Abstract
Background
Intranasal high flow of dehumidified (dry) air results in evaporative cooling of nasal passages. In this randomized clinical trial, we investigated the effect of dry gas induced nasal cooling on migraine headaches.
Methods
In this single-blind study, acute migraineurs were randomized to either nasal high-flow dry oxygen, dry air, humidified oxygen or humidified air (control) at 15 L/min for 15 min. All gases were delivered at 37°C. Severity of headache and other migraine associated symptoms (International Classification for Headache Disorders, 3rd edition criteria) were recorded before and after therapy. The primary endpoint was change in pain scores, while changes in nausea, photosensitivity and sound sensitivity scores served as secondary endpoints. A linear regression model was employed to estimate the impact of individual treatment components and their individual interactions.
Results
Fifty-one patients (48 ± 15 years of age, 82% women) were enrolled. When compared to the control arm (humidified air), all therapeutic arms showed a significantly greater reduction in pain scores (primary endpoint) at 2 h of therapy with dry oxygen (−1.6 [95% CI −2.3, −0.9]), dry air (−1.7 [95% CI −2.6, −0.7)]), and humidified oxygen (−2.3 [95% CI −3.5, −1.1]). A significantly greater reduction in 2-h photosensitivity scores was also noted in all therapeutic arms (−1.8 [95% CI −3.2, −0.4], dry oxygen; −1.7 [95% CI −2.9, −0.4], dry air; (−2.1 [95% CI −3.6, −0.6], humidified oxygen) as compared to controls. The presence of oxygen and dryness were independently associated with significant reductions in pain and photosensitivity scores. No adverse events were reported.
Conclusion
Trans-nasal high-flow dry gas therapy may have a role in reducing migraine associated pain.
Keywords
Introduction
Migraines are recurrent episodes of throbbing headache accompanied by nausea/vomiting, photosensitivity and sound sensitivity (1). Migraine affects more than 12% of the population in the United States, mostly females (2–4). The frequency of migraine attacks ranges from a few attacks each year to daily headaches in a large proportion of patients (1). Migraine headaches can result in significant impairment of quality of life and loss of productivity (5). Additionally, there is a significant healthcare expenditure for patients presenting to the emergency room with headaches (5). Current management of migraine headaches includes long-term prophylaxis with daily medication, followed by administration of rescue drugs during episodes of severe acute headaches (6,7). Various non-pharmacologic therapies have emerged to treat acute migraine attacks with limited success. Recent evidence suggests that sphenopalatine ganglion blocks using either lidocaine or cold exposure can reduce duration of migraine headaches (8).
Several clinical studies have clearly demonstrated the use of high-flow oxygen in cluster headache (9–14). In contrast, the evidence for the use of high-flow oxygen therapy in the treatment of acute migraine is still controversial, with some studies showing efficacy (13–15). Vasoconstriction of blood vessels in the scalp has been proposed as a possible mechanism of action. However, some researchers have suggested that the cooling effect of high flow dry oxygen may be in part responsible for the pain relief (16). A randomized clinical trial conducted in migraineurs using dry air and/or oxygen failed to show a difference in the primary end point of pain reduction between the two interventions (15). Furthermore, a recent study that made use of a device to deliver a pressured perfluorocarbons (PFC)/air mixture via intranasal catheter led to a significant reduction in headache, thereby demonstrating the benefits of evaporative cooling (17).
Based on these studies, we hypothesized that cooling might play a role in the pain relief provided by trans-nasal gas therapy. The overall objective of this study was to assess the relative contribution of cooling and oxygen therapy in alleviating pain of acute migraine headaches.
Methods
The study was conducted in the outpatient migraine clinic and the Institute for Clinical and Translational Research (ICTR) at the Johns Hopkins University. The study was approved by the Johns Hopkins Institutional Review Board (IRB00166706) and registered at ClinicalTrials.gov (NCT04129567).
Trial design
This single-center research study was designed as a randomized, placebo-controlled, parallel group, patient blinded superiority trial with unequal allocation ratios. The main aim of this study was to measure the efficacy of trans-nasal gas therapy in treating acute migraine headache episodes. This randomized controlled trial (RCT) sought to compare three different treatment arms to a control arm. Patients suffering from an acute headache episode were recruited at the headache clinic (eligibility criteria) if they i) had a definite diagnosis of migraine (>1 year), ii) suffered their first migraine episode before 50 years of age, iii) had at least one migraine attack per month, iv) had no changes in their migraine prophylaxis medications (daily oral drugs and monthly/quarterly biologics) for at least 3 months prior to enrolment, v) met International Classification of Headache Disorders, third edition (ICHD-3) criteria for diagnosis of episodic migraine (18), and vi) were able to attend a short treatment session right after their clinic visit. Patients were deemed ineligible if they a) had marked nasal septal deviation, b) had a history of recurrent epistaxis or chronic rhinosinusitis, c) were dependent on oxygen to maintain arterial oxygen saturation (SaO2) > 95%, and d) had concurrent sinus/intranasal surgery. A written informed consent was obtained and the risks and potential discomforts of the study explained.
Sample size determination
A good clinical response between the treatment and placebo arms was defined as an effect size of two units on the visual analog scale (VAS), with a common standard deviation of 1.5. Based on a comparison of the means of two independent groups with alpha = 0.05 and power = 0.80, employing a formula for a two-sided test 19, the sample size per study arm was determined to be equal to 10 subjects.
Patient recruitment
Patients who presented to the migraine clinic with an acute unremitting pain that was present at the time of the visit were included in the study. The exact time of onset of migraine headaches could not be controlled. All participants were recruited from Baltimore City/Baltimore County itself rather than different geographical areas as varying humidity levels could have affected the findings. Furthermore, the inlet conditions of the delivered air were carefully controlled so as to deliver either 100% humidified air or dry air (<3 parts per million). As a result, measuring the humidity of air was not deemed necessary.
Randomization and blinding
Simple randomization techniques (such as the flipping of a coin) led to the generation of treatment arms with unequal allocation ratios. This resulted in a skewed distribution of patients across different treatment groups. The treating neurologist and the research assistant evaluated eligibility together, obtained informed consent and enrolled participants. The entire random allocation sequence (list) was generated and held by an independent colleague situated off-site and otherwise uninvolved in the study. The investigators would contact this individual to enrol new participants and would subsequently be informed of the allocation arm at the time of the study. The ICTR research nurse opened up the sequentially numbered envelope containing the randomization assignment (the process of allocation concealment). Thus, the results of the allocation were completely concealed from the study personnel responsible for allocating patients to treatment arms until the moment of assignment. On the other hand, patients remained blinded to the interventions and the study hypothesis for the entire duration of the trial.
Data collection
Following an informed consent, a detailed patient history comprised past and current medications, and relevant medical and social history was taken. Baseline patient characteristics and Migraine Disability Assessment Scale (MIDAS) scores were recorded initially. The patients then completed a Visual Analog Scale (VAS) questionnaire rating their symptoms on a scale of 1–10 on a printed copy of the paper. The readings were subsequently measured using a ruler and the data was treated as continuous in nature. The following symptoms were rated on the VAS scale: Pain (headache), nausea, light sensitivity and sound sensitivity (noise sensitivity). The patient dataset was collected at different time points (baseline, 5 min, 15 min, 2 h and 24 h).
Interventions (gas therapy)
Subjects were consented for a study which tested two different concentrations of oxygen (21% oxygen at room air vs. 100% oxygen). Following an informed patient consent, subjects were randomized to receive either dry oxygen, dry air, humidified oxygen, or humidified air therapy (control) at flow rates of 15 L/min for 15 min. The independent variables were the type of gas used (oxygen or air) and the moisture content (dry or humidified) of the gas. Oxygen administration itself results in vasoconstriction of blood vessels and causes pain relief. However, the overall hypothesis of this proposal is that dry gas administered at a flow rate of 15 L/min will cause mucosal water evaporation with local cooling of the upper airways adjacent to the sphenopalatine ganglion, which in turn will reduce the pain of the migraine. Humidified air was used as a control intervention as it was devoid of both the treatment modalities that were being tested (i.e. oxygen and dryness). We have previously demonstrated that air temperature has a modest effect on cooling with better cooling noted using warm dry air as opposed to cool air (20). All gases (four different arms) were warmed to body temperature to avoid any potential bias as humidification of air itself involves warming of the gas.
A VapothermR device (Vapotherm Inc, USA) was used to warm up and/or humidify air/oxygen, which in turn was delivered via an Equate™ soft nasal cannula (Walmart Inc, USA) at 15 L/min. The Equate™ soft nasal cannula is a proprietary 62 cm long oxygen tubing (plastic) and is meant for single patient use. Air or oxygen from the cylinders was connected to a device via a long tubing and the cylinders were covered to blind the patients to the therapy. The device has a humidification element that was turned on or off based on the intervention. The specific intervention was chosen based on the results of the randomization assignment, delivered to a research nurse in an opaque envelope.
After collecting baseline questionnaires and VAS scores, the intervention was delivered to the patients in a seated position. Trans-nasal oxygen/air therapy was delivered through a nasal cannula. Nasal saline drops were provided to all patients and they were instructed to use it as needed. The VAS scores were noted at regular intervals after the start of the gas therapy (0, 5 min, 15 min and 2 h). The patients were provided with a 24-h scoring sheet at the end of the intervention and were subsequently followed up. Patients refrained from taking their acute anti-migraine medications until the end of the study period (24 h).
Primary and secondary endpoints
The primary endpoint was defined as a change in pain score at 2 h and 24 h post-therapy as assessed by the visual analog scale (VAS). Changes in nausea, photosensitivity and sound sensitivity were considered as measures of secondary endpoints and were similarly measured using the VAS. In addition, pain relief defined as a score of 0 or 1 at the end of 2 h was also evaluated. Relief from other symptoms such as nausea, photosensitivity and sound sensitivity were correspondingly defined.
Statistical analysis
Continuous variables were summarized as mean ± SD or median (interquartile range [IQR]) and categorical variables were presented as frequencies (percentages). Data was presented with 95% confidence intervals (95% CI) as appropriate. The changes in symptom scores for each therapeutic arm (dry oxygen, dry air and humidified oxygen) were compared with the control group (humidified air) using the Student’s t-test. Time-related changes in VAS scores were compared using the paired t-test. The independent effects of treatment components (gas type and moisture content) and their potential interactions on change in symptom score at 2 h were estimated using a linear regression model. Considering variable distribution, the baseline VAS scores were compared across arms using the Kruskal-Wallis test. The proportion of patients with symptom relief at 2 h were summarized and compared to the control arm using two-sided p-values derived from the Fisher’s exact test. Two-tailed p-values were calculated and p-values < 0.05 were considered statistically significant. IBM SPSS 25.0 was used to perform statistical analysis.
Results
A flowchart depicting the selection of participants through the clinical trial (CONSORT Statement) is shown in Figure 1. Patient recruitment and follow-up took place between September 2018 to September 2019. The baseline characteristics of patients randomized to individual treatment arms are presented in Table 1. Enrolment ceased when the targeted sample size was reached. Although 60 patients were eligible for this clinical study, nine patients declined to participate.

A flowchart depicting the selection of participants through the clinical trial (CONSORT Statement).
Baseline characteristics.

Fifty-one patients (48 ± 15 years of age, 82% females) were recruited. A majority of the patients presented with moderate-severe intensity migraines as indicated by recurring emergency department (ED) visits, elevated MIDAS scores (average score 69.2 ± 46.2) and the use of multiple anti-migraine drugs. Twenty patients were randomly assigned to the humidified oxygen arm, 11 to the dry air, 12 to the dry oxygen arm and the remaining eight patients were assigned to the humidified air arm, all of whom received the intended line of treatment. The analysis followed the intention to treat principle.
There were no significant differences between the four arms (dry oxygen/dry air/humidified air/humidified oxygen) in terms of the baseline pain scores (4.7 ± 1.2/4.4 ± 2.3/5.3 ± 1.9/5.5 ± 2.1, p = 0.38), baseline nausea scores (1.5 ± 1.5/1.2 ± 1.7/3.5 ± 3.4/2.5 ± 3.2, p = 0.50), baseline photosensitivity scores (4.3 ± 2.7/3.4 ± 2.6/5.1 ± 2.2/4.2 ± 3.4, p = 0.59) and baseline noise sensitivity scores (3.3 ± 3.7/3.0 ± 2.6/3.4 ± 3.0/3.1 ± 3.1, p = 0.99). Figure 2 is a schematic representation of mean symptom scores across study arms at baseline and after therapy.
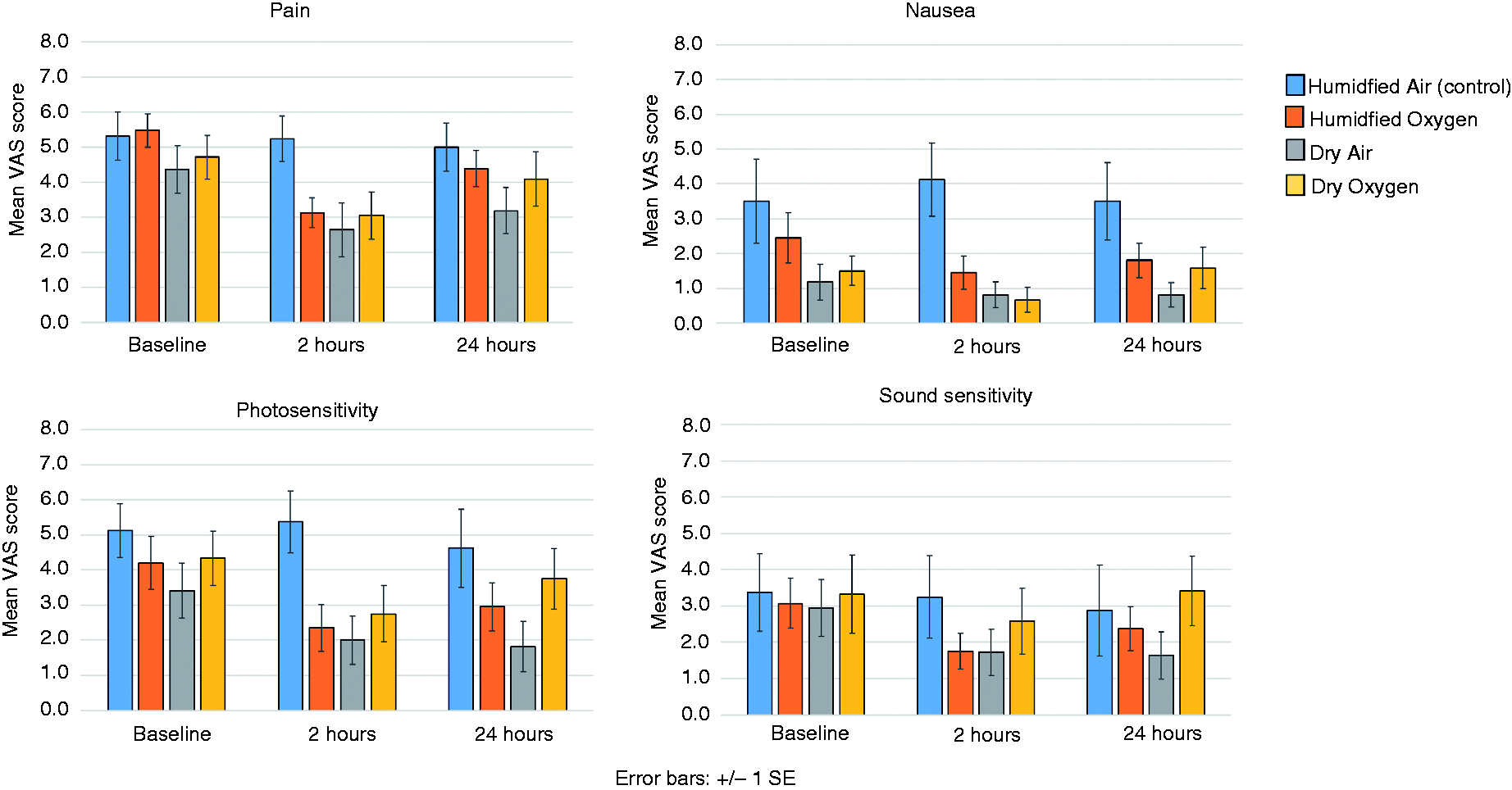
Schematic representation of mean symptom scores across study arms at baseline and after therapy.
Changes in pain scores (primary endpoint)
The mean pain scores of all arms at baseline, 2 h and 24 h are outlined in Table 2. Patients receiving humidified oxygen, dry air and dry oxygen showed a significant reduction in pain scores from baseline to 2 h post therapy (5.5 ± 2.1 to 3.1 ± 1.9, 4.4 ± 2.1 to 2.6 ± 2.5, and 4.7 ± 2.2 to 3.0 ± 2.3, respectively; p < 0.05 for all comparisons). In contrast, patients receiving humidified air did not demonstrate a significant reduction in pain scores during the same period (5.3 ± 1.9 to 5.2 ± 1.8, p = 0.763). A significant reduction in mean pain scores was also observed in patients treated with humidified oxygen and dry air at 24 h (5.5 ± 2.1 to 4.4 ± 2.3 and 4.4 ± 2.1 to 3.2 ± 2.2, respectively; p < 0.05 for both comparisons).
VAS scores before and after intervention for all study arms.
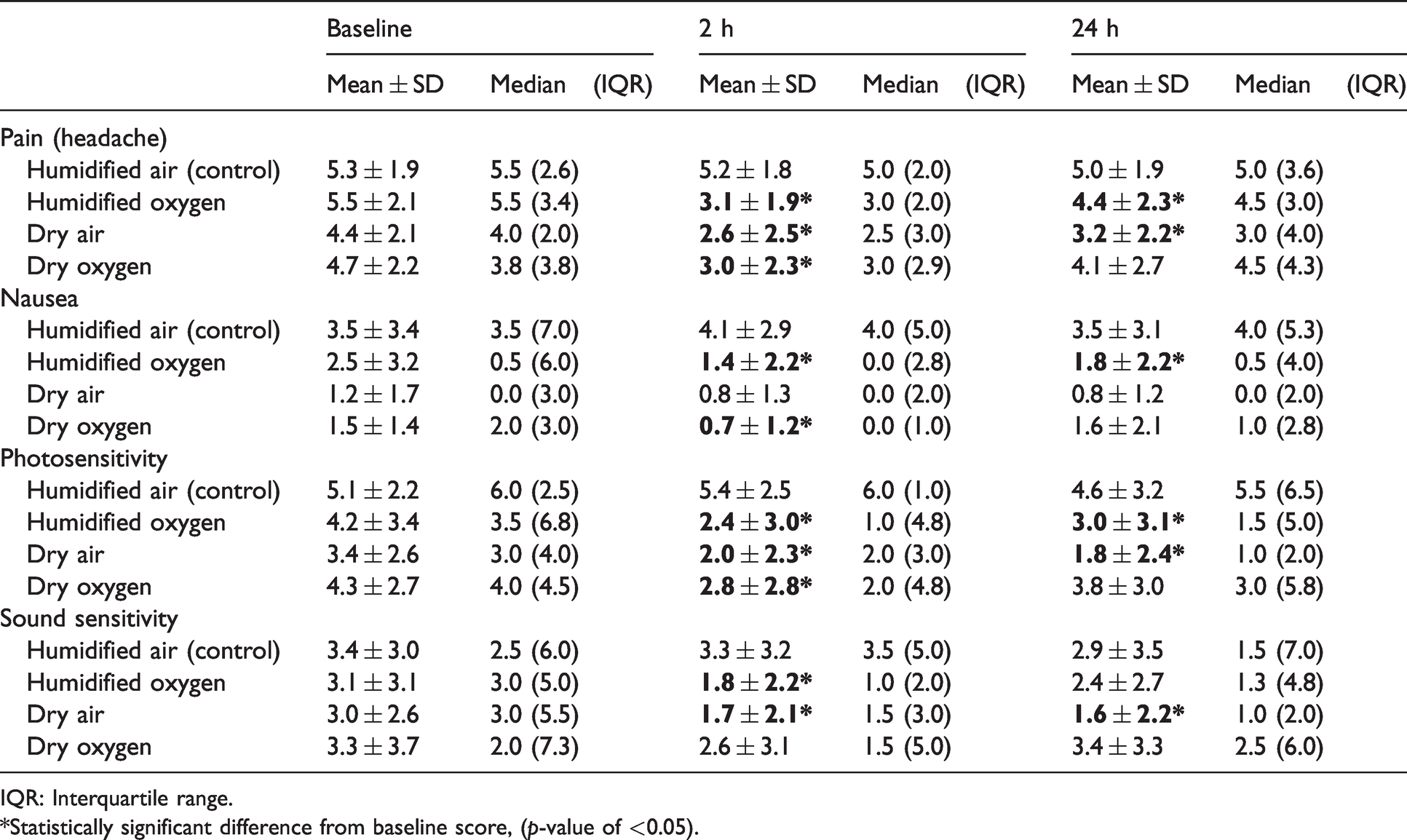
IQR: Interquartile range.
*Statistically significant difference from baseline score, (p-value of <0.05).
When compared to the control arm (humidified air), all therapeutic arms showed a significant reduction in pain scores (primary endpoint) after 2 h of therapy with dry oxygen (−1.6 [95% CI −2.3, −0.9]), dry air (−1.7 [95% CI −2.6, −0.7)]), and humidified oxygen (−2.3 [95% CI −3.5, −1.1]) (Table 3). The extent of change in mean pain scores (baseline to 2 h) was not significantly different between the three treatment arms.
Comparison of changes in symptom scores at 2 and 24 h between the treatment arms and the control arm (humidified air).

*Statistically significant based on t-test (p-value of < 0.05).Bold p-value of less than 0.05 is considered to be statistically significant.
None of the patients in the humidified air (control) group achieved pain relief at 2 h; however, 25% of patients in the dry oxygen group, 46% of patients in the dry air group, and 20% in the humidified oxygen group were relieved of pain at 2 h (Table 4).
Symptom relief at 2 h.

*p-values are based on 2-sided Fisher’s Exact test comparing the treatment arm to the humidified air arm (control).Bold p-value of less than 0.05 is considered to be statistically significant.
Changes in nausea, photosensitivity and sound sensitivity scores (secondary endpoint)
Nausea scores: A significant reduction in nausea scores was observed in the humidified oxygen and dry oxygen arms at 2 h compared to baseline (2.5 ± 3.2 to 1.4 ± 2.2 and 1.5 ± 1.4 to 0.7 ± 1.2, respectively; p < 0.05 for both comparisons) (Table 2). At 24 h, the nausea score reduction remained significant in the humidified oxygen group (2.5 ± 3.2 to 1.8 ± 2.2, p < 0.05). When compared to the control arm, a significant reduction in the mean nausea score was only noted in the dry oxygen arm at 2 h (−1.5 [95% CI −2.7, −0.2]. No significant change was observed at 24 h (Table 3).
While only 25% of patients in the humidified air (control) arm reported a relief of nausea symptoms at 2 h, over 70% of patients in all other treatment arms experienced relief from nausea (Table 4).
Photosensitivity scores: All arms except humidified air (control) experienced a significant reduction in photosensitivity scores at 2 h compared to baseline (4.2 ± 3.4 to 2.4 ± 3.0 [humidified oxygen]; 3.4 ± 2.6 to 2.0 ± 2.3 [dry air]; 4.3 ± 2.7 to 2.8 ± 2.8 [dry oxygen]; p < 0.05 for all comparisons). This benefit persisted in the humidified oxygen and dry air arms at 24 h (4.2 ± 3.4 to 3.0 ± 3.1 [humidified oxygen]; 3.4 ± 2.6 to 1.8 ± 2.4 [dry air]; p < 0.05 for all comparisons) (Table 2). When compared to the control arm, significant reductions in photosensitivity scores were observed in all active treatment arms at 2 h (−2.1 [95% CI −3.6, −0.6], humidified oxygen; −1.7 [95% CI −2.9, −0.4], dry air; −1.8 [95% CI −3.2, −0.4], dry oxygen) (Table 3).
Relief from photosensitivity was observed in 13% of patients in the humidified air arm (control) whereas the dry oxygen, dry air and humidified oxygen arms achieved a 42%, 46% and 55% improvement in symptoms (respectively) (Table 4).
Sound sensitivity scores: The humidified oxygen and dry air arms achieved significant reductions in sound sensitivity scores at 2 h compared to baseline (3.1 ± 3.1 to 1.8 ± 2.2 and 3.0 ± 2.6 to 1.7 ± 2.1, respectively; p < 0.05 for both comparisons) (Table 2). This significant reduction persisted at 24 h in the dry air arm (3.0 ± 2.6 to 1.6 ± 2.2; p < 0.05). When compared to the control arm, no significant benefit was observed with active treatment (Table 3).
In the humidified air arm, 38% of patients achieved relief from sound sensitivity at 2 h, while the dry oxygen, dry air and humidified oxygen arms experienced symptomatic relief in 50%, 55% and 60% patients (respectively) (Table 4).
Interaction between gas type and humidity
An estimation of the effects of individual treatment components and their interactions on symptom scores at 2 h was performed by employing a linear regression model (Table 5). The type of gas used (i.e. oxygen compared to air) significantly reduced pain, nausea and photosensitivity scores by 2.28, 1.62 and 2.10 points respectively at 2 h. Similarly, moisture content (i.e. dryness compared to humidification) was associated with significant reductions of pain and photosensitivity by 1.65 and 1.66 points respectively.
Linear regression estimates of the effect of treatment components and their interaction on change in symptom scores at 2 h.
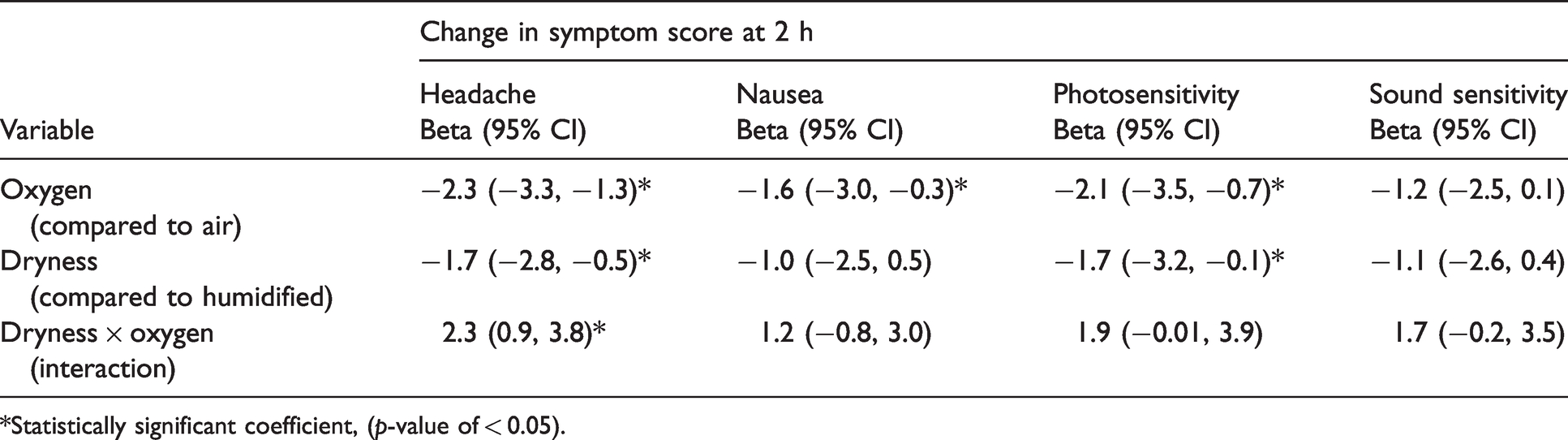
*Statistically significant coefficient, (p-value of < 0.05).
With regard to pain scores, the expected additive reduction with the combined use of both treatment modalities, dryness and oxygen, was 3.93. However, the actual reduction observed in pain score at 2 h was 1.6. This reflects the antagonistic interaction between gas type and moisture content (i.e. oxygen and dryness) (Table 5).
Adverse events and protocol deviations
The trial was conducted in accordance with the original protocol. All study participants completed the entire course of the study without any interruptions and were included in the analysis. There was no missing data or protocol deviation. No significant adverse effects were recorded in any intervention arm. None of the participants received anti-migraine drugs during the oxygen/air therapy. All study participants completed the 24-h post-treatment follow-up.
Discussion
We evaluated the effect of trans-nasal cooling in reducing symptoms in acute migraine headaches. All three treatment arms (dry oxygen, dry air and humidified oxygen) showed a significant reduction in pain scores at the end of 2 h, wherein significant pain relief at 2 h was achieved only in the dry air arm. The results of our randomized blinded study suggest that both the cooling effect of the dry air and the vasoconstrictive effects of oxygen may contribute to pain relief in acute migraine headaches. High-flow trans-nasal air cooling/oxygen therapy was well tolerated and there were no significant adverse events.
Up until 1980s the prevalent theory on migraine pathogenesis was initial vasoconstriction (focal cerebral ischemia/transient neurologic symptoms) and later vasodilation of the blood vessels supplying the brain (throbbing headache) (21). The advent of vasoconstrictors such as triptans and ergotamine strengthened the vascular theory initially, but soon it became evident that it was not true (22). Subsequently, the focus of attention was directed towards the components of the migraine circuit such as the trigeminal nucleus, trigeminal ganglion, hypothalamus, thalamus, pons and the periaqueductal gray matter; it was hypothesized that migraine triggers could interfere with the overall balance of the neuronal circuit concerned with modulation of sensory activity and precipitate a migraine attack (23,24). Increasingly, the release of calcitonin gene-related peptide (CGRP) from the neurons of the trigeminal ganglion is considered a plausible mechanism for triggering a migraine attack (25). Recent studies consider a wave of cortical spreading depression (CSD) with trigeminal nerve activation as a more plausible explanation in migraine with aura (26–29). Additional evidence suggests that CSD is in some way directly connected to tissue hypoxia, neuronal swelling and subsequent impaired neurovascular/neurometabolic coupling (30,31). Other less well accepted theories include sensitization and micro-embolic episodes contributing to migraine (32,33).
Some of the current therapies for migraine reverse vasodilation. Triptans are 5-HT1B/1D receptor agonists and act by promoting vasoconstriction as well as by inhibiting the stimulated release of CGRP (CGRP blockade), thereby blocking pain circuits in the brain (25). Hundred percent oxygen causes cerebral vasoconstriction and current guidelines have clearly demonstrated the role of oxygen as being a safe and effective agent for aborting attacks of acute cluster headaches (10–15,34). However, the role of oxygen in terminating acute migraine episodes was largely unknown. In 1930, Wolff and Lenox demonstrated that hyperoxia has vasoconstrictive effects on cerebral arteries in patients with migraine/aura. The initial set of experiments conducted in mice using 100% oxygen did not suppress CSD (35). More recently, experiments conducted by Shin et al. (36) found that peri-infarct spreading depressions were getting suppressed by 100% oxygen due to a reduction in CSD susceptibility at the ischemic penumbra. Additionally, normobaric oxygen therapy in rodent studies was found to be significantly effective in reducing brain injury following episodes of transient ischemia (37). Oxygen might thus help alleviate at least some of the causative factors in migraine by causing vasoconstriction, promoting greater oxygen utilization/decreasing tissue hypoxia in the brain and reducing peri-infarct spreading CSDs (35–39). Subsequently, Ozkurt and Cinar demonstrated the efficacy of high-flow oxygen therapy in aborting acute attacks of migraine (among other headache types) in an emergency department setting (14). In addition, Singhal and colleagues conducted a randomized, cross-over design placebo-controlled trial whereby adult migraine patients self-administered high-flow oxygen/medical air using face masks during attacks of acute migraine (15). The results were interesting in that there was no statistically significant difference in the mean change in pain scores between the oxygen and air-treated arms, although both study arms achieved a substantial relief of symptoms. Dry gas was directly used from the tank and as such the possibility of cooling of the nasal mucosa with subsequent relief of pain by the dry air was postulated. Recently a pilot study conducted by Vanderpol and colleagues using trans-nasal evaporative cooling resulted in significant relief of headache and other symptoms of migraine (17). Trans-nasal evaporative cooling in migraine might help attenuate trigemino-vascular system activation via the sphenopalatine ganglion (29). Additionally, temperature changes may be transmitted from the mucosal of nasal sinuses to the dura mater (meningeal coverings) of the brain via venous blood. This might aid in vasoconstriction of the blood vessels supplying the brain, reduce the release of inflammatory mediators and attenuate overall inflammation/local edema (17). Cooling also preferentially affects conduction mediated by small myelinated fibers (which act as pain afferents) via a gating mechanism and might help relieve pain (17).
Our randomized clinical trial confirms and extends the results of previous studies. One of the interesting observations of our study is that dry air was more effective than dry oxygen in terms of pain score reduction/pain relief. Surprisingly, we did not see an additive or synergistic effect of cooling and hyperoxygenation. It is quite possible that oxygen and air cooling act via similar pathways to reduce migraine symptoms, or vasoconstriction might impair the cooling efficacy by reducing the mucosal blood flow, although this is highly speculative. This can partly explain why the dry oxygen arm results were not superior to those of the dry air/humidified oxygen arms. In addition, a statistically significant interaction indicates that the effect the gas type has on pain score reduction is dependent on the level of the moisture content of the gas. We did not perform test dosing or duration effects, as this would have significantly complicated the study design.
Study limitations
The single-blinded nature of this study makes it susceptible to experimenter bias. However, patients were not directly asked questions; rather, they filled out a form and were blinded to the study hypothesis and the nature of the intervention. An error in the randomization procedure resulted in unequal allocation ratios between the groups, which was not realized until the study enrolment was completed. However, this did not materially affect the results of the study. Although it ultimately led to a higher number of patients in the humidified oxygen arm, clear differences were still observed between the therapeutic and control groups. Considering prior planning of the main analyses, no additional adjustments were made for multiple testing. However, this constitutes a major limitation of this study. Another major limitation of this study was the relatively small size of this sample study (n = 51). Further studies are required to determine whether results can be replicated across large, diverse patient groups.
Conclusion
Trans-nasal dry gas (air or oxygen) at a flow rate of 15 L/min is safe, feasible, and may be a potentially effective intervention in the acute treatment of migraine. The effect is mitigated by humidifying the air, suggesting that cooling of the mucosa might play a role in this effect. Trans-nasal cooling was also tolerated well and there were no documented adverse effects. Trans-nasal evaporative cooling can therefore be used as an effective adjunctive therapy in the treatment of migraine. However, further studies are required to clarify the optimal flow rate and the duration of this therapy for producing a maximal effect.
Clinical implications
Trans-nasal high flow dry gas results in significant reduction in headache scores and migraine associated symptoms. Further studies are required to elucidate the optimal flow rate/duration of this therapy for maximal effect. If established, trans-nasal cooling therapy using air may be a simple, inexpensive and widely available adjunctive treatment for acute migraine.
Footnotes
Ethics committee approval and informed consent
A written informed consent was obtained from all individual participants included in the study. All procedures performed in this study involving human participants were approved by the Johns Hopkins Research Ethics Committee.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The current study was funded by internal grants from the Institute for Clinical and Translational Research (ICTR) at the Johns Hopkins Bayview to the PI (NT).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HT is the co-founder of CoolTech LLC, which is developing a trans-nasal cooling device for use in out-of-hospital cardiac arrest and for neurogenic fevers and is funded by an NHLBI SBIR grant (R44HL108542). He was involved in the concept and design of the study but was not involved in direct patient interaction, consenting and conduct of the study.
