Abstract
Background
Erenumab was effective and well tolerated in a pivotal clinical trial of episodic migraine that included subjects both naïve to, and those who had failed, previous preventives. Here we evaluated the efficacy and safety of erenumab (70 mg or 140 mg) versus placebo in the subgroup of patients who had previously failed preventive treatment(s): ≥1 or ≥2 prior failed migraine preventive categories, and in patients who had never failed.
Methods
Prespecified subgroup analyses evaluated change from baseline to months 4–6 (the primary endpoint of the blinded study phase) in monthly migraine days, achievement of ≥50% and ≥75% reduction in monthly migraine days, and change from baseline in acute migraine-specific medication days. Adverse events were also evaluated.
Results
Treatment with both doses of erenumab resulted in greater reductions in monthly migraine days at months 4–6 (treatment difference versus placebo [95% CI], never failed subgroup: −0.9 [−1.5, −0.3] for 70 mg and −1.3 [−1.9, −0.7] for 140 mg; ≥1 prior failed medication categories subgroup: −2.0 [−2.8, −1.2] for 70 mg and −2.5 [−3.4, −1.7] for 140 mg; ≥2 prior failed medication categories subgroup: −1.3 [−2.6, 0.0] for 70 mg and −2.7 [−4.0, −1.4] for 140 mg). Similar results were observed in the monthly acute migraine-specific medication days endpoint, and in the achievement of ≥50% and ≥75% reduction in monthly migraine days. For the ≥50% reduction in monthly migraine day endpoint, placebo response in the no prior treatment failed group was 32.6%, in the ≥1 failed treatment 17.5%, and in the ≥2 failed treatments 11.1%.
Conclusion
Erenumab showed consistent efficacy in episodic migraine patients who had failed prior preventive treatments and was well tolerated across subgroups. The data suggest prior patients with prior treatment failures have lower placebo response rates.
Introduction
There is a pressing need for new preventive treatments in migraine. Of all patients with migraine, nearly one-third have a frequency of three or more days per month (1), and over half report severe impairment or the need for bed rest, which can result in patient absenteeism with social and economic consequences (2). Dissatisfaction with preventive treatments was identified as a leading unmet need by subjects with episodic migraine in the American Migraine Prevalence and Prevention Study survey (3). Further, while clinical practice and audit data (4) suggest failure of one preventive does not preclude a response to the next, there is no evidence from controlled trials as to whether another preventive treatment would be effective after one has failed.
Commonly used preventive medications, such as topiramate, propranolol, and amitriptyline, have been repurposed from other indications rather than designed to target a specific pathophysiological mechanism in migraine (5). These treatments are often limited by insufficient efficacy and poor tolerability (6,7), and may require dose titration during initiation in an attempt to mitigate the impact of adverse effects, which can delay the onset of efficacy (7). As a consequence of these limitations, currently used preventive medications are associated with high failure rates and low adherence (8). The development of treatments with a mechanism of action specific for migraine has the potential to provide efficacious and better-tolerated migraine therapies (9).
Calcitonin gene-related peptide (CGRP) is a 37-amino-acid neuropeptide that is widely distributed in human nociceptive pathways (10), and is strongly implicated in the pathophysiology of migraine (11). Erenumab is a fully human monoclonal antibody that selectively targets the canonical CGRP receptor (12). In STRIVE, a Phase 3, 6-month controlled trial in patients with episodic migraine, erenumab doses of 70 mg or 140 mg administered monthly by subcutaneous injection (sc) when compared to placebo significantly reduced migraine frequency, increased the ≥50% response rate, reduced the use of acute migraine-specific medication, and reduced the average daily impact of migraine (13).
There are limited data to inform clinicians on the efficacy and tolerability of preventive medications in patients who have previously failed treatment with other preventive therapies. The aim of the present analysis was to evaluate the efficacy, safety, and tolerability of erenumab in the subgroup of patients in STRIVE who had failed migraine preventive treatment(s), for whom there is a higher unmet need.
Methods
The STRIVE study in episodic migraine
The main study design, methods, and statistical analyses were reported previously (11). In brief, STRIVE (NCT02456740) was an international, randomized, double-blind, placebo-controlled, Phase 3 trial of erenumab 70 mg and 140 mg sc monthly in adult patients with episodic migraine. During the 1-month baseline phase and 6-month double-blind treatment phase, subjects completed an electronic diary daily with information about their migraine and non-migraine headaches and use of acute migraine-specific therapies and analgesic medications on a daily basis. There was no limit to the number of prior preventive medications a subject could have failed due to partial response or tolerability issues. However, there was a specific subset of patients who were excluded from the study: Those who had no therapeutic response to three or more migraine-preventive classes, no therapeutic response being defined as no benefit in migraine attack frequency, duration, or severity after ≥6 weeks of treatment at the appropriate dose, as judged by the investigator.
Detailed information on study design, population, and results are provided in the primary publication (13). The protocol and patient consent information were approved by all relevant ethical review boards, all subjects gave written informed consent, and the study was conducted in accordance with the principles laid down in the Declaration of Helsinki and Good Clinical Practice.
Subgroups
Subgroups were defined based on prior migraine preventive treatment failure for reasons of insufficient efficacy or unacceptable tolerability, or both, as recorded by the investigator at enrollment. The number of prior preventive treatment failures for any given subject was based on medication categories. The never-failed group included treatment-naïve patients and patients who had been exposed to a preventive treatment(s) without failure or who were continuously using preventatives throughout the study.
Medications were classified as migraine-preventive treatments based on European guidelines (14), American Headache Society (AHS) guidelines (5), and current practice (6). These were topiramate, serotonin-norepinephrine reuptake inhibitors (SNRIs) (i.e. venlafaxine, desvenlafaxine, duloxetine, and milnacipran), beta blockers (i.e. atenolol, bisoprolol, metoprolol, nadolol, nebivolol, pindolol, propranolol, and timolol), tricyclic antidepressants (i.e. amitriptyline, clomipramine), divalproex sodium/sodium valproate, flunarizine, verapamil, lomerizine, lisinopril, candesartan, butterbur/feverfew/magnesium (≥600 mg/day), riboflavin (≥100 mg/day), clonidine, guanfacine, cyproheptadine, methysergide, pizotifen, carbamazepine, and gabapentin.
Efficacy endpoints
Efficacy analyses of the subgroup with ≥1 prior preventive treatment failure were prespecified in the STRIVE protocol, whereas tolerability analyses for all subgroups and efficacy analyses of the subgroup with ≥2 prior preventive treatment failures were
Statistical methods
Efficacy analyses included all patients who received at least one dose of investigational agent and had at least one change-from-baseline measurement in monthly migraine days. Change from baseline in monthly migraine days and MSMD were analyzed using a linear mixed-effects model including covariates of treatment, visit, treatment by visit interaction, stratification factors (region and prior/current treatment with migraine preventive medication), and baseline value assuming a first-order autoregressive covariance, without any imputation of missing data.
Adverse events
Adverse events (AEs) were tabulated for each subgroup according to Common Terminology Criteria for Adverse Events (CTCAE) grading. Analysis of AEs included all subjects who received at least one dose of investigational agent.
Results
Baseline characteristics of subgroups
Of patients with prior treatment failure, 94% completed the 24-week treatment versus 89% of patients with no prior treatment failure.
Of 955 subjects with episodic migraine randomized in the STRIVE study, 405 (42.4%) reported prior use of migraine-preventive treatment; of these, 370 (91.4%) reported ≥1 and 161 (39.8%) reported ≥2 treatment failures due to insufficient efficacy and/or poor tolerability. The never-failed group included treatment-naïve patients (n = 540; 56.5%) and patients who had previously received a preventive treatment but did not fail or were continuously using the preventive treatment during study (n = 45; 4.7%).
Baseline demographics and clinical characteristics by treatment group.
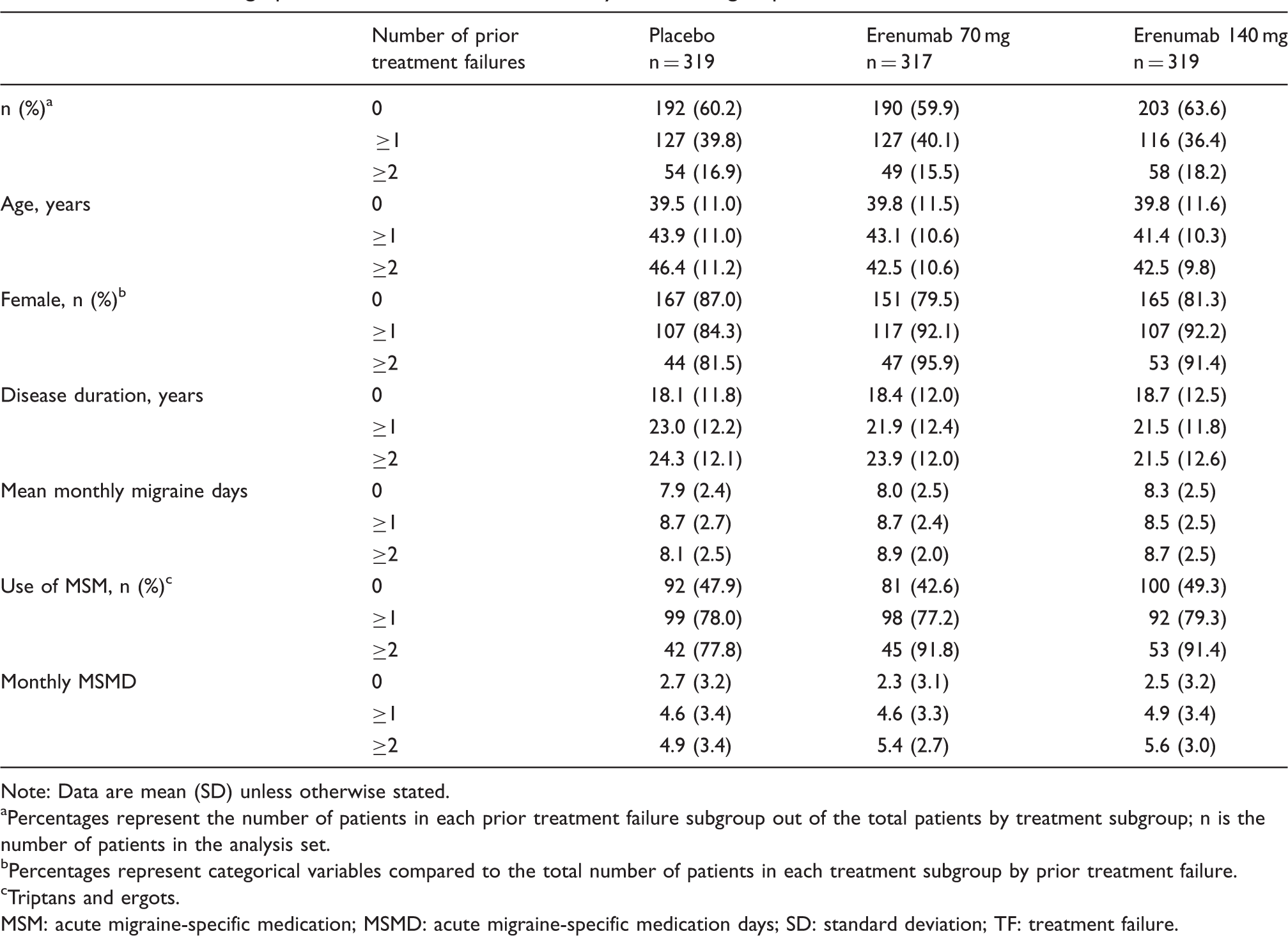
Note: Data are mean (SD) unless otherwise stated.
Percentages represent the number of patients in each prior treatment failure subgroup out of the total patients by treatment subgroup; n is the number of patients in the analysis set.
Percentages represent categorical variables compared to the total number of patients in each treatment subgroup by prior treatment failure.
Triptans and ergots.
MSM: acute migraine-specific medication; MSMD: acute migraine-specific medication days; SD: standard deviation; TF: treatment failure.
Characteristics of prior preventive treatment and treatment failure
Prior preventive treatment and failure. a

Note: SNRIs (serotonin-norepinephrine reuptake inhibitors) include venlafaxine, desvenlafaxine, duloxetine and milnacipran. Beta blockers include atenolol, bisoprolol, metoprolol, nadolol, nebivolol, pindolol, propranolol, and timolol.
Categories are not mutually exclusive and patients may contribute to more than one category.
Percentages represent the number of patients with any prior preventive treatment compared to the overall study population (placebo n = 319; erenumab 70 mg n = 317; erenumab 140 mg n = 319; total n = 955).
Category-specific failure the corresponding category of rate.
Other includes butterbur/feverfew/magnesium (≥600 mg/day)/riboflavin (≥100 mg/day), clonidine/guanfacine, cyproheptadine, methysergide, pizotifen, carbamazepine and gabapentin.
Efficacy
Mean monthly migraine days
Both doses of erenumab resulted in greater reduction in mean monthly migraine days than placebo in all subgroups (Figure 1). Patients with no prior treatment failure had a high placebo response, which resulted in lower treatment differences (least squares mean [LSM] at month 4–6, [95% CI], no prior treatment failure: Erenumab 70 mg vs. placebo, −0.9 [−1.5, −0.3], Change in monthly migraine days up to 6 months (a) and change in mean monthly migraine days versus placebo at month 4–6 (b).
Responder rates
More subjects reached ≥50% reduction in mean monthly migraine days with either dose of erenumab than with placebo across all subgroups (Figure 2). Patients with prior treatment failure had a lower placebo response, resulting in larger treatment differences in the proportion of ≥50% reduction and odds ratios (OR). The OR versus placebo of achieving ≥50% reduction in mean monthly migraine days averaged over months 4–6 was 1.8 and 2.7 for erenumab 70 mg and 140 mg, respectively, in the non-failed subgroup, 2.9 and 3.1 in subjects with ≥1 prior treatment failure, and 2.9 and 4.5 in subjects with ≥2 prior treatment failures.
Proportion of patients reaching ≥50% reduction in mean monthly migraine days up to 6 months.
A similar pattern was observed for subjects achieving ≥75% reduction in mean monthly migraine days (Figure 3): The OR of erenumab-treated subgroups versus placebo was larger among patients with prior treatment failure than among patients who had never failed; erenumab 140 mg showed consistent efficacy in both subgroups (≥1 and ≥2 treatment failures); and placebo responses were greatest in the non-failed subgroup.
Proportion of patients reaching ≥75% reduction in mean monthly migraine days up to 3 months.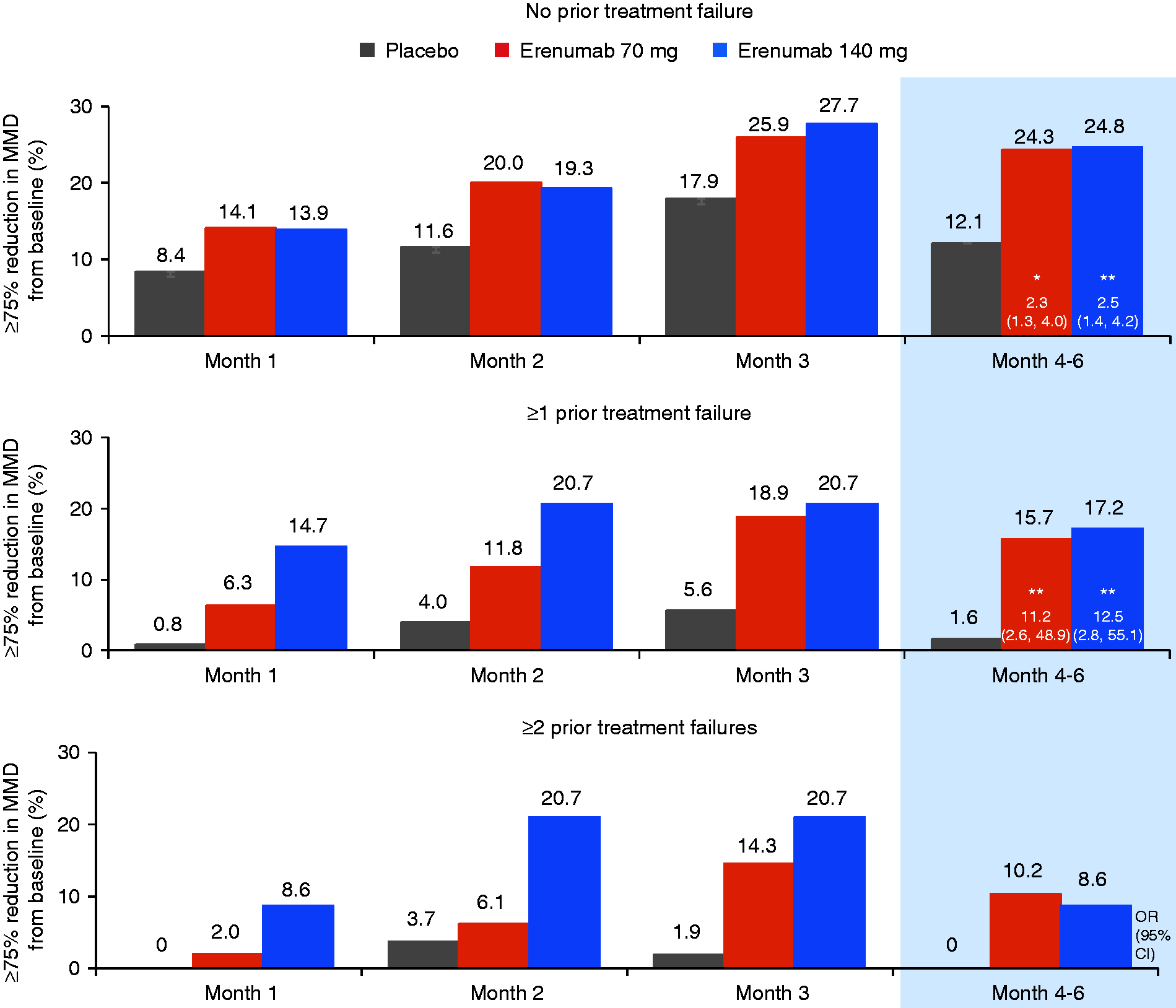
Mean monthly migraine-specific medication days (MSMD)
Treatment with erenumab 70 mg and 140 mg reduced monthly MSMD compared with placebo across all subgroups (0, ≥1, ≥2 failed prior medications; Figure 4). Greater reductions in monthly MSMD were observed in patients treated with erenumab 140 mg with ≥1 and ≥2 failed preventive medications versus patients with no prior treatment failure. Placebo responses were similar across subgroups for this endpoint.
Change in monthly acute migraine-specific medication days up to 6 months (a) and placebo-adjusted change from baseline in acute migraine-specific medication days versus placebo averaged over months 4–6 (b).
Tolerability
Adverse events.

AE: adverse event; SAE: serious adverse event.
Discussion
In this prespecified subgroup analysis, erenumab 70 mg and 140 mg were shown to be effective, safe, and well tolerated in episodic migraine patients with prior treatment failure compared with placebo. Consistent with the results of the main primary analysis (13), erenumab versus placebo significantly reduced monthly migraine days, and more patients achieved ≥50% and ≥75% reductions in mean monthly migraine days from baseline across all treatment failure subgroups. Patients across subgroups and treatment groups within each subgroup had similar monthly migraine days at baseline; however, patients who had failed prior preventive treatments had more MSMD, were slightly older, and had a longer duration of disease. Remarkably, placebo rates comparing ≥50% responder rates were one-third less in patients who had failed two classes of previous preventive.
In patients who had previously failed a preventive treatment, efficacy was consistent in all outcomes analyzed. While the change from baseline effects in erenumab groups were similar between all subgroups, the lower placebo response in the ≥50% and ≥75% response rates and mean monthly migraine days endpoints, possibly driven by lower expectations from subjects who have failed preventive treatment(s) (15), may have contributed to the larger placebo-adjusted treatment effect in the failure population. A lower placebo response in patients who have failed preventive treatments has recently been observed in other retrospective studies (16,17). This observation has been confirmed in a prospective study including episodic migraine patients who had failed 2–4 preventive treatments that also showed low placebo responses in this patient population (18). In contrast to the mean monthly migraine days-based endpoints, the MSMD endpoint exhibited a relatively uniform placebo response between all subgroups. These results support that a lower placebo response might be achieved in migraine trials by including a sufficient proportion of patients who have tried and failed migraine preventives. Further analysis of recent trial data may provide further insights into factors contributing to a low or high placebo response.
Importantly, the benefits associated with erenumab versus placebo observed in the subgroups with prior preventive treatment failure were not accompanied by an increased incidence of AEs. The overall rate of AEs (but not Grade ≥2 or SAEs) was slightly greater in the subgroup of patients with prior treatment failure than in the subgroup without prior treatment failure (including patients naïve to preventive treatment). As with the overall population (13), there was no imbalance between placebo, erenumab 70 mg, and erenumab 140 mg in the incidence of AEs in either the prior treatment failure subgroup or the no prior treatment failure subgroup.
The results of the current analysis with erenumab show significant improvements versus placebo in monthly migraine days, ≥50% and ≥75% responder rates, and MSMD in patients who had failed prior preventive treatments due to unacceptable tolerability or lack of efficacy. These data would suggest that erenumab is of potential use in patients that have failed current standard treatment options. The results of the current analysis of erenumab in episodic migraine are consistent with results of a study of erenumab in chronic migraine in which placebo effects reduce with the number of previous failed medications (15).
Limitations
It remains to be seen how these results translate to the real-world clinical setting. Reassuringly, the effects of CGRP pathway monoclonal antibodies are seen across the spectrum of episodic and chronic migraine with all antibodies tested. Further, this subgroup analysis had a
Conclusions
We report the efficacy and safety of a CGRP receptor monoclonal antibody in patients with episodic migraine with prior preventive treatment failures. Erenumab at both 70 mg and 140 mg doses demonstrated consistently greater efficacy than placebo in patients with prior treatment failures (≥1 and ≥2 categories). Our prespecified and
Clinical implications
Erenumab had consistent efficacy across clinical endpoints in patients with episodic migraine across subgroups, although patients in the non-failed subgroup had a higher placebo response, which resulted in lower treatment differences. Erenumab was effective in patients with prior preventive treatment failure. Erenumab was well tolerated in patients who had failed previous preventives. Placebo response rates are lower in patients who have failed previous preventive medications.
Footnotes
Acknowledgments
The authors thank the STRIVE study participants and investigators for their commitment to this study. The authors also thank Jackie L Johnson, PhD and Matt Lewis, PhD (Novartis Ireland Ltd, Dublin, Ireland) for providing medical writing support/editorial support, which was funded by Novartis Pharma AG, Basel, Switzerland in accordance with Good Publication Practice (GPP3) guidelines (![]() ).
).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PJG reports grants and personal fees from Amgen and Eli-Lilly and Company; personal fees from Alder Biopharmaceuticals, Allergan, Autonomic Technologies Inc., Dr Reddy’s Laboratories, Biohaven Pharmaceuticals, Electrocore LLC, eNeura, Novartis, Scion, Teva Pharmaceuticals, Trigemina Inc, MedicoLegal work, Journal Watch, Up-to-Date, Oxford University Press, Massachusetts Medical Society, and Wolters Kluwer; and a patent: Magnetic stimulation for headache, assigned to eNeura without fee. KP reports research support and/or consulting fees from Amgen, Novartis, Lilly, Allergan, TEVA, and Autonomic Technologies Inc. GB reports research support and/or consulting fees from Allergan, Menarini, Novartis, Pfizer, Reckitt Benckiser and Teva. JB reports research grants and/or consulting fees from Allergan, Amgen, Clinivest, Colucid, Zozano, Supernus, Depomed, Pernix, Teva, and Avanir. JK is an employee of and holds stock in Novartis. FZ, HP, DM, and RL are employees of and hold stock in Amgen. Employees of the sponsors were involved in study design, data collection, analysis and interpretation. All authors, including those who are employees of the study sponsors, drafted and/or revised the manuscript and approved the final version for submission.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Amgen, Thousand Oaks, CA, USA and Novartis Pharma AG, Basel, Switzerland.
Contributors
All authors participated in the study design, implementation, and/or conduct of the study. All authors contributed to the review of the protocol and approved the final manuscript.
Ethical approval and conduct
The study protocol was reviewed by the Independent Ethics Committee or Institutional Review Board for each center. The study was conducted according to the ethical principles of the Declaration of Helsinki.
Patient information and consent
Informed written consent was obtained from each subject before conducting any procedures. Additional information on the study was provided verbally by the study investigator or in a written format.
