Abstract
Introduction
Migraine is characterized by cycling phases (interictal, preictal, ictal and postictal) with differing symptoms, while in chronic tension type headache pain phases are fluctuating. The question we asked is whether these phases are associated with changes in parameters of somatosensation and axon-reflex erythema.
Methods
Patients with episodic migraine and chronic tension type headache were examined psychophysically in the interictal, preictal and ictal phase and healthy subjects on five different test days. Thresholds and suprathreshold ratings of pressure and electrical pain were assessed on three different regions of the head. In migraine patients and in healthy controls, electrically induced axon-reflex erythema was measured in the area of the first trigeminal branch. All migraine patients filled out questionnaires about prodromal symptoms at every visit.
Results
The axon-reflex erythema was always larger in patients with migraine in contrast to healthy subjects. The pressure pain threshold was lower in migraine patients and chronic tension type headache in comparison to healthy subjects. Electrical pain thresholds did not differ between headache patients and healthy subjects and showed no changes between the phases. However, suprathreshold pain ratings showed less habituation solely in the preictal phase of migraine. The number of prodromal symptoms in migraine patients was increased in the preictal and ictal phase.
Discussion
Reduced habituation was the unique sign of the preictal phase in migraine patients, independently of prodromal symptoms, whereas a larger axon-reflex erythema and higher pressure pain sensitivity are constitutional and non-phase dependent properties of migraine. Reduced inhibitory mechanisms in the preictal phase may contribute to trigger headache attacks in migraine.
Keywords
Introduction
In migraine, different phases are characteristic: (a) The interictal phase without headache or other symptoms; (b) the preictal phase, in which various prodromal neuropsychological symptoms like craving and fatigue occur; (c) the ictal phase, with headache accompanied by vegetative symptoms like nausea and vomiting, sensory hypersensitivity to light and sound, allodynia or neck pain, and (d) the postictal phase, in which neuropsychological symptoms similar to the preictal phase may occur (1). An aura with transient sensory dysfunctions such as scintillating scotomata usually precedes the ictal phase but can also occur during, or after, the ictal phase. In line with that, altered processing of external stimuli, recruitment of neuronal circuits and sensitization of central neurons are suggested to underlie the migraine symptoms including headache generation (2). Based on experimental and clinical studies, a temporary hyperexcitability of central nociceptive neurons caused by a deficient endogenous modulatory antinociceptive system has been suggested to be involved in migraine generation (3–6).
It has long been assumed that intracranial structures like the cranial dura mater and cerebral blood vessels are involved in nociceptive processes underlying the generation of primary headaches (7); however, since that time little progress has been achieved in finding intracranial variations that might be associated with migraine or chronic tension type haedache (CTTH). In addition to extracranial structures like the temporal artery (8), we know that afferent neuronal connections exist between intra- and pericranial tissues, suggesting a substantial influence of extracranial meningeal afferent projections on meningeal nociception and headache generation (9). Accordingly, several studies demonstrated changes in extracranial sensitivity in migraine patients, while others did not find any differences (10–12). This might be due to different stimulation and test methods, time of measurements or differing head regions that were examined. Due to technical and logistical reasons, tests were performed mostly within the interictal phase.
In the current prospective study, we compared migraine and CTTH patients with healthy volunteers. In migraine patients, we selected different time points within the migraine cycle; in CTTH patients, we compared phases of no or minor headache with phases of intense headache and phases before intense headache and probed different areas of the head surface for stimulation with two modalities. Our aim was to find out if there are changes in pain thresholds and habituation between these groups and in a temporal association with migraine attacks and CTTH fluctuations. As an objective measure of the function of primary afferents, we used in addition electrically induced axon-reflex erythema.
Methods
Participants
Thirty-three healthy controls (nine males and 24 females, aged between 19 and 56 years, median 24 years) without a history of chronic pain, headache or neurologic disorders, 21 migraine patients (one male, 20 females, between 20 and 56 years, median 30 years) as well as seven patients suffering from chronic tension type headache (CTTH) (six females, one male, between 19 and 46 years, median 28 years) with at least 15 headache days per month took part in the study. Headache patients were recruited via advertising in the university newsletter and on local Facebook groups and from the pain clinic at the University hospital. Healthy controls were recruited via advertising on Facebook groups and from the university staff. Migraine with or without aura and CTTH were diagnosed according to the International Classification of Headache Disorders, third edition (ICHD-3 beta 2013) by an experienced neurologist and headache therapist (13). The headache patients did not suffer from any other disorder that could have influenced somatosensory sensation or psychophysical compliance. Migraine patients were allowed to take antiemetics but no prophylactic medication or acute medication against headache. All participants gave their written informed consent prior to the experiments. The study was approved by the local ethics committee of the University of Erlangen-Nürnberg and conformed to the declaration of Helsinki.
Test protocol
All participants took part in a training session, in which they became accustomed to the stimuli, the experimental procedures, and the use of the rating scale. For individual participants, the test time within the day was kept constant with a maximal variance of 2 hours. All healthy volunteers came in for their test sessions on five different days with an interval of 1–2 days (Figure 1(a)). Test session 2 in healthy volunteers was used for comparison with data from the interictal phase in migraine patients, test session 3 in healthy volunteers was used for comparison with the preictal phase in migraine patients and test session 4 in healthy volunteers was used for comparison with the ictal phase of migraine patients. For the headache patients, an individual schedule was designed to increase the probability that one test session was within 24 hours before the headache day (preictal) and one session during headache (ictal). For migraine patients, the first two sessions were planned in the interictal phase at least 26 hours after or before their headache attack (Figure 1(a)).
Scheme of the experimental protocol and data analyses for psychophysical testing. (a) After a training session, psychophysical testing was performed at five days with threshold testing and rating of suprathreshold stimuli and assessing axon-reflex erythema. The schedule was individualized so that headache occurred ideally at the fourth test session. However, if headache occurred at test session 2, 3 or 5, the values were accepted and sorted to the respective migraine phase. In healthy volunteers, the testing was repeated on five days (days 1 to 5). (b) Pressure pain and electrical thresholds were assessed with the method of limits, and the values were z-normalised. (c) Suprathreshold stimulation was performed with electrical stimuli with 100 Hz for 5 s. All participants had to rate the beginning (black column) and end (grey column) of the 5 s stimulus. These two values were then subtracted. When the end of stimulation was rated higher, a positive value results and indicates sensitization, whereas a negative value indicates habituation to the stimulus. (d) Test regions were on both sides of the head in the frontal, temporal and occipital regions (marked with grey squares).
In CTTH patients, who mostly showed fluctuating pain states, it was often difficult to define a pain-free interval and particularly a postictal phase; therefore, only patients who showed an interval of at least 52 hours with virtually no, or significantly less pain than during the ictal phase, were included. If the session took place within 26 hours before the headache started, or was clearly aggravated, the test values were regarded as preictal data. If more than 26 hours had passed before headache started or was clearly aggravated, the values were regarded as interictal data. Postictal data were abandoned due to an insufficient number of clear cases. If within 24 hours of the first test session headache occurred in migraine patients or CTTH patients, the data were excluded and, if possible, a new test session was started. When headache occurred at test session 2, 3, 4 or 5, the data were accepted and, in the case of headache at the second test session, the interictal values were taken from a test session after the postictal phase.
Anatomical test regions at the head
The test regions at the head (pressure, electrical stimulation) were located as follows: One at the insertion of the upper neck muscles within the innervation territory of the greater occipital nerve; another at the insertion of the temporal muscle (overlapping area of the maxillary and mandibular division of the trigeminal nerve); and a third at the forehead within the innervation territory of the ophthalmic division of the trigeminal nerve (Figure 1(d)).
Pressure
Pressure pain thresholds (PPT) were tested using an electronic algometer (Somedic Algometer, Bo Johansson Somedic Sales AB®, Sweden) with a circular rubber probe area of 1 cm2 (probe diameter of 1.1 cm). In each subject, PPT was determined with a series of three ramp stimuli (30 s in between) with a slope of 50 kPa/s. Subjects were instructed to press the stop button of the algometer as soon as the pressure sensation changed to the sensation of pain. PPT was calculated as the mean of three trials. Values were z-normalized (see data analysis and Figure 1(b)). Suprathreshold pressure pain was assessed by applying a pressure of PPT + 10% for 5 s and the subjects were asked to estimate the painfulness (pain rating) of the pressure on an 11-point numeric analogue scale (NAS: 0 = no pain; 1 = minimal sensation of pain, i.e. pain threshold; 10 = maximum imaginable pain; see data analysis and Figure 1(b)).
Electrical stimulation
Electrical sensitivity was tested using a bipolar surface electrode. The electrode consisted of two stainless steel wire loops (Ø 0.1 mm) (distance 1.5 mm). The surface electrode was fixed to the subject’s head at the test site using adhesive tape (Peha-haft® 6 cm × 20 m, Paul Hartmann AG, Heidenheim, Germany). To determine the electrical pain threshold, repetitive stimuli at 2 Hz (triggered by UPG 200, ELV Electronic AG, Leer, Germany) were applied using a constant current stimulator (DS7A, Digitimer Ltd, Welwyn Garden City, UK). Stimulation intensity (mA) was increased until the subject announced verbally that the stimuli evoked a painful sensation. The threshold was determined as the mean threshold of three measurements. Suprathreshold stimulation was applied at threshold intensity with a frequency of 100 Hz for five seconds. It was rated at the beginning (first second) and at the end (fifth second) of the stimulus on the NAS between 0 and 10 as used for assessing pressure pain. The difference between the second pain rating and the first pain rating denoted either increased pain (positive values) or decreased pain (negative values) (see Figure 1(c)).
Laser doppler imaging
To visualize the electrically induced flare reaction of the skin at the right frontal side of the head in migraineurs in comparison to healthy volunteers, a Laser Doppler Imager (LDI, Moor Instruments Ltd, London, United Kingdom) was placed at a distance of 30 cm from the subject’s head. Laser Doppler imaging scans (estimated scan time: 56 s, area: 7.5 × 7.5 cm2) were recorded twice at baseline and once after the electrical stimulation pulses (5 s, 100 Hz, threshold intensity). Superficial blood flow was calculated for each pixel by means of the intensity of the Doppler shift of the backscattered laser light (arbitrary perfusion units). Size of erythema was evaluated offline by the total number of pixels (0.005 mm2 per pixel), in which flux values exceeded the mean flux of the second baseline image by two standard deviations.
Questionnaires
Characteristics and accompanying symptoms of the headache, such as allodynia, were documented with a short questionnaire, designed by the neurologist and headache specialist who diagnosed the headache disorder, in both migraine and CTTH patients. Additionally, the migraine patients had to fill in a questionnaire, which was adapted from the questionnaire used at the outpatient clinics of the University Hospital of Hamburg-Eppendorf (Professor A May) regarding prodromi of the migraine attack before each test session.
Data analyses and statistical analysis
Data from pressure and electrical pain thresholds and suprathreshold pain ratings of pressure were z-normalized according to the formula described in Figure 1(b). The control group consisted of the healthy volunteers without headache (see Figure 1(b)).
All data were evaluated with the Statistica 6.0 software (StatSoft, Tulsa, USA). Significant differences were calculated by analysis of variance (ANOVA) and post hoc Fisher’s least square difference (LSD) test. If applicable, repeated measures analysis was used. p values less than 5% were considered to indicate significant differences. All values are given as mean ±standard error of the mean (SEM).
Results
Headache patients
Clinical description of migraine patients, TTH and healthy controls.

Number of tested volunteers in the different test time points.

Number of prodromal symptoms in the different phases of migraine patients.
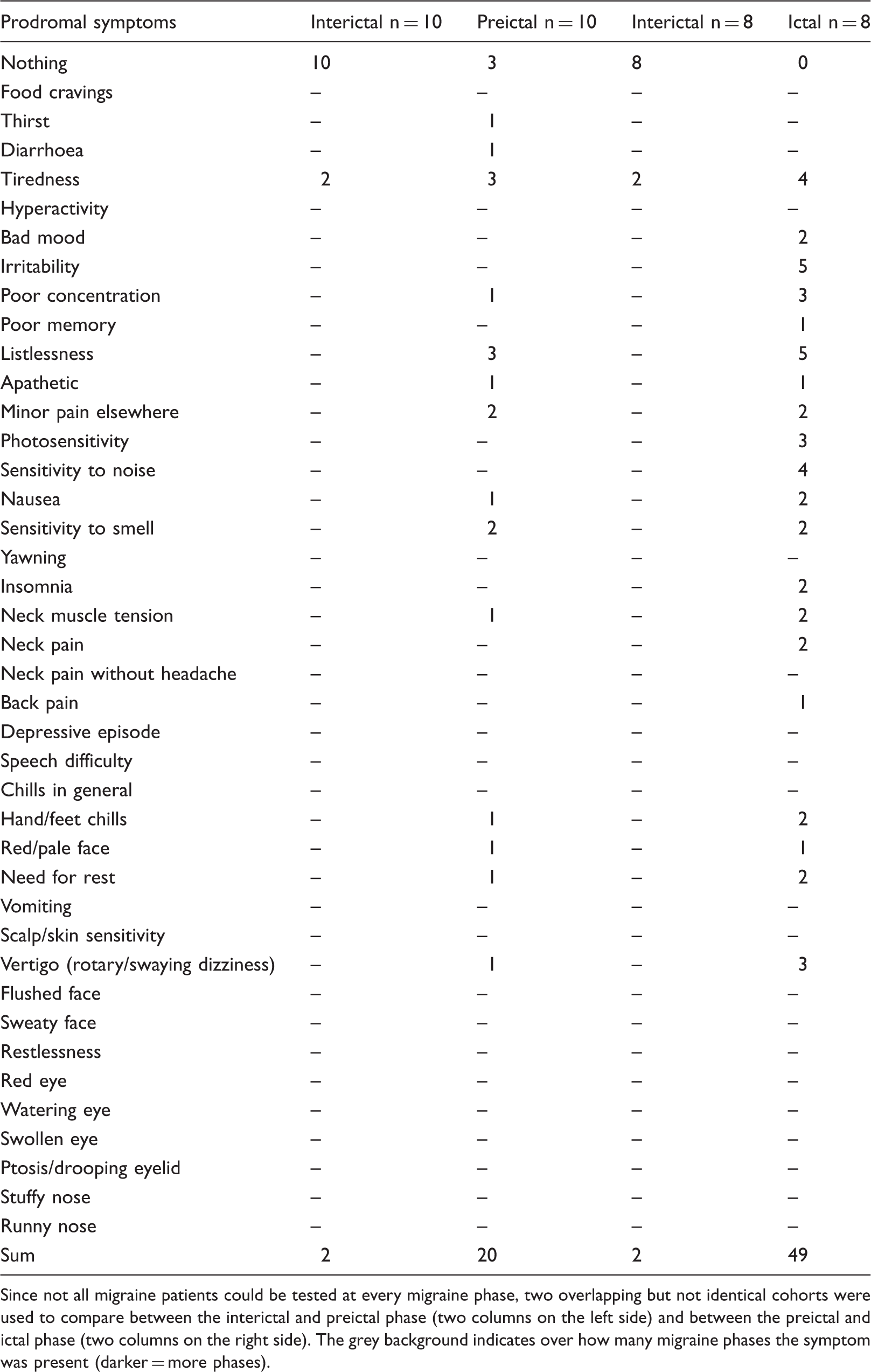
Since not all migraine patients could be tested at every migraine phase, two overlapping but not identical cohorts were used to compare between the interictal and preictal phase (two columns on the left side) and between the preictal and ictal phase (two columns on the right side). The grey background indicates over how many migraine phases the symptom was present (darker = more phases).
Migraine patients have larger axon-reflex erythemata
To objectively test the function of primary trigeminal afferents, we assessed the size of the axon-reflex erythema at the forehead. This is an indirect measure of the C-fiber function based on their release of neuropeptides causing vasodilation. A significantly larger electrically induced axon-reflex erythema area was observed at the forehead in migraine patients in comparison to healthy controls (F (1.27) = 12.186, p = 0.0017, ANOVA; see an example in Figure 2(a)). Using post-hoc testing, no significant difference between healthy and migraine patients was observed interictally, but preictally and ictally the migraine patients showed a significantly larger erythema size (ANOVA, repeated measures design, post hoc LSD, p = 0.011 and 0.05, respectively). However, within the group of migraine patients, no differences in erythema size between the migraine phases could be observed (ANOVA, F(2,16) = 0.26; p = 0.8; see Figure 2(b)).
Axon-reflex erythema. (a) Specimens of laser Doppler imaging recordings are shown for a healthy subject and one patient with migraine. Increased blood flow in superficial blood vessels is shown in light green, yellow red and white. Electrical stimulation (white arrows indicate electrodes) caused a larger axon-reflex erythema in the migraine patient in comparison to the healthy subject. (b) Area of axon-reflex erythema in healthy subjects (grey) and migraine patients (black). Bars and stars indicate significant differences of p < 0.05.
Migraine patients and CTTH patients are more sensitive to pressure pain
Analyses in healthy subjects and in both groups of patients showed no significant differences between the test sites in pressure pain ratings, either threshold or suprathreshold. Side differences were not observed in any of the groups. Specifically, in migraine patients no differences were found between the predominant headache side or the site at which headache occurred during the study. Therefore, the data from the six different test sites were pooled for further analyses of temporal changes.
In healthy subjects, a good reproducibility of pressure pain threshold was observed over the five test sessions with no significant differences (n.s. ANOVA with repeated measures design) (see Figure 3(a) for three test sessions from test day 2 to 4). Changes in pressure pain thresholds were not observed either in migraine or in CTTH patients between the different test sessions (interictal, preictal or ictal) within the respective subject groups (n.s., ANOVA with repeated measures design; see Figure 3(a)). The pressure pain thresholds were different between healthy controls, migraine patients and CTTH patients (ANOVA F (6.76) = 3.32, p = 0.007; post hoc LSD, p = 0.01). In particular, both migraine and CTTH patients had significantly lower pressure pain thresholds than healthy subjects (ANOVA post hoc LSD, healthy vs. migraine: p = 0.027; healthy vs. CTTH: p = 0.023). CTTH patients and migraine patients did not have significantly different pressure pain thresholds (ANOVA post hoc LSD, n.s.).
Psychophysical assessment of pressure pain. Values are z-normalized with the group of healthy subjects as control population. The range between −2 and +2 comprises the values of 95 % of the control population. Every grey line belongs to a single volunteer. Thick black lines are the mean values of the volunteers within a group. (a) Pressure pain thresholds. Values of migraine patients and tension-type headache patients are above 0, indicating sensitization in these patient groups in comparison to healthy subjects. No cyclic changes are observed. (b) Rating of suprathreshold pressure stimulation showed no cyclic changes. Since the stimulus intensity was adjusted to the threshold, the stimulation intensity was lower in migraine patients and tension type headache patients, but the rating was in a similar range in healthy subjects, which means it was relatively higher.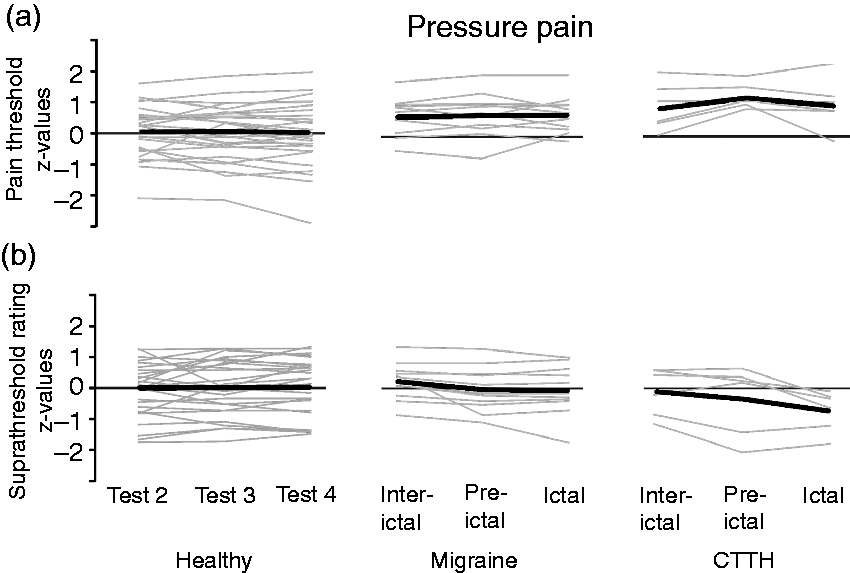
The ratings to suprathreshold stimulation in migraine patients were in a similar range to healthy controls (n.s. ANOVA; Figure 3(b)); however, due to the lower threshold, the stimulation intensity was lower to produce the same rating indicating a possible sensitization (compare Figure 3(a) and (b)). Cyclic changes could not be observed in suprathreshold pressure pain rating within any group (Figure 3(b)).
Migraine patients have cyclic changes in electrical pain sensitivity
Similar to pressure pain sensitivity, healthy subjects and both patient groups did not show significant differences, either between the test sites or between the five measurements in threshold or suprathreshold testing. In the migraine patients, no side difference was seen concerning the predominant headache site and the site at which headache occurred during the observed attack.
Therefore, the data from all six test sites were pooled for further analyses of temporal (possibly cyclic) changes and for comparison between the three groups.
In the group of healthy subjects, there was no significant difference in electrical pain thresholds between the five test sessions on five different test days (n.s. ANOVA with repeated measures design and post hoc LSD test; Figure 4(a)). The electrical pain thresholds were not significantly different between migraine patients, healthy subjects or CTTH patients, and no temporal changes were observed (n.s. ANOVA with repeated measures design and post hoc LSD test; see Figure 4(b)).
Electrical pain threshold. Values are z-normalized with the group of healthy subjects as control population. The range between −2 and +2 comprises the values of 95 % of the control population. Every grey line belongs to a single volunteer. Thick black lines are the mean values of the migraine patients within a group. Since not all migraine subjects could be measured in all migraine phases, the number of values varies between panels (b)–(c). (a) In healthy subjects, electrical pain threshold was assessed five times and shows good reproducibility. (b) Comparison of electrical pain threshold in migraine and tension-type headache patients between the inter-ictal and pre-ictal phase. (c) Comparison of electrical pain threshold in migraine and tension-type headache patients between the pre-ictal and ictal phase. (d) Comparison of electrical pain threshold in migraine and tension-type headache patients between the interictal and ictal phase.
The suprathreshold electrical stimulation with 100 Hz pulses for 5 s was rated at the beginning and the end of the 5 s stimulus train, since we were interested in the adaptation/habituation or sensitization of electrically induced pain sensation as an index of peripheral or central signal modulation (see Figure 1(c) data acquisition and analyses). Healthy subjects rated the stimulus at all test sites at the end of the electrical stimulation lower than at the beginning (see Figure 5). In healthy volunteers, no significant differences in the ratings of repetitive stimulations were found when comparing the differences between the beginning and end of electrical stimulation over the five test sessions (n.s. ANOVA with repeated measures design and with post hoc LSD test; see Figure 5). Interestingly, the end ratings were not significantly different between migraine patients and healthy subjects and did not differ over the three test sessions and migraine phases (Supplementary Figure 1).
Rating of suprathreshold electrical stimulation of healthy subjects. The beginning (black bar) and the end (grey bar) of the 5 s electrical stimulation with 100 Hz were rated separately. On all test areas and the sides of the head, and on all test days, the end of stimulation was rated less painful than at the beginning.
Similarly, no significant differences in pain ratings between the interictal, preictal and ictal phase could be observed in CTTH patients (see Figure 6(b) and (c), right hand panels). However, in migraine patients, the differences between the ratings at the beginning and the end of the stimulation were significantly smaller in the preictal phase than in the interictal phase. In the preictal phase, the end of the 5 s electrical stimulation was rated equally painful or more painful than the beginning (ANOVA, repeated measures design F (2.51) = 3.37, p = 0.042; post hoc LSD, p = 0.008; see Figure 6(a) and (b) middle panels). These preictal values in migraine patients were also significantly higher than the values of the third and the fourth test session in healthy subjects (p = 0.004 and p = 0.001, respectively; ANOVA repeated measures design and post hoc LSD). Interestingly, no statistically significant difference between the interictal and ictal phase was observed.
Rating of suprathreshold electrical stimulation of healthy subjects, migraine patients and tension-type headache patients. The differences between the rating of the end of stimulation and beginning of stimulation are depicted. Since not all volunteers could be measured in all migraine phases, the number of values varies. (a) Raw values of single volunteers are shown, meaning that every line shows the values of one single volunteer. (b) Bar graphs show the mean values ± standard error of means of the volunteer groups. The horizontal bar with two stars indicates a significant difference in electrical pain rating in patients with migraine between the interictal and preictal phase.
Discussion
Compared to healthy volunteers, we found higher pressure pain sensitivity in CTTH and migraine patients. There were no differences observed in electrical pain thresholds between headache patients and healthy subjects. Electrical pain thresholds were similar interictally, preictally and ictally within the respective patient groups. Independently of the migraine phase, in patients with migraine a larger electrically induced axon-reflex erythema was present in comparison to healthy volunteers. Habituation to a 5 s long electrical pulse was not different between healthy subjects and migraine patients in the interictal and ictal phases. However, higher pain ratings were found at the end of a 5 s lasting electrical stimulus only in the preictal phase of migraine, which we interpreted as less adaptation/habituation.
Changes in sensitivity on the head of patients with primary headache disorders have frequently been tested, and an increased sensitivity especially in pressure pain has been found in migraine patients and tension-type headache patients in contrast to healthy volunteers (14). Accordingly, we found reduced pressure pain thresholds in tension-type headache patients and in migraine patients in comparison to healthy volunteers.
In recent years, cyclic changes in migraine have become a focus of research. However, only in a few studies have changes in sensory perception been found (11,15–17). Accordingly, functional MRI studies have shown that cyclic changes in activity of the spinal trigeminal nucleus already occur in the preictal phase (18). These studies are logistically difficult to perform, since migraine attacks mostly occur irregularly so that fMRI or psychophysical test sessions cannot be scheduled in advance. One study solved this problem by performing fMRI in one patient every day over one month, showing that changes in hypothalamic activation due to nociceptive stimulation are altered in the preictal phase, while changed coupling to the spinal trigeminal nuclei and dorsal rostral pons occurs in the preictal phase, continuing into the ictal phase (19). In a retrospective study in a cohort of migraine patients, a positive correlation was found between heat pain thresholds and the time interval to the next migraine attack; however, this was not a longitudinal study (17). We found reduced pain rating at the end of a 5 s lasting electrical stimulus in the preictal phase in migraine patients in contrast to the interictal and ictal phase and in contrast to healthy subjects or CTTH patients.
However, in electrical threshold testing no changes were found either during the migraine cycle or in the pressure pain thresholds or the suprathreshold pressure pain testing. Thus, thresholds or short-lasting suprathreshold stimuli (2 s pressure) might be less suited to detecting sensory changes in single patients within the migraine cycle.
The reduction of pain rating during a longer lasting stimulus in the present (5 s) and other studies (15) (30 s) could be based on peripheral changes in receptor sensitivity – adaptation mechanisms that comprise, in our opinion, both attenuated transduction processes and desensitized axons; that is, decreased action potential firing during a continuous stimulus, which may be due to sodium channel inactivation. In contrast to heat stimulation, electrical stimulation bypasses the whole transduction process in the nerve endings. Peripheral nociceptive fibers (C-and A-delta fibers) are restricted in their following frequencies of action potentials. Microneurographic experiments show that human peripheral C-fibers cannot follow 100 Hz for longer than a few pulses (unpublished observation by the authors). Primary trigeminal and spinal afferents are known to have intracranial collaterals innervating the dura mater and extracranial collaterals innervating periosteum and deep muscle layers (9,20). To objectively test the function of these primary afferents that also innervate intracranial structures, we used electrically induced axon-reflex erythema in an area innervated by the first division of the trigeminal nerve. In humans, action potentials are needed to cause the widespread axon-reflex erythema via release of CGRP from C-fibers. Thus sensitization, expressed as increased following frequency of these primary afferents, could lead to less adaptation and subsequently also to a larger erythema due to more CGRP release (21,22). We indeed found a twofold larger axon-reflex flare size in migraine patients in comparison to healthy subjects. However, this increase in flare size was not dependent on the migraine cycle but was present in all migraine phases. This indicates that in migraineurs either a higher following frequency of primary afferents, facilitated CGRP release, a higher CGRP content or a higher vascular reactivity is present, independent of the migraine cycle. There are very few studies using axon-reflex erythema as a measure of CGRP release in respect to migraine (23,24). However, in these studies signal transduction via receptors like TRPV1 receptor channels, activated by capsaicin as a C-fiber stimulus, might have influenced the outcome (25). When testing axon-reflex erythema at the arm, the results may be influenced by the fact that trigeminal neurons may have a different efferent response function compared to neurons innervating the arm (26). Nevertheless, it has been shown that vasodilation was greater in female patients with migraine than in healthy women, which could be reduced by a CGRP antagonist (23,24). In future, the model of measuring axon-reflex erythema in trigeminal areas might be used in testing CGRP antagonists or antibodies. Changes in erythema size could be correlated in individual patients with the therapeutic response.
Taken together, in view of finding of changes in axon-reflex erythema, independent of the migraine phases, it is more unlikely that the phenomenon of reduced pain rating at the end of the electrical stimulation in the preictal phase is a matter of sensitization of primary afferents. Reduced central habituation during the preictal phase and a persisting sensitization of vasodilatatory mechanisms in migraine appear more likely.
Central mechanisms can modulate the perception of noxious stimuli in opposite directions, appearing as habituation or wind-up phenomena. In migraine patients, reduced interictal habituation has been observed in numerous studies with different modalities like visual, auditory, and somatosensory stimulation assessed with different methods such as evoked potentials, fMRI and psychophysics; for a review see (27). Reduced habituation may be due to a reduced initial response amplitude; however, we found no difference in ratings at the beginning of the stimulus between healthy subjects and migraine patients in any migraine phase (as shown in Supplementary Figure 1). During the migraine attack, an increased initial response or increased thresholds were observed, whereas habituation seemed normal (for review see (27)). Recently, a study analysing retrospectively different migraine phases within single patients showed changes in habituation to a 30 second heat stimulus in the preictal phase in comparison to the interictal period and, most interestingly, also in contrast to the ictal phase, in which a tendency to even more habituation was seen (15). Interestingly, this study, like our study, found that reduced habituation was typical for the preictal phase but not for the ictal or interictal phase. Thus, it might be hypothesized that not only do increasing changes occur in migraine from the interictal to preictal phase culminating in the headache attack, but also that specific changes are already present in the preictal phase. Reduced habituation may be an expression of reduced inhibitory control, which may lead to a greater impact of other pre-existing changes, which then may lead to headache.
Reduced central habituation might also underlie the prodromal symptoms of migraine. In our questionnaire, subjective sensations resembling typical prodromal symptoms were more often noted in the preictal phase than in the interictal phase. However, the reduced habituation to electrical stimulation was not present during the ictal phase, whereas the same prodromal symptoms were reported ictally more often than preictally. Thus, the reduced habituation seems to be mechanistically independent of the prodromal changes. The prodromi themselves are often unspecific, such as tiredness, making it hard for the migraineurs themselves to recognize them as signs announcing a migraine attack. However, together with reduced habituation, it may be possible to find a predictive model for migraine attacks, and therapeutic intervention might be effective even at this early stage of the migraine cycle to prevent the headache and vegetative symptoms.
Limitations and strengths of the study
This is the first study in which axon reflex erythema in the trigeminally innervated skin was measured in migraine patients. This might be used to monitor the action of the CGRP/CGRP receptor as a target of therapeutic actions. The use of electrical stimuli has the advantage that they are not dependent on transduction molecules, but depolarise the axon directly, and thus axonal excitability and central signal processing are tested alone. Another strong point of the study is the intra-individual comparison of sensitivity on the head in the different migraine phases, which might reveal a readout with individual predictive value for migraine attacks. Since this intra-individual comparison of migraine phases is logistically demanding, this causes most of the limitations of the study: The cohort is small for migraine patients and even smaller for CTTH patients, and the healthy volunteers were not perfectly age and gender matched. However, age and gender differences will most likely not have influenced our results, since there is a large inter-individual variance in psychophysical testing, which makes it unlikely that small age and gender effects in small groups will be found. Another limitation is that despite detailed individual planning, headache occurred sometimes not as scheduled at test session 4, but at another test session. Thus, the repetition count of test sessions was not the same for all headache patients as for healthy volunteers; that is, values from test session 3 in healthy volunteers were compared in some headache patients with pre-ictal values obtained in test session 4.
Conclusion
Migraineurs exhibit not only higher sensitivity to painful stimulation of facial areas and an increased axon-reflex erythema but also cyclic changes with reduced habituation of pain perception to longer lasting stimuli. This reduced habituation is typical for the preictal phase and is not present during the interictal and ictal phases. It might be hypothesized that during the preictal phase some inhibitory control is lost and the pre-existing changes are then effective in triggering mechanisms leading to headache. The clinical implication of this is that the method of testing habituation to nociceptive electrical stimulation might be used as a prediction for patients and clinician scientists to examine the effects of an early therapeutic intervention.
Key findings
Migraine patients have reduced habituation to electrically induced pain only in the preictal phase. Migraine patients in all phases have larger electrically induced axon-reflex erythema on the head in comparison to healthy subjects. Migraine patients and tension-type headache patients are more sensitive to pressure pain than controls. The reduced habituation (in the preictal phase) may make migraineurs more vulnerable to triggers precipitating headache.
Supplemental Material
Supplemental material for Cyclic changes in sensations to painful stimuli in migraine patients
Supplemental material for Cyclic changes in sensations to painful stimuli in migraine patients by Marion Strupf, Britta Fraunberger, Karl Messlinger and Barbara Namer in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was supported by the ELAN fund of the FAU Erlangen Nürnberg and the Dr. Robert Pfleger Stiftung; BN was supported by DFG grant NA 970 3-1.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
