Abstract
Background
Characteristics, disabilities, and optimal management of medication overuse headache remain uncertain. This study aimed to elucidate the clinical characteristics of patients with medication overuse headache enrolled in a medication overuse headache registry in Korea.
Methods
The Registry for Load and Management of MEdicAtion OveruSE Headache (RELEASE), a cross-sectional prospective observational study including seven referral headache centers in Korea, started enrolling adult patients with medication overuse headache in April 2020. Data included information on headache characteristics, burden on daily function, depression, anxiety, history of acute and preventive medications, and treatment strategies.
Results
A total of 229 patients (85.6% females; mean age, 45.5 ± 13.5 years) were enrolled by June 2021. The average durations of chronic headaches and medication overuse were 6.5 and 4.3 years, respectively. In the past month before enrollment, patients had headaches for 25 days and severe headaches for 12 days, and used acute medications for 20 days. Patients were disabled in 66.8 days in the past 3 months and had moderate/severe depression and anxiety in 56% and 35%, respectively. The proportion of patients on preventive treatments increased from 38% to 93% during the study period.
Conclusions
RELEASE study reflects the current management status and opportunities to improve the quality of care in patients with medication overuse headache.
Introduction
Medication overuse headache (MOH) is a challenging chronic disorder due to its relatively high prevalence (1–2%) in the general population (1,2) and its negative impact on individual and socioeconomic aspects (3,4). Thus, the 2015 Global Burden of Disease study ranked MOH among the top 20 disorders causing disability measured by years lived with disability (5).
Despite growing concerns and interest, several aspects of MOH management are a focus of long-standing debate, including the need and optimal duration of withdrawal therapy, role of steroids, and effective preventive medications. The European Headache Society recommends simultaneous implementation of education on MOH, preventive treatment, and withdrawal of overused medications (6), whereas Danish guidelines recommend initiation of preventive treatment only if needed after discontinuation of the causative medication for 2 months (7). Several clinical trials investigating MOH treatment did not yield a consensus on optimal strategies (8–12). Although a well-designed cross-sectional observational study (13) has shown numerous findings including clinical characteristics, outcomes, and burden in patients with MOH, the study was conducted exclusively in Europe and Latin America and did not include treatment with botulinum toxin A or anti-calcitonin gene-related peptide (CGRP) monoclonal antibodies. Few single-center, retrospective observational studies have addressed MOH in Asian populations (14–17).
We established a multicenter cooperative registry termed Registry for Load and Management of Medication Overuse Headache (RELEASE). The principal objective of RELEASE is to improve headache research and identify more effective, tolerable, and practical approaches in patients with MOH using data from real-world clinical practice.
In this report, we provide a summary of the key clinical features of 229 patients enrolled in the first year of enrollment in RELEASE.
Methods
Study design and population
RELEASE is a nationwide, prospective, multicenter registry that aims to enroll 500 consecutive patients with MOH visiting one of seven academic or regional headache centers in Korea from 1 April 2020 to 31 December 2021 (Figure 1). The RELEASE-MOH study group was established in January 2020 to facilitate multicenter collaborative clinical research for MOH in Korea, provide epidemiological profiles and current MOH management status, and evaluate practical and effective approaches to manage MOH. The local institutional review boards of all participating centers approved the study (Approval no. of the main center: Dongtan 2020-02-004). All patients provided written informed consent to participate in the study.
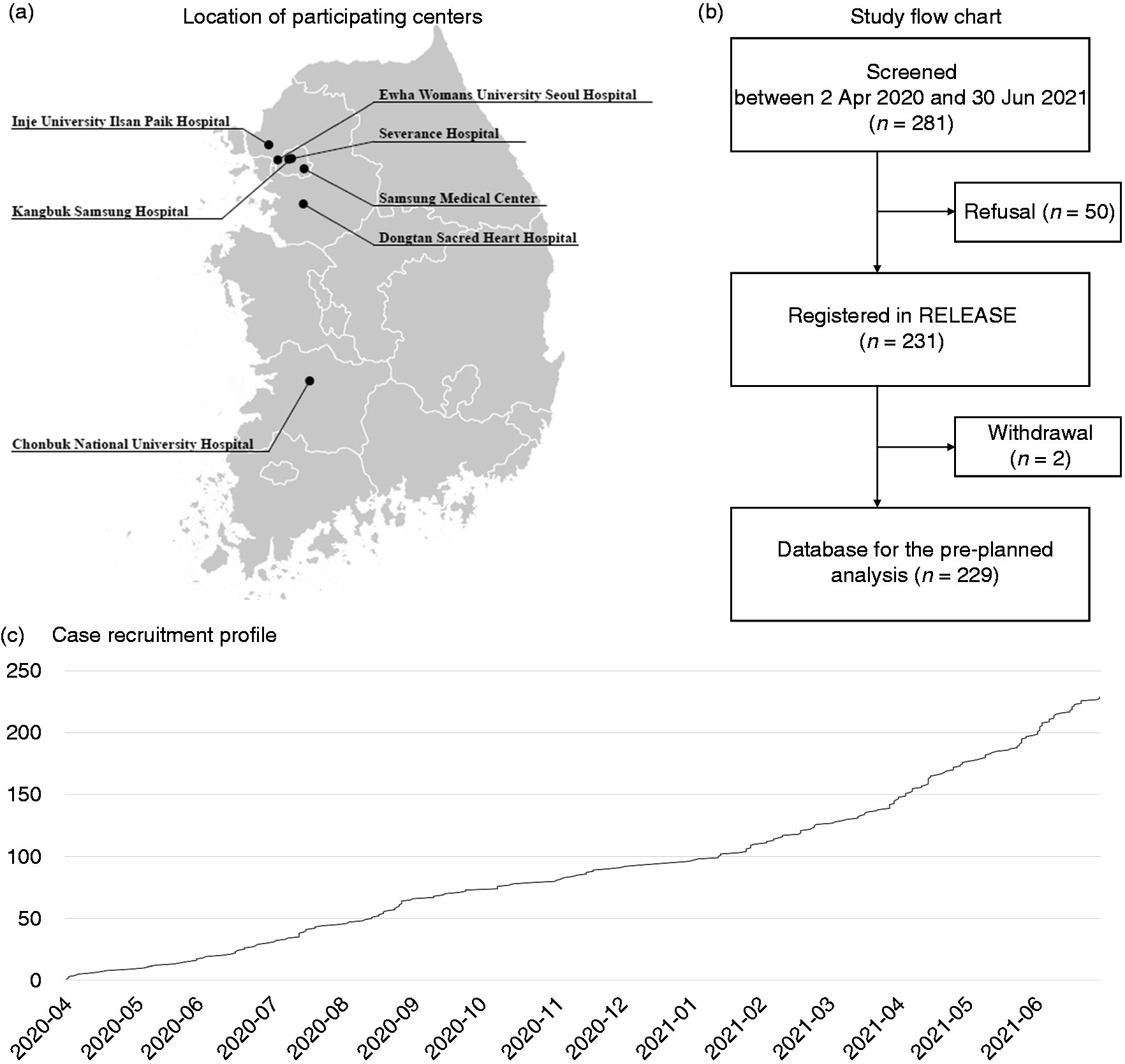
Registry for Load and management of Medication Overuse headache (RELEASE). (a) Locations of participating centers, (b) study flowchart, and (c) case recruitment profile.
On first visit, all patients were screened to ensure they met the eligibility criteria, which included age ≥19 years, willingness to provide informed consent, diagnosis of a primary headache disorder, and fulfillment of one of the following conditions: a) diagnostic criteria for MOH according to the International Classification of Headache Disorders, 3rd edition (ICHD-3) (18), b) use of migraine-specific drugs, combination analgesics, or opioids for symptomatic treatment of headache for ≥10 days in the past month, and c) use of simple analgesics for ≥15 days in the past month. Although the primary purpose of this registry is to obtain data on patients with MOH, there are some reasons we also decided to recruit the patients who overused acute medication for the past 1 month. First, physicians usually educate the patients about the risk of medication overuse headache and start preventive therapy, even if the patients overuse acute medication for just 1 month. Second, recent clinical trials evaluating the effect of anti-CGRP monoclonal antibodies have also performed the subgroup analysis in patients with medication overuse for the past 1 month (19–21). Patients who refused to participate in the study or withdrew their consent before completing questionnaires and case report forms were excluded.
Data collection
The RELEASE database comprised information on demographics including age, sex, body mass index, marital status, educational level (uneducated or illiteracy, ≤6 years, 7–9 years, 10–12 years, or >12 years), social history including smoking status (currently smoking, ever smoked, or never smoked), alcohol consumption, and amount of caffeine intake/day in the past 3 months (never, ≤200 mg/day, ≥200 mg/day). Data on age of headache onset, conversion to chronic daily headache, and overuse of acute medications were also collected. Data on the approach of discontinuation (admission to hospital, outpatient clinic, or self-discontinuation) and outcomes were also collected from patients who tried to quit overused medications. Data were also collected regarding the number of visits to a pharmacy or clinic, admission to hospital for headache treatment, and visits to an emergency department. Data on the type (computed tomography or magnetic resonance) and number of neuroimaging studies that had been administered to the patients were also collected.
At first visit, patients provided information on the durations of headache, severe-intensity headache, and acute medication use for headache in the past 30 days, 31–60 days, and 61–90 days, with guidance provided by the research team. All patients were queried regarding any acute and preventive medications used (duplicate selection allowed). The type of symptomatic therapy for headache relief and days of use for each therapy were collected and categorized into six groups as follows: Ergotamine (1 g ergotamine with 100 mg caffeine is the only formula available in Korea), triptans, simple analgesics (acetaminophen, aspirin, naproxen, ibuprofen, and others), opioids, combination analgesics (acetaminophen/tramadol, over-the-counter pill with caffeine, over-the-counter pill without caffeine, and others), and other acute medications. Whether the patients were on preventive therapy were queried, and the category, effect, and tolerability of each preventive therapy were recorded. The categories of preventive therapies were as follows: Antiepileptic drugs (AED), beta-blockers, calcium channel blockers, tricyclic antidepressants (TCA), serotonin-norepinephrine reuptake inhibitors (SNRI), angiotensin receptor blockers/angiotensin-converting enzyme inhibitors (ARB/ACEi), botulinum toxin A, monoclonal antibodies for CGRP (galcanezumab is the only anti-CGRP monoclonal antibody available in Korea as of June 2021), transcutaneous electrical nerve stimulation, greater occipital nerve block, and others. The effects of each preventive therapy were also measured based on the proportion of reduction in headache days: ≥50%, 30–49%, ≤30%, no change or worsening, and unknown/unmeasurable.
Corresponding physicians selected one or more of primary headache types and one category for chronic daily headache (if applicable) according to the ICHD-3 (18). Since the most common category of chronic daily headache that MOH patients have is chronic migraine (13), we collected the components of diagnostic criteria for migraine.
Data on the following comorbid conditions were also collected: Hypertension, diabetes mellitus, hyperlipidemia, heart disease, kidney disease, hepatic disease, gastric ulcer disease or reflux esophagitis, depression, fibromyalgia, and herniated disc in cervical or lumbar spine. In patients with neck pain/discomfort, data were collected on its degree, duration, and severity as well as its relationship with the headache.
The following information on discontinuation strategies utilized in cohort patients were collected: Method of discontinuation of overused medication (abrupt discontinuation, tapering down, or maintenance), location of treatment (inpatient or outpatient), and medication class and timing of initiation (not started, started at initial visit, or started ≥2 weeks after initial visit) of preventive therapy.
Questionnaires
A set of nine structured questionnaires was used to obtain detailed data on clinical features including depression, anxiety, stress, allodynia, efficacy of acute treatment, and quality of daily life. The impact of headache on functional ability at work, school, and home was measured using the previously validated Korean version of the Headache Impact Test-6 (HIT-6) (22). The sum of scores in HIT-6, ranging from 36 to 78 points, was used to categorize impact as follows: 36–49, little/no impact; 50–55, some impact; 56–59, substantial impact; and 60–78, severe impact. The number of affected days was evaluated using the Korean version of the Migraine Disability Assessment (MIDAS) (23). The Korean version of the Patient Health Questionnaire-9 (PHQ-9) (24) and Generalized Anxiety Disorder-7 (GAD-7) (25) scales were used to screen depression and anxiety, respectively. A total PHQ-9 score, ranging from 0 to 27, was used to define depression level as follows: 0–4, no/minimal depression; 5–9, mild depression; 10–14, moderate depression; 15–19, moderately severe depression; and 20–27, severe depression. For the GAD-7 scale, total scores ranged from 0 to 21, and scores of 0–4, 5–9, 10–14, and 15–21 defined no, mild, moderate, and severe anxiety, respectively. Overall health status, headache-related quality of life, and disability were assessed using the Korean versions of the EuroQol (EQ-5D) (26) and the Migraine-Specific Quality of Life Questionnaire (K-MSQ) version 2.1 (27). The EQ-5D descriptive system considers five dimensions: mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. Each dimension has three levels: No issues, some issues, and extreme issues (26). The equation with the time trade-off method was applied to transform responses to the EQ-5D, which ranged from −1 to 1 (0, equivalent to death; <0, worse than death; and 1, full health). The K-MSQ comprises 14 items that measure the impact of migraine on three dimensions (the role function-restrictive, role function-preventive, and emotional function dimensions) over the past 4 weeks. Each item was scored using a six-point Likert-type scale to calculate the total score for each dimension, which ranged from 0 to 100, with higher score meaning better quality of life (27). Psychological stress was assessed using the short-form Perceived Stress Scale (PSS-4) (28), and allodynia was assessed by the Allodynia Symptom Checklist-12 (ASC-12) with a score of ≥3 indicating the presence of allodynia (29). Efficacy of acute medication was evaluated by the Migraine Assessment of Current Therapy (migraine-ACT) questionnaire (30).
At each visit (1, 3, 6, and 12 months after the first visit), headache profiles, acute medication use days, disability in daily life, and number of visits to medical facilities in the past month were recorded. Additionally, neck pain/discomfort, caffeine intake, compliance with preventive therapy, and categories of acute and preventive therapies that were prescribed were recorded. The same set of nine structured questionnaires was filled by patients at all visits (Supplemental Table 1).
Outcomes
The primary outcome was number of patients without medication overuse for the past month at 3 months. The key secondary outcomes were a) change in the proportion of patients with a MIDAS score of ≥21; b) the proportion of patients with at least 50% reduction in a MIDAS score from baseline at each visit; c) change in the proportion of patients with a HIT-6 score of ≥61; and d) number of patients with at least 50% reduction among those with a HIT-6 score of ≥61.
Statistical analysis
For a sample size calculation for the present registry, we expected the proportion of patients without medication overuse for the past month at 3 months of 70% since corresponding outcomes in the previous studies were ranged 62–72% (8,13,31). The target sample size would be 341 when we set a two-sided 95% confidence interval with a width of 0.1. We assumed loss of follow-up rate at 3 months of 32% which is higher than that of previous studies (13–20%) (8,13,32) since there is no obligation to participate in the present study. Therefore, enrollment sample size was estimated as 500. The present study was planned for database locking and data cleaning to address issues and queries and to improve data quality after the first year of recruitment, and the next database cleaning will be performed after the recruitment of approximately 400 patients. To estimate the rate of primary outcome at 3 months, we only included the patients who were enrolled in this registry until March 15, 2021 which is 3 months prior to the date of database locking (30 June, 2021). We also compared the rate of primary outcome according to the detoxification methods.
Results
Among 281 patients who met the eligibility criteria, 50 (17.7%) patients were unwilling to participate in the study. Therefore, the registry comprised 231 patients from seven headache centers in Korea between 2 April 2020 and 30 June 2021. The present descriptive study included the data of 229 patients after the removal of two patients who withdrew their consent (Figure 1).
Patient demographics
The key demographic characteristics at baseline are presented in Table 1. The mean age at enrollment was 45.5 ± 13.5 years, and 196 patients (85.6%) were female. The proportions of patients with lifetime education of >12, 10–12, and 7–9 years were 59%, 28.4%, and 9.6%, respectively. Thirty-eight percent of the patients took <200 mg caffeine per day whereas 21.0% of the patients had >200 mg caffeine per day.
Baseline characteristics of the study population.

Note: Data are presented as numbers (%) or mean ± standard deviation, unless otherwise indicated.
Headache characteristics
In the study cohort, 207 patients (90.4%) were diagnosed with MOH and 22 patients overused acute medications for the past month before enrollment but did not meet the ICHD-3 criteria for MOH. Most of the patients (99.1%) had chronic migraines, whereas the remaining two patients (0.9%) had chronic tension-type headaches. In the past 30 days before enrollment, the patients experienced an average of 25.5 ± 5.5 headache days with 10.8 ± 7.9 severe headache days, and 48.9% of the patients experienced headaches every day. Overall, these data were comparable to those reported in large studies conducted in the USA, Europe, and Latin America (Table 2). The mean age of headache onset was 24.4 ± 12.5 years. The mean durations of chronic daily headache and medication overuse were 6.5 and 4.3 years, respectively (Table 2).
Comparison of headache characteristics of the current study cohort with previous studies on medication overuse headache.
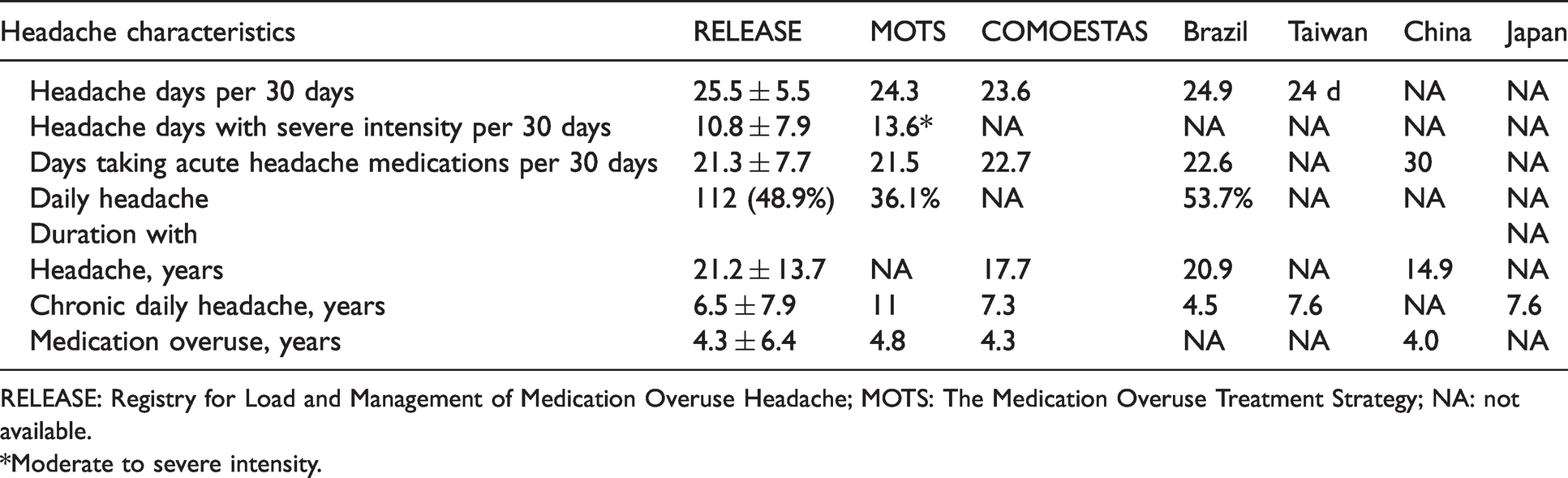
RELEASE: Registry for Load and Management of Medication Overuse Headache; MOTS: The Medication Overuse Treatment Strategy; NA: not available.
*Moderate to severe intensity.
Sixty-nine patients (30.1%) have tried to quit overused medications, and only 10% succeeded for more than two consecutive months. Among 227 patients with chronic migraine, unilaterality, pulsating quality, moderate or severe pain intensity, and aggravation by or causing avoidance of routine physical activity were reported by 62.1%, 83.7%, 96.5%, and 81.5% of the patients, respectively. Nausea or vomiting, sensitivity to light and sound, and aura were reported by 88.5%, 67.4%, and 8.8% of patients.
Headache medications at baseline
In the study cohort, acute medications for headaches were used for 21.3 ± 7.7 days in the past month. The most common class of overused medications was simple analgesics, followed by triptans, combination analgesics, ergotamine/caffeine, and opioids (Table 3). Preventive treatment at baseline was used in 87 patients (38.0%) (Figure 2). AED (n = 58, 25.3%) were the most common preventive treatment, followed by beta-blockers (n = 33, 13.5%), TCA (n = 28, 12.2%), botulinum toxin A (n = 21, 9.2%), anti-CGRP monoclonal antibodies (n = 17, 7.4%), calcium channel blockers (n = 14, 6.1%), ARB/ACEi, (n = 5, 2.2%) and SNRI (n = 3, 1.3%) (duplicate selection allowed).
Comparison of acute medications at baseline among the current studies.

Note: Data are presented as numbers (%). Duplicate selection allowed.
*Actual numbers of the patients were estimated using the total number of the patients and percentage provided.
†Ergotamines.
References: MOTS: Headache 2021; 61: 351–362; COMOESTAS: J Headache Pain 2016; 17: 20, Carlsen et al.: JAMA Neurol 2020; 77: 1069–1078.
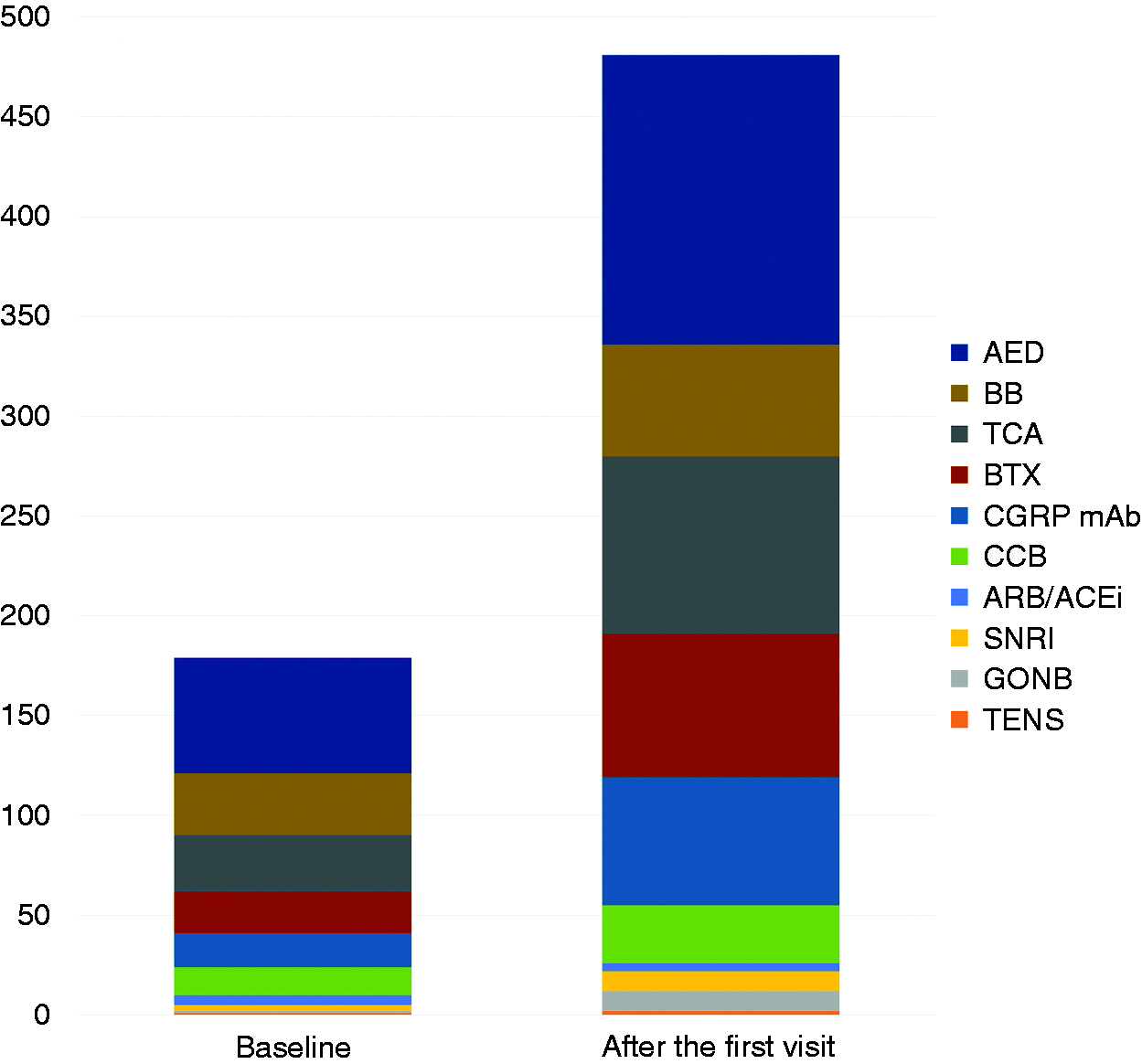
Comparison of proportions of preventive therapies between baseline and the first visit. Duplicate selection allowed, and 155 (72.8%) patients were on multiple classes of preventive therapies.
Comorbidities
Seventy-one patients (31.0%) reported at least one vascular risk factor. The rates of depression and fibromyalgia were 20.1% and 12.7%, respectively. Additionally, 64.2% of the patients reported neck pain/discomfort and neck pain/discomfort was usually present in the prodromal (36.7%) and headache (47.6%) phases.
Questionnaires
Nearly 90% of the patients were severely impacted by headache measured by the HIT-6 (Table 4). The patients stated that they experienced disability on two-thirds of the past 3-month period measured by the MIDAS score. Based on the PHQ-9 and GAD-7 scales, 55.9% and 35.3% of the patients had moderate/severe depression and anxiety, respectively. The mean PSS-4 score was 8.1 ± 3.0. Thirty-four percent of the patients experienced allodynia measured by the ASC-12. The mean migraine-ACT score was 2.4 ± 1.6. The overall health status and headache-related quality of life and disability measured by the EQ-5D and K-MSQ are summarized in Table 4.
Headache-associated impact on daily life, disability, and psychological issues.

HIT-6: Headache Impact Test-6; MIDAS: Migraine Disability Assessment; PHQ-9: Patient Health Questionnaire-9; GAD-7: Generalized Anxiety Disorder-7; PSS-4: Perceived Stress Scale-4; ASC-12: Allodynia Symptom Checklist-12; Migraine-ACT: Migraine Assessment of Current Therapy.
Treatment strategies
Tapering down and abrupt withdrawal strategies were used as discontinuation methods in 42.7% and 41.0% of the patients, respectively, whereas the remaining patients continued the overused medications. Seventy-eight percent of the patients started discontinuation treatment in the outpatient clinic.
After the first visit, 213 (93.0%) patients were on preventive treatment, 155 (72.8%) were prescribed multiple classes of preventives: AEDs (64.2%), TCAs (38.9%), botulinum toxin A (31.4%), anti-CGRP monoclonal antibodies (28.4%), and beta-blockers (24.5%), whereas calcium channel blockers, SNRI, and ARB/ACEi were used less frequently (Figure 2) (duplicate selection allowed). Short-term steroids were used in 17.9% of the patients.
Primary outcome
Among 132 (57.6%) patients enrolled until 15 March 2021, 91 (68.9%) patients were followed up at 3 months after enrollment. Sixty-three (69.2%) out of 91 patients did not overuse acute medications for the past month at 3 months. The proportion of the patients without medication overuse was highest in the abrupt withdrawal group (28/35, 80.0%) than in the tapering down (33/48, 68.8%) and maintenance (2/8, 25.0%) groups (p = 0.01).
Discussion
The present study using the prospective, nationwide, multicenter MOH registry database in Korea demonstrates several key findings. First, the patients with MOH had experienced long-term chronic daily headaches and medication overuse, and exhibited poor headache profiles. Second, a substantial proportion of the patients was severely impacted by headaches, was disabled in daily living, and suffered moderate/severe depression and anxiety. Third, only about one-third of the patients were on preventive medications at baseline despite a considerable burden on headache and psychological aspects. Moreover, the proportions of patients on preventive therapies with proven efficacy, such as botulinum toxin A and anti-CGRP monoclonal antibodies were low.
RELEASE has some differences from other cohorts included in previous MOH trials (9,13,33). RELEASE is the most recent MOH registry that enrolled participants starting in April 2020 after the approval and availability of anti-CGRP monoclonal antibodies in Korea. Second, this is the first multicenter MOH registry representing the Asian headache population. Finally, RELEASE is not a randomized trial for specific treatment strategies and reflects all available therapeutic options for MOH, including botulinum toxin and combination treatments in real-world clinical practice.
The duration of chronic daily headache in the present study (6.5 years) was longer than that reported in the Brazilian MOH study (4.5 years) (33) and was markedly shorter than that reported in the MOTS study (11 years) (Table 2) (9). The average of 4.3 years for medication overuse in the present study was comparable to that reported in the COMOESTAS study (4.3 years) (34), and was slightly shorter than that reported in the MOTS study (4.8 years) (Table 2) (9). The differences in duration of headaches before MOH diagnosis among these studies might be driven by differences in culture, ethnicity, and availability of healthcare services in regions where the studies were conducted (9,33,34). In the present study, headaches were present 80% of the month, with severe headache and acute medication use rates of 40% and 70%, respectively, which were comparable to those reported in other large-scale studies (Table 2) (9,33,34). In a recent study in China, the patients with MOH used acute medications every day in that study (17), which was markedly higher than that reported in other studies (21–22 days) (Table 2). Comparisons with other Asian countries were not possible due to the substantial heterogeneity in target outcomes among the studies (14–16).
The impact of MOH on disability in daily living was comparable between the present study and the previous studies. The mean HIT-6 score and the proportion of severely impacted patients (HIT-6 score of 60–78) in the present study (Table 2) were comparable to those in the MOTS study (approximately 88%) (9). The mean MIDAS score in the present study (66.8) was comparable to that reported in the COMOESTAS study (60.8) (34). The percentage of patients reporting depression (PHQ-9 score ≥5) was 83% in the present study and 74% in the MOTS study (9). Depression is common, affecting 39–80% patients with MOH despite difference in the measures used to evaluate depression (the Hospital Anxiety and Depression Scale in the COMOESTAS study and Beck Depression Inventory in Indian study) (14,34), In the present study, anxiety (GAD-7 score ≥5) was present in 62% of the patients, which was similar to that reported in the MOTS study (68%) (9) but was higher than that reported in the COMOESTAS study (49.5%) (34). Although the direction between psychological comorbidities and MOH is unclear, the burden of psychological comorbidities and headache profiles should be considered since the rates of these comorbidities are associated with increased odds of MOH (35) and can be reduced by the treatment of MOH (36).
In the present study, only 38% of the patients were on preventive treatment at baseline, which might be low given the burden of MOH. Compared to the present study, 45% of the patients were receiving preventive treatment in the MOTS study (9). Unfortunately, there is only one review article on MOH management in Korea (37), and national guidelines or consensus recommendations specifically for MOH are lacking. However, most headache experts in clinics or referral headache centers follow major headache guidelines (6,38); therefore, all patients in the present study might be candidates for preventive treatment. The proportion of patients on preventive treatment increased to 93%, more than twice the baseline proportion of 38%. Considering that most patients in the present study had chronic migraines, the proportions of patients administered medications with proven efficacy for chronic migraines, such as AED, botulinum toxin A, and anti-CGRP monoclonal antibodies, increased significantly after enrollment in the study (Figure 2). A recent clinical trial demonstrated that patients with MOH received the most benefit when preventive treatment was combined with the discontinuation of overused medication (8); we therefore expect to achieve positive results in the current study.
The present study has several limitations. First, selection bias might be inevitable since all participating headache centers functioned as referral centers in each region and six of the seven centers were located in Seoul and Gyeonggi province. Thus, the study patients might have had more severe and frequent headaches with more acute medication use compared to the patients with MOH in the general population in Korea. However, we assume that the differences between referral centers and community hospitals or clinics might not be significant since access to specialists in community, general, and even university hospitals in Korea is easy without the need of a referral letter and nearly half of the population of Korea lives in Seoul and Gyeonggi province. Some study participants were from other regions in Korea who used the fast transportation system within the country. Second, some patients were not using headache diaries before study enrollment and there might be recall bias regarding headache profiles at baseline. However, the current study findings are in close agreement with previous MOH studies from other countries (9,33,34). Third, some patients did not want to be engaged in the study despite the absence of potential harm to the participants with participation. However, the rate of patients who declined to participate in the study was 17.7%, which might not significantly impact the results.
Conclusion
The results of this comprehensive, multicenter MOH registry study in Korea reveal that patients with MOH had considerable headache-related burden, with impact on daily lives and psychological comorbidities, and highlight the opportunities to improve the quality of headache care by thorough application of acute and preventive strategies based on current guidelines and evidence.
Clinical implications
Medication overuse headache is a challenging headache disorder related to disability in daily life and psychological comorbidities. The management strategies for MOH might be insufficient in Korea. Given recent growing evidence on the benefits and adverse effects of various therapeutic strategies, well-designed nationwide MOH registries should provide real-world clinical evidence for headache clinicians and researchers.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024211057184 - Supplemental material for Interim analysis of the Registry for Load and Management of Medication Overuse Headache (RELEASE): A multicenter, comprehensive medication overuse headache registry
Supplemental material, sj-pdf-1-cep-10.1177_03331024211057184 for Interim analysis of the Registry for Load and Management of Medication Overuse Headache (RELEASE): A multicenter, comprehensive medication overuse headache registry by Hong-Kyun Park, Min Kyung Chu, Sun-Young Oh, Heui-Soo Moon, Tae-Jin Song, Mi Ji Lee, Jin-Ju Kang, Yooha Hong, Soo-Jin Cho and on behalf of the RELEASE investigators in Cephalalgia
Footnotes
Acknowledgements
This study was supported by a grant by the Korea Headache Society.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S-JC was involved as a site investigator of a multicenter trial sponsored by Otsuka Korea, Allergan, Ildong Pharmaceutical Co., LTD, Novartis International AG, Eli Lilly and Company, Hyundaipharm. Co. Ltd., Biohaven Asia Pacific Ltd, H. Lundbeck A/S (Lundbeck), and Parexel Korea Co., Ltd., and received lecture honoraria from Allergan Korea, WhanIn Pharm Co., LTD, Shinpoong Pharma. Co., Ltd, and SK chemicals in the past 24 months.
MKC was a site investigator for a multi-centre trial sponsored by Otsuka Korea, Novartis, International AG, Lundbeck Korea Co., Ltd and Eli Lilly and Co. He functioned as an advisory member for Teva, and received lecture honoraria from Allergan Korea, Handok-Teva, and Yuyu Pharmaceutical Company over the past 24 months. He received grants from the Yonsei University College of Medicine (2018–32-0037) and National Research Foundation of Korea (2019R1F1A1053841).
H-KP, S-YO, H-SM, T-JS, MJL, J-JK, and YH declare no conflict of interest.
Funding
This study is supported by the Research Group Support Fund of Korean Headache Society.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
