Abstract
Background
Migraine shows a cyclic pattern with an inter-ictal-, a pre-ictal, an ictal- and a post-ictal phase. We aimed to examine changes in psychophysical parameters during the migraine cycle.
Methods
The perception of nociceptive and non-nociceptive stimuli and an electrically induced axon-reflex-erythema were assessed in 20 healthy controls and 14 migraine patients on five consecutive days according to different phases of the migraine cycle. Pain was rated three times during a 10-second electrical stimulus. The size of the axon-reflex-erythema was determined using laser-Doppler-imaging. Intensity and hedonic estimates of odours presented by Sniffin’ Sticks were rated.
Results
In healthy controls, no significant changes over the test days were observed. In migraine patients pain thresholds at the head decreased with an ictal minimum. Less habituation after five seconds of stimulation at the head was found pre-ictally, whereas reduced habituation to 10-second electrical stimulation was present in all phases. The axon-reflex-erythema size showed an inter-ictal-specific minimum at the head. odours were perceived ictally as more unpleasant and intense.
Conclusions
Somatosensory functions, pain thresholds and habituation as predominantly central parameters, axon-reflex-erythema as a peripheral function of trigeminal neurons and odour perception as a predominantly extra-thalamic sensation change specifically over the migraine cycle indicating complex variations of neuronal signal processing.
Keywords
Introduction
Migraine shows a cyclic variation pattern of different phases: a pain-free inter-ictal phase, a still pain-free pre-ictal phase with changes in perception and behaviour, an ictal phase with headache and autonomic symptoms and a post-ictal phase without headache but still some sequelae (1). Electrophysical and neuroimaging studies assessing differences in the experimental pain perception between migraine patients and healthy controls yielded contradictory results both in trigeminal and extra-trigeminal body regions (2 –13). Also, inconsistent olfactory alterations were described in the ictal versus inter-ictal phase ranging from hypersensitivity to microsmia and osmophobia (14,15). Since prospective investigations on migraine phases bear technical and logistical difficulties, only a few studies tackled temporal fluctuations over the whole migraine cycle. In one of these studies, less adaptation to heat pain was observed at the pre-ictal test session compared to the inter-ictal phase (16). This finding correlated with our previous psychophysical pilot study, in which habituation to a five-second lasting suprathreshold electrical stimulus in the trigeminally innervated region was found to be reduced solely in the pre-ictal phase (17). It has been hypothesised that during the pre-ictal phase the impact of the inhibitory control systems in the brain is reduced, so that based on a pre-existing vulnerability of the migraine brain external stimuli are more effective in triggering an attack (17). In both studies, patients’ examination times were retrospectively classified into different migraine phases and thus examinations were not necessarily performed within the same migraine cycle. Schulte and May (18) tested migraine patients for at least 30 consecutive days prospectively performing phase-specific measurements temporally related to spontaneous, untreated migraine attacks (19). As a result of painful trigeminal stimulation they confirmed increased ictal pain perception intensity, but more interestingly, they found an activation of the hypothalamic region within the last 48 hours prior to the headache attack (19). With respect to this new findings it was assumed that the pre-ictal phase starts earlier than 24 h prior to the headache (1).
In the present study, we examined temporal changes in different subtests for assessing trigeminal, extra-trigeminal, thalamic and extra-thalamic sensory signalling pathways. We selected one test for objective peripheral nerve fibre function (axon-reflex-erythema) and one test for central nervous function in form of habituation of nociceptive input. Both tests were performed in a trigeminally innervated region and at the arm as a spinally innervated area. For probing extra-thalamic pathways, we chose an olfactory test. We focused on performing phase-specific, temporal-related measurements on five consecutive days to assess changes in pre-ictal habituation more precisely.
Methods
Participants
Twenty-two migraine patients without any neurological disorders, diagnosed headaches or other forms of chronic pain were included after an interview and passed through the regular sequence of five test sessions. However, only in 14 patients did the different phases of the migraine cycle take place in the planned order. Thus we present here the data of 14 migraine patients with episodic migraine (female, aged between 19 and 55 years, mean age 26.1 ± 2.6 years) and 20 healthy subjects (19 females, one male, aged between 18 and 32 years, mean age 23.7 ± 0.7 years). Inclusion criteria for control subjects were age >18 years and normal olfactory function as tested by the odour identification test at the first test session. Control volunteers were excluded if they suffered from any chronic diseases, chronic pain or headache disorder, if they took any acute medication except contraceptive, or if headache or another acute disease occurred during the test days.
The patients were recruited via an advertisement on local social media groups and the Pain Centre of the University Hospital of Erlangen. Diagnosis of migraine and exclusion criteria (comorbidities that could influence somatosensory sensation and psychophysical evaluation) were assessed by an experienced neurologist B. F. according to the criteria of the International Classification of Headache Disorders (20). Migraine patients were not allowed to take acute or prophylactic pain medication during the test sessions. All participants gave their written informed consent to participate in the examinations. The study was reviewed by the local ethics committee of the University of Erlangen-Nürnberg (Nr 246-14B) and all study procedures were in accordance to the Declaration of Helsinki.
Test protocol
Before the experimental sessions, all participants took part in a training session. The ability to smell was verified by an olfactory identification test. As a control, 20 healthy volunteers were tested on five consecutive days (Monday to Friday), whereby the first session was the training session. In the group of migraineurs, the test sessions were planned individually depending on the next expected migraine attack, and the training session took place before the regular test sequence consisting of the inter-ictal, pre-ictal, ictal and post-ictal test session. Migraineurs were tested during all four migraine phases within their individual migraine cycle, defined as follows:
Inter-ictal phase: period more than 24 h before the start and later than 24 h after the end of a headache attack. The testing took place within 24 h prior to the pre-ictal phase. Pre-ictal phase: period within 24 h before the beginning of a typical migraine attack (pain state). Ictal phase: period during painful migraine attack. Post-ictal phase: period after end of headache attack until 24 h after the end of a migraine attack (pain state).
On each day the test time was kept constant to either morning, noon or afternoon. We defined the pre-ictal phase as the period within 24 hours before headache started. At the time of our study, it was not yet published that the pre-ictal phase might start as early as 48-hours before headache (1). As seen in Figure 1, the second test session in healthy controls was used for comparison with the inter-ictal phase in migraineurs, the third session for comparison with the pre-ictal phase, the fourth session for comparison with the ictal phase and the fifth session for comparison with the post-ictal phase.
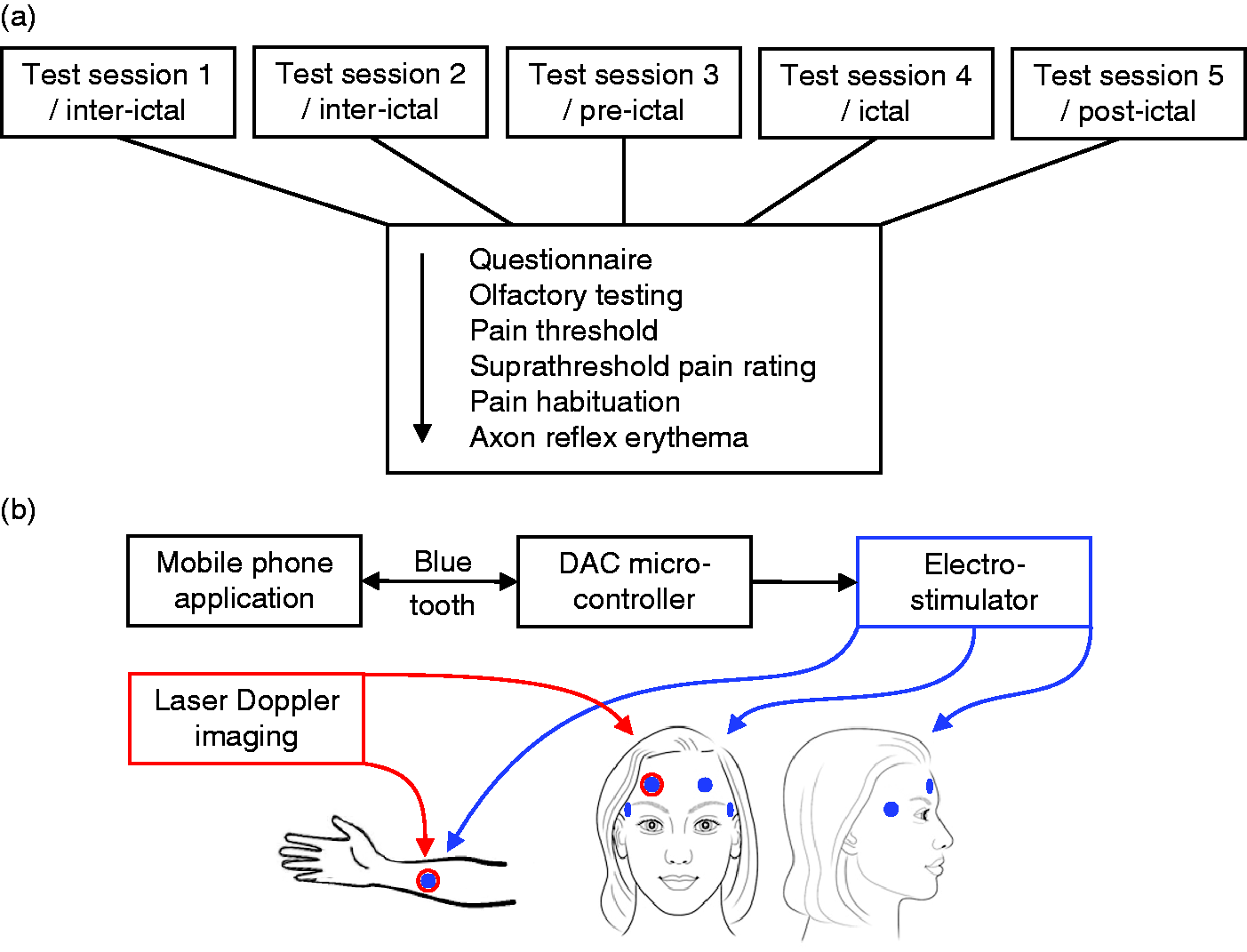
(a) Workflow of psychophysical experiments: Overview of the different test modalities for psychophysical experiment. Every test day started with filling a questionnaire, followed by the olfactory testing with Sniffin’ Sticks. After determining electrical pain threshold and suprathreshold pain rating for second one, five and 10, pain habituation was calculated by the change in pain rating in the first and the final five seconds of stimulation. Images of laser Doppler scans represent the size of the electrically induced axon-reflex-erythema. (b) Technical test equipment and test regions: Mobile phone application for electrical stimulation, connected via Bluetooth sensor to a microcontroller with a digital-analog-converter (DAC), which forms the communication to the electrostimulator. The DAC converts a digital input signal into a corresponding analog output voltage Uout. The stimulus intensity in mA is given by the formula Istimulus = Uout * (I/U), whereby I/U = 10mA/5V was set as required current-voltage ratio on the DS5 electrical stimulator.21 Stimuli were applied via different surface electrodes at the skin of the head (forehead and temple) or the inner side of the right forearm, laser Doppler scans were applied at the forehead.
Each test session started with filling in a questionnaire followed by the olfactory test, rating of the electrical stimulation and finally laser Doppler imaging (Figure 1a).
Anatomical test regions
Electrodes for electrical stimulation were placed on both sides of the forehead (ophthalmic branch) and both temples (overlap of ophthalmic and maxillary branch). The inner side of the right forearm served as a spinally innervated test area (Figure 1b).
Electrical stimulation
We used two different bipolar surface electrodes adapted to the respective needs for assessing electrical sensitivity (two punctiform stainless steel buttons, Ø 0.2 mm, distance 2,5mm) and for axon-reflex erythema (two stainless steel wire loops, Ø 0.1 mm, distance 1.5 mm). Electrical pain thresholds were determined using a constant current stimulator (DS5, Digitimer Ltd, UK), which was controlled by a mobile phone application via Bluetooth connection operated by the subjects themselves (Figure 1a) (21). The pain threshold was determined by repetitive stimuli at 2 Hz, whereby the current intensity (mA) increased continuously. The mean of three trials was used to calculate the pain threshold. This threshold intensity was then applied using 100 Hz stimulation resulting in higher pain ratings due to the higher frequency. Thus stimulation at the individual pain threshold intensity with a frequency of 100 Hz for 10 seconds served as a suprathreshold test stimulus.
Pain was rated by subjects on a numerical rating scale from 0 (no pain) to 10 (strongest imaginable pain) at the beginning (at 1 second), middle (at 5 seconds) and end (at 10 seconds) of stimulation. Ratings were normalised to the mean pain ratings of all trials on all five test days of one individual volunteer.
Laser Doppler imaging
We used a laser Doppler imager (LDI, Moor Instruments Ltd, UK) to analyse the size of the electrically induced axon-reflex-erythema (17). Scans (20 seconds, area 3x3 cm) were taken before and after the electrical test stimulation. The first baseline scan was subtracted from all other scans. The size of erythema was calculated offline by the total number of pixels (0,005 mm2 per pixel), in which flux values exceeded the mean flux of the baseline image by two standard deviations (17). The area of the erythema was standardised with reference to the daily frontal suprathreshold stimulation intensity of each subject (Unit: cm2/mA).
Hedonic and intensity estimates of odours
For olfactory experiments we used Sniffin’ Sticks (Burghart Messtechnik GmbH, Holm, Germany). All subjects performed an identification test with 16 odours, from which at least 12 had to be identified correctly to include the subjects in the study. For olfactory hedonic testing, the participants were exposed to 12 odours (peppermint, garlic, fish, banana, apple, n-butyric acid, pineapple, iso-butyric acid, peach, indole, vanilla/caramel, valeric acid). Odours were rated verbally on a numerical rating scale based on hedonic values (−4 most unpleasant, 0 neutral, +4 most pleasant) and intensity (between 0 and 10) (22).
Questionnaires
At the beginning of each test session, a questionnaire was filled by all participants stating their current health condition and possibly migraine preceding or accompanying symptoms (MPA symptoms) over the migraine cycle (23). In the following we use the term ‘MPA symptoms’ for all symptoms and conditions that migraine patients are usually asked about as occurring in the time before the headache starts and are usually seen as ‘prodromal’. The questionnaire was used to document the end of the migraine attack retrospectively.
Data analysis and statistical analysis
All data was analysed using Statistica 6.0 software (StatSoft, Tulsa, USA). Normality of data was tested by the Shapiro-Wilks-test and normally distributed values were evaluated by analysis of variance (ANOVA). Differences between the subject groups or between stimulation regions were tested by one-way ANOVA, between the different test days by repeated-measures ANOVA. For analyses of interactions, we used two- or three-way repeated-measures ANOVA, for post-hoc testing the Fisher’s least square difference (LSD) test. For analysis of MPA symptoms, we used a non-parametric chi-square test with significant differences at a p value of less than 5%.
Significant differences were assumed at a p value of less than 5%. Normally distributed values are presented as mean ± standard error of the mean (SEM), other data as median ± quartiles.
Data for electrical pain threshold were z-normalised (z = [test value – mean value of healthy controls]/standard deviation of healthy controls). Data for pain ratings were normalised to the mean of all pain rating data across all test sessions for each subject and given as percentage value.
Results
Participants
Twenty-two migraine patients passed through the regular sequence of five test sessions.
In 14 patients the different phases of the migraine cycle took place in the planned order: inter-ictal, pre-ictal, ictal and post-ictal phase. For details about migraine characteristics see Table 1.
Clinical features in migraine patients and healthy controls. Epidemical data of healthy controls and migraine patients as well as clinical information of migraine attacks in patients based on anamnesis.

*localization of headache during experiment, r = right head side, l = left head side.
**symptoms before or during headache attack.
***Headache attacks manifesting before or during menstrual bleeding or in pill-free interval under oral contraceptive.
Electrical pain thresholds are decreased in migraine patients only on the headache day
For further analysis, the electrical pain threshold of the right and left head side were averaged, as there were no significant differences between right and left side (i.e., in migraine patients headache side versus non-headache side) through the four test sessions.
We found a significant reduction of the electrical threshold in the ictal phase compared to the inter-ictal phase in migraineurs (ANOVA: F(1,32) = 8.698, p = 0.006; LSD, migraine, inter vs. ictal: temple p = 0.004; forehead p < 0.001; Figure 2). In the post-ictal phase the thresholds were also higher than in the ictal phase (ANOVA: F(1,32) = 5.44, p = 0.027; LSD, ictal vs. post-ictal: temple p < 0.0001; forehead p = 0.0001) but no difference was observed between pre-ictal and ictal phase (ANOVA: F(1,32) = 3.54, p = 0.069).

Electrical pain threshold. Graphs show the z-normalized electrical thresholds at the temple and arm for migraine patients (■) and healthy controls (○). The values are given as mean +/− standard error of mean, while the range between +2 and −2 represents the values of 95% of the healthy control population. In terms of QST evaluation, positive values indicate a gain of sensory function (sensitisation) and negative values indicate a loss of sensory function (desensitisation). In this case, higher pain thresholds in migraine patients in inter-ictal phase in comparison to healthy controls mean less sensitivity to electric stimuli, reflected by values under zero. *p < 0.05.
At the arm, no significant difference in pain threshold between migraineurs and healthy controls was found when all test days were included (ANOVA: F(1,32) = 0.43, p = 0.52). Migraine patients showed significantly reduced pain thresholds also at the arm in the ictal phase in comparison to the inter-ictal phase (ANOVA: F(3,39) = 4.34, p = 0.01; LSD, p = 0.008, Figure 2).
Habituation at the head changes with the migraine phases
Consistent with our previous study (17), changes in habituation at shorter duration of stimulation were observed to be specific for the pre-ictal phase of the migraine cycle. At the temple, less habituation was found in migraine patients within the first five seconds of stimulation at the pre-ictal test day in comparison to the inter-ictal and ictal phase (ANOVA: F(3,39) = 4.02, p = 0.014, LSD, pre-ictal vs. inter-ictal: p = 0.009; pre-ictal vs. ictal: p = 0.01, Figure 3a). This is reflected by significant higher pain ratings at second 10 in migraine patients compared to healthy controls (ANOVA: F(1,32) = 7.24, p = 0.011; see online supplementary material).

Habituation to electrical painful stimulus. Pain habituation in healthy controls (grey) in contrast to the migraine group (black) during the first (a) and last (b) five seconds of electrical stimulation at the temple and the arm in different phases of the migraine cycle/test sessions. Participants rated pain at the beginning, the middle and the end of a 10-second stimulus. (a) Pain habituation in the first five seconds was calculated by subtracting pain scores for second five and second one and multiplying this result with −1. This change in rating is given as column representing the mean “decrease” from second one to second five in % ± standard error of mean. *p < 0.05 (b) For calculating habituation in the last five seconds, pain scores for second 10 and second five were subtracted and the result was multiplied with −1. *p < 0.05.
In the last five seconds of stimulation, migraine patients showed significantly less pain habituation than healthy controls independently of the migraine phase (ANOVA, temple: F(1,32) = 4.842, p = 0.035 Figure 3b). At the forehead habituation was no different between migraine and control group.
At the arm, no significant difference in habituation either between the migraine group and healthy controls nor between the different phases in the migraine group were observed for the first or the last five seconds of stimulation (Figure 3a, b).
Pain rating of electrical stimuli
A suprathreshold electrical stimulus at 100 Hz was used to test for habituation. All participants rated painfulness at second one, five and 10.
At the forehead, pain ratings were not significantly different between healthy controls and migraine patients and did not change significantly over the migraine cycle. At the temple, pain ratings were significantly higher in migraine patients compared to healthy controls (ANOVA: F(1,32) = 4.429, p = 0.0433) but no significant difference was found at the frontal test region (data not shown). More specifically, pain ratings at second 10 were significantly higher in migraine patients compared to healthy controls (ANOVA: F(1,32) = 7.24, p = 0.011; Figure 4a), in contrast to pain ratings at second one and second five. The difference at second 10 indicates a lack of habituation in migraine patients at the temple.

Axon-reflex-erythema. (a) Example of a laser Doppler image in a healthy control and a migraine patient after electrical stimulation. The intensity of electrically induced increased blood flow in the superficial skin blood vessels is represented by different colours. Migraine patients showed larger areas and more pronounced erythema compared to healthy controls. (b) Size of axon-reflex-erythema at the forehead (left) and the forearm (right) in migraine patients (black) and healthy controls (grey) at different phases of migraine cycle/test sessions. *p < 0.05.
At the arm, pain ratings were significantly higher in healthy controls compared to migraine patients (ANOVA: F(1,32) = 8.155, p = 0.007). In detail, significant differences between the test groups were found at the arm at second one, second 5, but not at second 10 (ANOVA, second one: F(1,32) = 10.36, p = 0.003; second five: F(1,32) = 4.62, p = 0.037; second 10: F(1,32) = 2.26, p = 0.14, Figure 4b).
Axon-reflex-erythema is larger in the pre-ictal test session only at the head
The electrically induced axon-reflex-erythema (Figure 4a) was recorded only at the forehead because laser Doppler scanning can only reliably measure changes in blood flow in planar skin.
Migraine patients showed phase-dependent changes in the erythema size in comparison to healthy controls (ANOVA: F(3,96) = 4.74, p = 0.004): The axon-reflex-erythema was significantly larger pre-ictally (LSD p = 0.028), ictally (LSD p = 0.016) and post-ictally (LSD p = 0.023) compared to the inter-ictal phase (Figure 4b). At the forehead, flare reactions were significantly larger than at the forearm (ANOVA: F(1,32) = 43.1, p < 0.0001) and appeared more intense compared to the forearm (not analysed quantitatively).
At the arm, we found no significant differences either between the phases/test session nor between the groups in general.
Changes in olfactory sensitivity in the ictal migraine phase
Ratings for all odours were pooled for analyses. There was no significant difference in hedonic or intensity ratings between the migraine group and healthy controls on average.
However, the olfactory perception followed different time courses in healthy controls and patients (ANOVA: F(3,96) = 3.35, p = 0.022): The unpleasantness score to olfactory stimulation habituated only in healthy controls, while migraineurs did not habituate. On the contrary, unpleasantness increased with a maximum at the ictal test session in migraine patients (LSD, ictal vs. inter-ictal: p = 0.008, vs. pre-ictal: p = 0.033; vs. post-ictal: p = 0.03, Figure 5a).

Olfactory testing. (a) Mean hedonic estimates (−4 to +4) in migraine group (black) and healthy controls (grey), whereas estimates of all odours were pooled. *p < 0.05. (b) Intensity ratings (0–10) were pooled in the same way for further analysis. *p < 0.05.
Also, intensity ratings followed cyclic changes during the migraine phase in contrast to the ratings in healthy controls during the test days (ANOVA: F(3,96) = 2.712, p = 0.049; Figure 5b): in the ictal test session migraine patients experienced odours more intensely than in the other migraine phases (LSD, ictal vs. inter-ictal: p = 0.009, vs. pre-ictal: p = 0.04, vs. post-ictal: p = 0.002).
Cumulative symptoms correspond to a developing migraine attack
Results of the questionnaires show a continuous accumulation of MPA symptoms, which are usually regarded as ‘prodromal’ symptoms preceding a migraine attack (Table 2). In the inter-ictal test session (session two in healthy controls) we found no difference in the number of MPA symptoms between migraine patients and healthy controls. In the pre-ictal test session, migraine patients reported significantly more MPA symptoms compared to healthy controls (X2(1) = 19.21, p < 0.0001, Figure 2). Migraine patients in ictal test sessions showed significantly more “MPA symptoms” compared to the pre-ictal phase (X2(1) = 12.64, p = 0.0004).
Overview of symptoms pronounced in migraine patients. Symptoms according to the different phases of migraine cycle. Five of 14 migraine patients did not recognise any prodromal symptom in the last 24 hours before the attack, while at ictal test session all patients showed at least one of the mentioned symptoms.

Discussion
In this study, migraine patients showed phase-dependent changes in different parameters assessing trigeminal, extra-trigeminal, thalamic and extra-thalamic sensory signalling pathways during the migraine cycle. Our results underline the hypothesis that migraine is based on fluctuating neurological mechanisms involving different inhibitory and excitatory structures of the central and peripheral pain modulation systems.
Only a few studies focused on performing phase-specific measurements during one individual migraine cycle including a migraine attack, because this is logistically extremely challenging. In the present prospective study, we achieved a high success rate of predicting a migraine attack, which was based on the strong personal commitment of the experimenter being flexible and available at any time, seven days a week. It was also helpful that most of the patients were students and thus also available most of the time. Furthermore, the high rate of menstrual migraine in our cohort provided particular predictivity. Using this difficult approach, we observed decreasing electrical pain threshold towards the migraine attack (pre-ictal, ictal) both for trigeminal and extra-trigeminal stimulation. This is in line with recent results for mechanical pain, where the study design and cohort size are comparable to the present study (24). While significantly lower thresholds in migraineurs in comparison to healthy controls were described in all phases of the migraine cycle (24), we found no significant differences between migraineurs and healthy controls, which corresponds to a metanalysis of electrophysical studies in recent years (25). However, we saw the tendency for migraineurs to have inter-ictally higher thresholds than healthy controls, decreasing towards the attack and approaching the level of healthy controls on the headache day, similar to another study with mechanically evoked punctate pain (26). Due to sufficient habituation mechanisms in the pain-free period and due to the repeated experience of extreme pain with a similar character during migraine attacks, migraineurs might experience experimental pain in the interictal phase as less intensely than healthy subjects who are rarely confronted with painful stimuli. When habituation gets reduced during the migraine cycle towards the ictal phase, as shown by our other results, the pain thresholds of migraineurs and healthy volunteers become more similar, as seen in the convergence of pain threshold towards z = 0. Nevertheless, the contradictory results to our previous study (17) and to other studies (10,12,27) show that pain sensitivity depends not only on the phases but also on the frequency of attacks (26), the study design and the temporal definition of migraine phases (1).
However, lack of habituation seems to be impaired early in the migraine cycle, as we found reduced habituation to a five-second lasting stimulus exclusively in the pre-ictal phase, similar to our previous study confirming this observation (17). Alterations in habituation to repeated stimuli are already described for evoked potentials of various modalities (28 –31), but most of the studies focused on comparison between the inter-ictal and the ictal phase. Since phenomena of habituation indicate a certain ‘filter function’ for incoming sensory inputs (32 –35), deficits in the immediate hours before the attack could promote the generation of pain. Alternatively, this might indicate that the ictal phase already starts some hours before the actual pain starts. It has been hypothesised that lack of habituation is the result either of a hyper-excitability of excitatory neurons or hypo-excitability of inhibitory interneurons (35). This could lead to vulnerability of the filter function in the pre-ictal phase, followed by pain-triggered counter-regulatory mechanisms in the ictal phase and an extensive regeneration of habituation in order to limit the duration and intensity of the headache. This explanation fits to results of a metanalysis from Copolla et al. (36), which shows that habituation continuously decreases the closer the approaching migraine attack is, reaching a maximum in the period directly before the headache (i.e., pre-ictal) and then normalises during the headache. An increased beta event-related desynchronisation (beta-ERD) response, a higher baseline ERD, as well as enhanced cortical responsivity after visual stimulation were found in the pre-ictal phase, which could be interpreted as increased excitability of the sensorimotor cortex compared to the inter-ictal phase (37,38). These findings are in line with the hypothesis of thalamocortical dysrhythmia (39), a functional disconnection of the thalamus leading to decreased intracortical lateral inhibition (36). By continuous functional magnetic resonance imaging (fMRI) scanning of migraine patients over 30 days, it was shown that immediately before the headache attack the hypothalamus was significantly more active and more intensely coupled with the activation of the spinal trigeminal nuclei (18,19).
Interestingly, the longer the painful electrical stimuli were applied the more migraine patients had difficulties habituating to it, independently of the migraine phase. In other studies in the inter-ictal period lack of habituation and even facilitation to longer-lasting stimuli was observed in migraine patients in contrast to healthy controls (8,25). This might be due to increased temporal summation in migraine patients in general, indicating that sensitisation is associated with activation-dependent plasticity of neural structures, independently of migraine phases or trigeminally innervated structures (10). We speculate that habituation differences between the forehead and the temple might be influenced by both variances in innervation and also skin structure that influence the function of afferent fibres. The skin at the forehead resembles thick glabrous skin whereas the temple has hairy skin. In glabrous skin less tactile C-fibres (C-touch fibres) are found, which might be involved in endogenous pain reduction (40 –42). Also, the differential proportion of the three divisions of trigeminal nerve might be differentially involved in pain modulation (32).
Olfactory perception is changed in the ictal phase
Olfactory perception is the only sensory modality that largely bypasses the thalamus and follows different processing pathways than trigeminal pain (43). An fMRI study showed that in migraine patients olfactory sensitivity did not change in the migraine cycle while habituation to painful trigeminal stimuli was impaired (44). This supports the idea that the pre-ictal phase is specifically associated with thalamic activation, which is not observed for olfactory stimulation. Activation of the amygdala could be responsible for the increased ictal intensity ratings in migraineurs (15,45). In physiological conditions, coupling between the amygdala and the superior frontal gyrus seemed to be involved in integrating odour perception and pain experience (46). Other brain regions like the temporal pole are more activated during odour stimulation in migraine patients with olfactory hypersensitivity, while typical regions for pain perception like the cingulate gyrus or, for inhibitory pain control, the locus coeruleus show lower activation at the same time in comparison to healthy controls (47). This odour-triggered deactivation could be seen as an abnormal brain function related to the migraine disease or as top-down regulation (47), so that other, potentially incoming noxious events, are less likely to cause symptoms. While others found a general unpleasantness towards odours only in patients with olfactory hypersensitivity (48), we found this phenomenon in our patients in the ictal phase.
Efferent function of primary afferents is altered in migraine patients
Recently Calcitonin gene related peptide (CGRP) antagonists and CGRP binding antibodies came into the focus as migraine prophylactic treatment. The axon-reflex-erythema in the skin is thought to be induced by CGRP, which is released from the endings of electrically activated nociceptors. Although it is not fully understood whether a larger axon-reflex-response is a result of a higher basic concentration of CGRP, a stronger CGRP secretion or an increased receptor sensitivity, the increased flare size may point towards migraine-specific changes of the primary afferents. Alternatively, the responsiveness of the arteries to CGRP may change subserving an increased flare reaction.
Since at the arm the axon-reflex erythema was no different between healthy and migraine patients and did not vary over the migraine cycle, we conclude that peripheral changes in migraine patients are restricted to the trigeminally innervated area.
Conclusions
Temporal plasticity and involvement of specific systems in the migraine cycle are reflected by different cyclic changes of various sensory modalities (see Figure 6). Reduced habituation towards thalamic-cortical processed modalities in the pre-ictal phase might play the key role for triggering the migraine attack (44). The impaired sensory filter function might result in a widespread enhanced sensitivity in the following ictal phase, which could influence different simultaneously incoming sensory inputs due to the activation of various subcortical brain regions (49,50). This could be the reason why non-thalamic sensory modalities like olfaction show alterations only in the ictal phase, because atypical functional connections are extremely activated in this period (49). Alterations in axon-reflex-flare size at the head suggest cyclic changes in the function of trigeminal primary afferents in contrast to spinal afferents within the migraine cycle, but more prospective studies with higher cohort sizes are needed to dissect peripheral mechanism in the migraine pathophysiology.
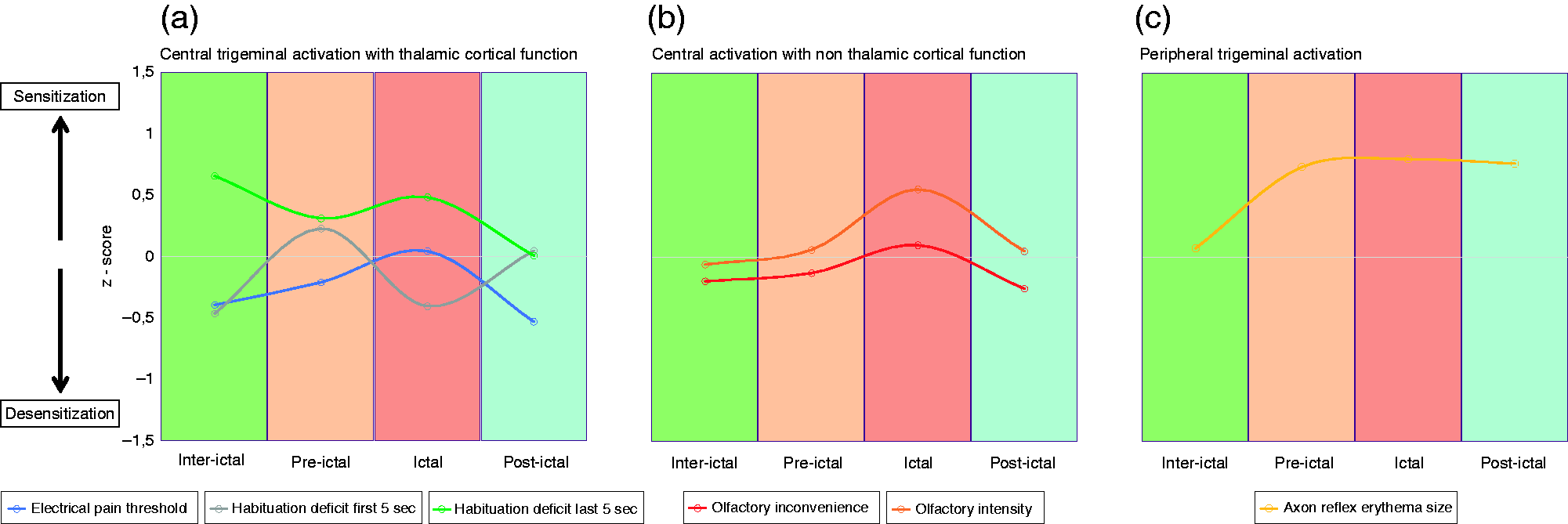
Time model for cyclic changes of psychophysical parameters at the head. Temporal changes of different parameters tested over the course of the migraine cycle. The different sensory signalling pathways are represented by three different graphs (a-c), whereby each curve shows one parameter. To compare all parameters with different units, z-normalisation of the raw data was applied for each parameter, using the values of the healthy controls over all test days as control group. A movement upwards (more than 0) means sensitisation, a movement downwards (less than 0) means desensitisation. (a) Central trigeminal activation with thalamic – cortical activation: The graphs represent the alteration of electrical sensitivity over the migraine cycle. While habituation to a short pulse train is reduced only pre-ictally (movement upwards), habituation to a longer lasting stimulus is generally impaired and pain threshold is decreased at the ictal day. (b) Central activation with non-thalamic cortical activation: Olfaction show sensitisation only in die ictal phase. (c) Peripheral trigeminal activation: Trigeminal electrical stimulation leads to higher peripheral sensitivity in the pre-ictal, ictal and post-ictal phase, represented by an enlarged axon-reflex-erythemata.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024221097932 - Supplemental material for Cyclic changes of sensory parameters in migraine patients
Supplemental material, sj-pdf-1-cep-10.1177_03331024221097932 for Cyclic changes of sensory parameters in migraine patients by Carolin Helfenstein, Marion Strupf, Andrea Stefke, Britta Fraunberger, Bertold Renner, Insa Suchantke, Markus Rothermel, Karl Messlinger, Roberto DeCol and Barbara Namer in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_03331024221097932 - Supplemental material for Cyclic changes of sensory parameters in migraine patients
Supplemental material, sj-pdf-2-cep-10.1177_03331024221097932 for Cyclic changes of sensory parameters in migraine patients by Carolin Helfenstein, Marion Strupf, Andrea Stefke, Britta Fraunberger, Bertold Renner, Insa Suchantke, Markus Rothermel, Karl Messlinger, Roberto DeCol and Barbara Namer in Cephalalgia
Supplemental Material
sj-pdf-3-cep-10.1177_03331024221097932 - Supplemental material for Cyclic changes of sensory parameters in migraine patients
Supplemental material, sj-pdf-3-cep-10.1177_03331024221097932 for Cyclic changes of sensory parameters in migraine patients by Carolin Helfenstein, Marion Strupf, Andrea Stefke, Britta Fraunberger, Bertold Renner, Insa Suchantke, Markus Rothermel, Karl Messlinger, Roberto DeCol and Barbara Namer in Cephalalgia
Supplemental Material
sj-pdf-4-cep-10.1177_03331024221097932 - Supplemental material for Cyclic changes of sensory parameters in migraine patients
Supplemental material, sj-pdf-4-cep-10.1177_03331024221097932 for Cyclic changes of sensory parameters in migraine patients by Carolin Helfenstein, Marion Strupf, Andrea Stefke, Britta Fraunberger, Bertold Renner, Insa Suchantke, Markus Rothermel, Karl Messlinger, Roberto DeCol and Barbara Namer in Cephalalgia
Supplemental Material
sj-pdf-5-cep-10.1177_03331024221097932 - Supplemental material for Cyclic changes of sensory parameters in migraine patients
Supplemental material, sj-pdf-5-cep-10.1177_03331024221097932 for Cyclic changes of sensory parameters in migraine patients by Carolin Helfenstein, Marion Strupf, Andrea Stefke, Britta Fraunberger, Bertold Renner, Insa Suchantke, Markus Rothermel, Karl Messlinger, Roberto DeCol and Barbara Namer in Cephalalgia
Footnotes
Key findings
Patients have reduced electrical pain thresholds and increased olfactory sensitivity in the ictal phase. Migraineurs show lacking habituation towards the first five seconds of a 10-second lasting stimulus in the pre-ictal phase and a phase-independent dysfunction in habituation towards the last five seconds. Migraineurs have larger axon-reflex-erythemata in all migraine phases (except inter-ictally) only in trigeminal areas.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The current work was performed in fulfilment of the requirements for obtaining the degree Dr. med. for CH at the Friedrich-Alexander-University Erlangen-Nürnberg.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was supported by the Dr. Robert Pfleger Stiftung, BN was supported by a grant from the Interdisciplinary Center for Clinical Research within the faculty of Medicine at the RWTH Aachen University and by the DFG grant NA 970 3-1, MR by the Deutsche Forschungsgemeinschaft RO4046/2-1/2-2 and/5-1, the Research Training Group 2416 (“MultiSenses – MultiScales: Novel approaches to decipher neural processing in multisensory integration”; 368482240/GRK2416), and the Interdisciplinary Center for Clinical Research within the Faculty of Medicine at the RWTH Aachen University, Grant IZKF TN1-7532007.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
