Abstract
Background
This meta-analysis evaluated the real-world effectiveness of onabotulinumtoxinA (BOTOX®), the first preventive treatment FDA-approved specifically for chronic migraine in 2010.
Methods
We systematically reviewed onabotulinumtoxinA observational data in chronic migraine published between 1 January 2010 and 31 March 2021. Random-effects models evaluated available data for primary and secondary endpoints defined in onabotulinumtoxinA pivotal trials at approximately 24 weeks and 52 weeks.
Results
Of the 44 full-text eligible studies (29 prospective; 13 retrospective; 2 other), seven evaluated change from baseline (mean[confidence interval]) at ∼24 weeks and ∼52 weeks, respectively, for onabotulinumtoxinA in: number of headache days/month: (–10.64 [–12.31, –8.97]; –10.32 [−14.92, –5.73]); number of days of acute headache pain medication intake per month (–7.40 [–13.04, –1.77]; overlapping CIs at 52 weeks); total Headache Impact Test-6 score (–11.70 [–13.86, –9.54]); –11.80 [14.70, –8.90]); and Migraine-Specific Quality-of-Life v2.1 score (MSQ; 23.60 [CI: 21.56, 25.64]; 30.90 [CI: 28.29, 33.51]). At ∼24 weeks onabotulinumtoxinA showed total Migraine Disability Assessment score of 44.74 [28.50, 60.99] and ≥50% reduction in migraine days response rate of 46.57% [29.50%, 63.65%]. A sensitivity analysis at study-end suggested durability of onabotulinumtoxinA effectiveness on MSQ.
Conclusion
The meta-analysis reflecting real-world practice broadly corroborated with evidence from pivotal and long-term open-label studies of onabotulinumtoxinA in chronic migraine preventive treatment.
Keywords
Introduction
Migraine is characterised by both painful symptoms, including attacks of intense throbbing headache that are often accompanied by nausea, vomiting and sensitivity to light and sound, and other sensory symptoms, such as tiredness, numbness, and allodynia (1,2). The symptoms of chronic migraine cause a level of functional and emotional impact in people with migraine (3) that ranks the disease second among worldwide causes of disability (4).
When headache occurs on 15 or more days per month beyond a period of three months and has the features of migraine occurring at least eight days per month, the presentation is termed chronic migraine (5), a complex, progressive, neurological disease (6) that prevails in 1–2% of the global population (7). Chronic migraine is associated with considerable disease burden; people with chronic migraine experience important impairments to daily functioning (8), health-related quality of life (HRQoL), and productivity (9,10). The disease also poses substantial direct costs to healthcare systems (11,12) and indirect costs to society (8).
Preventive therapeutic strategies aim to reduce frequency, duration, and intensity of migraine attacks (13–15). Among oral medications used and studied in the prevention of migraine, only topiramate 100 mg/day has clearly demonstrated efficacy in the prophylaxis of chronic migraine (16,17), but no treatments are licensed for preventive treatment of chronic migraine specifically. Moreover, poor efficacy and tolerability of oral preventive medications has led to low treatment persistence (14% at 12 months), and overuse of acute prescription medication for migraine (18–26). Calcitonin gene-related peptide (CGRP) monoclonal antibodies and gepants, which act on the CGRP pathway, have been more recently licensed by the US Food and Drug Administration (FDA) as preventive treatment options for migraine indications, including atogepant (27,28) and rimegepant (29,30) for episodic migraine; and eptinezumab (31,32), erenumab (33,34), fremanezumab (35,36) and galcanezumab (37,38) for both episodic and chronic migraine.
OnabotulinumtoxinA (BOTOX®) is an intramuscularly-injected acetylcholine release inhibitor and neuromuscular blocking agent that was the first prophylactic treatment specifically indicated for chronic migraine; onabotulinumtoxinA was approved in 2010 by the FDA (39) and by some national medicines agencies in Europe (40,41). Since its approval in the chronic migraine indication, the effectiveness of onabotulinumtoxinA has been captured within a large body of evidence from the real-world collected from the open-label prospective studies (42,43) (such as FORWARD, COMPEL) in addition to observational studies of treatment effectiveness. This real-world evidence (RWE) has complemented gold-standard evidence (44) from the onabotulinumtoxinA Phase 3 REsearch Evaluating Migraine Prophylaxis Therapy (PREEMPT 1 [NCT00156910] and PREEMPT 2 [NCT00168428]) pivotal randomised controlled trial (RCT) programme (6,45) in the chronic migraine population.
RWE is of increasing interest for both initial and ongoing regulatory and payer assessments in healthcare markets across the world (46–48) and such evidence on onabotulinumtoxinA effectiveness and safety representing real-world practice is particularly crucial for optimising chronic migraine treatment pathways given the heterogeneity of the patient population (49) (not reflected in RCTs) and complex patient management in the real world (50,51). Hence, given the ten-year experience with onabotulinumtoxinA since the treatment was licensed in chronic migraine, a systematic identification and evaluation of observational data around its real-world effectiveness in reducing headache symptoms, headache-related disability, and improving HRQoL in patients with chronic migraine is warranted for informing payer and clinical decision-making.
Objectives
The purpose of this study was to conduct a meta-analysis to evaluate the real-world effectiveness of onabotulinumtoxinA for the treatment of chronic migraine and to discuss these results in comparison with those obtained in the onabotulinumtoxinA PREEMPT pivotal trials and long-term open-label studies, where feasible.
Methods
The meta-analysis followed methodological guidelines (52–56) and was reported in accordance with the Meta-analyses Of Observational Studies in Epidemiology (MOOSE) Checklist (57).
Data collection
We conducted a systematic literature review (SLR) to inform the meta-analysis; the SLR was performed in accordance with the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) reporting guidelines (58,59) and included studies published between 1 January 2010 and 31 March 2021. We formulated the research questions and PICOS (Population, Intervention, Comparison, Outcomes, and Setting) framework based on the specific chronic migraine disease area, disease-modifying factors, interventions, and study types, which in turn informed the search strategies (Supplementary Table 1A).
Our searches spanned Medical Literature Analysis and Retrieval System Online (MEDLINE), Excerpta Medica database (Embase), and the Cochrane Library/Cochrane Central Register of Controlled Trials to identify relevant published real-world data. The searches applied controlled vocabulary and keywords, limited to articles involving human participants and published in English. Both MeSH and Emtree (Embase subject heading) terms were used in the search strategy (Supplementary Table 1B). MeSH terms included migraine, migraine disorders. Keywords for the condition were combined with those used for study design; examples of MeSH for study design were: retrospective studies, health survey, observational study, etc. Finally, we applied a filter to exclude publications in the form of letters, editorials or notes.
We also conducted bibliographic searches and gray literature searches of published systematic reviews and narrative reviews, and conducted manual searches within PubMed, Google, and Google Scholar. To retrieve the latest data from conference proceedings, manual searching was conducted for the previous 3 years (January 2017 to March 2020 and during the update from March 2020 to January 2021).This search identified relevant abstracts and presentations made at relevant scientific congresses, including: International Headache Society (5), Migraine Trust (60), American Headache Society (61), European Headache Federation (62), American Academy of Neurology (63) and International Society for Pharmacoeconomics and Outcomes Research (64). In addition, trial registry searches (Clinicaltrials.gov and the EU Clinical Trials Register [65]) retrieved ongoing and planned real-world observational studies for onabotulinumtoxinA in the target population of adult patients with chronic and unspecified migraine.
Research analysts with master’s degree in a pharmacy qualification conducted the study selection, initially reviewing the abstracts and titles of each citation and identifying a list of potentially relevant studies following a screening flowchart (Figure 1). Only those studies that met all pre-defined criteria for inclusion (Supplementary Table 1A) were considered for final extraction, while excluded citations were given a code justifying the reasons for exclusion. The lists obtained from the two reviewers were combined; a third reviewer (EO) then resolved any discrepancy. Any article that could not be clearly excluded based on the abstract was progressed for full-text review. Full-text versions of relevant studies were examined in more detail to determine a final list of included studies based on the Newcastle–Ottawa Scale (NOS) (66); a score of 4 (out of a maximum of 9) was the minimum cut-off point we applied for inclusion of full journal publications only (66,67). Assessment criteria varied according to study type (cohort studies or case–control studies; Supplementary Table 2). For meta-analysis, we selected studies of patients with chronic migraine specifically.

PRISMA flowchart. CFB: change from baseline; NOS: Newcastle–Ottawa Scale. *1 case–control study (82), 1 subgroup analysis (116).
Analysis
The meta-analysis considered the key endpoints examined in the onabotulinumtoxinA PREEMPT RCT programme in patients with chronic migraine (6,45,67). Both PREEMPT 1 and 2 comprised a 24-week, randomised, double-blind phase followed by a 32-week, open-label phase. The two PREEMPT RCTs were identical in design in terms of inclusion/exclusion criteria, randomisation, visits, double-blind phase, open-label phase, safety assessments, and treatment; in both studies, eligible patients were randomised (1:1) to onabotulinumtoxinA (155–195 U) or placebo injections every 12 weeks and assessed every four weeks. The primary study endpoint of both RCTs was Week 24 change from baseline (CFB) in number of headache days per month (a headache day was defined as any headache lasting at least 30 minutes; a month was defined as a 28-day period). Secondary efficacy variables included: CFB in number of migraine days per month, moderate/severe headache days per month, number of cumulative hours of headache on days when headache was experienced, proportion of patients scoring severe (≥60 points) on the Headache Impact Test (HIT)-6, number of headaches per month, number of migraine attacks (meeting ICHD-2 criteria 1.1 migraine without aura or 1.2 migraine with aura according to the patient's diary) per month, and number of days of acute headache pain medication intake per month. The definition used for the two headache/migraine attack frequency outcomes was a patient-reported headache/migraine with a start and end time indicating that the pain lasted four or more continuous hours. A further response analysis for PREEMPT included the rate of patients with ≥50% reduction in the number of headache days per month (6,67,68).
The patient-reported outcomes in PREEMPT included functioning, and HRQoL, as measured through CFB in total HIT-6 and Migraine-Specific Quality of Life v2.1 (MSQ) scores. HIT-6 evaluates the impact of headache on patient general well-being and daily activity, with scores ranging from 36 (no impact) to 78 (maximum impact); ≥60 is considered a major impact on general well-being and daily activity (69). The MSQ 14-item questionnaire measures the impact of migraine on the respondent’s physical and emotional functioning based on three domains: 1) role-function restrictive (limitations to daily social and work-related activities); 2) role-function preventive (how migraine prevents these activities); and 3) emotional function (migraine-associated emotional functioning) (70). All item scores are reverse-coded, and both MSQ domain and total score (evaluated in the present meta-analysis) are scaled 0–100, with higher score indicating better migraine-related quality of life (70).
We also conducted a meta-analysis on the Migraine Disability Assessment (MIDAS) score, if available in the selected studies. The MIDAS assesses headache-related disability, whereby the total score is graded across four levels: Grade I (0–5; minimal or infrequent), Grade II (6–10; mild or infrequent); Grade III (11–20, moderate), and Grade IV (≥21; severe disability) (71). Although MIDAS was not administered in the PREEMPT clinical programme, we evaluated the endpoint in meta-analysis as it has been deemed an important outcome by the International Headache Society for assessing the impact of preventive migraine treatments in clinical practice (72).
We conducted primary meta-analyses on PREEMPT endpoints for which there were enough studies available, on assessments ‘close to 24 weeks’ (∼24 weeks; up to six months) and ‘close to 52 weeks’ (∼52 weeks; up to 13 months), and a sensitivity analysis at ‘study-end’, whatever the duration of follow-up (Table 1). We applied both fixed-effects (inverse variance) and random-effects approaches. For the latter approach, we followed the DerSimonian and Laird method (73), adjusting standard errors of the study-specific estimates to incorporate a measure of study heterogeneity (i.e., Tau-squared [τ2]). However, given this high heterogeneity (both clinical and statistical) across studies, the random-effects approach was considered to be much more appropriate for interpretation of results and as such, reported in this study (74). In those studies, for which the input data in the required format were incomplete, we imputed missing values, estimating means and standard deviations from median, interquartile range and number of patients, based on the method described by Wan et al. (2014) for n > 200 (75):
Primary and sensitivity analyses.

*With durations of least 24 weeks; N/A: not applicable,
The primary analysis on available data for PREEMPT endpoints and MIDAS score included populations from all studies/all designs (i.e., prospective and retrospective) to allow for comprehensiveness and large sample sizes, and full, peer-reviewed publications only (Table 1; Figure 1). Data from conference abstracts were not included in this analysis as the granularity of information published in abstracts did not allow for assessing the quality of the study using the NOS score. To minimise the heterogeneity observed across studies, we then narrowed the primary analysis to prospective studies that had durations of at least 24 weeks. (Table 1; Figure 1).
Following primary analysis, we conducted sensitivity analysis 1 at study end, whatever the study duration, to provide higher analytical power and to allow evaluating the durability of treatment effect beyond ∼52 weeks on all available PREEMPT endpoints and MIDAS score (Table 1; Figure 1). Two further sensitivity analyses were conducted on Change from baseline in number of headache days per month. Sensitivity analysis 2 first considered the totality of full publications and abstracts on this outcome at 52 weeks; then sensitivity analysis 3 examined manufacturer-sponsored versus unsponsored studies to assess potential ‘sponsored bias’ at ∼24 weeks (Table 1; Figure 1).
All analyses were conducted on R statistical software (version 4.0.4).
Results
In total, the SLR identified 193 real-world studies in migraine, among which 157 were published as full papers and 36 as conference abstracts. After removing duplicate publications and studies in unspecified migraine, 44 full-text publications and 36 abstracts on chronic migraine passed screening and eligibility assessment and were considered for inclusion in the primary meta-analysis (Table 2; Figure 1). Of the 44 studies in full-text form, 29 were prospective studies (of which 15 had durations of 24 weeks or longer and were thus included in the primary analysis restricted to prospective studies); 13 were retrospective studies; two were classified as “other”. Geographically, most of the included studies were conducted in Italy (n = 15) and Spain (n = 11), with other countries across Europe, North America and Australia (Supplementary Figure 1).
Characteristics of studies included in meta-analysis.

CA: conference abstract; CGRP: calcitonin gene-related peptide; Dec: December; EMR: electronic medical record; FU: follow-up; Jan: January; JP: journal paper; N/A: not applicable; NOS: Newcastle–Ottawa Scale; N/R: not reported; onabotA: onabotulinumtoxinA; PCS: prospective cohort study; RCS: retrospective cohort study; UK: United Kingdom.
Note: *Germany, Italy, Norway, Russia, Sweden, Spain, and the United Kingdom. ^Erenumab, galcanezumab, fremanezumab. ‡NOS N/A for abstracts or secondary references
Outcomes assessed across studies (any time points).

CFB: change from baseline; HIT-6: Headache Impact Test-6; MSQ: Migraine-Specific Quality-of-Life Questionnaire; VAS: visual analog scale.
Bold font indicates outcome evaluated through meta-analysis. No studies reporting the endpoints of MSQ or migraine severity (VAS) were identified.
*Insufficient data (e.g., lack of standard deviation; standard error for CFB) to enable inclusion within meta-analysis.
PREEMPT endpoints, assessed where sufficient data were available in eligible studies, included mean CFB in number of headache days per month, number of days of acute headache pain medication intake per month, total HIT-6 score, and MSQ score. Studies also evaluated incidence of ≥50% reduction in the number of headache days per month. In addition to PREEMPT endpoints, some included studies also assessed the absolute MIDAS score. However, not all outcomes were assessed across all studies (Table 3).
Baseline clinical characteristics of patients within the included onabotulinumtoxinA real-world studies indicated heterogeneous patient populations (Supplementary Table 3). Mean (±standard deviation [SD]) age at study inclusion ranged from 39.3 (±10.2) (76) to 51.6 (±10.3) (77). Across the studies patients were, however, predominantly female (71% [78] to 92.5% [79]).
Duration of chronic migraine diagnosis ranged from a mean of 2.4 (±3.5) (80) to a mean of 25.7 (±12.3) years (81). The baseline number of headache days per month ranged from a mean of 18.9 (±5.5) (82) to a mean of 27.3 days (±4.7) (83) (Supplementary Table 3).
On patient-reported outcomes, mean HIT-6 scores at baseline ranged from a mean of 61.2 (SD not reported) (84) to a mean of 72.1 (±6.0) (85) (Supplementary Table 4). Mean baseline MSQ scores for role-function restrictive domain ranged from 36.2 (±17.8) (86) to 36.7 (SD not reported) (87). On the role-function preventive domain, mean scores ranged from 50.2 (±22.8) (REPOSE study) (86) to 51.4 (SD not reported) (87). On the emotional function domain, mean scores ranged from 38.0 (SD not reported) (87) to 42.4 (±25.6) (86) (Supplementary Table 4). Mean MIDAS scores at baseline ranged from 35.9 (±29.6) (88) to 117.13 (±94.8) (89) (Supplementary Table 4).
We also observed broadly comparable differences across studies for onabotulinumtoxinA dosage and the treatment protocol followed (Supplementary Table 5). Consequentially, the I2 statistics confirmed high overall study heterogeneity (Figures 2 –5) indicating that the random-effects models were the appropriate approach for interpretation of the meta-analysis results. However, we also reported the results of the fixed-effect models for completeness.
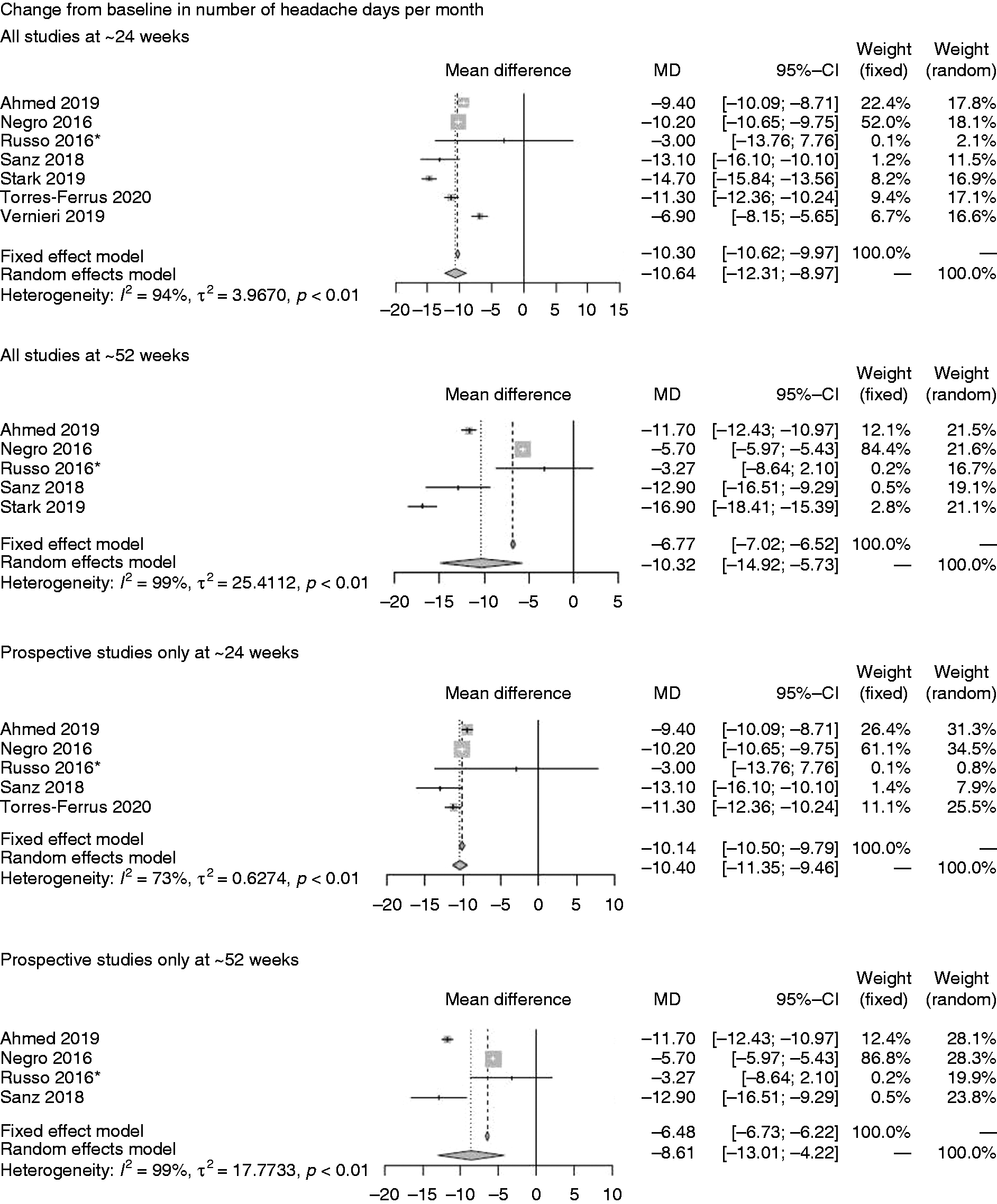
Primary analysis *Imputation of missing data applied. ¶ Comprises one prospective study only. CI: confidence interval; HIT: Headache Impact Test; MIDAS: Migraine Disability Assessment; MSQ: Migraine-Specific Quality of Life.

Sensitivity analysis 1: outcomes at study end *Imputation of missing data applied. ¶Comprises one prospective study only. CI: confidence interval; HIT-6: 6-item Headache Impact Test; MSQ: Migraine-Specific Quality-of-Life.

Sensitivity analysis 2: broad publication criteria (i.e., inclusion of conference abstract/poster data).

Sensitivity analysis 3: Unsponsored versus sponsored studies *Imputation of missing data applied. CI: confidence interval.
Primary analysis
Seven studies assessed onabotulinumtoxinA on mean CFB in number of headache days per month at ∼24-weeks and five at ∼52-weeks (Figure 2A) (77,86,90–94). The random-effects models revealed mean treatment effects of onabotulinumtoxinA of –10.64 (95% confidence interval (CI; –12.31, –8.97)) at ∼24 weeks and –10.32 (CI: –14.92, –5.73) at ∼52 weeks. When considering prospective studies only, similar mean treatment effects were achieved at ∼24 weeks (–10.40 (CI: –11.35, –9.46)) but slightly lower at ∼52 weeks (–8.61 (CI: –13.01, –4.22)) on this endpoint.
On CFB in number of days of acute headache pain medication intake per month (Figure 2B), onabotulinumtoxinA showed a mean of –7.40 (CI: –13.04, –1.77) on the endpoint at ∼24 weeks but the confidence intervals crossed zero at ∼52 weeks (77,91,93).
Seven studies reported the incidence of ≥50% reduction in migraine days per month (Figure 2C) at ∼24 weeks only and revealed an incidence of 46.57% (CI: 29.50%, 63.65%) for all studies and 38.52% (CI: 22.23%, 54.81%) for the group of prospective studies (88,91,93,95–98).
Data on mean CFB in total HIT-6 score (Figure 2D) were available in one study only (Stark 2019) (93) at ∼24 weeks (–11.70 [CI: –13.86, –9.54]) and ∼52 weeks (–11.80 [CI: 14.70, –8.90]). For mean CFB in MSQ score, we identified a single study only, which was also prospective (86) reporting the outcome at ∼24 weeks (23.60 [CI: 21.56, 25.64]) and ∼52 weeks (30.90 [CI: 28.29, 33.51]) (Figure 2E). On MIDAS score (Figure 2F), four studies (76,89,91,99) were available at ∼24 weeks only and showed onabotulinumtoxinA treatment effects based on a random-effects model of 44.74 (CI: 28.50, 60.99). Three prospective studies at ∼24 weeks showed similar treatment effects (44.26 [CI: 13.84, 74.68]) (76,89,91).
Sensitivity analysis
Sensitivity analysis 1, which assessed outcomes at study end, indicated that durability slightly waned over time for onabotulinumtoxinA on mean CFB in number of headache days per month (–9.21 [CI: –12.48, –5.93]) based on a random effects model considering all studies and compared with earlier assessments. However, the waning of effectiveness was less so considering prospective studies only (–8.85 [CI: –12.83, –4.88]) (Figure 3A) (79,83,86,90–94,100–102).
On number of days of acute headache pain medication intake per month, mean CFB was slightly lower at study end (–5.40 [CI: –10.22, –0.58]) (Figure 3B) (77,83,91,93) compared with earlier assessments. Only two studies provided data collected prospectively, Ahmed et al. (83) (4 weeks) and Russo et al. (91) (39 weeks) revealing less of a reduction on number of days of acute headache pain medication intake per month (−3.50 [CI: –4.01, –2.99]) for onabotulinumtoxinA (Figure 3B).
Data on mean CFB in total HIT-6 score (–9.84 [CI: –13.14, –6.55]) was only available for one study (83) at 4 weeks and 60 weeks (93) (Figure 3C), but a decrease over the long term was observed only over the 60-week study (93). The prospective study (4-week study) (83) assessed mean CFB in total HIT-6 score at the end of the study (Figure 3C) showing a mean CFB of –8.40 (CI: –9.69, –7.11). On CFB increase in MSQ score, the single prospective study (86) indicated a mean of 33.60 (CI: 30.03, 37.17) at 84 weeks (Figure 3D), an overall improvement compared with earlier assessments.
Sensitivity analysis 2 based on all publications (full articles and abstracts) indicated CFB in number of headache days per month at ∼52 weeks of –8.61 (CI: –11.69, –5.62) (77,86,90–93,103,104); no data were available for ∼24 weeks (Figure 4). In sensitivity analysis 3, comparing data for this endpoint at ∼24 weeks indicated slightly less favourable results for unsponsored studies (–9.93 [CI: –11.78, –8.08]) versus sponsored studies (–12.03 [CI: –17.22, –6.84]) (Figure 5) (77,90–92,94).
Discussion
This meta-analysis indicates that outcomes for onabotulinumtoxinA in the real world measured at ∼24 weeks and ∼52 weeks are broadly comparable to the outcomes observed in the PREEMPT pivotal studies of onabotulinumtoxinA. For example, the n = 347 adults randomised to onabotulinumtoxinA every 12 weeks for two cycles in PREEMPT 2 experienced statistically significant and clinically meaningful improvements on the primary endpoint, mean CFB on number of headache days per month (–9.0 onabotulinumtoxinA vs –6.7 placebo, p < 0.001) (45). Our meta-analytic results showed a comparable magnitude of treatment effect for onabotulinumtoxinA on this measure at ∼24 weeks (–10.64 [CI: –12.31, –8.97]) and ∼52 weeks (–10.32 [CI: –14.92, –5.73]).
Moreover, whereas both PREEMPT 1 and 2 indicated statistically significant results for onabotulinumtoxinA versus placebo on selected secondary endpoints evaluated in our analysis (e.g., incidence of ≥50% reduction in migraine days per month, CFB in total HIT-6 scores, and CFB in MSQ scores) across all time points, our primary meta-analytic results pointed to similar levels of changes on outcomes for onabotulinumtoxinA in the real-world (67). Our analysis also showed similar CFB decrease in number of days of acute headache pain medication intake per month as in PREEMPT 2 (45).
In terms of real-world, long-term treatment effects compared with those observed in the COMPEL long-term, open-label clinical study of onabotulinumtoxinA, our findings were variable. For example, COMPEL observed a mean reduction in number of headache days per month (–10.7 [CI: –11.0, –10.0]) at 108 weeks for onabotulinumtoxinA (43). However real-world data suggested lower treatment effect on this outcome which had also slightly waned over time (–9.21 [CI: –12.48, –5.93]) from among the 11 studies that ranged from four weeks (84,93) to 105 weeks (90). Conversely, compared with the CFB for HIT-6 scores in COMPEL (–7.1 [CI: –7.8, –6.9]) at 108 weeks, two real-world studies (of four weeks [83] and 60 weeks [93] duration, respectively) together showed better improvement at study end (–9.84 [CI: –13.14, –6.55]). However, in considering these comparisons, it is notable that the COMPEL study evaluated the fixed-dose, fixed-site injection paradigm, which may not necessarily reflect real-world practice. Similarly, long-term mean CFB in HIT-6 score in our analysis was better than observed in the FORWARD multicenter, randomised, parallel-group, open-label prospective study of onabotulinumtoxinA 155 U every 12 weeks for three cycles. The comparator in this study was topiramate immediate-release 50–100 mg/day. Patients randomised to onabotulinumtoxinA exhibited significant (p < 0.001) mean (±SD) CFB in HIT-6 score of –5.6 (±7.2) (compared with –1.3 [±3.9] for topiramate) (42) to week 30 on the secondary endpoint of CFB in HIT-6 score. In terms of overall durability of treatment effect found across our studies, apart from a mean CFB increase in MSQ score in the single study by Ahmed et al. (86) at 84 weeks, the real-world data showed waning durability on other outcomes at end-of-study assessments.
A few important limitations to our meta-analysis must be considered for interpreting our findings. Firstly, there was high heterogeneity of results reported between studies (I2 ≥ 90% for most analysis) for many scenarios. High heterogeneity is expected in meta-analyses that include a large number of studies to enrich the sample (55). The meta-analyses presented in the current study are mostly based on single-arm trials with relatively narrow CIs, which influences the values of the statistical heterogeneity parameters. In particular, the narrower CIs of included studies, the higher I2. The fact that the CIs are narrow can be explained by potential overpowering, which is common in RWE. Indeed, given the same total sample size, the arm-wise sample size is larger for single-arm trials, hence the CIs are usually narrower (52–55). For example, a 99% I2 was estimated in the real-world meta-analysis of intravitreal aflibercept and intravitreal ranibizumab in age-related neovascular macular degeneration (105), and a 97% I2 in a meta-analysis on real-world statin use and risk of new-onset diabetes (106). Other comparable heterogeneities have also been reported elsewhere (105,106).
Nevertheless, we took the following steps to resolve the problem of high heterogeneity. First, we applied a random-effects model to account for the uncertainty of the pooled results (54). In this case, we did not expect a common ‘true effect', but rather a distribution of effects, as the population is heterogeneous across different observational studies (per age, disease severity, previous treatment, origin, ethnicity, socio-demographic data). Indeed, the CIs in the random-effects model reflected the range of results observed in all included studies. Hence, we presented the results across three different observation periods.
We also evaluated a series of scenarios and assessed potential sources of study heterogeneity by applying specific inclusion criteria to the meta-analysis, these included: retrospective versus prospective design, sponsorship status, and more technical sources allowing inclusion of results based on abstracts or studies for which data imputation procedures for missing data have been applied. We observed no substantial differences in the results following sensitivity analyses.
Many of the meta-analysis estimates (e.g., reduction in the number of days with migraine, at six or 12 months, or of moderate/severe headache days at 12 months, CFB in the total HIT-6 score at six months, CFB at six and 12 months of the MSQ score) were also based on only one or two studies (e.g., reduction in the number of moderate/severe headache days at six months, proportion of patients at six and 12 months with HIT-6 score of 60 or more). This is a result of the data identified, and some of the parameters were rarely investigated/reported in the selected studies (e.g., HIT-6 and MSQ scores); hence, this reduced the number of included studies and limited the ability to analyse the outcomes. Another limitation is the missing information to characterise the patients included in the meta-analysis, especially for some variables such as the number of previous prophylactic treatments received.
This meta-analysis has, nonetheless, systematically considered the totality of empirical evidence and provided insights regarding the agreement or disagreement of the results from different observational study designs (54). In addition, all studies were selected based on a NOS score of at least 4 out of a maximum of 9 (66). For example, for the primary endpoint (change from baseline in number of headache days per month) analysed in the primary analysis, only one of 11 studies rated 4, three rated 5, and seven out of 11 studies rated ≥6 or more on the NOS. Of the 56 full-text studies identified in the SLR, eight studies were of high quality, with a score ranging from 7–8 on NOS; 48 studies were of moderate quality with a score of 4–6 on NOS. The two studies deemed low quality (i.e., NOS score of 2–3) were excluded from the meta-analysis. Hence, given these study limitations and strengths, while no strong conclusions can be made on the findings, our results provide a broad indication of the treatment effects associated with onabotulinumtoxinA in the real world as observed in the RCTs.
It can be noted that the NOS is one of the most widely used scales for assessing quality and risk of bias in observational studies (66,107), as other tools, such as the ROBINS-I, are more commonly used to only assess the risk of bias of observational studies; therefore, each assesses different aspects in the design, execution, validity, and precision of a study (108).
While a cutoff threshold for the NOS distinguishing between ‘good’ and ‘poor’ quality studies is yet to be validated (66), we defined a score of 4 as the cutoff for our approach to selecting studies of adequate quality. We applied this for our primary analysis, including only the studies that satisfied this criterion. We further conducted a sensitivity analysis which included the studies with low NOS (i.e., <4 points) to assess the influence on the results of relaxing the inclusion criteria, including one study based on full text for the endpoint, Migraine Disability Assessment (MIDAS) score. We then included studies for which only the abstracts were available; for this analysis, the endpoints available for assessment included: CFB in number of headache days per month; CFB in number of days of acute headache pain medication intake per month; and ≥50% reduction in migraine days per month. It is notable that, in the results for second sensitivity analysis including abstract-only studies, we found that, not only were the differences unsubstantial, the small differences only further favoured onabotulinumtoxinA. Hence, by excluding these studies from the primary analysis, we have ensured that our main analysis was not only based on robust data, but also conservative. Finally, we also assessed the risk of publication bias by conducting a sensitivity analysis considering sponsored versus non-sponsored studies and found that the results were comparable.
Conclusions
This meta-analysis of onabotulinumtoxinA, in the context of clinical practice that reflects the heterogeneity of populations treated with onabotulinumtoxinA for preventive treatment of chronic migraine, broadly corroborates with the efficacy that has been observed in the pivotal RCTs and long-term, open-label studies of onabotulinumtoxinA. Ongoing research collecting real-world data on onabotulinumtoxinA and the newer treatments for chronic migraine will provide further insights into the effectiveness of chronic migraine preventive treatments in clinically generalisable patient populations.
Article highlights
A meta-analysis of onabotulinumtoxinA was conducted to provide context for the heterogeneity of populations prophylactically treated with onabotulinumtoxinA for chronic migraine in clinical practice. In total, these data corroborate the efficacy that has been observed in the pivotal RCTs and long-term, open-label studies of onabotulinumtoxinA.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024221123058 - Supplemental material for Effectiveness of onabotulinumtoxinA (BOTOX®) for the preventive treatment of chronic migraine: A meta-analysis on 10 years of real-world data
Supplemental material, sj-pdf-1-cep-10.1177_03331024221123058 for Effectiveness of onabotulinumtoxinA (BOTOX®) for the preventive treatment of chronic migraine: A meta-analysis on 10 years of real-world data by Michel Lanteri-Minet, Anne Ducros, Clement Francois, Elzbieta Olewinska, Mateusz Nikodem and Laure Dupont-Benjamin in Cephalalgia
Footnotes
Authors’ contributions
All authors read, contributed to, and approved all the drafts and the final manuscript:
CF was involved in the review of the systematic literature review (SLR) protocol, the meta-analysis plan, and interpretation of results.
MN was involved in the review of the SLR protocol, meta-analysis plan development, statistical analysis and interpretation of results.
EO was involved in the development of the SLR protocol, the extraction grid, and the reporting of the SLR.
LDB was involved in the review of the SLR protocol, the meta-analysis plan, and interpretation of results.
ML-M was involved in the review of the meta-analysis plan and interpretation of results.
AD was involved in the review of the meta-analysis plan and interpretation of results.
Acknowledgements
Medical writing assistance, under the direction of the authors, was provided by Gauri Saal, MA Economics (funded by AbbVie according to Good Publication Practice guidelines [![]() ]) and Inès Bouajila, Health Economics & Outcomes Research Fellow from AbbVie.
]) and Inès Bouajila, Health Economics & Outcomes Research Fellow from AbbVie.
The initial systematic literature review was performed by Bridge Medical, updated by Creativ-Ceutical, and funded by AbbVie.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ML-M reports personal fees from Allergan; personal fees from Amgen; grants, personal fees from Eli Lilly; personal fees from Grunenthal; grants and personal fees from Lundbeck; grants and personal fees from Medtronic; grants, personal fees from Novartis; personal fees from Pfizer; personal fees from Reckitt Benckiser; personal fees from Sun Pharmaceutical; grants, personal fees from Teva Pharmaceuticals; personal fees from UPSA; personal fees from Zambon. AD reports personal fees from AbbVie/Allergan, Eli Lilly, Lundbeck, Novartis, and Teva. FC, MN, and EO work for Creativ-Ceutical, a consultancy company commissioned by AbbVie who provided the funding to perform the SLR update and the meta-analysis. LDB is an employee of AbbVie and holds stock options from GSK and AbbVie.
Funding
The authors disclose receipt of the following financial support for the research, authorship, and publication of this article: The literature review and editorial assistance for the manuscript was funded by AbbVie. AbbVie participated in the review of the SLR protocol, the meta-analysis plan, interpretation of results; and in the writing, reviewing and approval of the final version. No honoraria or payments were made for authorship.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


