Abstract
Background
Migraine attacks are unpredictable, precluding preemptive interventions and leading to lack of control over individuals' lives. Although there are neurophysiological changes 24–48 hours before migraine attacks, so far, they have not been used in patients' management. This study evaluates the applicability and the ability to identify pre-attack changes of daily “at home” electroencephalography obtained with a portable system for migraine patients.
Methods
Patients with episodic migraine fulfilling ICHD-3 beta criteria used a mobile system composed of a wireless EEG device (BrainStation®, Neuroverse®, Inc., USA) and mobile application (BrainVitalsM®, Neuroverse®, Inc., USA) to self-record their neural activity daily at home while resting and while performing an attention task, over the course of 2 weeks. Standard EEG spectral analysis and event-related brain potentials (ERP) methods were used and recordings were grouped by time from migraine attacks (i.e. “Interictal day”, “24 h Before Migraine”, “Migraine day” and “Post Migraine”).
Results
Twenty-four patients (22 women) recorded an average of 13.3 ± 1.9 days and had 2 ± 0.9 attacks. Twenty-four hours before attack onset, there was a statistically significant modulation of relative power in the delta (decrease) and beta (increase) frequency bands, at rest, and a significant reduction of the amplitude and inter-trial coherence measures of an attention event-related brain potential (P300).
Conclusions
This proof-of-concept study shows that brain state monitoring, utilising an easy-to-use wearable EEG system to track neural modulations at home, can identify physiological changes preceding a migraine attack enabling valuable pre-symptom prediction and subsequent early intervention.
Introduction
Migraine is a common brain disorder characterised by episodes of severe headache, nausea, vomiting and intolerance to sensory stimuli that may last up to three consecutive days (1). Attacks interfere with work, family and social activities, leading to absenteeism and poor quality of life. According to the Global Burden of Disease Study 2010 (2), it is one of the leading causes of years lived with disability, with estimated costs of $13 billion per year in the USA alone, primarily due to missed workdays (3). One of the most disturbing aspects of migraine is the unpredictability of the attacks, contributing to high anxiety levels and poor control over patients' lives. This is one of the main factors associated with a low quality of life among migraine sufferers, who report difficulty in making plans, assuming commitments and making decisions related to their lives due to fear of developing a migraine attack. In fact, migraine patients are three times more likely to suffer from generalised anxiety disorders (4). Although migraine sufferers are able to identify some triggers and early symptoms, most attacks are still difficult to predict. This limitation precludes preemptive or timely pharmacological intervention, which can improve therapeutic response (5,6).
Migraine attacks are complex neurovascular events characterised by the activation of the trigeminovascular system, with a release of CGRP and other neuropeptides in the cranial circulation leading to cranial vasodilatation, neurogenic inflammation and pain. While the mechanisms responsible for this periodic activation remain poorly understood, there is evidence of a neurogenic basis for migraine (7), and that attacks are preceded by asymptomatic brain changes taking place hours or days before the onset of pain. Recently, functional brain imaging studies performed during spontaneous (8) and provoked (9) migraine attacks have shown a changing pattern of brain modulations across the migraine cycle. The day before the attack, there is an activation of the hypothalamus and an increased neuronal coupling between the hypothalamus and the trigeminocaudal nucleus. However, with the onset of pain this pattern of connectivity is substituted by an increased coupling between the hypothalamus and the dorsal pons. These findings are in agreement with the well-known pattern of premonitory symptoms experienced by some patients before the attacks (10,11). A study, performed with an electronic diary, has shown that migraine attacks can be preceded, by up to 48 hours, by a subjective decline of health perception and poorly defined physical symptoms, suggesting that the underlying process begins well before the headache stage (11). This is in agreement with the metabolic changes found in brain functional imaging performed in the prodromal stage (8), and with neurophysiological brain state fluctuations in migraine, some of which occur before an upcoming attack (12). Yet, so far, it has not been possible to translate this knowledge into patients' benefit. Current roadblocks include the lack of both an adequate monitoring system and reliable correlated neural measures.
Most neurophysiological data related to migraine attacks has been collected in small samples of patients, with only a few datasets per patient and limited to controlled laboratory settings (12). Besides, most studies have focused on investigating “trait” neural aspects that differentiate migraine patients from controls. By taking advantage of the mobility and ease of use of a new system, here we raise a different question focusing on “state”, and not “trait”, changes throughout the migraine cycle in patients. We build upon research on the few studies investigating changes during the migraine cycle, such as Schulte and May's landmark study (8) and aim at understanding how these functional anatomy modulations are reflected on well-established neurophysiological signals.
This proof-of-concept study evaluates the applicability and value of daily “at home” electroencephalographic records obtained with a new portable system for the acquisition and analysis of neurophysiologic signals to detect incoming migraine attacks. It aims to: i) evaluate the usability of this daily neurophysiologic monitoring system in migraine sufferers at home, away from a laboratory or clinical setting, and ii) identify reliable neurophysiological markers that characterise neural changes associated with incoming attacks, namely during resting state EEG and an attention task. Based on previous studies, we expect to find a decrease in ERP amplitude and a change in EEG power towards a decrease in delta power (12–14) before the attacks.
Methods
Study design
This study was designed as a prospective observational two-week longitudinal study consisting of the daily recording of EEG/ERPs during the performance of simple cognitive tasks and the daily recording of migraine attacks and other symptoms. The study design and the protocol were approved by the local Ethics Committee of the Lisbon Faculty of Medicine and Centro Hospitalar Universitário Lisboa Norte.
Participants
Subjects with migraine were recruited from the Headache Outpatient clinic of a university hospital, after signed informed consent. Patients fulfilled the following inclusion criteria: a) age above 18 years; b) more than 9 years of formal education and ability to use a mobile/smartphone in daily life; c) diagnosis of episodic migraine, with or without aura, for more than 1 year, fulfilling the ICHD-3 beta criteria (1); d) frequency of migraine attacks of between 1 and 5 per month, in the last 3 months; e) ability to differentiate migraine from non-migraine headaches; f) absence of prophylactic medication that could interfere with EEG activity (antiepileptics, antidepressives); and g) no history of other neurologic disorders.
Demographic and clinical data, obtained by a semi-structured interview, included present age, age at migraine onset, migraine classification (with aura, without aura, or both), present attack frequency and duration of attacks, family history of migraine, co-morbidity and current treatment.
Headache record
Patients were instructed to fill in a daily record (in paper or electronic) of any headaches, migraine attacks (moderate to severe pain and associated gastrointestinal symptoms, photophobia and phonophobia symptoms), and to signal the presence and severity of 13 prodromal symptoms considered most specific to predict migraine attacks (11) that were classified in five degrees from absent (0) to severe (4). In headache days, patients were asked if the headache was identical to their usual migraine attacks, and a positive response was diagnosed as a migraine day. During attacks, patients could take their usual migraine medication and were asked to record all medication taken. Patients receiving prophylactic medication for migraine could not change it during the study period. Use of oral contraceptives was not contra-indicated.
Data categorisation
During neurophysiological data analysis, daily sessions were categorised according to the following criteria: Interictal – data was recorded on a day that was both more than 48 hours before migraine onset and more than 48 hours after the end of a migraine event; Before Migraine – data was recorded within 24 hours of the onset of a migraine event; Migraine – data was recorded on the day/days of reported migraine event; After Migraine – data was recorded within 48 hours following migraine. Since we cannot trace the onset of the prodromal phase exactly, we did not attempt to classify the period between 48 hours and 24 hours before the attack, and we discarded it from the analysis. That time period may contain hours belonging to the preictal phase and hours of the interictal phase. If a participant had more than one session in a particular category (i.e. the participant reported more than one migraine day over the course of the study), the session with the maximum amount of data remaining after pre-processing and artifact rejection (see details below) was selected for analysis.
Neurophysiological data
Equipment
EEG data was collected using the Neuroverse®, Inc. system composed of a three-channel wireless, portable EEG device (BrainStation®, Neuroverse®, Inc., USA) (Figure 1) coupled with a mobile iOS application (BrainVitalsM®, Neuroverse®, Inc., USA) controlling both stimuli presentation and EEG data recordings. The EEG device was self-applied to the subject's forehead, using a disposable adhesive, at the beginning of each recording session. The EEG device communicated via Bluetooth with the mobile application implemented on an Apple® iPad® mini. In this study, the EEG system used patches of three disposable flexible electronic sensors (including the reference and ground sensors) for EEG recordings. These sensors are heterogenous ultrathin film structures comprised of 3M Tegaderm™ and stretchable circuits made of biologically inert materials (silicon and gold). EEG data were written into a text file at a sampling rate of 500 Hz, with time-stamped markers for visual stimuli onsets generated by the BrainVitalsM® mobile app.
Wearable, wireless, forehead electroencephalographic (EEG) system. This figure illustrates the Neuroverse® EEG system used in this study, depicting back, side and frontal views of the device, as well as the disposable adhesive track and flexible electronics (three sensors) patch. This system is composed of a wireless forehead EEG device and a software mobile application.
Data collection
Study participants were trained to affix the BrainStation® EEG device to their forehead and to operate the BrainVitalsM® app. They were asked to record brain activity at home daily for a 14-day period and to do it at the same time of the day, preferably in the evening at a time they could rest. After the participant put on the EEG device and visually checked the mobile app for ongoing device connectivity (i.e. visual display of EEG ongoing signal on the mobile app), EEG data was collected during resting state (two blocks, 6 min duration each) and a visual attention task (three blocks, 3 min 20 sec duration each). During the resting state blocks, participants were instructed to sit quietly and look at a black dot at the center of the iPad screen for six minutes. A small countdown timer at the top of the screen indicated the amount of time left in the block.
During the visual attention blocks, a randomised sequence of images was displayed on screen. Patients were instructed to count the number of images from a designated target category (either cars or people) and ignore non-target images (other commonplace objects such as books, electronic devices, or furniture). The designated target category was constant for all blocks of a single day of testing (i.e. on a single day, participants would count images of cars interspersed with images of other commonplace objects). Stimulus duration was 100 ms with an interstimuli interval (ISI) of 500 ms. There were 75 target and 225 non-target stimuli during a single block.
EEG and ERP recording
EEG was collected from a single forehead sensor. The continuous signal was sampled at 500 Hz, then the digitised EEG was imported to MATLAB 2015a (MathWorks Inc., Natick, MA, USA), where all subsequent processing was performed. The data was high-pass filtered at 0.1 Hz, notch-filtered at 50 Hz, then low-pass filtered at 40 Hz.
Analysis of resting state EEG
The continuous data from the two 6 min resting conditions were segmented into overlapping 1024 ms epochs (the segmenting window was moved by 512 ms steps). Epochs were detrended and those containing artifacts (such as amplifier saturation or eye or muscle movement) were rejected.
Artifact rejection was a two-step process. During the initial, automated step, epochs with sections of flat or ‘saturated’ values or values exceeding a ±50 µV threshold were rejected. During the second, manual step, a single researcher visually inspected all remaining epochs and rejected any that contained activity consistent with either an eye blink or a saccade. A single daily session had to yield at least 50 epochs after artifact rejection to be considered for inclusion. After artifact rejection of the resting state data, there were on average 666.8 epochs (sd = 276.8) per subject per day. There was no difference in number of trials according to recording day F(3,27) = 0.458, p = 0.714.
The averaged power spectrum was calculated using a Fast Fourier Transform with a Hanning window applied to each epoch. Absolute power was calculated for the following frequency bands: delta (0.5–4.0 Hz), theta (4.0–8.0 Hz), alpha (8.0–12.0 Hz), beta (12.0–30.0 Hz), and gamma (30.0–40.0 Hz). Absolute power in each frequency band was transformed according to 10*log10(x), where x = absolute power in the band of interest (15). Log-transformed absolute power was analysed in a two-way repeated measures ANOVA with factors of day type (Interictal, Before Migraine, Migraine, and After Migraine) × frequency (delta, theta, alpha, beta, gamma).
Relative power was calculated as the ratio of summed power in a particular frequency band to summed power from 0.5–40 Hz. Relative power in the band of interest, in each of five frequency bands (delta, theta, alpha, beta, and gamma), was calculated according to 10*log10(x/1-x), where x = relative frequency in band of interest (15). The relative power was analysed in a two-way repeated measures ANOVA with factors of day type (Interictal, Before Migraine, Migraine, and After Migraine) and X frequency (delta, theta, alpha, beta, and gamma).
Analysis of visual attention ERPs
We investigated if the migraine cycle was affecting neurophysiological functional modulations of common everyday cognitive processes, such as attention. For this purpose, we evaluated neural modulations of a well-known event-related brain potential, the P300 (16), while performing a standard visual attention task. Epochs time-locked to target and non-target stimuli presentation onset were extracted from the continuous EEG data recorded during the visual attention blocks. Epoch length was 1200 ms epochs, extending from 300 ms prior to onset of stimulus presentation to 900 ms post-stimulus onset. Epochs were detrended, baseline corrected (the average value of the pre-stimulus interval −300 to 0 ms was subtracted from the epoch) and epochs contaminated by artifacts were rejected. Again, artifact rejection was a two-step process as described above. A single daily session had to yield at least 20 epochs per stimulus type after artifact rejection to be considered for inclusion. After artifact rejection of the short window epochs from the visual attention task, there were an average of 136.4 target epochs (sd = 51.8) per day. There was no difference in number of trials according to recording day F(2,16) = 0.153, p = 0.859. The remaining epochs were median-averaged according to stimulus type (target, non-target). The P300 measurement was area under the curve from a window extending from 350−550 ms post-stimulus. This analysis was performed using the STUDY framework implemented in EEGLAB v13.4.4b (17). A 1 × 3 paired design with levels of day type was created (‘Normal Day’, ‘Day Before Migraine’, ‘Migraine Day’).
Time-frequency analysis of visual event-related activity
In order to further investigate the processes underlying the amplitude decrease in the response to attentional targets preceding a migraine attack, we analysed event-related spectral perturbations (ERSP) and inter-trial coherence (ITC) for day types associated with migraine cycle. The event-related potential (ERP) only captures neural activity that is both time- and phase-locked to the time-locking event (i.e. target and non-target stimuli). Inter-trial coherence (ITC) measures capture that phase-locked activity at the single-trial level. Oscillatory activity that is consistently phase-aligned over many individual trials results in a visible ERP; ITC analysis helps reveal which frequencies are the major contributors to that ERP. Event-related spectral perturbation (ERSP) measures show how strongly event-related power at different frequencies either increase or decrease relative to the baseline. Significant ERSPs, while they provide information about event-related modulations of the power spectrum in different parts of the brain, may be more difficult to interpret, given that they are not always directly reflected in the ERP. Epochs time-locked to target and non-target stimuli onset were extracted from the continuous EEG data recorded during the visual attention blocks. For the purpose of increasing frequency resolution in the lower bands, a longer epoch window (1750 ms windows, from −500 to 1250 ms post-stimulus) was used. After artifact rejection of the long window epochs from the visual attention task, there were an average of 116.4 target epochs (sd = 40.9) per day. There was no difference in number of trials according to recording day F(2,12) =0.723, p = 0.51. The newtimef function in EEGLAB (17) was used to calculate event-related spectral perturbation (ERSP) and inter-trial coherence (ITC) values from the single-trial epochs. The time-frequency transforms were computed using a Morlet wavelet rising linearly from three cycles at 3 Hz to eight cycles at 40 Hz. Trials were baseline-corrected with a −300 ms to 0 ms pre-stimulus window. ITC values were exported from EEGLAB and the mean ITC over the 350–550 ms window was calculated for delta (3–4 Hz) and theta (4–8 Hz) bands.
Statistical analysis
Statistical analyses were performed using SPSS Statistics for Windows, version 17.0 (SPSS Inc., Chicago Ill., USA), with the exception of the significance threshold masking described in the “time-frequency measures” of the results section. Comparison between the four phases was performed by two-way repeated measures ANOVAs with a post hoc analysis with a Bonferroni adjustment. For all ANOVAs, the assumption of sphericity was assessed using Mauchly's test of sphericity; for any test that failed the assumption of sphericity at a level of p < 0.05, the Greenhouse-Geisser correction was used to determine significance and the estimated epsilon (ɛ) is reported. Confidence intervals are reported as mean ± standard error.
Results
Brain monitoring.

Resting state EEG power
In order to evaluate possible neural changes at rest, and correlated biomarkers associated with the migraine cycle, a statistical comparison of absolute power for each frequency band per migraine cycle period was performed. Since a single daily session had to yield at least 50 epochs after artifact rejection to be considered for inclusion, only 10 patients met the criteria for inclusion in this analysis (e.g. sufficient number of epochs for each of four day types); and yielded an average of 666.8 (SD = 276.8) epochs per day type.
Absolute power: There were no outliers, as assessed by examination of studentised residuals for values greater than ± 3 standard deviations. Log-transformed absolute power was normally distributed (p > 0.05) as assessed by Shapiro-Wilk's test of normality on the studentised residuals.
There was no statistically significant interaction between day type and frequency band, (F(12,108) =2.817, p > 0.05, ɛ = .263). There was no main effect of day type on absolute power, (F(3,27) = 0.843, p > 0.05). There was a main effect of frequency band (F(4,36) =116.53, p < 0.001, ɛ = .403). This effect reflects typical spectral distribution, with each frequency band having significantly greater power than higher frequency bands and significantly less power than lower frequency bands, with the only exception being that alpha and beta bands were not significantly different from each other, (2.101 ± 0.785, p > 0.05), as shown by Bonferroni post-hoc tests. Thus, absolute power analysis did not yield discriminating results associated with migraine cycle.
Relative power analysis: There were no outliers, as assessed by examination of studentised residuals for values greater than ±3 standard deviations. Log-transformed relative power was normally distributed (p > 0.05) except for delta power on the Interictal day (p = 0.025) and theta power Before Migraine (p = 0.27), as assessed by Shapiro-Wilk's test of normality on the studentised residuals. Contrary to absolute power, relative distribution of power across frequency bands was affected by day type. Frequency changes at rest correlated with migraine cycles (F(12,108) = 3.558, p < 0.05). Namely, effects in the relative power of the delta (F(3,27) = 5.022, p < 0.01) and beta (F(3,27) = 4.577, p < 0.02) bands (the Bonferroni correction for multiple effects testing lowered the significance threshold to p < 0.025). Post hoc analysis with a Bonferroni adjustment revealed (Figure 2) that there is a statistically significant decrease of relative power in the delta band 24 h before migraine onset (Interictal day (dark blue bar) vs. 24 h Before Migraine (orange bar) (2.285 ± 0.604, p < 0.05)), followed by a return to a higher level after the migraine attack (Migraine day (red bar) vs. Up to 48 h after Migraine (light blue bar) (−1.276 ± 0.361, p < 0.05)). Conversely, relative beta power increases 24 h before migraine onset (Interictal day (dark blue bar) vs. 24 h Before Migraine (orange bar) (2.093 ± 0.617, p < 0.05).
Resting state relative frequency analysis. This figure shows the relative power values across five EEG frequency bands, during resting state, for each migraine cycle period (*p < 0.05, statistics performed on log-transformed relative power values, see online Methods). Error bars show standard deviation.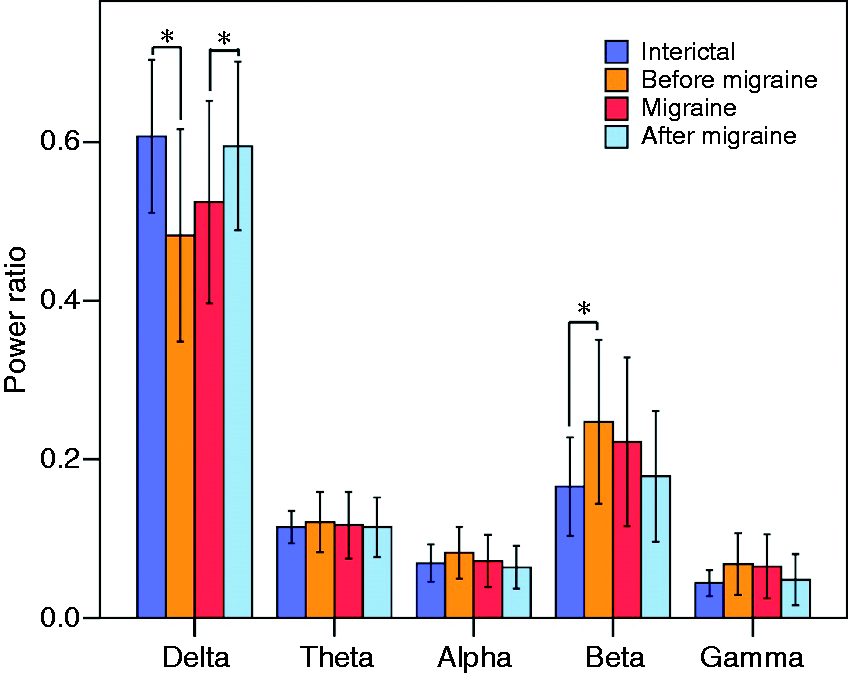
Attention event-related brain potentials (P300 ERP)
Furthermore, we investigated possible effects of the migraine cycle affecting functional modulations of common everyday cognitive processes, like attention, by examining a well-established visual attention ERP, the P300. For this analysis, a single daily session had to yield at least 20 epochs per stimulus type after artifact rejection to be considered for inclusion and nine patients met this criteria for three day types (e.g. sufficient number of epochs per stimulus type for each of the day types); yielding an average of 133.2 (STD 51.8) target and 408.4 (STD 132.5) nontarget epochs per day type.
The P300 area was analysed in a two-way repeated measures ANOVA with factors of day type (Normal, Day Before, Migraine) and X stimulus type (Target, Nontarget). Analysis of the studentised residuals showed that there was normality, as assessed by examination of studentised residuals for values greater than ±3 standard deviations. P300 area was normally distributed (p > 0.05), as assessed by Shapiro-Wilk's test of normality on the studentised residuals.
Importantly, our results show that a statistically significant interaction between day type and stimulus type, (F(2,16) = 14.431, p < 0.001) was found for the amplitude modulations of the P300 ERP, as such a simple main effects analysis was performed. Although there is no day type (i.e. migraine cycle) effect in the processing of nontarget visual stimuli (F(2,16) = 3.679, p > 0.025 (Bonferroni adjustment for multiple effects testing)) (Figure 3, left panel), there is a significant effect for the processing of target stimuli (F(2,16) = 14.227, p < 0.001). We found a significant reduction of the P300 ERP amplitude to target images 24 h before migraine onset (Interictal day (dark blue line) vs. 24 h Before Migraine (orange line) as indicated by a post hoc analysis with a Bonferroni adjustment (198.217 ±23.024 µV2, p < 0.001)) (Figure 3, right panel).
Modulations of an attention event-related brain potential (P300 ERP). The left panel of this figure shows the amplitude modulations of the ERP response to non-target visual stimuli, while the right panel shows the responses to the target stimuli, for each of three analysed migraine cycle periods (i.e. interictal: blue line; 24 h before migraine attacks: orange line; and migraine periods: red line). * = p < 0.001.
P300 ERP time-frequency measures
In order to further investigate the mechanism underlying the amplitude decrease in the neural response to attentional targets preceding a migraine attack, we analysed spectral perturbations (ERSP) and inter-trial coherence (ITC) of this ERP. The same participants and sessions from the P300 analysis (above) were used for this analysis. In order to increase frequency resolution in the lower bands, a longer epoch window (1750 ms windows, from −500 to 1250 ms post-stimulus) was used. The longer epoch led to higher rejection rates due to artifact, so seven patients remained after this procedure with an average of 116.4 (STD 40.9) target and 378.8 (STD 112.8) nontarget epochs per daily recording session.
The effects on ERSP and ITC values were calculated using parametric statistics. Figure 4 shows time-frequency points that were significant at a level of p < 0.05 after correction for multiple comparisons using the False Discovery Rate (FDR) algorithm. We found no significant effect of the migraine cycle in the ERSP analysis (Figure 4, mid row). The mean log-transformed ITC values over the P300 time window (between 350–550 ms) were analysed with a two-way repeated measures ANOVA with factors of day type (Normal, Day Before, Migraine) and X frequency (delta, theta). No variable had studentised residuals greater than ± 3 standard deviations. Mean ITC was normally distributed (p > 0.05), as assessed by Shapiro-Wilk's test of normality on the studentised residuals. Mauchly's test of sphericity indicated that the assumption of sphericity was met for the two-way interaction, χ2(2) = 0.733, p = 0.338. There was no statistically significant interaction between day type and frequency, F(2, 16) = 1.711, p > 0.05. However, the main effect of day type showed that there was a statistical significance in mean ITC between different days, F(2, 16) = 19.782, p < 0.01. Post hoc analysis with a Bonferroni adjustment showed that mean ITC during the P300 window suffers a significant neural firing coherence loss 24 h before a migraine attack (Interictal Day vs. 24 h Before Migraine, (−0.333 ±0.055, p < 0.01)), remaining below coherence levels in the migraine period, as compared to the normal period (Interictal Day vs. Migraine Day (0.153 ± 0.058, p > 0.05)). Furthermore, coherence was significantly more reduced 24 h before a migraine attack even when compared to the Migraine Day (−0.179 ± 0.044, p < 0.05)). Finally, the main effect of frequency showed a statistically significant difference in ITC between delta and theta bands, F(1, 8) = 29.875, p < 0.01, with greater coherence in the delta than the theta band (0.205 ± 0.038, p < 0.01).
Time-frequency and inter-trial coherence analysis of P300 ERP responses to target stimuli. This figure depicts results from event-related amplitude modulations ERP (upper row) event-related spectral perturbations (ERSP) (mid row) and inter-trial coherence (ITC) (lower row) analysis for the neural responses to target stimuli in three of the analysed migraine cycle periods (i.e. interictal period; 24 h before migraine attacks; and during migraine periods). The right-most column shows significant differences between days. Gray bars on the top row indicate the time points where the ERP is significantly different, red blobs on the middle and bottom row indicate time and frequency points where the ERSP and ITC differ significantly across days.
Discussion
The present study reports three neurophysiological changes anticipating the onset of the attacks. Compared to the interictal period, there was a significant change in the relative distribution of resting state delta and beta power bands, a decrease in P300 amplitude and a decrease in signal coherence, especially in the low frequency range, in the P300 ERP responses to target images during an attention task. These changes began preictally, continued into the ictal period and reverted to baseline after the attack. The change in slow frequency delta power and coherence suggests a mechanism common to both resting state and visual attention results.
There is previous evidence of altered functional connectivity between the hypothalamus and the locus coeruleus (LC) in migraine patients (18); furthermore, functional connectivity between the hypothalamus and other brain structures (including the dorsal pons) changes over the migraine cycle (8). Given the importance of the LC-NE (norepinephrine) system in P300 generation (19), it is possible that the EEG/ERP changes in this study are neurophysiologic correlates of the hypothalamic modulation, thus converging with the findings of brain imaging previously reported (8,20).
A variety of previous neurophysiological studies in migraine have identified signals that fluctuate along the migraine cycle (21). For instance, reports have shown that outside the attacks there is an increase of slow frequency activity in the theta or delta bands, in time intervals ranging from the interictal phase (22,23) to 1–4 days (13) and 36 hours before migraine attacks (12). Moreover, a wide range of measures of cortical potentials, such as the P300 (24), contingent negative variation (CNV) (25,26) and the visual N2 (27–30; however, see 31) have indicated that migraine patients present a consistent pattern of impaired habituation to sensory stimuli interictally, expressed by a lack of decremental response to repeated stimulation that reverts to normal before the attacks. Although the present results seem to converge with those findings, they cannot be directly compared due to both methodological and scientific enquiry differences. A significant number of these studies aimed at distinguishing neural responses between healthy subjects and migraine patients and, in addition, previous data resulted mostly from group or pairwise comparisons between patients tested during a limited number of sessions that were retrospectively divided across the migraine cycle. The present study, however, aimed at identifying neural correlates that could reliably predict an incoming migraine attack and for that purpose performed within-subject comparisons of four migraine cycle phases (10) observed during 14 consecutive daily records, enabling a unique capture of the complete spectrum of events taking place in the migraine cycle of each patient. In addition, previous recordings were obtained in medical and research centre laboratories, limiting the data collection ability, and often with considerable consumption of time and specialised services, while the present data was collected at home, enabling easy and systematic data collection, as well as the much-needed future use as a treatment efficacy and management tool.
We would like to acknowledge some limitations to this study. The first is related to the smaller than expected patient sample size. Although we were able to include 35 compliant patients and most recorded a total of 14 days, both a high frequency of migraine occurrence, leading to cases of no interictal days between attacks, and a strict artifact removal protocol, did not always allow for sufficient sampling across each of the four migraine periods per subject, which led to exclusion from the final analysis (five patients dropped out, one patient had a technical problem, five patients had no migraine attacks during the study period, 13 patients' data did not provide an adequate sample of each period considered, 11 patients were included in the final neurophysiological data analysis (10 women and one man (average age of 33.8 ± 10.9 years)). Additionally, since the exact timing of attack onset is unknown, the time intervals were arbitrarily established by periods of 24 h. Yet, there may be variations within and among subjects in the precise timing and speed of the attacks' development, and the exact timing of medication intake was not recorded. Finally, the data collection period was not long enough to enable reliable data acquisition across multiple migraine attacks per patient. Notwithstanding those limitations, patients were able to effectively use this technology at home and these preliminary results produced significant signals that can be conceived as biomarkers of upcoming attacks. Furthermore, currently ongoing predictive modelling work is yielding extremely encouraging preliminary results using the integration of multiple features of the acquired neural data in the prediction of incoming attacks in most of the 35 studied patients.
In this study, patients did not receive any feedback. As the predictive model is refined, the next step for clinical application, which we are currently preparing, is to reproduce this study in a larger population with a probabilistic feedback warning of an incoming migraine attack, as well as a refinement for testing time reduction, and evaluation of the impact of this information in migraine management and the patient's quality of life.
Migraine is a cyclic brain disorder affecting 20% of females and 10% of men in their most productive years. Its severe impact on individuals' lives leads to its ranking at eighth place among the leading causes of disability (2). Despite the development of new intervention strategies, there is a dire need for novel predictive and intervention methodologies that can enable better management and prevention (6). In this study, we report a demonstration that a daily EEG, self-recorded at home with a wearable mobile system, allows for the monitoring of neural changes anticipating migraine attacks before the onset of pain. The current result suggests that this assessment and prediction technology may open new paths for migraine management and research, namely the much-needed possibilities of attack prediction, early intervention and attack abortion, which so far have not been met.
This is a pilot study and this technology must be applied to a larger sample of individuals over a longer period, with analysis of sensitivity and specificity for the prediction of migraine attacks. If it proves to be a good attack predictor we believe that these findings, enabling a low cost and easy to use at home technique to predict incoming migraine attacks with reliable neurophysiologic markers, can lead to significant translational applications within three distinct avenues: i) patients' management, ii) therapeutic research and drug development, and iii) basic research into migraine physiology. The first relates directly to patients' use as an “at home” attack prediction system, allowing for both: i) a much-needed better control of their lives, facilitating life planning and reducing related anxiety and importantly ii) a potential intervention aid, enabling early attack detection and treatment with potential avoidance or reduction of the severity of the attack, as has been demonstrated in those cases where there is a predictable trigger (32). Secondly, it may contribute to the differentiation between migraine attacks and other headaches that warrant other types of pharmacological intervention and could enable clinical trials about drug efficacy at the earliest stages of attacks, with current or new medications, or about short-term preventive treatments to avoid migraine attacks. Lastly, it can also be of valuable use in research, enabling “at home” larger population studies with systematic neural data collection to allow a better understanding of migraine brain states, their sources, fluctuations, triggers and stabilisation.
Clinical implications
Migraine attacks are preceded by silent neural changes. This proof-of-concept study demonstrates that “at home” EEG, recordings with a portable device, can detect neurophysiologic changes before the attacks. This may enable migraine patients “at home” timely treatment of incoming attacks, resulting in better control over their lives.
Footnotes
Acknowledgements
The authors would like to thank all patients who participated in the study, Melissa Caswell for assistance in logistics and Raynard Fung, Carson Whitsett and Michael Bajema for their contributions to technology development and implementation.
Author contributions
IPM contributed to the conception and design of the study, patient selection and recruitment, data collection, analysis and writing of the article, MW contributed to data analysis and writing of the article; ML contributed to data collection, analysis and technology development; CM contributed to the instruction and monitoring of patients and data collection; and RGC contributed to the conception and design of the study, data collection, analysis, technology development and writing of the article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Devices, electrodes and apps were provided by Neuroverse Inc.
