Abstract
Background
Functional connectivity of brain networks may be altered in migraine without aura patients. Functional magnetic resonance imaging (fMRI) studies have demonstrated changed activity in the thalamus, pons and cerebellum in migraineurs. Here, we investigated the thalamic, pontine and cerebellar network connectivity during spontaneous migraine attacks.
Methods
Seventeen patients with episodic migraine without aura underwent resting-state fMRI scan during and outside of a spontaneous migraine attack. Primary endpoint was a difference in functional connectivity between the attack and the headache-free days. Functional connectivity was assessed in four different networks using seed-based analysis. The chosen seeds were in the thalamus (MNI coordinates x,y,z: right, 22,–24,0 and left, –22,–28,6), pons (right, 8,–24,–32 and left, –8,–24,–32), cerebellum crus I (right, 46,–58,–30 and left, –46,–58,–30) and cerebellum lobule VI (right, 34,–42,–36 and left, –32,–42,–36).
Results
We found increased functional connectivity between the right thalamus and several contralateral brain regions (superior parietal lobule, insular cortex, primary motor cortex, supplementary motor area and orbitofrontal cortex). There was decreased functional connectivity between the right thalamus and three ipsilateral brain areas (primary somatosensory cortex and premotor cortex). We found no change in functional connectivity in the pontine or the cerebellar networks.
Conclusions
The study indicates that network connectivity between thalamus and pain modulating as well as pain encoding cortical areas are affected during spontaneous migraine attacks.
Introduction
Migraine is a multiphasic disorder that is characterised by recurrent attacks of head pain (1) and multiple sensory symptoms (2). The pathophysiological processes that constitute a migraine attack are not fully understood. Clinical features, such as premonitory symptoms (3) and central sensitisation (4), suggest altered function of the central nervous system (CNS) during migraine attacks.
Neuroimaging evidence accumulated over the past decade indicates abnormal function of specific brain areas in migraine patients during and outside of attacks compared to healthy controls (5). More recently, the neuroimaging focus has advanced from alterations of single regions of interest (ROIs) to delineate changes in the complex intrinsic architecture of the brain (6). Thus, resting-state functional magnetic resonance imaging (rsfMRI) has emerged as a powerful, non-invasive technique for investigating abnormalities of intrinsic brain connectivity (7). Studies utilising rsfMRI reported abnormal thalamocortical functional connectivity in migraineurs compared with healthy controls (8,9). One study suggested decreased connectivity between the thalamus and the visuospatial system in migraine patients compared with healthy controls (8). Another study associated altered connectivity between the thalamus and several cortical and subcortical areas with cutaneous allodynia in migraine without aura compared with healthy volunteers (9). Notably, these studies compared patients with healthy controls. Therefore, possible specific migraine attack-related functional connectivity patterns are unknown (i.e. patterns not found in general pain conditions).
The dorsal pons has been suggested as the generator of a migraine attack (10), while the cerebellum has emerged as a modulator of pain (11). Most recently, a significant role of the cerebellar connectivity in trigeminal nociception has been suggested (12). However, functional connectivity of the dorsal pons and cerebellum has never been investigated during migraine attacks.
Here, we investigated the network connectivity of thalamic, pontine and cerebellar ROIs during spontaneous attacks of migraine without aura. We hypothesised that functional connectivity is altered during migraine attacks compared to outside of attacks.
Methods
Study design and participants
We recruited participants via advertisements on a Danish website for recruitment of volunteers for health research (http://www.forsoegsperson.dk) and via announcement on the intranet for hospital workers in the Capital Region of Denmark. Patients were eligible for inclusion if they were aged 18–65 years and had a verified diagnosis of episodic migraine without aura in accordance with the International Classification of Headache Disorders-II criteria (13). Exclusion criteria were as follows: tension-type headache more than five days per month; a history of any other primary headache; pregnant or breast-feeding women; contraindication for MRI; signs or verified diagnosis of any cardiovascular or cerebrovascular disease; uncontrolled (i.e. active) psychiatric disorder or drug abuse; and if the participant did not accept information about potential incidental findings during the experiment. Enrolment was done at Glostrup Hospital from 2012 to 2014. The first patient was scanned in May 2013 and the last patient in September 2014. The present study was a substudy of a larger study investigating the blood–brain barrier permeability in migraine with (14) and without aura (15).
The Ethical Committee of Copenhagen (H-3-2012-073) approved the study. All participants gave their written consent after receiving detailed oral and written information. The study was done in accordance with the Helsinki II Declaration of 1964, with later revisions.
The study was registered at ClinicalTrials.gov (NCT02202486).
Procedures
A member of the research team (FMA) instructed the study participants to call in as early as possible after the onset of a migraine attack. All patients were informed about the IHS migraine without aura criteria (13). In case of an attack, the participants were advised to travel to the hospital by a taxi or public transport (fare reimbursed) 4–24 h after the onset of attack. On arrival at the hospital, we performed MRI on a 3.0 Tesla Philips Achieva Scanner (Philips Medical Systems, Best, Netherlands) using a 32-element phase-array receive head coil. To minimise head movements, foam pads were placed in the head coil. High-resolution anatomical images were acquired using high-resolution T1-weighted (T1W) sequences (ADNI sequence). Functional imaging was performed using a gradient-echo echo planar imaging sequence (32 slices of 4.0 mm thickness; slice gap, 0.1 mm; field of view, 230 × 230 mm; in-plane resolution, 2.9 × 2.9 mm; repetition time, 3.0 s; echo time, 35 ms; flip angle, 90°; and SENSE (SENSitivity Encoding) factor 2; total scan duration, 600 s). The participants were instructed to keep their eyes closed during the scan, but to stay awake. We scanned all participants again on a headache-/migraine-free day. On this day, all participants had to be headache-free (48 h) and migraine-free (72 h) free. Painkillers or triptans were not allowed on and 48 h before both experimental days. All T1W images were assessed by a neuroradiologist (FW) for pathological abnormalities, which were not found in any of the patients included in the present study.
MRI data analysis
The entire data analysis was performed by co-author AH. Data were initially processed using FMRIB’s Software Library (FSL) including motion correction and spatial smoothing with a 5-mm full width at half maximal Gaussian kernel. In order to remove motion-related, scanner-related and physiological noise, we applied FSL’s ICA-based Automatic Removal of Motion Artifacts (ICA-AROMA) (16). We chose this method of data denoising because it is particularly suited for identifying motion artifacts (17) and since migraine patients could be expected to exhibit more movement during attacks, due to pain and discomfort, which could theoretically influence the results. Following this general denoising, white matter and cerebrospinal fluid was delineated by automated segmentation using FSL’s FAST (18). Using these segmentations, signals specifically from white matter and cerebrospinal fluid segmentations were extracted and removed from the data by including them as nuisance regressors in a general linear model (GLM). Nonlinear registration using FMRIB’s Nonlinear Image Registration Tool (FNIRT) was applied between the subject’s structural image and the Montreal Neurological Institute (MNI) standard space. We performed seed-based resting state correlation analyses using 5 mm (radius) spherical ROIs in the cerebellum crus I (MNI coordinates x,y,z: right, 46,–58,–30 and left, –46,–58,–30), cerebellum lobule VI (right, 34,–42,–36 and left, –32,–42,–36), dorsal pons (right, 8,–24,–32 and left, –8,–24,–32) and thalamus (right, 22,–24,0 and left, –22,–28,6). The cerebellar crus I and lobule IV are associated with pain processing (19) and the coordinates were determined using Cerebellar Atlas in MNI152 space after normalisation with FLIRT for FSL. The thalamic ROI (posterior parietal) was selected using the Oxford Thalamic Connectivity Probability Atlas for FSL. The pontine ROI (dorsal pons) was chosen manually using standard brain (MNI152, T1, 2 mm) in FSL. Average time courses were extracted from these seeds and fitted with a voxel-wise general linear model (GLM) consisting of an explanatory variable representing the demeaned extracted time courses. Statistical testing comparing the networks produced by the seeds in the ictal versus the interictal phase was performed using paired statistical testing by permutation-based non-parametric testing (20) (5000 permutations) with threshold-free cluster enhancement (21), family-wise error (FWE) corrected for multiple voxel comparisons. Correction for multiple seeds was not done. In addition, average connectivity maps were calculated separately for each state (ictal/interictal) and for each ROI. A one-sided FWE-corrected P < 0.05 was considered statistically significant.
Results
We recruited 74 patients, of whom 17 (15 women, 2 men) completed scans both during and outside of migraine attacks and were included in the final analysis (Figure 1). Median age was 37 years (age range 24–54 years), median duration of disease was 17 years (range 6–35 years) and median attack frequency was three per month (range 1–8 attacks). Thirteen (76%) of the 17 patients had usually unilateral pain, two (12%) reported bilateral headache and two (12%) had unilateral headache but with shifting sides from attack to attack. Median time from onset of attack to MRI examination was 6 h (range 4.0–15.5 h). Median time between attack and attack-free scans was 27 days (range 12–87 days). Characteristics of migraine attacks in 17 patients during the scan sessions are shown in Table 1. Four patients used migraine-preventive drugs. None of the participants reported a migraine attack within 72 h before or during 24 h after the baseline scan. The cerebellar and pontine networks are shown as Supplementary Figure 1. Patients did not exhibit more head motion during the ictal scan compared to the interictal scan (ictally 0.59 mm vs. interictally 0.57 mm absolute displacement, 95% CI of difference: –0.12 to 0.15 mm, P = 0.71, paired T-test).
Flow chart of the study. Characteristics of migraine attacks in 17 patients during the MRI scanning sessions. Verbal rating scale 0–10: mild = 1–3; moderate = 4–6; severe = 7–10.

Functional connectivity changes during migraine attacks
Baseline (attack-free day) thalamus connectivity networks are shown in Figure 2. The seed-based analysis comparing the ictal with interictal state revealed increased functional connectivity between the right thalamus and several brain regions (Figure 3a), including left superior parietal lobule (SPL), insular cortex, primary motor cortex, supplementary motor area and orbitofrontal cortex (Table 2). We also found decreased functional connectivity between the right thalamus and right primary somatosensory cortex (Figure 3b) and premotor cortex (Table 3). We found no change in connectivity between the left thalamus and any other brain area. In addition, there were no connectivity changes in the pons or cerebellum networks. Explorative analysis revealed no correlation between the connectivity changes and headache intensity or duration.
Baseline whole-brain intrinsic connectivity networks for seeds placed in the right and left thalamus. The blue spheres mark the seed location. R marks the right hemisphere side; x, y, and z gives MNI coordinates for the slices. Colour code gives the statistical significance expressed as P values (corrected for multiple comparisons). The image is thresholded to show statistically significant voxels only (i.e. P values between 0.05 and 0). Differences in intrinsic connectivity for seed placed in the right thalamus during migraine attacks compared to outside of migraine attacks. (a) Areas of increased connectivity during attacks. (b) Areas of decreased connectivity during attacks. R marks the right hemisphere side; x gives MNI coordinates for the slices. Colour code gives the statistical significance expressed as P values (corrected for multiple comparisons). The image is thresholded to show statistically significant voxels only (i.e. P values between 0.05 and 0). Peak locations, cluster size and significance values of brain areas with increased connectivity to the right thalamus in 17 patients with migraine without aura attacks compared to the interictal condition. MNI: Montreal Neurological Institute. P value: one-sided FWE-corrected. Peak locations, cluster size and significance values of brain areas with decreased connectivity to the right thalamus in 17 patients with migraine without aura attacks compared to the interictal condition. MNI: Montreal Neurological Institute. P value: one-sided FWE-corrected.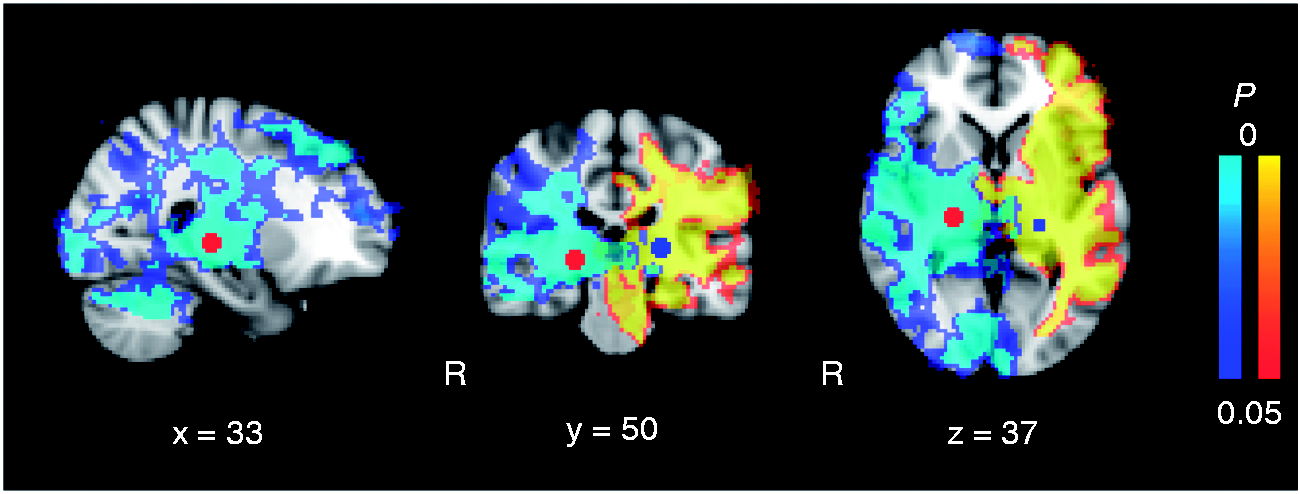



Discussion
The major finding of the present study was alteration of the right thalamocortical network during spontaneous attacks of migraine without aura relative to the interictal state. Collectively, areas responsible for pain modulation (e.g. insular and orbitofrontal cortex) and visual perception (i.e. superior parietal lobule) were more connected with right thalamus, whereas pain-encoding areas (i.e. primary somatosensory cortex) were less connected with right thalamus during attack.
A strength of the current study lies in self-controlled design (each patient served as his or her own control), which allows to compare functional brain connectivity during and outside of attacks. Moreover, this design minimises potential influence of migraine preventive drugs, which were used by four patients. We focused on the posterior parietal thalamus, which is the main relay station of ascending pain pathways travelling from peripheral sites to the cortex (22). We found altered connectivity between the right thalamus and several pain relevant areas. Lateralisation of head pain was almost equal between the two sides (right side, seven patients [41%] and left side, six patients [35%]). The median headache intensity was slightly higher in those with right-sided pain (VRS 8) compared with left-sided (VRS 6). Interestingly, pain syndromes are more common after right thalamic lesions (23,24) and the right somatosensory part of thalamus contains high concentration of norepinephrine (25), which is involved in intrinsic control of pain (26). Our network-based analysis revealed increased functional connectivity between the right thalamus and the contralateral superior parietal lobule, frontal orbital cortex (OFC), insula, primary motor cortex and the supplementary motor area. The fundamental question is whether these patterns of functional connectivity are migraine-attack specific. All areas with altered connectivity are involved in general pain processing or modulation, but some of the areas may be migraine specific. Thus, the SPL is responsible for visual perception (27) and 76% of our patients reported photophobia exclusively during migraine attacks. We further recorded an increased synchronicity of the OFC and thalamus during attacks. There is some evidence supporting a role of OFC in headache, but it is unlikely to be migraine specific. Increased activity in the OFC was reported in episodic migraine (outside of attacks) and acute myogenous facial pain patients (28). A PET study showed decreased glucose metabolism in OFC in episodic migraine patients who were efficiently treated with external trigeminal nerve stimulation (29). Furthermore, changed metabolism in the OFC has been reported in cluster headache (30) and in medication overuse headache (31). The insula has an important role in pain experience (32,33), as its activation and deactivation has been associated with pain. Patients with a lesion in the insula report elevated pain ratings on experimentally induced pain (34). Also, the primary and supplementary motor areas are involved in pain modulation (35). Increased excitability in these areas has been reported in phantom pain (36). Taken together, the present data suggest that increased connectivity during migraine attacks is likely due to increased modulation of the nociceptive information transmitted through thalamus.
We found that the connectivity was decreased between the right thalamus and the ipsilateral primary somatosensory cortex (S1) and premotor cortex. The S1 cortex is responsible for pain processing, including encoding of the location, quality and duration of pain (37–39). The S1 receives nociceptive signals from trigeminal afferents via the thalamus (40). The decreased connectivity (i.e. reduced synchronicity) between thalamus and right S1 may reflect uncoupling of S1 from the thalamic network. Thus, alteration of the thalamic modulation of trigeminal nociceptive input may contribute to pain perception during the attack. In support, the central pain syndrome is associated with increased activity of the S1 and suppressed thalamic inhibition of the ascending nociceptive inputs (41).
The present study did not include cognitive and emotional tests during and outside of the migraine attacks. We acknowledge that these factors might affect not only pain perception but also the connectivity networks. In addition, we did not include non-migraine controls and therefore, we cannot exclude possible interictal altered activity in the dorsal pons and cerebellum in patients compared to controls.
In conclusion, the present study indicates that network connectivity between the right thalamus and both pain modulating and encoding cortical areas are affected during spontaneous attacks of migraine without aura. Studies of functional connectivity during experimental, non-migrainous trigeminal pain are necessary to determine if these findings are migraine-specific. Moreover, future studies of the different phases of a migraine attack can reveal whether the network connectivity normalises before or after amelioration of the migrainous headache.
Clinical implications
The migraine attack is associated with change in the intrinsic brain connectivity. Change in thalamic network connectivity may be a marker of pain. Further studies may reveal whether altered thalamic networks are migraine-specific.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: FM Amin has received travel grant or payment for speaking from Novartis and Allergan; A Hougaard has received payment for speaking for Allergan; T Sprenger’s current or previous institutions have received payments for speaking or consultation from Eli Lilly, Allergan, Actelion, ATI, Novartis, Genzyme and Teva; M Ashina is a consultant or scientific adviser for Allergan, Amgen, Alder, ATI, Eli Lilly and Teva.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Lundbeck Foundation (R155-2014-171), the Novo Nordisk Foundation (NNF11OC1014333), University of Copenhagen, the Research Foundation of the Capital Region of Denmark (A4620) and the Danish Council for Independent Research-Medical Sciences (DFF-4004-00169B).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
