Abstract
Objective
“Pain interference” and “headache impact” refer to negative consequences that pain and headache have on one’s life. This study investigated determinants of these negative impacts in a large patient cohort who have chronic migraine with medication overuse.
Methods
Six hundred and eleven adults were enrolled from 34 headache, neurology, and primary care clinics. Negative consequences of chronic migraine with medication overuse were determined using the Patient-Reported Outcomes Measurement Information System (PROMIS) Pain Interference 6b questionnaire and the Headache Impact Test 6. Relationships between PROMIS-6b and Headache Impact Test 6 scores with demographics, headache characteristics, medication use, anxiety symptoms, and depression symptoms were assessed with linear regression. Elastic Net regression was used to develop a multiple regression model.
Results
PROMIS-6b T-Scores averaged 65.2 (SD 5.4) and Headache Impact Test 6 scores averaged 65.0 (SD 5.3), indicating severe negative consequences of chronic migraine with medication overuse. Chronic migraine with medication overuse interfered with enjoyment of life, concentration, daily activities, doing tasks away from home, and socializing. Depression symptom severity had the strongest relationship with pain interference and headache impact. Moderate-to-severe headache frequency, headache intensity, and anxiety symptoms were also associated with pain interference and headache impact.
Conclusions
Chronic migraine with medication overuse is associated with substantial negative consequences, the extent of which is most strongly related to depression symptoms.
Introduction
Chronic migraine (CM), which has an estimated prevalence of about 2% in the general population, manifests with at least 3 months of headaches on at least 15 days per month, including at least 8 days per month that meet International Classification of Headache Disorders, 3rd edition (ICHD-3) criteria for migraine with or without aura (1–3). Approximately half of people with CM take symptomatic migraine medications with a high enough frequency that it is classified as “medication overuse” (MO) (1,4). Those affected by CM have multiple domains of their lives negatively impacted, an impact that is even greater amongst those who have CM with MO (5–7). The patient demographics, headache characteristics, medication use, and psychological symptoms that impact the extent to which CM with MO interferes with daily life, and the relative importance of such factors, have been inadequately investigated.
The impact of CM with MO on daily life was measured in the Medication Overuse Treatment Strategy (MOTS) trial, a randomized, pragmatic clinical trial testing two treatment strategies for patients who have CM with MO. Validated questionnaires were used to measure “pain interference”, a term that describes the consequences that pain of any kind has on different aspects of a person’s life, as well as the impact of headache on daily life were measured (8). In the MOTS trial, patients reported the impact that pain and headache caused on their day-to-day activities, doing tasks away from home, social functioning, ability to concentrate, mood, and their enjoyment of life and recreational activities. The objective of this analysis was to investigate pain interference and headache impact in patients who have CM with MO and to determine the demographics, headache characteristics, medication use factors, and psychological symptoms that are most strongly associated with pain interference severity.
Methods
Patient enrolment, eligibility, and consent
MOTS trial patients (n = 611) were enrolled from 34 healthcare sites in the USA, including headache specialty, general neurology, and primary care clinics.
Men and women who were at least 21 years old and had CM with MO according to ICHD-3 beta diagnostic criteria were eligible (9). Patients were willing to be randomized to either of the two clinical trial treatment strategies (migraine prevention medication with immediate switching from the overused medication to an alternate acute therapy that can be used with a limited frequency or migraine prevention medication without immediate switching from the overused medication), agreed to provide information in a daily headache diary, and could not have had changes to their migraine preventive therapy within four weeks of enrolment. Potential participants were not offered enrolment if there were safety concerns related to either of the two treatment strategies, such as opioid or barbiturate withdrawal syndrome if a patient was randomized to immediate discontinuation of the overused medication.
All patients participated in an informed consent process and signed Institutional Review Board approved consent documents. The MOTS trial was registered with clinicaltrials.gov (https://clinicaltrials.gov/ct2/show/NCT02764320) prior to the enrolment of the first patient.
Patient assessments
Guidance on data collection for headache studies provided by the National Institutes of Health (NIH) National Institute of Neurological Disorders (NINDS) common data elements (available at: https://www.commondataelements.ninds.nih.gov/headache), as well as patient reported outcomes from the NIH Patient-Reported Outcomes Measurement Information System (PROMIS), were used when developing MOTS methods. Assessments completed prior to randomization, and that are included in this manuscript, included: demographics, headache characteristics, medication use, Generalized Anxiety Disorder 7 (GAD-7), Patient Health Questionnaire 9 (PHQ-9), PROMIS Pain Interference 6b, and Headache Impact Test 6 (HIT-6).
Using a structured interview, patients self-reported their baseline headache frequency (i.e. average number of days per month (30 days) with headache of any kind/severity during the prior 3 months), frequency of moderate to severe intensity headaches (i.e. number of days during the prior 30 days during which headache become moderate or severe intensity at any time), average headache intensity (i.e. on days with headache, average headache intensity on a scale from 0 = no pain to 10 = most severe pain), number of years with migraine (calculated from patient report of the month and year of their first migraine), number of years with CM (calculated from patient report of the month and year during which they first developed CM), number of years with MO (determined by research team using patient-reported data), and types and frequency of medications used (determined by research team using patient-reported data).
Symptoms of anxiety were assessed using the GAD-7, a seven-question screening instrument for assessing symptoms of anxiety during the prior 2 weeks (10). Each question is scored from 0 (“not at all”) to 3 (“almost all the time”) and the sum of the answers yields a total score from 0–21: 0–4 = no anxiety, 5–9 = mild anxiety, 10–14 = moderate anxiety, and 15–21 = severe anxiety.
Symptoms of depression during the prior 2 weeks were assessed using the PHQ-9. Each question is scored from 0 (“not at all”) to 3 (“nearly every day”) and the sum of the answers yields a total score of 0 to 27 for depression severity: 0–4 = minimal depression, 5–9 = mild depression, 10–14 = moderate depression, 15–19 = moderately severe depression, and 20–27 = severe depression (11,12).
The PROMIS Pain Interference 6b questionnaire assesses the extent to which pain interfered with a patient’s enjoyment of life, ability to concentrate, day-to-day activities, recreational activities, doing tasks away from home (e.g. getting groceries, running errands), and socializing with others during the prior 7 days (13). For all questions except socializing with others, respondents choose one of five possible responses to how much pain interfered (e.g. how much did pain interfere with your enjoyment of life?): “Not at all”, “a little bit”, “somewhat”, “quite a bit”, or “very much”. For the question asking about how often pain kept them from socializing with others, respondents choose one of five possible responses: “Never”, “rarely”, “sometimes”, “often”, or “always”. Raw scores on a scale from 6 to 30 points are converted into a standardized score, with a mean of 50 and a standard deviation of 10 in the United States general population. The PROMIS Pain Interference questionnaire has been tested and validated across numerous pain conditions and in healthy individuals (8).
The HIT-6 is a six-item questionnaire that assesses the impact headaches have on a person’s ability to function on the job, at home, at school, and in social situations (14). The HIT-6 also asks about the frequency of severe headaches and the frequency of wanting to lie down due to headache. Three of the questions refer to the prior 4 weeks, while the other three questions do not specify a time period. Each question has five response options: “Never”, “rarely”, “sometimes”, “very often”, and “always”. The answers are summed to obtain a total score that ranges from 36 to 78, with higher numbers indicating more impact from headache: 36–49 = little or no impact, 50–55 = some impact, 56–59 = substantial impact, 60–78 = severe impact.
Statistical Analyses
Descriptive statistics were used to characterize the MOTS patients, their headache characteristics, medication use, symptoms of anxiety and depression, pain interference, and headache impact.
The relationships between the PROMIS 6b T-score and patient demographics (age, race, ethnicity, sex), headache characteristics (number of headache days, number of moderate to severe headache days, average headache intensity, number of years with migraine, number of years with CM), medication use (medication class overused, overuse of one vs. multiple classes of migraine medication, number of days using an abortive medication, number of years in a MO pattern, use of migraine preventive medication, use of non-medicinal migraine therapies), anxiety symptoms (GAD-7 total score), and depression symptoms (PHQ-9 total score) were assessed by using linear regression. Relationships of each of these variables with the HIT-6 score were also assessed. Multivariable modeling was performed for pain interference and headache impact in order to determine which additional variables contributed to pain interference and headache impact beyond that already explained by an individual variable. All variables in Table 1 were potential candidates for inclusion in each multivariable model. A subset of these variables was selected by using linear regression with Elastic Net regularization. The Elastic Net method prevents overfitting the sample by minimizing a penalty term along with the model residuals. Elastic Net method was used because it selects a subset of variables without entirely ignoring variables that are somewhat correlated with other variables.
Summary statistics, relationship with PROMIS 6b T-Score, and relationship with HIT-6 score. Mean (SD), N; or n/N (%).

PHQ-9: Patient Health Questionnaire 9; GAD-7: Generalized Anxiety Disorder 7.
Results
Patient enrolment and demographics
Patients were enrolled between February 20, 2017 and June 16, 2020. Headache specialty clinics enrolled 412 (66%) of the patients, 159 (25%) patients were enrolled from general neurology clinics, and 54 (9%) patients were enrolled from primary care clinics. Baseline PROMIS 6b Pain Interference scores were provided by 611 (98%) of the patients.
Mean age was 45 (SD 13) years with a median of 44 and a range from 21–83 years (Table 1). Sex at birth was female for 532 (87%), male for 78 (13%), and sex data were missing for one patient. Race was reported as White 492 (81%), Black 40 (7%), Asian 7 (1%), American Indian or Alaskan Native 2 (0.3%), chose not to answer 7 (1%), and other race 63 (10%). Hispanic ethnicity was reported by 86 of 601 (14%). Race-ethnicity was categorized into a group other than White-non-Hispanic for 25% of the total sample.
Headache characteristics
Mean number of headache days per 30 days was 24.2 (SD 5.5), including 13.7 (SD 7.2) days on which there was moderate to severe headache intensity. Mean headache intensity was 6.3 (SD 1.7) on a scale ranging from 0 = no pain to 10 = most severe pain imaginable. On average, patients had migraine for 23 (SD 14) years and CM for 12 (SD 11) years.
Headache medication
Patients reported using symptomatic medication that could be used to treat a headache on an average of 23.5 (SD 6.3) days per 30 days. They had been in an MO pattern for an average of 4.9 (SD 6.0) years. The overused medications were from the following pharmacologic classes: Simple analgesics = 383 (63% of patients), combination analgesics (not containing opioids) = 244 (40%), triptans = 131 (21%), multiple classes not individually overused = 84 (14%), opioids = 25 (4%), and ergotamines = 6 (1%). A single medication class was overused by 69% of patients, while 31% overused medications from two or more medication classes. At baseline, migraine preventive medication was being used by 278 (46% of patients).
Anxiety and depression
The mean GAD-7 score was 7.8 (SD 5.5), suggestive of “mild” anxiety symptoms. Scores were consistent with “no” anxiety for 33%, 31% had symptoms suggestive of “mild” anxiety, 22% had “moderate” anxiety, and 14% had severe anxiety. The mean PHQ-9 score was 8.9 (SD 6.0) suggestive of “mild” depression symptoms. Scores were consistent with “no depression” for 27% of patients, “mild” depression for 32%, “moderate” depression for 22%, “moderately severe” depression for 13%, and “severe” depression for 6%.
Pain interference
PROMIS 6b T-Scores ranged from 41 to 78 points, with a mean of 65.2 points (SD 5.4). The mean score was 1.5 standard deviations worse than the reference population from the United States general population (mean 50, SD 10), suggestive of substantial pain interference in this CM with MO population. The six items contributed approximately equally to the total score. The most frequent score was a “4” for all six items, and the mean item score ranged from 3.4 to 3.7 among the six items (Table 2).
Summary statistics for each item on the PROMIS 6b and HIT-6, and their relationships with PHQ-9 score. The table reports the number of patients who responded to each question with an answer of 1 (“not at all “or “never”) to 5 (“very much” or “always”) and the average response for the entire study cohort. Average responses were between 3 and 4, demonstrating substantial pain interference and headache impact in each of the domains. Depression symptom severity, measured by the PHQ-9, was significantly associated with pain interference and headache impact in every domain.

The PHQ-9 had the strongest relationship with PROMIS 6b (Table 1). Each point higher on the PHQ-9 was associated with 0.441 points higher on the PROMIS 6b T-Score. Variation in PHQ-9 scores accounted for 24% of the variation in PROMIS 6b T-Score (Figure 1). The GAD-7 was also associated with PROMIS 6b. Each point higher on the GAD-7 was associated with 0.365 points higher PROMIS 6b T-Score, and variation in GAD-7 scores accounted for 14% of the variation in PROMIS 6b T-Score.

Relationship between PROMIS 6b and PHQ-9. Overlapping points were moved slightly in a random direction so that all points are visible.
Relationships amongst the PHQ-9 total score and the individual items on the PROMIS 6b were approximately equal for all six items (Table 2). The percentage of variation explained by the PHQ-9 ranged from 15% to 20% among the six items, and the slope of the relationship ranged from 0.06 to 0.07. A 10-point change in PHQ-9 score corresponded to approximately 0.6 points change on the individual items of the PROMIS 6b.
The headache characteristics with the strongest relationship with PROMIS 6b were the frequency of moderate-to-severe headaches and the average headache intensity. Each additional day of moderate-to-severe headache per thirty days was associated with 0.222 points higher PROMIS 6b T-Score, and each additional point of headache intensity accounted for 0.81 points higher PROMIS 6b T-Score.
No other characteristic accounted for more than 1% of the variation in PROMIS 6b T-Score.
A multivariable score with terms for PHQ-9, moderate-to-severe headache frequency, and average headache intensity (57.09 + 0.39 PHQ-9 + 0.49 Intensity + 0.12 Frequency) accounted for 29% of the variation in PROMIS 6b T-Score (Figure 2).
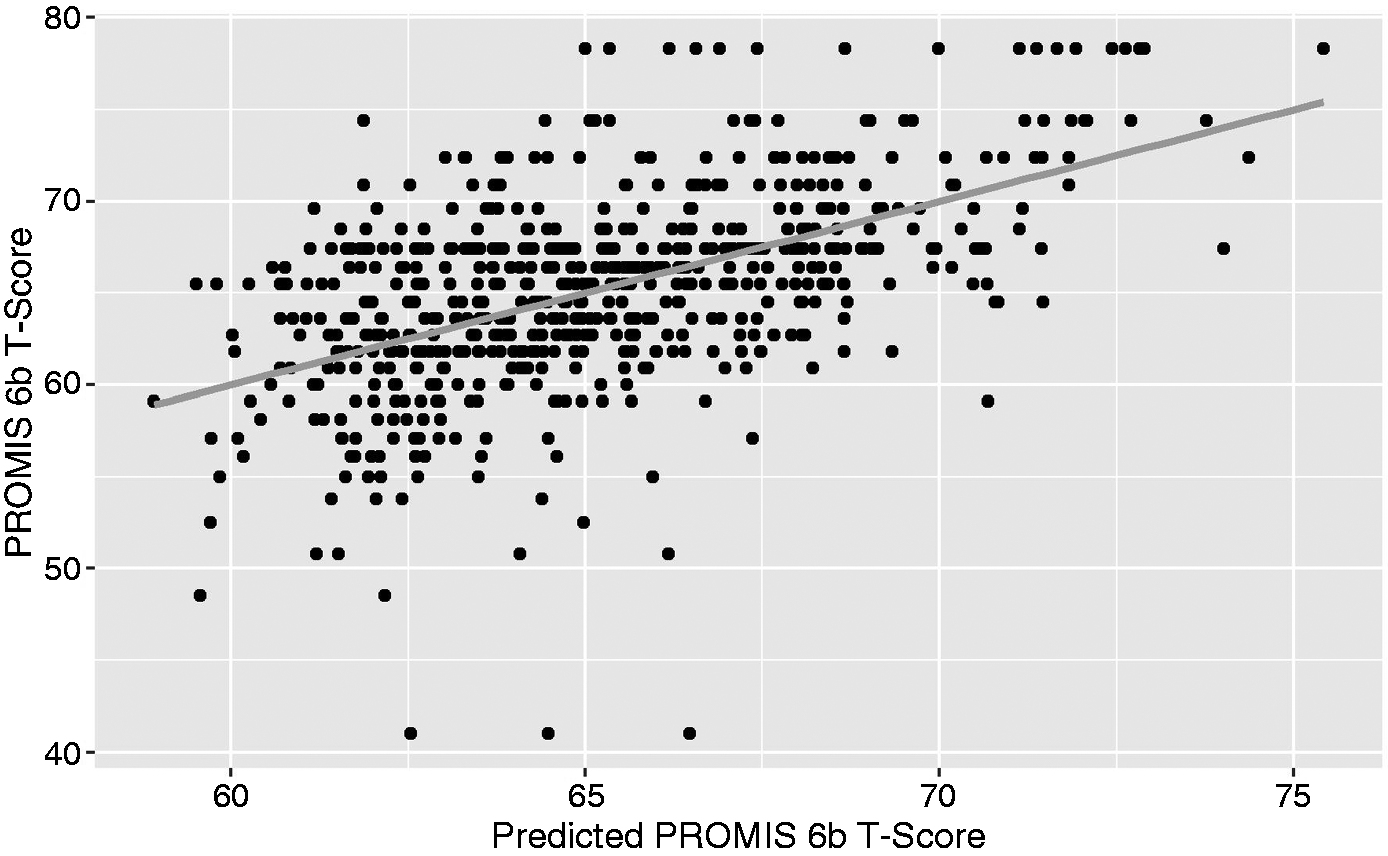
Relationship between PROMIS 6b and a multivariable predictor with terms for PHQ-9, frequency of moderate-to-severe headaches, and average headache intensity.
Headache impact
HIT-6 scores ranged from 44 to 78, with a mean and median of 65.0 (SD 5.3) indicating severe impact from headache. HIT-6 scores of at least 60 points were reported for 562 (89%) of the patients. HIT-6 scores were strongly correlated with PROMIS 6b T-Score (Figure 3). One point difference on the HIT-6 was associated with 0.750 points on the PROMIS 6b T-Score (SE 0.028, p < 0.001). The HIT-6 scores accounted for 54% of the variation in PROMIS 6b T-Scores (r = 0.73).

Relationship between PROMIS 6b and HIT-6. Overlapping points were moved slightly in a random direction so that all points are visible.
Relationships with the HIT-6 were similar to those for the PROMIS 6b (Table 1). Like the PROMIS 6b, the PHQ-9 also had the strongest relationship with HIT-6. (Figure 4) Each point higher on the PHQ-9 was associated with 0.405 points higher on the HIT-6. Variation in PHQ-9 scores accounted for 21% of the variation in HIT-6 scores. The GAD-7 was also associated with HIT-6. Each point higher on the GAD-7 was associated with 0.379 points higher HIT-6, and variation in GAD-7 scores accounted for 16% of the variation inn HIT-6 scores.
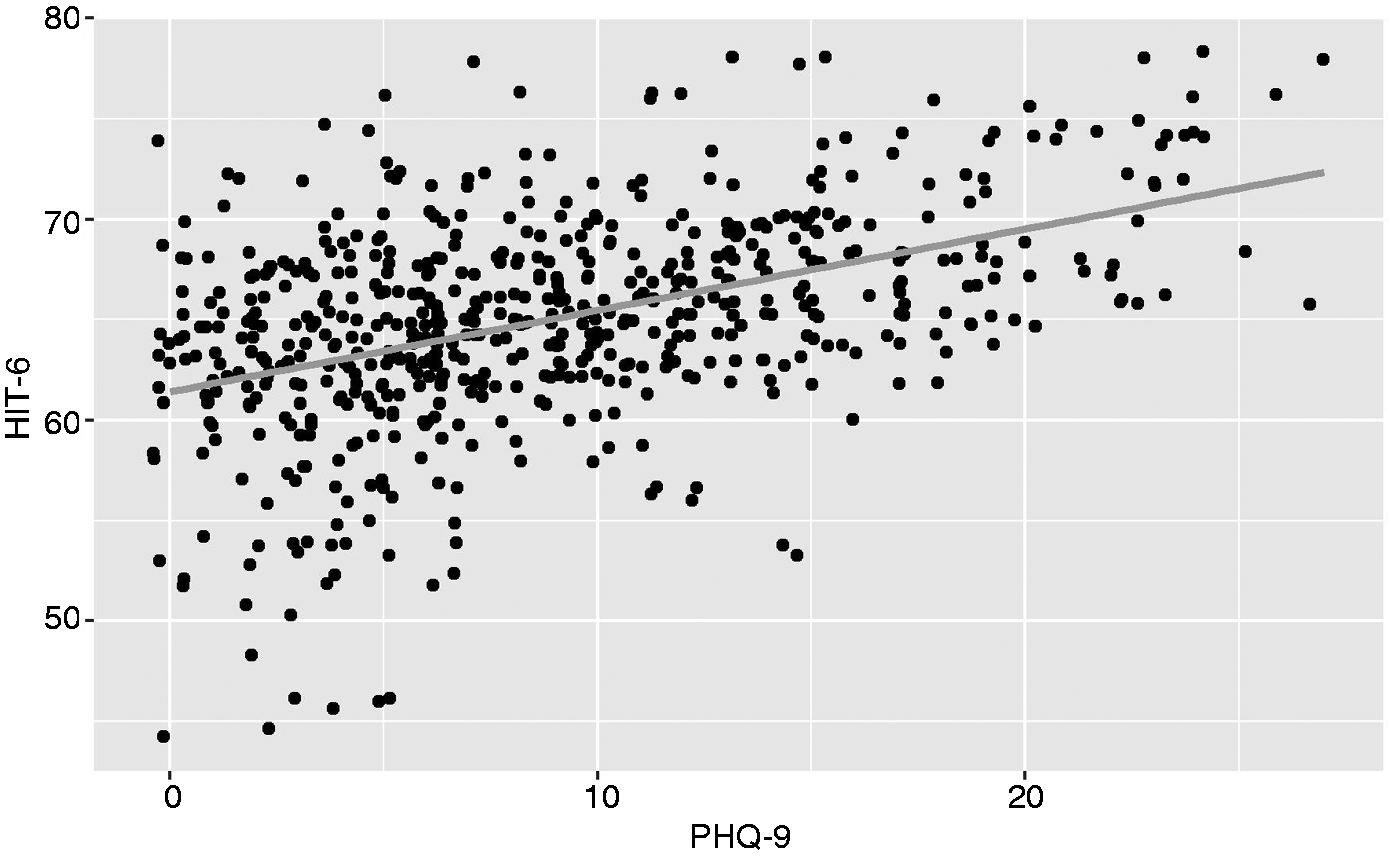
Relationship between HIT-6 and PHQ-9. Overlapping points were moved slightly in a random direction so that all points are visible.
The percentage of variation explained by the PHQ-9 ranged from 3% to 19% among the individual items of the HIT-6 (Table 2). The PHQ-9 had the strongest relationships with the frequency of tiredness and difficulty concentrating, and the weakest relationship with the frequency of severe pain.
Similar to PROMIS 6b, the headache characteristics with the strongest relationship with HIT-6 were the frequency of moderate-to-severe headaches and the average headache intensity. Each additional day of moderate-to-severe headache per 30 days was associated with 0.222 points higher HIT-6 score, and each additional point of headache intensity accounted for 0.10 points higher HIT score.
No other characteristic accounted for more than 5% of the variation in HIT-6 score. A multivariable score for HIT-6 also included terms for PHQ-9, moderate-to-severe headache frequency, and average headache intensity. Age and GAD-7 also made slight contributions with relatively small coefficients. This multivariable model (57.47 + 0.28 PHQ-9 + 0.66 Intensity + 0.13 Frequency – 0.075 Age + 0.063 GAD-7) accounted for 33% of the variation in HIT-6 scores (Figure 5).
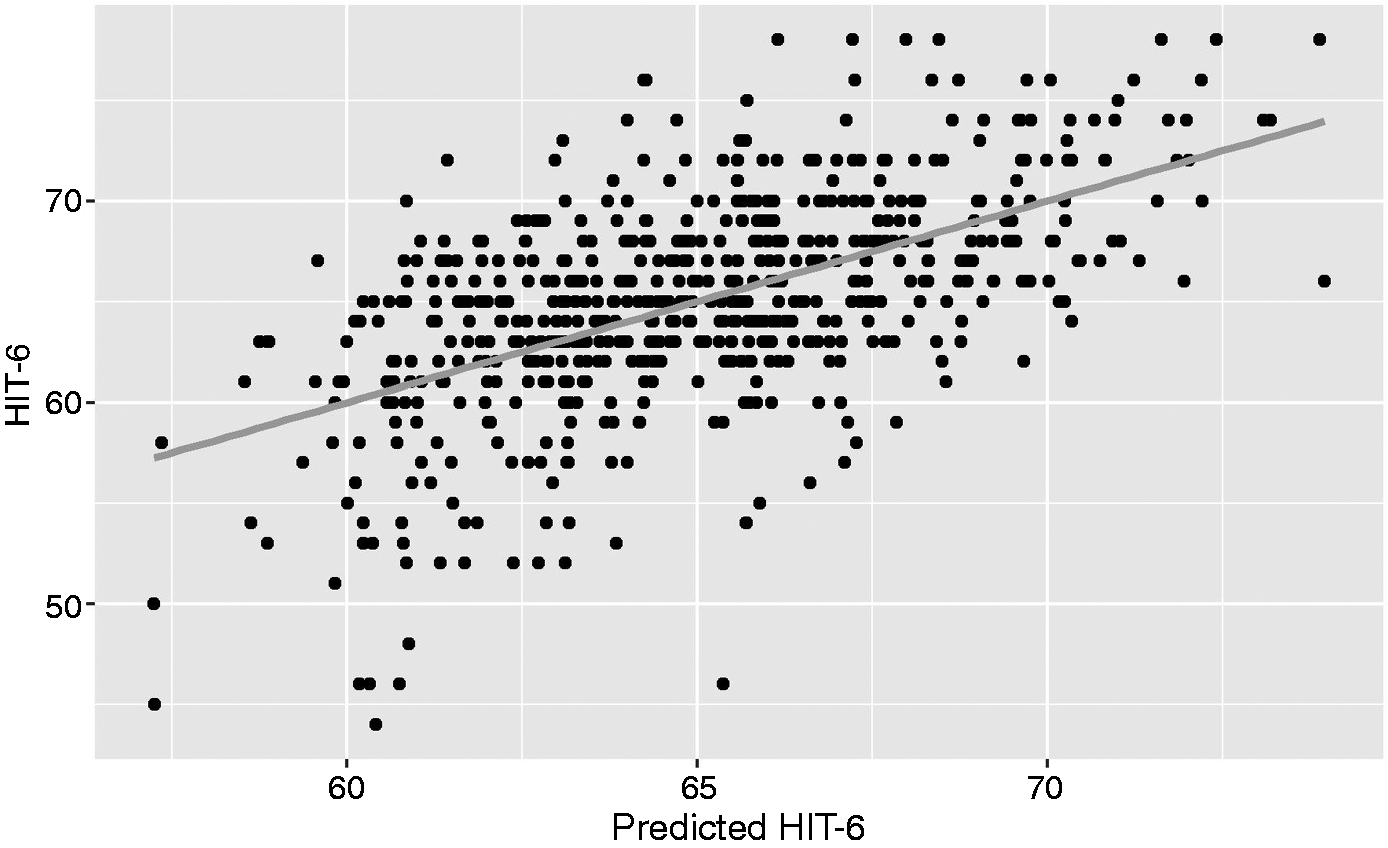
Relationship between HIT-6 and a multivariable predictor with terms for PHQ-9, frequency of moderate-to-severe headaches, average headache intensity, age, and GAD-7.
Discussion
The main findings of this analysis are that: a) CM with MO is associated with substantial pain interference and headache impact in multiple life domains; and b) the extent of these negative consequences is associated with symptoms of depression, symptoms of anxiety, frequency of days with moderate to severe headaches, and average headache intensity. The results demonstrate the enormous impact that CM with MO has on multiple aspects of one’s life, and that the severity of that impact is related to specific headache characteristics and psychological symptoms.
“Pain interference” measures the extent to which pain negatively impacts multiple different aspects of a person’s daily activities. “Pain interference” is a highly relevant health-related outcome in chronic pain disorders such as CM with MO. The PROMIS Pain Interference questionnaire was developed based on extensive review of the pain literature, existing published measures, expert clinical input, and qualitative research with patients who have chronic pain conditions (13). The PROMIS Pain Interference questionnaire is a robust, standardized method for quantifying the effects of chronic pain on patients’ lives and function, and the effects of treatment (13). Several prior studies have demonstrated that migraine is associated with disability and reduced quality of life, often using the Migraine Disability Assessment (MIDAS) questionnaire, HIT-6, Short-Form Health Survey (SF-36), and Migraine Specific Quality of Life questionnaires (7,15). Although disability and quality of life are outcomes related to pain interference, there are meaningful differences between the outcomes measured by these questionnaires with those measured by the PROMIS Pain Interference 6b. Few studies have specifically interrogated pain interference amongst those with CM. A study of 667 adults with CM, 41% of whom had MO, who were enrolled into a clinical trial of erenumab had average baseline PROMIS Pain Interference scores of 63.4, with significant post-treatment improvements compared to placebo (16,17). Patients in the American Registry for Migraine Research (ARMR), 67.2% of whom had CM, had an average PROMIS pain interference score of 63.2 (18). Participants in the MOTS trial, all of whom had CM with MO, had baseline PROMIS Pain Interference scores that were slightly higher than those in the ARMR and erenumab studies. Furthermore, analysis of the individual pain interference subdomains assessed by PROMIS 6b demonstrated that CM with MO impacts all measured aspects of life (enjoyment, concentration, daily activity, recreation, errands, socializing) substantially and largely equally.
Whereas the PROMIS Pain Interference questionnaire assesses the negative impacts from pain of any kind, the HIT-6 specifically ascertains negative consequences from headache. Thus, the use of HIT-6 is complementary to the PROMIS Pain Interference questionnaire. However, as anticipated, this analysis demonstrated that PROMIS Pain Interference scores are highly correlated with HIT-6 scores. Furthermore, the headache and psychological characteristics that explain variance in PROMIS Pain Interference scores are similar to those that explain HIT-6 score variance. Unlike the PROMIS Pain Interference questionnaire, the HIT-6 has been used commonly in migraine studies, including several studies of CM with MO (19–22). The HIT-6 scores in the patients from the MOTS trial reported herein, demonstrating severe headache impact, are comparable to those reported in other studies of individuals who have CM with MO (19–22).
Depression symptoms were most strongly associated with the extent of pain interference and headache impact amongst those with CM with MO. Prior migraine studies have demonstrated relationships between depression with headache-related disability and pain interference (18,23–27). Our findings expand upon prior publications by demonstrating the relationship between depression symptoms with pain interference and headache impact specifically amongst a large U.S. clinical patient population who have CM with MO and by showing that depression symptoms explained a larger amount of variance in pain interference and headache impact scores compared to patient demographics, anxiety, headache characteristics, and medication use factors. In addition, this analysis demonstrates that depression symptoms are associated with each subdomain of the pain interference to a similar extent, including concentration, socializing, enjoyment of life, daily activities, enjoyment of recreation, and doing tasks away from home. Depression symptoms were also associated with each item of the HIT-6, most strongly associated with difficulty concentrating and tiredness.
In our study, anxiety symptoms were also associated with pain interference and headache impact. Prior migraine studies have found relationships between anxiety with poor quality of life and disability (24,26,28,29). Like our study, the analysis of ARMR data by Pearl and colleagues specifically investigated the relationship between GAD-7 scores and PROMIS Pain Interference scores (18). After controlling for age, sex, headache frequency, years with migraine, and average headache intensity, no relationship was found between anxiety symptoms and pain interference. However, only 67.2% of patients in ARMR had CM (vs. 100% in this MOTS analysis) and the patients in ARMR had less severe anxiety symptoms (ARMR mean on GAD-7 = 5.5 vs. MOTS mean on GAD-7 = 7.8), perhaps explaining the differences in study results. Prior studies of the HIT-6 have found relationships between symptoms of anxiety and headache impact, including studies using the GAD-7 (10,26).
Average headache intensity and the number of days with moderate to severe headache were associated with pain interference and headache impact. Prior studies in migraine have demonstrated the important relationship of headache intensity with disability and pain interference (5,25,30–32). It seems intuitive that more severe average headache intensity would be associated with greater negative consequences from CM. The concept of measuring average headache intensity is highly related to measuring the number of moderate-to-severe headache days. Moderate-to-severe headache days are a good indicator of days on which individuals have ICHD-defined migraine attacks. It is expected that a higher frequency of days with full-blown migraine attacks would be associated with greater pain interference and headache impact.
It is notable that participant demographics, years with migraine, years with CM, years with MO, the frequency and types of acute medications overused, and the use of migraine-preventive medications did not significantly contribute to the variance in pain interference and headache impact scores. It could be hypothesized that the overuse of certain migraine medications, like opiates, would be associated with greater negative consequences. Although such a relationship was not identified in this study, given the small number of study patients overusing opiates, firm conclusions on this relationship cannot be drawn. It is hoped that the use of effective migraine preventive medication would be associated with less pain interference and headache impact. Reductions in headache-related disability and improvements in quality of life attributed to effective treatment have been demonstrated in numerous migraine prevention trials (15,33–35). The lack of an association between the use of migraine preventive therapy and negative consequences from CM with MO in our study might be explained by the preventive medication not being particularly effective, an assumption supported by the fact that the patient population consisted of individuals who had CM with MO with high frequency severe headaches. The longitudinal component of the MOTS trial will allow for analyses that investigate relationships between the use of effective headache preventive therapy and reductions in pain interference and headache impact among patients who have CM with MO.
Although this analysis was cross-sectional and the directionality of relationships can therefore not be determined, results suggest the importance of managing psychological symptoms as well as headache frequency and intensity in patients who have CM with MO. Presumably, a comprehensive therapeutic approach that reduces the frequency of migraine headaches, lessens the average intensity of recurrent headaches, and reduces symptoms of depression and anxiety, would maximize reductions in pain interference and headache impact. This might be best accomplished through migraine-targeted preventive and symptomatic therapies in combination with psychological interventions such as counseling, cognitive behavioral therapy, biofeedback, stress reduction techniques, medications and other therapeutic interventions.
A limitation of this analysis is that much of the baseline data, such as headache frequency and intensity, and number of years with CM and MO, were ascertained during a structured interview that relied on patient recall. Another limitation is that the PROMIS Pain Interference 6b questionnaire asks about the impact of “pain”, as opposed to asking specifically about the impact of “headache” or “migraine”. Since individuals who have CM with MO often have comorbid non-headache related pain in addition to migraine, the pain interference in this cohort likely reflects that caused by migraine and other types of pain that might all contribute to MO. Data on other pain conditions were not available for these analyses. However, the analyses also included the HIT-6 as a second outcome measure, a questionnaire that specifically ascertains negative impacts related to headache. Strong correlations between PROMIS Pain Interference scores and HIT-6 scores and the high similarities between the models that explained variance in PROMIS Pain Interference scores and variance in HIT-6 scores suggest that both questionnaires are useful for studying negative impacts of CM with MO. A minority of the variance in PROMIS Pain Interference scores and HIT-6 scores was explained by the models that included variables such as depression, anxiety, frequency of moderate to severe headache days, and average headache intensity. This indicates that there are factors not included in our modeling that explain a substantial amount of the variance in pain interference and headache impact We hypothesize that these other factors might include non-migraine pain, cephalalgiaphobia (i.e. fear of the next headache), medication side effects, sleep quality, pain acceptance, pain catastrophizing, and others (32,36). Future studies need to collect data that measure these other factors and determine the extent to which they might associate with pain interference and headache impact. Finally, since this is a cross-sectional analysis, the direction of the relationships between CM with MO, pain interference, headache impact, depression, and anxiety cannot be proven. One condition could cause another unidirectionally, or there could be a bidirectional relationship, and other unmeasured factors might mediate the relationship.
Conclusions
Patients in the USA who have CM with MO have substantial pain interference and headache impact in multiple life domains, including enjoyment of life and recreational activities, ability to concentrate, daily activities, doing tasks away from home, and socializing. The extent of these negative consequences can be partially explained by symptoms of depression and anxiety, average headache intensity, and the frequency of days with moderate-to-severe headache. It is likely that a comprehensive patient management approach that targets headache and psychological symptoms would optimally limit the impact that CM with MO has on a person’s life.
Key findings
Patients who have chronic migraine with medication overuse have substantial pain interference and headache impact in multiple life domains. The extent of pain interference and headache impact is most strongly associated with symptoms of depression. It is also associated with symptoms of anxiety, average headache intensity, and the frequency of moderate to severe headaches. Results suggest that a comprehensive patient management approach that targets headache and psychological symptoms would optimally limit the impact that CM with MO has on a person’s life.
Footnotes
Data availability statement
MOTS trial data will be made available within clinicaltrials.gov according to the rules and regulations set forth and/or within the Patient Centered Outcomes Research Institute’s designated data repository.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The institutions for each of the authors received a sub-award from Mayo Clinic for participating in the MOTS trial.
TJS reports a grant from PCORI during the conduct of the study; grants from American Migraine Foundation, Amgen, Henry Jackson Foundation, National Institutes of Health, and United States Department of Defense, personal fees from Allergan, Alder, Amgen, Biohaven, Cipla, Click Therapeutics, Dr. Reddy’s, Eli Lilly, Equinox, Ipsen Bioscience, Lundbeck, Novartis, Teva, Weber and Weber, and XoC, stock options from Aural Analytics and Nocira, and royalties from UpToDate, outside the submitted work.
SS-S has served as a paid speaker for Eli, Lilly and Teva and received funding for a fellowship program from Allergan.
NM, MB, KD, KL, WM, MTB, KP, PR, JL, JW and MMC report nothing to disclose.
ZA reports grants from Teva, personal fees from Allergan, Eli Lilly, Electrocore, and Amgen, outside the submitted work.
FMC reports personal fees from Biohaven, outside the submitted work.
JDL reports personal fees and non-financial support from Amgen, outside the submitted work.
HS reports personal fees from Allergan, Amgen, Biohaven, and Teva, and personal fees and non-financial support from Novartis, outside the submitted work.
VTM reports personal fees from Amgen, personal fees and other from Allergan, personal fees from Biohaven, personal fees from Theranica, personal fees and other from Lilly, personal fees from Teva, personal fees from Lundbeck, outside the submitted work.
NMS reports personal fees from AMGEN/Novartis, personal fees from Supernus, personal fees from TEVA, personal fees from Biohaven, personal fees from Abbvie (Allergan), outside the submitted work.
JGH has received research funding from PCORI.
TR has received personal compensation from the Patient-Centered Outcomes Research Institute for serving as co-investigator of the MOTS trial.
DWD reports personal fees from Amgen, AEON, Autonomic Technologies, Axsome, Allergan, Alder BioPharmaceuticals, Biohaven, Clexio, Dr Reddy's Laboratories/Promius, Cerecin, Eli Lilly, eNeura, Neurolief, Novartis, Ipsen, Impel, Satsuma, Supernus, Theranica, Teva, Vedanta, WL Gore, Nocira, XoC, Zosano, Upjohn (Division of Pfizer), Pieris, Revance, Equinox, Salvia, Lundbeck. He has received speaking fees from Eli Lilly, Novartis Canada, Amgen, Lundbeck and CME fees or royalty payments from HealthLogix, Medicom Worldwide, MedLogix Communications, Mednet, Miller Medical, PeerView, WebMD Health/Medscape, Chameleon, Academy for Continued Healthcare Learning, Universal Meeting Management, Haymarket, Global Scientific Communications, Global Life Sciences, Global Access Meetings, Catamount, UpToDate (Elsevier), Oxford University Press, Cambridge University Press, Wolters Kluwer Health. He has stock options in Precon Health, Aural Analytics, Healint, Theranica, Second Opinion/Mobile Health, Epien, Nocira, Matterhorn, Ontologics, King-Devick Technologies. He is on the Board of Directors for Paranet North America, Precon Health, Epien, Matterhorn, Ontologics, King-Devick Technologies. He holds a patent: 17189376.1-1466:vTitle: Botulinum Toxin Dosage Regimen for Chronic Migraine Prophylaxis without fee. He receives research funding from American Migraine Foundation, US Department of Defense, PCORI, and Henry Jackson Foundation. He has received professional society fees or reimbursement for travel: American Academy of Neurology, American Brain Foundation, American Headache Society, American Migraine Foundation, International Headache Society, Canadian Headache Society.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this manuscript was funded through a Patient-Centered Outcomes Research Institute (PCORI) Award (PCS-1504-30133). The views in this publication are solely the responsibility of the authors and do not necessarily represent the views of the Patient-Centered Outcomes Research Institute (PCORI), its Board of Governors or Methodology Committee.
