Abstract
Background
Chronic migraine (CM) is associated with high impact and reduced health-related quality of life (HRQoL).
Methods
Patients with CM from PREEMPT (Phase 3 REsearch Evaluating Migraine Prophylaxis Therapy) were randomized (1:1) to receive onabotulinumtoxinA or placebo for two 12-week cycles in the double-blind (DB) phase, followed by three 12-week cycles of open-label (OL) onabotulinumtoxinA (onabotulinumtoxinA/onabotulinumtoxinA (O/O) and placebo/onabotulinumtoxinA (P/O) groups, respectively). HRQoL endpoints were assessed over 56 weeks using the Headache Impact Test (HIT-6) and the Migraine-Specific Quality of Life Questionnaire (MSQ). HIT-6 score reductions ≥2.3 and ≥5 denoted between-group minimally important difference and within-patient clinically meaningful response, respectively.
Results
A total of 1236 participants (O/O, n = 607; P/O, n = 629) participated in both phases. The DB phase showed significantly reduced HIT-6 and MSQ for onabotulinumtoxinA versus placebo (all p < 0.001). The OL phase showed significantly reduced HIT-6 for O/O versus P/O at weeks 28, 36, and 48, but not 56. All three MSQ domains showed improved HRQoL relative to baseline, but only the role restrictive domain showed a significant difference between O/O and P/O at week 56.
Conclusions
Benefits of onabotulinumtoxinA on HRQoL versus baseline persisted throughout the OL phase. Statistical superiority in favor of O/O was demonstrated for HIT-6 through 48 weeks and for MSQ (role restrictive) at 56 weeks.
Introduction
Chronic migraine (CM), characterized by ≥15 headache days per month, is highly debilitating and associated with significant personal, social, and economic burden. Individuals with CM experience greater headache impact (1) and disability (2), reduced productivity at work, home, and school (3), decreased health-related quality of life (HRQoL) (1), and increased comorbid medical conditions, including obesity, vascular disease, and psychiatric disorders (1,2,4) compared with those with episodic migraine (EM; <15 headache days/month). In addition, those affected by CM make more visits to health care providers than those with EM (1,3), and direct and indirect costs of CM are greater than four times higher than those associated with EM (5).
The clinical safety and efficacy of onabotulinumtoxinA as headache prophylaxis in adults with CM was demonstrated in the Phase III REsearch Evaluating Migraine Prophylaxis Therapy (PREEMPT) clinical trials program (6–10). In the pivotal trials, PREEMPT 1 and PREEMPT 2, eligible patients were randomized to double-blind treatment with onabotulinumtoxinA or placebo for 24 weeks followed by open-label treatment with onabotulinumtoxinA for 32 weeks, for a total treatment of up to 56 weeks (6,7). In addition to evaluating the efficacy of onabotulinumtoxinA vs. placebo on headache-related variables, the PREEMPT trials also evaluated treatment effects on headache impact using the 6-item Headache Impact Test (HIT-6) and on HRQoL using the Migraine-Specific Quality of Life Questionnaire (MSQ), version 2.1. Primary results from the 24-week, double-blind phase have been published separately for PREEMPT 1 (6) and PREEMPT 2 (7). Data from the two trials were subsequently pooled to examine the durability and precision of the individual study results and to explore other potentially relevant findings by virtue of the added statistical power (8,9). Pooled results from the double-blind period have been reported for the HIT-6 and MSQ at weeks 12 and 24 (8–10). Herein, we report the pooled HRQoL outcomes for the entire 56-week treatment period, which includes the double-blind and open-label periods.
Methods
Study design and patient population
PREEMPT 1 and 2 were conducted concurrently at 122 sites (106 in North America and 16 in Europe) between January 2006 and August 2008. In each study, eligible adults (aged 18–65 years) were required to meet the International Classification of Headache Disorders, 2nd edition (ICHD-2) diagnostic criteria for CM (11). To be eligible for inclusion, patients must have had ≥15 headache days during the 28-day screening period (baseline), during which ≥4 hours of each headache day were continuous headache and ≥50% were migraine or probable migraine days. At the start of the double-blind period, qualified individuals were randomized (1:1) to receive onabotulinumtoxinA or placebo for 24 weeks (two injection cycles), after which all patients received onabotulinumtoxinA in the 32-week (three injection cycles) open-label period.
Randomization was stratified by baseline medication overuse in blocks of four for each site. OnabotulinumtoxinA (155 U) or matching placebo (2 ml of 0.9% sodium chloride (saline)) was administered as 31 fixed-dose, intramuscular injections across specific head/neck muscle areas every 12 weeks. At their discretion, investigators could inject an additional 40 U (maximum total dose, 195 U) of study drug for a maximum of 39 injection sites each cycle. Complete study design details for PREEMPT 1 and PREEMPT 2 have been published (6,7).
The PREEMPT clinical trial program was approved by an independent ethics committee or local institutional review board at each participating site, and written informed consent was obtained from all enrolled patients. The studies were conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines.
Outcome measures
The validated HIT-6 is a self-administered tool that assesses the impact of headache using widely measured, functionally relevant domains: pain, social and role limitations, cognitive functioning, vitality, and psychological distress (12,13). Each item assesses how often headaches interfere with activities or cause distress; response options are “never” (6 points), “rarely” (8 points), “sometimes” (10 points), “very often” (11 points), and “always” (13 points). Points for all six items are summed to obtain a total score (36–78); higher total scores indicate greater negative impact. Categorically, scores ≤49 indicate little to no headache impact, 50–55 indicate some impact, 56–59 indicate substantial impact, and ≥60 indicate severe headache impact (13). The HIT-6 has been validated both in EM and CM (13,14). The between-group minimally important difference (MID) for the reduction in HIT-6 total score is ≥2.3 points (15). At the patient level, a score reduction ≥5 points is considered to be a clinically meaningful response (13). HIT-6 was assessed at baseline and at 4, 8, 12, 16, 20, 24, 28, 36, 48 and 56 weeks.
The MSQ is a migraine-specific assessment for quality of life that was developed to assess the effect of migraine on daily functioning across three domains. The Role Restrictive (RR) domain measures the effect of migraine on daily social and work-related activities, the Role Preventive (RP) domain assesses whether migraine prohibits performing these activities, and the Emotional Functioning (EF) domain measures the emotions associated with migraine (16). Response options for the 14 items in version 2.1 are: 1 = “none of the time,” 2 = “a little bit of the time,” 3 = “some of the time,” 4 = a good bit of the time,” 5 = “most of the time,” and 6 = “all of the time” (17). Domain scores are calculated as the sum of their item scores, and then transformed to a 0–100 reversed scale in which higher scores indicate better HRQoL (17,18). The within-group MIDs for MSQ version 2.1 domains are RR = 10.9, RP = 8.3, and EF = 12.2 (19), and the between-group MIDs are RR = 3.2, RP = 4.6, and EF = 7.5 (20). The MSQ has been shown to reliably measure HRQoL among patients with migraine in the outpatient setting (18). Among patients receiving prophylactic migraine therapy, the MSQ has shown stable and reliable psychometric properties, lending support for its use as an outcome measure in clinical trials (21), and was recently validated in the CM patient population (17). MSQ was assessed at baseline and at 12, 24 and 56 weeks.
Statistical analysis
Data from PREEMPT 1 and 2 were pooled for an integrated efficacy analysis (8,9). All efficacy data were analyzed using the intent-to-treat (ITT) population, which included all randomized patients. Statistical comparisons for the open-label phase were based on the patients’ double-blind-phase treatment; thus, the groups compared were those who received onabotulinumtoxinA in both double-blind and open-label phases (O/O) vs. those who received placebo in the double-blind phase and onabotulinumtoxinA in the open-label phase (P/O).
Between-group comparisons of HIT-6 and, separately, HIT-6 response categories were made using Wilcoxon rank-sum tests. Between-group binomial comparisons of HIT-6 response dichotomies were made using Pearson’s chi-square (or Fisher exact tests if ≥25% of the expected cell counts were <5). Missing HIT-6 data were imputed using a prespecified, modified last observation carried forward technique (6,7). All observed MSQ data were analyzed without imputation for missing values.
Most HIT-6 and MSQ outcomes were tertiary variables in the PREEMPT study; therefore, analyses reported herein should be considered exploratory. Significance is reported to indicate noteworthy differences in support of previously reported primary and secondary outcomes at a two-sided alpha level of ≤0.05. All variables and statistical methods were prespecified prior to unblinding.
Results
Patient disposition and demographics
Baseline demographic and clinical characteristics.

CM: chronic migraine; HIT-6: 6-item Headache Impact Test; MSQ: Migraine-Specific Quality of Life Questionnaire; O/O: onabotulinumtoxinA in double-blind phase and open-label phase; P/O: placebo in double-blind phase and onabotulinumtoxinA in open-label phase; HRQoL: health-related quality of life; y: years; h: hours.
HIT-6 scores of 36–49 indicate little or no impact; 50–55, some impact; 56–59, substantial impact; 60–78, severe impact.
MSQ v2.1 scores range from 0 (poor HRQoL) to 100 (good HRQoL).
Headache impact: HIT-6 results
The mean total HIT-6 scores at baseline were similar between treatment groups (O/O 65.5 (95% confidence interval (CI) 65.21–65.81) versus P/O 65.4 (95% CI 65.07–65.71), p = 0.64). Most patients were severely debilitated by their migraines, with 93% (95% CI 91.7–94.4, p = 0.57 between groups) reporting a total HIT-6 score ≥60 (severe headache impact) and another 5% reporting a score of 56–59 (substantial impact; p = 0.54 across the four score categories). During the 24-week double-blind phase, both treatment groups showed notable reductions (i.e. improvements) in mean total HIT-6 score; however, these changes were significantly larger in the onabotulinumtoxinA group than in the placebo group at all visits (Figure 1(a)). In both groups, HIT-6 precipitously dropped over the 4 weeks following active or placebo treatment; the decline in HIT-6 at 4 weeks was significantly greater with onabotulinumtoxinA than placebo. Smaller subsequent improvements continued throughout the double-blind period, with onabotulinumtoxinA being superior to placebo at all time points. At the start of the open-label phase, after patients in both treatment groups received onabotulinumtoxinA injections at week 24, mean HIT-6 total scores showed another steep reduction in both treatment groups (Figure 1(a)). Not surprisingly, the change from week 24 to week 28 in the P/O group was larger (3.7 points) than the change in the O/O group (2.7 points). However, despite statistically significant within-group changes from baseline during this period, the between-group differences remained statistically significant and in favor of O/O through week 48. The between-group HIT-6 MID (≥2.3 points) was achieved starting at week 16, and lasted through the end of the double-blind period (week 24). This MID was not observed during the open-label period when both treatment groups received onabotulinumtoxinA.
Efficacy of onabotulinumtoxinA vs. placebo on changes in (a) mean change (standard error) in total HIT-6 scorea and (b) percentage of patients with severe or substantial headache impact (HIT-6 score ≥56) at each time pointb.
Similarly, early during the double-blind phase, the percentage of patients with substantial (HIT-6 score 56–59) or severe headache impact (HIT-6 score ≥60) dropped markedly in both treatment groups (Figure 1(b)), although significantly smaller percentages of patients in the onabotulinumtoxinA group than in the placebo group were considered substantially or severely affected. At week 28 (first visit after all P/O patients were switched to onabotulinumtoxinA), 67% of O/O and 72% of P/O patients had mean HIT-6 scores ≥56. By the end of the open-label phase, after all patients had received ≥3 cycles of onabotulinumtoxinA, the O/O and P/O groups had indistinguishable HIT-6 scores, with 66% and 68%, respectively, with substantial or severe headache impact (Figure 1(b)).
Throughout the double-blind phase, the percentage of patients with a clinically meaningful individual response on the HIT-6 (i.e. ≥5-point decrease in total score) was significantly greater in the onabotulinumtoxinA group than in the placebo group (Figure 2(a)). During the open-label phase, >50% of patients in both groups had clinically meaningful individual responses at all time points after receiving onabotulinumtoxinA. The between-group difference at week 36 (57.3% (CI 53.6–61.0) versus 50.6% (CI 46.9–54.3), p = 0.013) was the only contemporaneous comparison to reach statistical significance (Figure 2(a)).
Efficacy of onabotulinumtoxinA vs. placebo on the proportions of patients achieving (a) individual HIT-6 MIDa during DB and OL phases of PREEMPT trials (*p < 0.001; ‡p = 0.002), (b) individual ≥50% reduction in headache days during DB and OL phases of PREEMPT trials (*p < 0.001; ‡p < 0.05), and (c) individual HIT-6 MID and ≥50% reduction in headache days during DB and OL phases of PREEMPT trials (*p < 0.001; †p = 0.002; ‡p < 0.05).
The percentage of patients achieving a ≥50% headache-day reduction is shown in Figure 2(b). At the end of the double-blind phase (week 24), a significant between-group difference was observed (p < 0.001) in favor of onabotulinumtoxinA. In the open-label phase, improvements in ≥50% headache-day reduction became similar for the P/O group and the O/O group by week 32.
Figure 2(c) shows the percentage of patients who were not only treatment responders in terms of an objective headache variable (≥50% reduction in headache days) but also had a clinically meaningful change in headache impact (≥5-point reduction in total HIT-6 score). Although the overall percentages are lower in both treatment groups, the same pattern emerges: significant between-group differences in the double-blind phase disappear during the open-label phase after all patients have received ≥3 cycles of onabotulinumtoxinA.
HRQoL: MSQ scores
Baseline mean MSQ domain scores were similar between the treatment groups (p ≥ 0.81 for all), and all mean scores were ≤56 points (Table 1), indicating poor HRQoL. At the end of the double-blind phase (week 24), between-group differences were significant for all MSQ domains (all p < 0.001) in favor of onabotulinumtoxinA (Figure 3(a)–(c)) By week 56, mean changes from baseline in the three functional domain scores increased significantly in both the O/O and the P/O groups, reflecting improvements in HRQoL (Figure 3(a)–(c)). At week 56, the between-group difference in MSQ change scores was statistically significant for the RR domain only (3.4; p = 0.043), but was only slightly larger than the between-group MID for the domain (3.2). The between-group differences in change scores for the other domains were notably smaller (RP = 1.7; EF = 2.9) than their respective MIDs (RP = 4.6; EF = 7.5) at week 56. By contrast, mean change scores for patients both in the O/O and P/O groups were markedly higher than the established within-group domain MIDs (Figure 3(a)–(c)).
Efficacy of onabotulinumtoxinA versus placebo on the MSQ v2.1a by functional domain during DB and OL phases of the PREEMPT trials. (a) Role Function – Restrictive, (b) Role Function – Preventive, and (c) Emotional Function (*p < 0.001; †p < 0.05).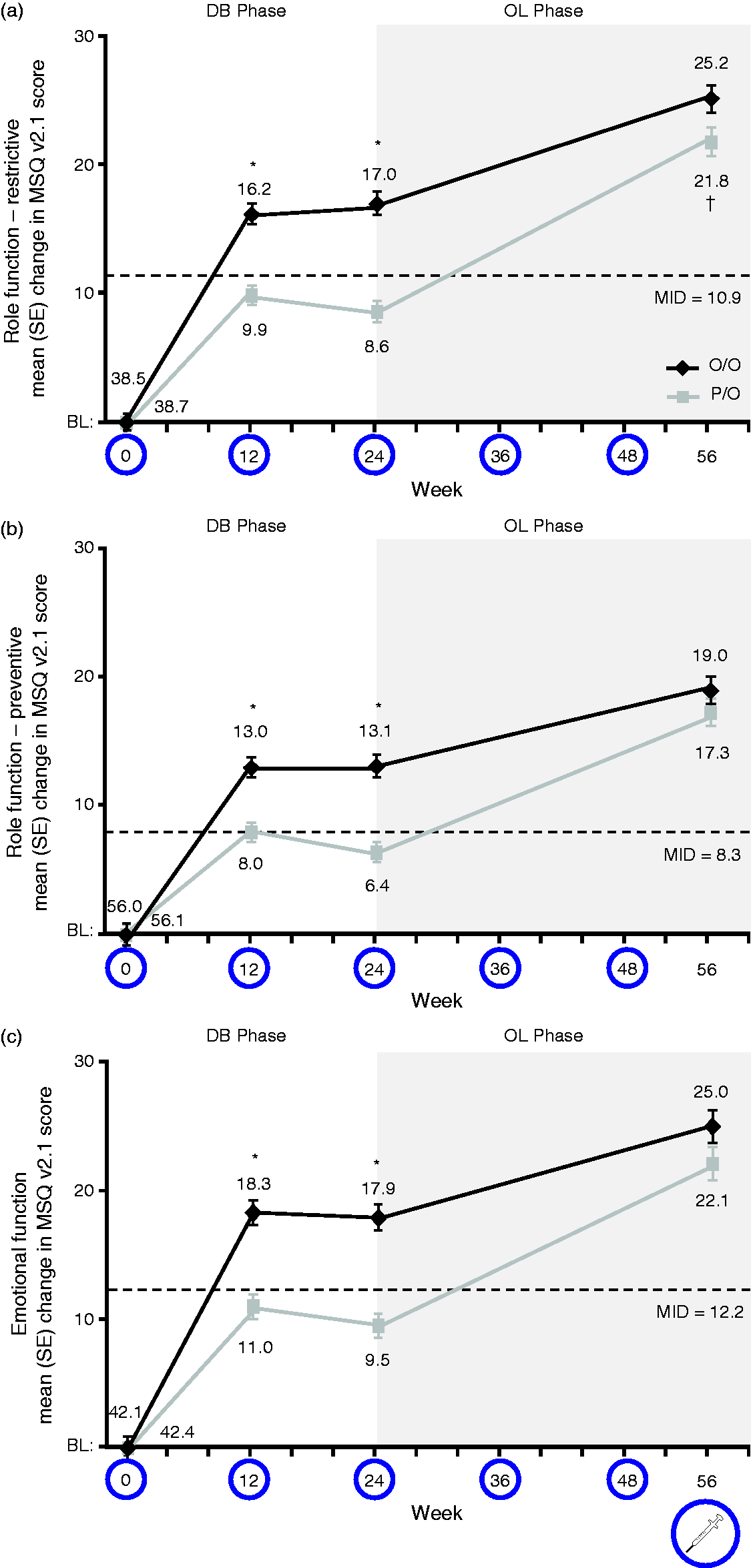
Discussion
The data reported herein examine the long-term effects of onabotulinumtoxinA on measures of headache impact and HRQoL, supporting and extending previous reports (6–10). Data from the double-blind phase of the studies demonstrate significant improvement with onabotulinumtoxinA versus placebo observed over 24 weeks of treatment. During the 32-week open-label phase of the trials, when all patients were receiving onabotulinumtoxinA, headache impact and HRQoL continued to improve both in the O/O and P/O groups. These data demonstrate that the benefits of onabotulinumtoxinA persist over 56 weeks of treatment using measures of impact (HIT-6) and HRQoL (MSQ questionnaires). Patients who made the switch from placebo to onabotulinumtoxinA (P/O) at 24 weeks experienced significant improvements from baseline at a rate of change not that different from what was observed among patients who received onabotulinumtoxinA from the start of the double-blind period. This indicates that the efficacy of onabotulinumtoxinA persists even if treatment is delayed. However, for several endpoints, statistically significant differences in favor of the O/O group versus the P/O group persisted for months, suggesting that, even in a group with a considerable response to placebo treatment during the double-blind phase, it takes a long time to achieve outcomes equivalent to the O/O group.
Headache impact
By week 16 (and lasting to week 24) of the double-blind phase, the between-group mean difference in the change in HIT-6 score exceeded the between-group MID (2.3 points; Figure 1(a)) (10). In the open-label phase, when both groups were receiving onabotulinumtoxinA, the between-group difference did not achieve the MID of 2.3 points, though there were statistically significant differences in favor of the O/O group versus the P/O group at 24, 36 and 48 weeks. Despite the delay in starting treatment, the P/O group experienced reductions in headache days after onabotulinumtoxinA was started. At each assessment during the double-blind phase, significantly more patients receiving onabotulinumtoxinA showed a clinically meaningful (≥5-point) reduction in total HIT-6 score (p < 0.001; Figure 2(a)) (10). At the end of the double-blind phase (week 24), 41% of patients receiving onabotulinumtoxinA and 25% of patients receiving placebo had achieved the individual MID in HIT-6 score (10). At the end of the open-label phase (week 56), P/O patients caught up with O/O patients in achieving this MID (O/O: 59%, P/O: 57%; p = 0.46).
HRQoL
During double-blind treatment, between-group differences in MSQ change scores exceeded the established between-group MIDs for each domain and within-group MIDs were achieved only by patients in the onabotulinumtoxinA group (10). At the end of open-label treatment (week 56), changes from baseline in all three domains exceeded the within-group MIDs for both treatment groups, but between-group differences in MIDs were achieved only for the RR functional domain. Therefore, treatment with onabotulinumtoxinA resulted in statistically significant and clinically meaningful reductions in headache impact and HRQoL that persisted and continued to improve throughout the 56-week trial.
The specific diagnostic criteria and nomenclature for CM have evolved considerably since the condition was first termed “transformed migraine” in the 1980s (23,24). For example, diagnostic criteria for CM in the ICHD-2 (11) were stricter than current ICHD third edition beta (ICHD-3b) criteria (25), resulting in fewer patients fulfilling CM criteria (26), and ICHD-3b was created to address these issues. Variation in case definition contributes to the varying estimates of CM prevalence (ranging from 0 to 5%) in epidemiologic studies (27). Although the PREEMPT studies were conducted using criteria that differ from ICHD-2 and ICHD-3b criteria, the overlap in diagnostic criteria is very high (28). Baseline demographic data in the PREEMPT trials reported herein are generally similar to those reported for individuals with CM in the American Migraine Prevalence and Prevention (AMPP) Study (29) and the Chronic Migraine Epidemiology and Outcomes (CaMEO) Study (2), a large epidemiologic study that used ICHD-3b diagnostic criteria, suggesting the populations obtained using these criteria are not substantively different.
Substantial early improvement both in O/O and P/O groups warrants discussion. At the first post-injection assessment (week 4), both O/O and P/O groups showed declines in HIT-6 scores, though there were significant differences between groups favoring onabotulinumtoxinA (Figures 1 and 2). Similarly, MSQ scores showed marked improvement in both O/O and P/O groups at the first post-baseline assessment (Figure 3) at 12 weeks, with mean changes nearly achieving MID in some domains. At 24 weeks, all patients knew they were receiving active treatment, possibly contributing to the observed HIT-6 decline and MSQ increase from week 24 to the next assessment. Graphically, the gap between the O/O and P/O groups at the end of the double-blind phase narrows more quickly in the open-label phase for HIT-6 and MSQ than the previously reported efficacy endpoints (8), suggesting that patients may achieve improvements in HRQoL before observing improvements in headache-day frequency. Clinically, this may be observed in patients who do not have an immediate reduction in headache days, but return to improved functioning levels after starting treatment with onabotulinumtoxinA.
CM can be a highly refractory and tremendously disabling disorder. In this pooled analysis, long-term treatment of CM with onabotulinumtoxinA resulted in robust reductions on headache impact, as measured by HIT-6 scores. Approximately one-third of patients had HIT-6 scores indicating the absence of severe or substantial headache impact at the end of the 56-week study. Nevertheless, approximately two-thirds of all patients (O/O, 66%; P/O, 68%) still had HIT-6 scores in the severe or substantial headache impact range at the end of open-label treatment with onabotulinumtoxinA. The results demonstrate sustained reductions in headache impact as well as persistent unmet medical needs. This information can be used by clinicians to help shape realistic patient expectations regarding onabotulinumtoxinA therapy for the treatment of CM.
An inherent strength of this study is the extended duration of effect both for the HIT-6 and HRQoL measures. This is of particular interest clinically because it shows a consistent long-term benefit, despite the strict dosage and administration guidelines as per the PREEMPT paradigm. It would be of interest to better understand the influence of variations with regard to dosage or injection techniques with onabotulinumtoxinA use.
Conclusions
Taken together, these findings from the PREEMPT trials highlight the long-term efficacy of onabotulinumtoxinA for reducing headache impact and improving HRQoL.
Clinical implications
OnabotulinumtoxinA has rapid and sustained benefits on a long-term (>1 year) basis, reducing headache impact and improving health-related quality of life (HRQoL). During 36-week open-label treatment with onabotulinumtoxinA, patients who had previously received 24 weeks of double-blind treatment with onabotulinumtoxinA maintained HRQoL improvements (i.e. reduced headache impact and improved migraine-specific QoL) obtained during double-blind treatment. During the open-label onabotulinumtoxinA treatment period, patients who had received double-blind treatment with placebo ultimately attained HRQoL improvements similar to those who had received double-blind onabotulinumtoxinA. Differences in the onabotulinumtoxinA in double-blind phase and open-label phase (O/O group) versus the placebo in double-blind phase and onabotulinumtoxinA in open-label phase (P/O group) suggest increasing benefits with longer-term therapy.
Footnotes
Acknowledgment
ClinicalTrials.gov registration numbers: NCT00156910 and NCT00168428.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Richard B Lipton, MD, has received grant support from the National Institutes of Health, the National Headache Foundation, and the Migraine Research Fund. He serves as consultant, serves as an advisory board member, or has received honoraria from Alder, Allergan, American Headache Society, Autonomic Technologies, Boston Scientific, Bristol-Myers Squibb, CogniMed, CoLucid, Dr Reddy’s Laboratories, Eli Lilly, eNeura Therapeutics, Merck, Novartis, Pfizer, and Teva. Noah L Rosen, MD, FAHS, is on the Speaker’s Board for Allergan, has been on advisory boards for Avanir and Teva, and has performed consulting work for Curelator. Jessica Ailani, MD, is a speaker for Allergan and Teva and has served on an advisory board for Avanir. Ronald E DeGryse, MS, is a full-time employee of Allergan plc, and owns stock in the company. Patrick J Gillard, PharmD, MS, is a full-time employee of Allergan plc, and owns stock in the company. Sepideh F Varon, PhD, is a full-time employee of Allergan plc, and owns stock in the company.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Allergan plc (Dublin, Ireland). Writing and editorial assistance was provided to the authors by Amanda M Kelly, MPhil, MSHN, of Complete Healthcare Communications LLC (Chadds Ford, PA), Kristine W Schuler, MS, and Dana Franznick, PharmD, with funding by Allergan plc (Dublin, Ireland). All authors met the International Committee of Medical Journal Editors authorship criteria. Neither honoraria nor payments were made for authorship.
