Abstract
Introduction: A population-based observational study was used to assess the prevalence, demographics, risk factors, and costs of triptan overuse, defined as more than 30 (International Headache Society criteria) or 54 (stringent criteria) defined daily doses per 3 months.
Methods: Analysis of the Dutch Health Care Insurance Board Database for 2005, which included prescriptions for 6.7 million people (46% of the total Dutch population).
Results: Triptans were used by 85,172 (1.3%) people; of these, 8,844 (10.4%; 95% CI 10.2–10.6) were overusers by International Headache Society and 2,787 (3.3%; 95% CI 3.2–3.4) were overusers by stringent criteria. The triptan-specific odds ratios for the rate of International Headache Society overuse compared with sumatriptan were: 0.26 (95% CI 0.19–0.36) for frovatriptan; 0.34 (95% CI 0.32–0.37) for rizatriptan; 0.76 95% CI 0.68–0.85) for naratriptan; 0.86 (95% CI 0.72–1.02) for eletriptan; 0.97 (95% CI 0.88–1.06) for zolmitriptan; and 1.49 (95% CI 1.31–1.72) for almotriptan. Costs for overuse were 29.7 million euros; for the International Headache Society criteria this was 46% of total costs and for stringent criteria it was 23%.
Discussion: In the Dutch general population, 1.3% used a triptan in 2005, of which 10.3% were overusers and accounted for half of the total costs of triptans. Users of frovatriptan, rizatriptan and naratriptan had a lower level of overuse.
Introduction
Migraine is a common (1,2), highly disabling (3,4), and costly (5) episodic brain disorder (6). Attacks typically last 1–2 days and strike with a median frequency of 1.5 per month; 10% of patients have two or more attacks per week (6). A high attack frequency is associated with a substantial increase of disability (4,7,8), costs (5,9), and risk of ischemic brain lesions (10,11). Overuse of acute headache agents is increasingly recognized as a paradoxical but major reason for sometimes dramatic increases in the attack frequency (medication-overuse headache) (7,12–15). Medication-overuse headache is increasingly recognized as a problem worldwide, with an estimated prevalence in the general population of 1–4% (12,14).
Triptans, selective 5-HT1B/1D (serotonin) receptor agonists, are specific, effective, and well tolerated agents to treat migraine attacks (16–18). Regular use of a triptan on ten or more days a month for more than three consecutive months may cause triptan overuse headache (13). Studies in headache clinics and open populations suggest an average critical dosage of 18 single doses per month (19,20). With the increasing availability of triptans over the counter and the currently often advocated (21) but still unproven instruction to treat attacks as early as possible while the headache is still mild, the prevalence of triptan overuse headache is likely to increase. This might have a major impact on the quality of life of migraine patients (22,23) and will cause a considerable increase in costs (9).
Overuse of triptans does not suddenly emerge; it starts with increasing use, develops in to more use, even more use and then sometimes results in overuse. To describe this phenomenon these different levels of triptan use are relevant, starting from a lower threshold of triptan overuse (overuse or seriously at risk of overuse, which may give an overestimation) up to a strict threshold (which may result in underestimation). To this end, we had access to a unique database of the national Dutch Health Care Insurance Board, which monitors, at the individual level, the dispensation of prescribed drugs at pharmacies for all inhabitants of the Netherlands.
In the present study, we sought to assess: (i) the prevalence and associated costs of triptan overuse in the Dutch general population; (ii) the demographic characteristics of triptan overusers to identify possible risk factors; and (iii) whether the level of overuse differs among the seven available triptans.
Methods
Study setting
Data were obtained from the Drug Information Project (GIP database) of the Health Care Insurance Board (CVZ). The CVZ is a public authority in the domain of drugs. As an independent non-profit governing body, it monitors conditions of the health insurance scheme in the Netherlands. In 2005, more than 10 million people (65% of all inhabitants of the Netherlands) were mandatorily insured as a result of the Sickness Fund Act. People were eligible for sickness fund insurance if they had a yearly income of less than €33,000. The GIP database registered the prescribed drugs that were dispensed at pharmacies for patients that are insured by sickness funds. All prescription drugs are coded according to the Anatomical, Therapeutic and Chemical (ATC) classification (24). Each registered patient has an anonymous unique identification number, which allows complete observation of medication use over time per patient. Each prescription also includes information on what insurance companies pay to the pharmacist, allowing an exact calculation of the costs. For migraine patients in the Netherlands, there were no financial restrictions in using acute or prophylactic headache therapy in the study year 2005 as long as a physician prescribed the medication. In the Netherlands over-the-counter sales of triptans is prohibited.
Definitions
A triptan user was defined as a patient for whom at least one prescribed triptan was dispensed in 2005. We used two definitions for triptan overuse, one based on the criteria of The International Headache Society (IHS), that is, use of a triptan on more than 10 days a month for ≥3 months (13), and a second more stringent definition, based on studies in headache clinics, that is, use of 18 single doses or more per month for ≥3 consecutive months (19). When patients use 18 defined daily doses (DDDs) or more over a long period, chronic daily headache based on medication overuse is evident (19).
Defined daily dose per triptan according to the World Health Organization (22)

First year of full availability of tablets without any surcharge.
Use of migraine prophylactic medication was defined as the dispensation of at least one prescription in 2005 of any medication that is registered in the Netherlands for migraine prophylaxis. Most migraine prophylactic agents have multiple disease indications. As the indication for a prescription is not recorded in the database, we could not establish whether the medication was prescribed for migraine or another disorder.
Statistical analysis
Data are summarized as means with 95% confidence intervals (95% CIs) for continuous variables and as numbers and percentages of subjects for categorical variables. Differences between groups are presented with 95% CIs. For differences among the triptans for the rate of overuse, we calculated the odds ratios (OR) with 95% CIs using sumatriptan as the reference. As sumatriptan is the longest available and most widely prescribed triptan, we adjusted the OR for duration of availability, by the method of indirect standardization. For this duration we took the number of years the drug was on the market without any surcharge (corresponding to the start of substantial use in the Netherlands). For statistical analyses we used SAS Enterprise Guide version 4.1.
Results
We found that 10% of all triptan users, which is 0.1% of the total Dutch general population, were overusing triptans. They were responsible for nearly half of the total costs for triptans.
We could assess the medication use of 6.7 million people, covering approximately 46% of the total Dutch population and 67% of all people that were insured with the sickness fund. The remaining part of the population could not be included in this study because of reasons unlikely to have introduced bias, such as non-connectable databases and other means of health insurance.
Clinical characteristics and demographics of triptan users compared with the total population
Medication that can be prescribed as prophylactic therapy for migraine. Amitriptyline is not registered and not prescribed as migraine prophylaxis in the Netherlands. Methysergide can only be prescribed for a short period to prevent adverse events and was therefore excluded. Source: GIP database/Health Care Insurance Board.
Clinical characteristics and demographics of triptan overusers compared with non-overusers

Stringent overusers are a subgroup of IHS overusers. †Medication that can be prescribed as prophylactic medication for migraine, but may have been prescribed for other co-morbid disorders. Source: GIP Database/Health Care Insurance Board.
Sumatriptan is available in four different formulations and two oral doses. The vast majority used just one formulation, although overusers (IHS: 17.2%; stringent: 24%) more often used multiple formulations than did non-overusers (5.8%). The majority exclusively used tablets: 64.2% of the total sample; 62.4% of the non-overusers; 75.4% of the IHS overusers; and 74.1% of the stringent overusers. Subcutaneous injections were used by 10.3% of the non-overusers, 6.6% of the IHS overusers and 4.6% of the stringent overusers.
Use of the various triptans in non-overusers versus overusers

Values are numbers (%) of subjects. *Stringent overusers are a subgroup of IHS overusers. Source: GIP Database/Health Care Insurance Board.
Level of overuse for each triptan relative to sumatriptan

Values are numbers (%). *Stringent overusers are a subgroup of IHS overusers. †Adjusted odds for duration of availability of the drug, by method of indirect standardization. §When using more than one triptan, the adjusted odds cannot be calculated (#) because of the variation in the duration of the availability of the triptan. Source: GIP Database/Health Care Insurance Board.
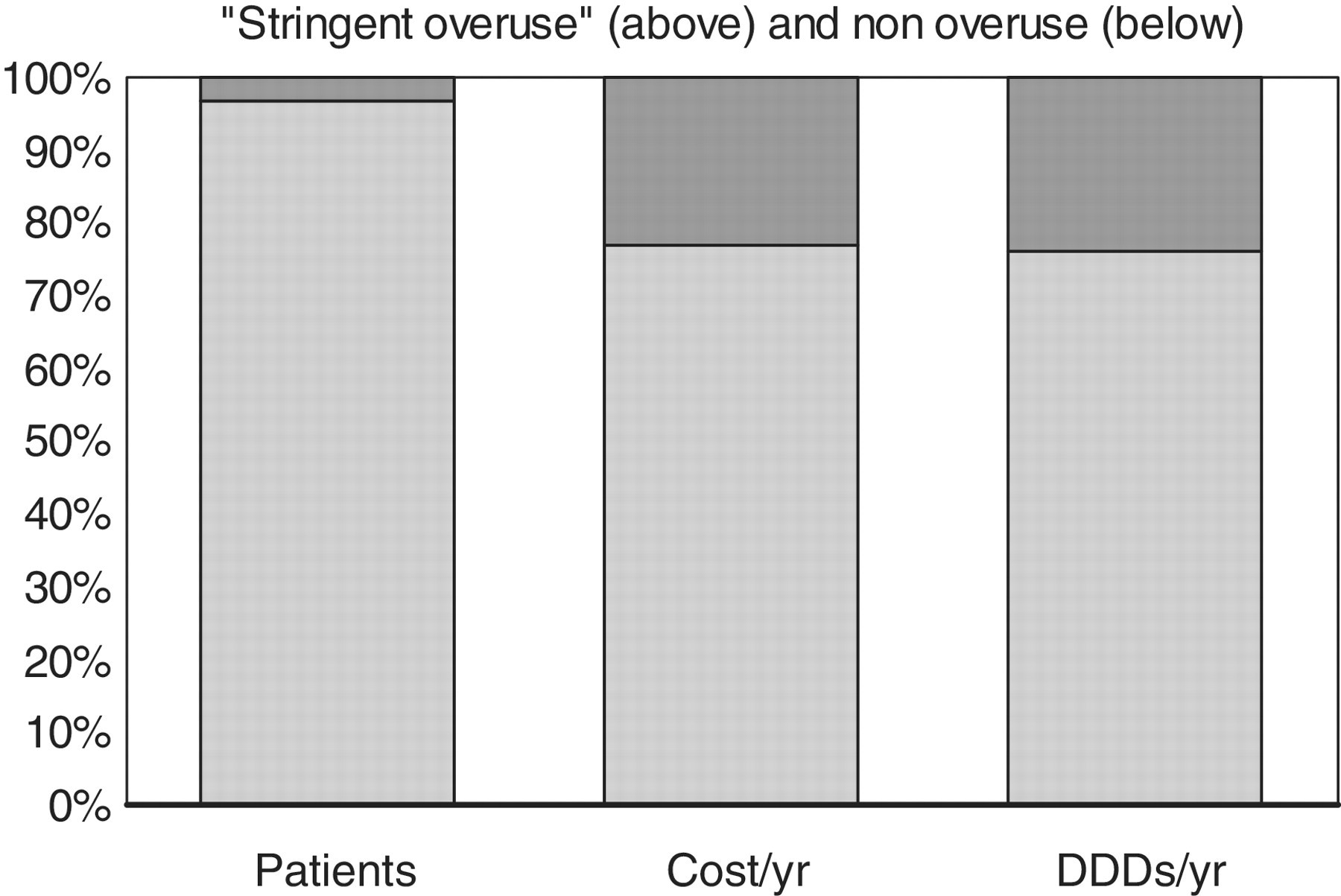
The costs of triptan use and overuse are shown in Figures 1 and 2. Total costs of triptan use in 2005 were €29.7 million, €349 per triptan user and €4.43 per inhabitant. Patients overusing triptans accounted for 46% (IHS criteria) and 23% (stringent criteria) of the total costs, €1543 per IHS overuser and €2468 per stringent overuser.
The proportion of patients with and without triptan overuse according to IHS criteria, with the associated cost and drug use. Percentage of patients with IHS overuse, generated cost and DDDs (daily defined dose). The top part of each bar indicates the IHS overusers (≥30 DDDs/qtr) and the bottom indicates the non-overusers. Source: GIP Database/Health Care Insurance Board. The proportion of patients with and without triptan overuse according to stringent criteria, with the associated cost and drug use. Percentage of patients with stringent overuse, generated cost and DDDs (daily defined dose). The top part of each bar indicates the stringent overusers (≥54 DDDs/qtr) and the bottom indicates the non-overusers. Source: GIP Database/Health Care Insurance Board.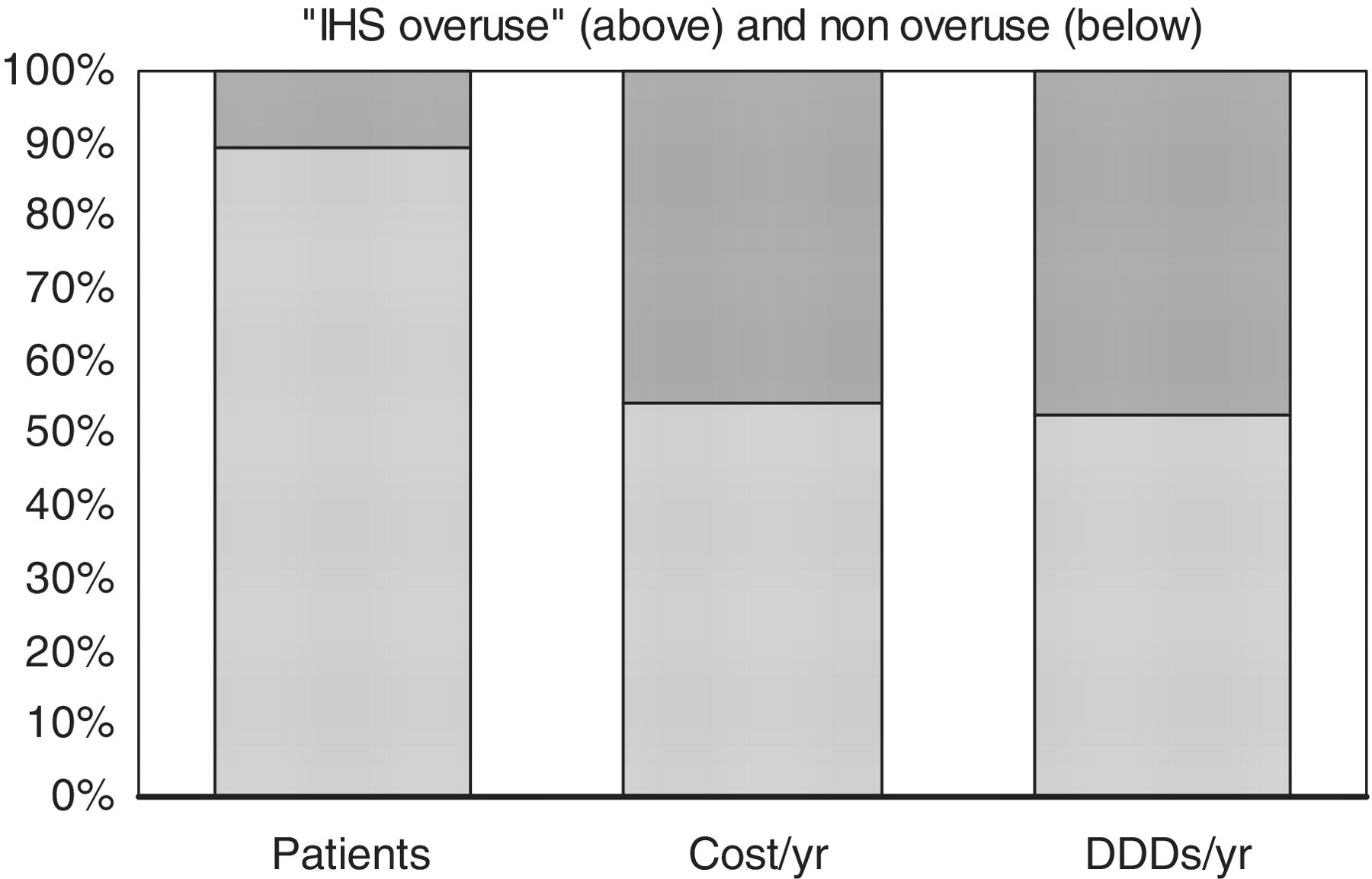
Discussion
We analysed the use and overuse for all seven available triptans in the Dutch general population in the year 2005. We could make use of a unique national Health Care Insurance Board Database, which covered the medication use of 6.7 million people. Of these, 1.3% had used a triptan at least once in 2005 and 0.13% (10.4% of all triptan users) were overusing triptans. Overusers accounted for almost half of the total costs for triptans. Remarkably, the level of overuse differed significantly per triptan. Users of rizatriptan, and possibly of frovatriptan and naratriptan, showed substantially lower levels of overuse. When patients use more than one triptan, the level of overuse is additional high: a doubling of levels of overuse compared with when the patient is only using one kind of triptan.
The results of the present study appear robust and representative because of the large number of patients included. We used an independent and unbiased nationwide database with an accurate count of actual dispense of triptans at pharmacies, covering nearly half of the total population of the Netherlands. A potential limitation is that we could not measure the actual use of triptans by patients. It seems, however, very unlikely that with an average use of 210 DDDs in IHS overusers and of 420 DDDs in stringent overusers, many patients would not have used the dispensed medication.
With regard to these thresholds we can argue that overuse lies somewhere in between the two presented thresholds. The lower limit, the IHS criteria, may give an overestimation, and the upper threshold, the stringent criteria, is probably an underestimation. With these stringent criteria, the existence of medication overuse headache is evident. With the IHS criteria there is either overuse or a high risk of overuse.
The results are in agreement with a smaller population-based study in Denmark. Here, 5% of sumatriptan users used >30 DDDs per month and were responsible for 38% of the total sumatriptan consumption and costs (26). In two French studies, 25–30% of triptan users were overusers (27) and 12% became overusers (defined as ≥180 DDDs/yr) within one year from starting using triptans (28). In an Italian study a much lower rate of overuse was found (3.2%), but this is probably due to a low overall use of triptans in this country (29).
One might argue that the prevalence of triptan overuse was overestimated given the nature of sickness fund-based databases. Patients included in such databases generally tend to come from relatively lower socio-economic classes, compared with patients with private health insurance. Low educational level is known to be associated with a higher risk of medication overuse (12,30).
Given the nature of our sickness-fund-based database, our population had a relatively lower socio-economic status than the population as a whole. However, our population represents 65% of the Dutch population in 2005 and other studies in this database revealed that data from these patients are similar to those in the general population (31,32). We also compared our data with those of a smaller database of dispensed drugs in pharmacies, including all income classes. Those data were very similar to those in the present study. In particular, there was no evidence for socio-economic class major difference in use of triptans.
In our analysis we calculated the average triptan consumption in a timeframe of 12 months, using the criteria based on a 3-month period. In this way we were able to provide more stable and reliable estimates than for shorter periods, given that use and overuse of acute anti-migraine medication are known to fluctuate substantially (12). Indeed, we found 9,120 IHS overusers (30 DDDs or more) in the first trimester of the year, 10,287 in the second trimester, 10,128 in the third and 11,088 in the final trimester; the average was 10,156 overusers. Of all overusers in the first quarter, only 63–65% were also overusers in at least one of the subsequent three trimesters.
The most striking finding of our study was that level of overuse differed among the triptans. In particular, use of frovatriptan, rizatriptan, and to a lesser degree naratriptan, was associated with remarkably lower proportions of overusers compared with the reference agent sumatriptan and the other triptans. Several confounding factors could potentially explain this finding and need to be discussed.
First, for practical reasons, we used the number of DDDs to define overuse. However, triptan overuse is in fact defined by the number of days on which at least one dose of a triptan is taken, irrespective of the total number of dosages per day or the milligrams per dose. In 2001, the DDD for sumatriptan was changed from 100 mg to 50 mg (http://www.whocc.no/atcddd/) (24). Thus, one tablet of 100 mg sumatriptan was suddenly equal to 2DDD. As a result, patients using 61–107 tablets of 100 mg per year were assigned as ‘IHS overusers’ (122–214 DDDs/yr), without necessarily fulfilling the IHS criteria for triptan overuse (use of at least one dose on ≥120 days per year). This could have potentially biased the results against sumatriptan in these patients. However, the risk of incorrectly assigning someone (this applies to each type of triptan) to the overusers group is negligible when applying the stringent criteria (≥216 DDDs/yr). The relative degree of overuse in this group was very similar to that when using the less stringent IHS criteria, with the understanding that the differences with sumatriptan are magnified to some extent. Use of almotriptan, sumatriptan and zolmitriptan was in both groups clearly associated with the highest level of overuse and use of rizatriptan, frovatriptan, and naratriptan was associated with the lowest level of overuse. Furthermore, from clinical experience we know that, for a variety of reasons, many patients divide 100 mg tablets into two of 50 mg, thereby doubling the actual number of doses used. Another reason that the extent of any possible bias by an erroneous classification of the WHO-DDD is small is that only 27.6% of all sumatriptan is delivered by pharmacies as 100 mg tablets. Consequently, any possible bias by an erroneous classification of the WHO-DDD is small in relation to the difference we found. We are, therefore, confident that the applied approach provides a reliable estimate of the relative levels of overuse for the various triptans.
A second potential confounding factor is that patients with cluster headache may sometimes use very high quantities of subcutaneous sumatriptan to treat their attacks, without necessarily being an overuser (33,34). This could have biased the results towards overuse of sumatriptan. However, use of the subcutaneous formulation of sumatriptan made up for only 8.8% of the total use of sumatriptan in the IHS overuse group and for only 5.7% in the stringent overuse group. This is less than in the non-overuse group (10.5%), making a major impact of overuse of subcutaneous sumatriptan unlikely. In other countries than the Netherlands, cluster headache patients are sometimes treated with high dosages of oral sumatriptan because of cost considerations. All triptans are carefully reimbursed in the Netherlands, so this is unlikely in present study.
A third potential bias that we need to discuss is the difference in duration of availability of the various triptans (Table 1). This might have led to preferential use of the agents available earlier by the most disabled patients, who are likely to have a higher level of overuse. Sumatriptan was the first available triptan (1991), but because of complicated reimbursement issues in the Netherlands, the oral formulation became fully reimbursed only in 1996. Sales for sumatriptan really started only then. Overuse before that time was very rare in the Netherlands (35). The other triptans were always fully reimbursed from the date of introduction. After adjusting for duration of availability of the drug (for sumatriptan from year of full reimbursement), the rate differences compared with sumatriptan disappeared for zolmitriptan, eletriptan, and almotriptan, but clearly remained for frovatriptan and rizatriptan, and to a lesser extent for naratriptan (Table 5).
Given the nature of the present observational study, we can only speculate as to (i) what the possible reasons and mechanisms are for the observed reduced levels of overuse; (ii) whether these represent a true clinical benefit; and (iii) whether this benefit applies to all three triptans. Frovatriptan was introduced 10 years after sumatriptan and consequently the absolute numbers for users (N = 957) and overusers (N = 17) in 2005 were very small compared with those for sumatriptan (N = 41,352 users and N = 5,554 overusers) and most other triptans; the user numbers for naratriptan were also very small. Furthermore, frovatriptan and naratriptan were marketed as ‘gentle’ triptans, with fewer adverse events and a slower onset of action, best suited for milder migraine attacks (16). Combined with their significantly lower 2-hr and 24-hr efficacy rates (16), this might well have led to a preferential use of frovatriptan and naratriptan by patients with milder migraines, which are known to be associated with a lower level of overuse. These considerations do not seem to apply to rizatriptan, whose user numbers were very high (N = 25,796 users and N = 1,026 overusers) and whose 2-hr and 24-hr efficacy rates are among the highest of all triptans (16).
One can ask whether the package size of the triptans can explain the differences. However, triptans are used in such large quantities that each prescription usually involves multiple packages. The package size of the various triptans varies. Nevertheless, we found no correlation between the size of the package and the association with overuse for the various triptans (data not shown).
Whether the observed differences also reflect a true clinical benefit can only be tested in prospective randomized clinical trials.
Overusers accounted for almost half of the total costs of triptans. These costs could be significantly reduced if physicians monitored prescriptions better and considered prophylactic treatment earlier in case of increasing headache frequency to prevent overuse. Once overuse is established, withdrawal of overused medication is the most appropriate therapy (36).
To our knowledge, this is the first extensive study reporting the prevalence of overuse of all currently available triptans in the general population. Although the overall prevalence of overuse was low, overuse accounted for a large health burden and a substantial proportion of total costs of migraine therapy. The level of overuse differs per triptan. Whether this reflects a true clinical benefit needs to be investigated in prospective studies.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interests
This study was initiated, designed, conducted, analysed and reported by the authors of the study, fully independent from any third party. MDF has received unrestricted research grants from, or served as a consultant to Almirall Prodesfarma, AstraZeneca, Glaxo SmithKline, Merck, Pfizer, Menarini, Johnson and Johnson, and Pharmacia. FD has received an unrestricted research grant from Janssen-Cilag, with no relation to the present study.
