Switched” to different CGRP mAb(s) after having taken a CGRP mAb(s) preventive medication (defined as ever using ≥2 CGRP mAbs and having taken a CGRP mAb within the last three months); and “Discontinued” and currently not using a CGRP mAb. CGRP mAbs available in the USA in 2019 and 2020 were erenumab, fremanezumab and galcanezumab, which are included in the current analyses; eptinezumab became available in 2020 and was not included in the baseline survey fielded in 2020 (17–20). The CGRP targeted oral preventive medications were approved in May and September of 2021 and were not included in the survey (21,22). The overall flow and disposition of participants included in this analysis is provided in the supplementary material (Figure S1).
Assessments
The sociodemographic, clinical, migraine-related and migraine treatment variables included in the analysis are detailed the supplementary material (Table S1).
The OVERCOME (US) survey also collected data regarding reasons for starting, switching or discontinuing a CGRP mAb. Respondents were able to select from pre-populated lists of options across various categories clinical, access, economic, etc. We did not collect data based on direct response/statements from patients but instead relied on pre-coded responses. Details on response options and how they were categorized are listed in the supplementary material (Table S2).
Statistical analysis
Descriptive statistics were used to summarize the data. Dichotomous or ordinal categorical variables were presented as numbers or percentages. Continuous variables were summarized as the mean ± SD. All continuous variables were standardized to maintain a uniform scale among predictors, enhancing the interpretability and stability of the logistic regression model. Standardized mean differences (SMD) were used to compare groups and interpreted as follows: values <0.20 = no difference, 0.20–0.49 = small difference, 0.50–0.79 = moderate difference and ≥0.80 = large difference (23).We chose to summarize the effect size with SMDs rather presenting p-values as the large sample size may lead to p-values indicating statistical significance when these are not clinically meaningful. This approach also allows a straightforward comparison of the coefficient magnitudes across predictors.
Machine learning techniques (random forest and least absolute shrinkage and selection operator (LASSO)) (24–27) were used to identify which sociodemographic, clinical, migraine-related and migraine treatment utilization variables (among 60 assessed; see supplementary material, Table S2) were most strongly associated with the following outcomes: (i) CGRP mAb EVER vs. NEVER use; (ii) Continued CGRP mAb use vs. Switching; and (iii) Continued CGRP mAb use vs. Discontinuing. The Random Forest and LASSO methods can handle high-dimensional data and have a built-in feature selection (24–27); this was beneficial given the large number of variables assessed in this study. The factors identified by machine learning were then included in logistic regression models to understand the directionality and magnitude of the associations (reported as odds ratio (OR) with 95% confidence intervals (CI)) of the association). SAS Enterprise Guide, version 8.2 (SAS Institute Inc., Cary, NC, USA) was used for all analyses.
Results
Study population
Of the 39,113 people with migraine, 25.6% (n = 10,011) reported having ever used a migraine preventive medication. Among these, 80.5% (n = 8059) had NEVER and 19.5% (n = 1952) had EVER used a CGRP mAb. Of all people with migraine included in this analysis, 5.0% (n = 1952/39,113) had ever used a CGRP mAb. The CGRP mAb EVER users were further categorized into three mutually exclusive subgroups defined at the time of the baseline survey: those who Continued their initial CGRP mAb at the time of the survey (n = 899/1952; 46.1%), those who Switched from their initial to another CGRP mAb (n = 286/1952; 14.7%) and those who Discontinued their CGRP mAb and were currently not taking one (n = 767/1952; 39.3%) as depicted in the supplementary material (Figure S1).
Sociodemographic and clinical characteristics
There were some sociodemographic, clinical and migraine-related characteristic differences between CGRP mAb EVER and CGRP mAb NEVER groups (Table 1). Most notable were that CGRP mAb EVER users were younger than NEVER users (mean age: 37.5 vs. 42.7 years, SMD = 0.41) and less frequently female (57.5% vs. 76.6%, SMD = 0.41). Other small differences (SMD = 0.2–0.49) were observed for race, ethnicity, education, annual household income and employment (Table 1).
Sociodemographic, clinical and migraine-related characteristics of people with migraine, stratified by CGRP use status.

Abbreviations; ASC-12 = 12-item Allodynia Symptom Checklist-12, CGRP = calcitonin gene-related peptide, mAb = monoclonal antibody, MSSS = Migraine Symptom Severity Scale, SD = standard deviation, SMD = standardized mean difference.
Note: The SMD values of <0.20 indicate no difference, 0.20–0.49 indicate small difference, 0.50–0.79 indicate moderate difference and ≥0.80 indicate a large difference. Absolute values of SMD are presented.
Category includes Asian American, Asian Indian, Chinese, Filipino, Japanese, Korean and Vietnamese.
Other includes American Indian only, Native Hawaiian only and two or more races.
Regions: Northeast (Maine, New Hampshire, Vermont, Rhode Island, Connecticut, New York, New Jersey and Pennsylvania), Midwest (Wisconsin, Illinois, Michigan, Indiana, Ohio, North Dakota, South Dakota, Nebraska, Kansas, Minnesota, Iowa and Missouri), South (Kentucky, Tennessee, Mississippi, Alabama, Florida, Georgia, South Carolina, North Carolina, Virginia, West Virginia, District of Columbia, Maryland, Delaware, Texas, Oklahoma, Arkansas and Louisiana), West (Montana, Idaho, Wyoming, Nevada, Utah, Colorado, Arizona, New Mexico, Washington, Oregon, California, Alaska and Hawaii).
Those who selected the options “I do not have health insurance” or “not sure”.
Joint or pain comorbidities included chronic back pain, fibromyalgia, osteoarthritis and rheumatoid arthritis.
Cardiovascular comorbidity included self-reported medical diagnosis of pre-diabetes, diabetes, high cholesterol, hypertension.
Comorbidities contraindicated for triptan use included aneurysm, angina, cerebral hemorrhage, claudication, myocardial infarction, stroke, transient ischemic attack and blood clots in legs/lungs.
Psychiatric comorbidities included self-reported anxiety, depression and panic disorder.
The MSSS composite index of seven International Classification of Headache Disorders-3 criteria (unilateral pain, pulsatile pain, moderate or severe pain intensity, routine activities worsen pain, nausea, photophobia and phonophobia) scores ranging from 0 to 21.
Other headache types included cervicogenic headache, chronic daily headache, cluster headache, new daily persistent headache, medication overuse headache or rebound headache, menstrual headache or menstrual migraine, post-traumatic headache or post-concussion headache, sinus headache stress headache and tension type headache or tension headache.
Clinical characteristics also differentiated CGRP mAb EVER users from CGRP mAb NEVER users. In particular, CGRP mAb EVER users were more likely to report two or more cardiovascular comorbidities (61.2% vs. 40.7%, SMD = 0.42), contraindications to triptan use (56.5% vs. 27.8%, SMD = 0.61) and current use of cannabis (59.8% vs. 36.1%, SMD = 0.49). Smaller differences were observed for other comorbidities (Table 1).
Among migraine-related characteristics, CGRP mAb EVER users vs. NEVER users had lived with migraine for fewer years (13.9 vs. 20.0 years, SMD = 0.43). Small or negligible differences were observed for all other migraine-related characteristics. (Table 1).
The CGRP mAb EVER users were more likely than the CGRP mAb NEVER users to be currently using any migraine preventive medication (90.2% vs. 62.1%; SMD = 0.70) and any AHS recommended acute prescription medication (85.6% vs. 60.4%; SMD = 0.59). A lower proportion of CGRP mAb EVER users vs. NEVER users reported currently using acute over the counter only (4.6% vs. 23.5%; SMD = 0.57) or one AHS consensus statement recommended preventive medication for migraine (15.9% vs. 70.4%; SMD = 1.32). Full details are presented in Table 2.
Acute and preventive migraine medication use by CGRP use status.

Abbreviations; AHS = American Headache Society, CGRP = calcitonin gene-related peptide, mAb = monoclonal antibody, mTOQ = Migraine Treatment Optimization Questionnaire, NA = not applicable; SMD = standardized mean difference.
Note: The SMD values of <0.20 indicate no difference; 0.20–0.49 indicate small difference; 0.50–0.79 indicate moderate difference and ≥0.80 indicate a large difference.
mTOQ assesses four items and scores ranging from 0 to 8 with scores <6 indicating poor/very poor acute treatment satisfaction.
Includes cardiovascular, anti-seizure, anti-depressant and botulinum toxin.
Devices such as Cefaly, Spring TMS, gammaCore.
Among the three subgroups of CGRP mAb EVER users, no large effect was observed and only a few contrasts had SMDs that indicated small effect. A lower proportion of CGRP mAb Continuers had contraindications to triptan use (52.7% vs. 64.0%, SMD = 0.23) compared to CGRP mAb Switchers. A higher percentage of CGRP mAb Continuers had no allodynia (19.6% vs. 12.2%, SMD = 0.20) compared to CGRP mAb Switchers. CGRP mAb Continuers had lower average headache pain intensity (7.5 vs. 7.1, SMD = 0.22) and Migraine Symptom Severity Scale (MSSS) sum score (17.9 vs. 17.0, SMD = 0.29) vs. CGRP mAb Discontinuers.
Migraine treatment-related characteristics were comparable across the subgroups of EVER users. However, a higher proportion of CGRP mAb Continuers vs. Discontinuers reported currently using ≥2 AHS consensus statement listed preventive medication (100% vs. 55%, SMD = 1.28) for migraine. A larger proportion of CGRP mAb EVER users who discontinued the treatment vs. those who continued were currently using one AHS consensus statement listed preventive medication (45% vs. 0%, SMD = 1.28).
Logistic regression models
Factors associated with CGRP mAb EVER use vs. CGRP mAb NEVER use
Of the 60 sociodemographic, clinical, migraine-related and migraine treatment utilization variables, four variables were shown to be significantly associated with CGRP mAb EVER use. The top two variables most significantly associated with higher odds of CGRP EVER mAb use vs. NEVER use were currently using recommended acute medication for migraine (OR = 2.43; 95% CI = 2.09–2.82) and having contraindications for triptan use (OR = 2.32; 95% CI = 2.06–2.60) (Figure 1).
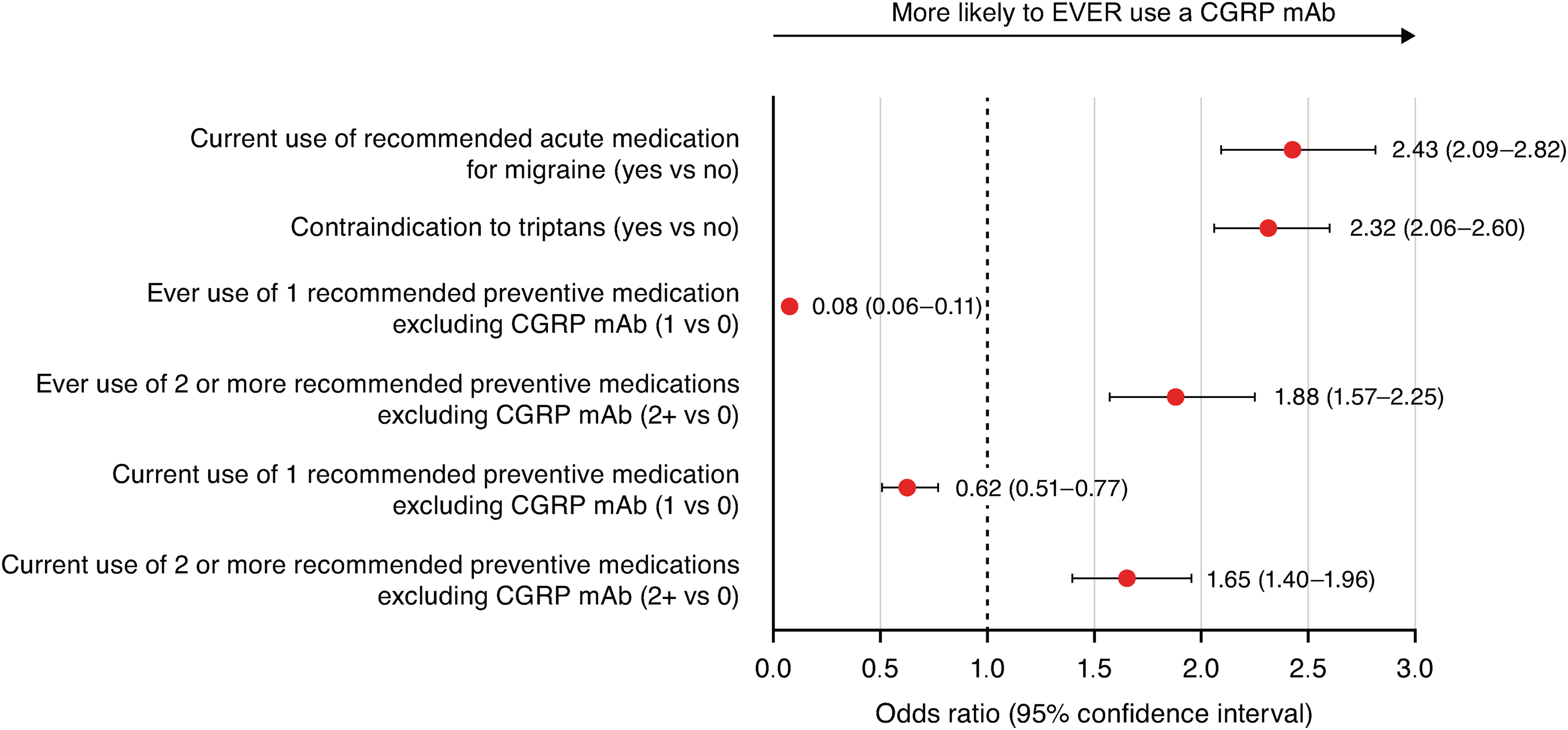
Factors associated with CGRP mAb EVER Use vs. CGRP mAb NEVER Use. Abbreviations; CGRP = calcitonin gene-related peptide, LASSO = least absolute shrinkage and selection operator, mAb = monoclonal antibody. Footnote: Of the 60 sociodemographic, clinical, migraine-related and migraine treatment utilization variables assessed, random forest identified three variables and LASSO identified six variables. After applying a pre-specified thresholds for the absolute value of the coefficient estimates, four variables were included in the logistic regression.
Factors associated with CGRP mAb EVER users who continued vs. switched
As shown in Figure 2, the two most significant factors associated with higher odds of CGRP mAb continuation vs. switching were current use of botulinum toxin for migraine prevention, which indicates at diagnosis of CM at initiation, (OR = 2.21; 95% CI = 1.42–3.44) and having a lower number of monthly headache days (4–7 vs. ≥15; OR = 1.86; 95% CI = 1.13–3.06). By contrast, the two most significant factors associated with lower odds of CGRP mAb continuation vs. switching were MSSS scores 11–14 vs. ≥19 (OR = 0.51; 95% CI = 0.34–0.78) and current use of consensus statement recommended preventive and acute medication(s) (OR = 0.51; 95% CI = 0.34–0.77).

Factors associated with CGRP mAb EVER users who continued vs. switched their CGRP mAb medication. Abbreviations; CGRP = calcitonin gene-related peptide, LASSO = least absolute shrinkage and selection operator, mAb = monoclonal antibody, MSSS = Migraine Symptom Severity Scale. Footnote: Of the 60 sociodemographic, clinical, migraine-related and migraine treatment utilization variables assessed, random forest identified five variables and LASSO identified 15 variables. After applying a pre-specified thresholds for the absolute value of the coefficient estimates, nine variables were included in the logistic regression. Monthly headache days (“8–14”) and MSSS group (“15–18” and “10 or less”), which were not statistically significant, are not shown.
Factors associated with CGRP mAb EVER users who continued vs. discontinued
The two most significant factors associated with higher odds of CGRP mAb continuation over discontinuation were current use of a botulinum toxin for migraine (OR = 4.53; 95% CI = 3.14–6.55) and ever using blood pressure medication for migraine (OR = 1.54; 95% CI = 1.21–1.96) (Figure 3). Conversely, the two most significant factors associated with lower odds of CGRP mAb continuation over discontinuation were ever using a botulinum toxin for migraine (OR = 0.34; 95% CI = 0.26–0.46) and ever using an implanted stimulator (OR = 0.59; 95% CI = 0.42–0.82) (Figure 3).

Factors associated with CGRP mAb EVER users who continued vs. discontinued. Abbreviations; CGRP = calcitonin gene-related peptide, LASSO = least absolute shrinkage and selection operator, mAb = monoclonal antibody, MSSS = Migraine Symptom Severity Scale, mTOQ = Migraine Treatment Optimization Questionnaire. Footnote: Of the 60 sociodemographic, clinical, migraine-related and migraine treatment utilization variables, random forest identified two variables and LASSO identified 11 variables. After applying a pre-specified thresholds for the absolute value of the coefficient estimates, 11 variables were included in the logistic regression. Age group (“40–49”) and MSSS group (“10 or less”), which were not statistically significant, are not shown.
Reasons for starting, continuing, switching or discontinuing a CGRP mAb
Perceived effectiveness of a CGRP mAb was the prevailing reason for starting the first CGRP mAb for the EVER group overall (61.0%), followed by dosing/delivery (47.5%) and recommendation/request (43.3%). The percentages of people with migraine in the Continued CGRP mAb use group endorsing each of the reason categories mirrored the priorities of those of the EVER group as a whole. Among those who switched CGRP mAb treatment, access/economics (59.1%) was the most frequent reason, followed by efficacy (45.1%) and recommendation/request (40.2%). The reasons for discontinuing CGRP mAb treatment were spread across categories with the greatest proportion selecting access/economics (35.7%), but to a lesser degree than that seen in the Continued or Switched groups. Approximately 32% and 28% of people with migraine in the Discontinued group endorsed tolerability and efficacy, respectively as reasons for discontinuing (Figure 4).

Reasons for starting, switching or discontinuing a CGRP mAb. Abbreviations; CGRP = calcitonin gene-related peptide, mAb = monoclonal antibody. Footnote: The order of reasons for mAb use behavior in the EVER group (a) are used for the subgroups Continued (b), Switched (c) and Discontinued (d). Participants could select one or more reason for starting, switching, or discontinuing a CGRP mAb. For the EVER group as a whole (A), the prevailing reason for CGRP mAb use was efficacy. The reasons for the Continued group mirror those for the EVER group as a whole. Access or economic reasons were the prevailing reason for people with migraine in the Switched group (59.1%) and to a lesser degree in the Discontinued group (35.7%). In the Discontinued group, 13.2% of people with migraine indicated disease resolution as the reason for discontinuation. Refer to the supplementary material (Table S1) for details on each category/reason for starting, switching or discontinuing a CGRP mAb.
Discussion
This observational population-based study demonstrated that at baseline 25.6% of people with migraine had ever used a preventive medication and only 5.0% had ever used CGRP mAbs. Of EVER CGRP mAbs users, 46.1% continued their original mAb, 14.7% switched to a different mAb and 39.3% discontinued mAbs altogether, at the time of survey (2019 and 2020).
We found that among the factors distinguishing these groups were various sociodemographic, clinical and migraine-related characteristics. CGRP mAb EVER users had a higher likelihood of cardiovascular comorbidities, contraindications to triptan use and substance use. With respect to migraine-related factors, CGRP mAb EVER users reported fewer years living with migraine, indicating that duration of illness is not associated with access to mAbs. The CGRP mAb EVER users were more likely to be currently using any preventive migraine treatments and acute treatments recommended by the AHS guidelines. Our findings suggest that CGRP mAb users may be more proactive or have better access to healthcare resources and information about novel treatments. The extent of insurance coverage of CGRP mAbs vary depending on the specific insurance plan and some patients may opt for private funding to access these medications. This information supports our assumption that individuals with better incomes might have more comprehensive insurance coverage or the financial means to afford these treatments out of pocket. Furthermore, a higher incidence of cardiovascular comorbidities and contraindications to triptan use in our study might have led them to seek newer preventive treatments with a better tolerability and safety profile. Moreover, their use of both acute treatments recommended by the AHS guidelines and preventive treatments may indicate more severe and frequent migraine episodes.
Using a logistic regression model, we found several factors associated with EVER vs. NEVER using a CGRP mAb. Pronounced use of recommended acute medication for migraine among ever users suggests access to higher quality care which may be associated with better access to newer preventive treatments. The presence of contraindications for triptan use may indicate the limitations of acute treatment options and a need for effective preventive modalities. Moreover, the use of two or more recommended non-CGRP mAb preventive medication, either currently or in the past, may reflect step-edit requirements before gaining access to CGRP mAbs. By contrast, individuals who are currently or have previously used only one recommended non-CGRP preventive medication tend to show lower odds of ever use of CGRP mAbs. This could reflect adequate control with a non-CGRP mAb preventive medication or lack of access due to step-edits.
Our study identified several factors associated with continuing vs. switching CGRP mAb treatments. The top factors associated with continuation of CGRP mAb (vs. switching) were current use of a botulinum toxin. Use of a botulinum toxin is a marker of access to specialists since most physicians are not trained to inject it. Because botulinum toxins are currently indicated only for CM, this may be a proxy for CM. We also note that having 4–7 monthly headache days (vs. ≥15 monthly headache days) is associated with continuation of the CGRP mAb (vs. switching). This could reflect partial treatment success, if monthly headache day frequency fell from ≥15 monthly headache days to this lower range, coupled with ongoing treatment need. In addition, current use of a botulinum toxin for migraine and ever use of blood pressure medications for migraine prevention were the key drivers associated with continued CGRP mAb use vs. discontinuation. Individuals currently using a botulinum toxin in combination with CGRP mAbs, were more likely to maintain their regimen, possibly due to the observed enhanced benefits of this combination therapy (28–30). Interestingly, the current use of botulinum toxin for migraine was identified as a factor associated with increasing the odds of continuing a CGRP mAb while ever use of a botulinum toxin was associated with decreasing the odds of continuing a CGRP mAb vs. switching or vs. discontinuing the treatment. The opposite direction of association from “current use of botulinum toxin” could be explained by the inclusion of a mixed group of people with migraine in the ever use of botulinum toxin group (both botulinum toxin responders and non-responders). Current users of a botulinum toxin might have chronic migraine at treatment initiation which remits with treatment, whereas ever users of botulinum toxin might include those with chronic migraine who may have failed to respond to the botulinum toxin and were discontinued; this refractory group may be more likely to not do well on a CGRP mAb.
The use of an implanted stimulator was a factor identified as decreasing the odds of continuing vs. switching and discontinuing CGRP mAbs. In addition, current use of recommended preventive and acute medications was a factor identified as decreasing the odds of continuing vs. switching. This may reflect people with migraine who are severely disabled by migraine and have not found benefit from multiple treatment modalities before utilizing an implant.
The continuation of CGRP mAbs also appears to be associated with factors suggestive of a successful treatment response like lower frequency of monthly headache days and mild ictal cutaneous allodynia. These findings suggest that individuals experiencing fewer headache days or mild allodynia perceive their current treatment as effective, reducing the necessity for alternative options.
Our analysis also indicated that people with migraine aged 18–29 years and 30–39 years are less likely to continue vs. discontinue a CGRP mAb compared to those 50 years or older. This may be reflective of issues of pregnancy, adherence, economic or access issues and history of migraine. Perhaps these individuals either have had migraine for a shorter duration, and thus are potentially less disabled by the disease, or, alternatively, have disease that is less tractable to CGRP mAbs. People with migraine in the MSSS group 11–14 vs. ≥19 and 15 to 18 vs. ≥19 were more likely to discontinue a CGRP mAb because those who are in these severity categories are less likely to continue mAb and may prefer managing their symptoms with alternative treatments. Current use of tobacco was associated with increased odds of CGRP mAb continuation compared to CGRP mAb switching. This was an interesting finding because smoking is associated with an increased risk of migraine and can cause and worsen migraine, especially in a dose-dependent manner (31).In addition, smoking is associated with developing comorbidities that present a contraindication to triptans, which, in turn, are associated with CGRP mAb use (as observed in this analysis) (31,32).
Our data showed that the most common reasons for starting a CGRP mAb were perceived or expected effectiveness, dosing or delivery, and recommendation by one's doctor/healthcare provider or family/friend, or request from the person with migraine. In the OVERCOME survey in the European Union (EU) conducted in 2020, Pascual et al. (32) evaluated 20,756 respondents who had migraine. Their findings showed a slightly higher rate of CGRP mAb (erenumab, fremanezumab, galcanezumab) EVER use (10.4%). Among the CGRP mAb EVER users, a similar rate of switching (15.4%) and a higher rate of discontinuation (54.9%) were observed when compared to results from the OVERCOME (US) (11). The reasons for switching were mainly recommendations from a doctor (27.0%) or a friend/family member (26.7%) and preference for the route of administration (26.7%). The most frequent reasons for discontinuing the treatment included improvement in headaches, recommendations from others, dosage or tolerability. Lack of efficacy was reported as the reason for discontinuing medication in only 11.5% of cases (33). Although these comparable studies did not reveal any dominant reasons for the different rates of starting mAbs during the initial years of introduction of these therapies in these jurisdictions, it is important to consider that there are significant differences in access to CGRP mAb treatments between the EU and US.
The general limitations of the OVERCOME (US) study have been previously discussed in detail (11). The utility of cross-sectional survey responses is inherently limited when attempting to capture the dynamic patterns of disease progression and the changing landscape of therapeutic options because diseases often follow dynamic trajectories and treatment approaches frequently advance or shift based on emerging evidence and a single point in time survey may fail to reflect these complexities. Specifically, our analyses regarding factors associated with CGRP mAb use only considered the cross-sectional characteristics of people with migraine and may not have captured possible time-varying factors such as changes in comorbid conditions, migraine severity and compliance to therapies over time. We present cross sectional data on EVER vs. NEVER used a mAb and among ever users those who continued, switched or discontinued. The proportion of people who continue, switch or discontinue is likely to vary with the time interval since the initial mAb was first started. In this cross-sectional study, we cannot fully control for time since initiation of the first mAb. Longitudinal studies are warranted to consider the time-varying factors and provide a comprehensive understanding of treatment trends. Also, the pre-populated response options were created by the research team and may not reflect all possible responses. Moreover, these data were collected early in the introduction of CGRP mAbs in the US when they were not fully endorsed as first-line treatment for migraine prevention. The utilization rates, knowledge and access have likely changed since then. The availability of samples and coupons for CGRP mAbs for migraine prevention in 2019 and 2020 may have influenced use. In addition, it was too early to collect data on eptinezumab, and open ended data for reasons for starting, switching or discontinuing a CGRP targeted mAb were not collected and therefore some reasons were likely missed. Finally, we must consider confounding by indication; people with migraine who are prescribed CGRP mAbs most likely have more frequent, severe, refractory and impactful migraine and this may show in comparisons between NEVER and EVER users. Those on CGRP mAbs may experience reduction in frequency and severity as a consequence of treatment; those changes may be reflected in the baseline assessment. For the subgroup analysis of the Continuers, Switchers and Discontinuers, a noteworthy limitation is a low number of participants in each of these subgroups.
Conclusions
Data from the presewnt study highlight the under-utilization of consensus-recommended first-line migraine-specific preventive therapies during the early years following the launch of CGRP mAbs, despite expert recommendations advocating their use (3,5,7), especially in those who have failed previous preventive treatments or are on concurrent oral preventive treatments. Differences in the pattern of CGRP mAb use observed in this study reflects the impact of different subgroups’ characteristics (age, contraindications to triptans, monthly headache days, allodynia, current/ever use of migraine treatments, implant use, etc.) on decisions to continue, switch, or discontinue treatment. It may help better characterize migraine treatment usage since the introduction of CGRP mAbs and provide valuable insight into the potential factors and patient-reported reasons associated with CGRP mAb use, and thus, may inform clinician's decision-making when considering preventive treatment options for people with migraine. However, further longitudinal studies are needed to better understand the underlying characteristics of people with migraine, treatment use patterns, behaviors and beliefs associated with selecting a specific migraine treatment, including CGRP mAbs. By gaining a deeper understanding of these factors, healthcare providers may develop more personalized and effective treatment plans to improve patient outcomes.
Clinical implications
While recommended as first-line treatments, CGRP-targeted mAbs remain underutilized for migraine prevention, particularly in those who have failed previous preventive treatments or are on concurrent oral preventive treatments. Differences in the pattern of CGRP mAb use observed in this study reflects the impact of different subgroups’ characteristics (age, contraindications to triptans, monthly headache days, allodynia, current/ever use of migraine treatments, implant use, etc.) on decisions to continue, switch or discontinue treatment. CGRP mAbs ever use versus never use was most associated with the current use of recommended acute medication and contraindications for triptan use. Continuing CGRP mAb use was most associated with current use of botulinum toxin for migraine. Identifying factors associated with the use of CGRP mAbs can assist clinicians in creating better preventive treatment plans for their patients.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251341243 - Supplemental material for Patterns of calcitonin gene-related peptide monoclonal antibody use in people with migraine: Results of the OVERCOME (US) study
Supplemental material, sj-docx-1-cep-10.1177_03331024251341243 for Patterns of calcitonin gene-related peptide monoclonal antibody use in people with migraine: Results of the OVERCOME (US) study by Sait Ashina, Gilwan Kim, E. Jolanda Muenzel, Dawn C. Buse, Anthony J. Zagar, Armen Zakharyan, Robert E. Shapiro, Robert A. Nicholson, Eric M. Pearlman and Richard B. Lipton in Cephalalgia
Footnotes
Acknowledgments
Data were presented in part as an oral presentation at the American Headache Society 65th Annual Meeting, 15–18 June 2023; Austin, TX, USA and as a poster presentation at the International Headache Congress, 14–17 September 2023; Seoul, South Korea. Medical writing support was provided by Millie Hollandbeck and Preetinder Kaur of Certara and was funded by Eli Lilly and Company, Indianapolis, IN.
Data availability
Lilly will provide access to anonymized individual participant data collected during the study. The data will be available to request on vivli.org after the study team has completed analyses and publications. Access will be provided after a proposal has been approved by an independent review committee identified for this purpose and after receipt of a signed data sharing agreement. After a proposal is approved, data and documents, including the study protocol, will need to be provided in a secure data sharing environment. For details on submitting a request, see the instructions provided at ![]() .
.
Declaration of conflict of interest
S. Ashina: Consulting/Advisory Board, Teaching, Honoraria: Allergan/AbbVie, Eli Lilly and Company, Teva. Consulting and Advisory Board: Amgen, Biohaven Pharmaceuticals, Impel NeuroPharma, Linpharma, Lundbeck, Novartis, Satsuma, Supernus, Percept, Theranica and Pfizer. D. Buse Consulting Honoraria, Research Grants: AbbVie/Allergan, Amgen, Biohaven Pharmaceuticals, Collegium Pharmaceutical, Eli Lilly and Company, Lundbeck, Teva, Theranica. G. Kim, E. Muenzel, A. Zagar, R. Nicholson and E. Pearlman are employees and minor stockholders of Eli Lilly and Company. A. Zakharyan is employed by TechData Service Company, a company contracted by Eli Lilly and Company (sponsor) to perform the statistical analysis. R. Lipton has received research support from the National Institutes of Health, the FDA and the National Headache Foundation. He serves as consultant, advisory board member, or has received honoraria or research support from AbbVie/Allergan, Amgen, Biohaven Pharmaceuticals, Dr Reddy's Laboratories (Promius), electroCore, Eli Lilly and Company, GlaxoSmithKline, Lundbeck, Merck & Co., Novartis, Teva, Vector and Vedanta Research. He receives royalties from Wolff's Headache, 8th edition (Oxford University Press, 2009) and Informa. He holds stock/options in Axon, Biohaven Pharmaceuticals, CoolTech and Manistee Therapeutics. R. Shapiro serves as consultant, advisory board member, or has received honoraria or research support from Eli Lilly and Company, Lundbeck, Theranica, Amneal, Pfizer and AbbVie.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Funding for the study and medical writing support were sponsored by Eli Lilly and Company.
Ethical statement
The Sterling Institutional Review Board (IRB ID #6425-001) approved this non-interventional survey study. Participation was voluntary and all respondents provided electronic informed consent to take part in the survey.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
