Abstract
Objective
To investigate if calcitonin gene-related peptide infusion induces migraine-like attacks in chronic migraine patients.
Methods
Fifty-eight patients with chronic migraine, either with or without headache on the experimental day, were assessed for the incidence of migraine-like attacks after an intravenous infusion with calcitonin gene-related peptide 1.5 µg/min over 20 min. The primary endpoint was the incidence of migraine-like attacks after calcitonin gene-related peptide. Exploratory endpoints were the association between the incidence of migraine-like attacks and presence of headache on the experimental day, and headache frequency in the past month. Migraine-like attack data was compared to a historic cohort of 91 episodic migraine patients without headache on the experimental day. Total tenderness score, pressure-pain threshold and supra-threshold pressure pain at baseline were investigated in relation to incidence of migraine-like attacks and presence of headache on the experimental day.
Results
In total, 83% of the 58 chronic migraine patients developed migraine-like attacks after calcitonin gene-related peptide infusion. Migraine-like attacks were found in 92% of chronic migraine patients with headache on the experimental day compared to 65% of chronic migraine patients without headache on the experimental day (p = 0.035). No differences were observed in total tenderness score and pressure-pain threshold between chronic migraine patients with and without headache on the experimental day. The incidence of migraine-like attacks following calcitonin gene-related peptide in chronic migraine patients without headache (65%) was equal to the historic cohort of 91 episodic migraine patients without headache (67%) on the experimental day.
Conclusions
Chronic migraine patients are hypersensitive to calcitonin gene-related peptide. The potency of calcitonin gene-related peptide as a migraine inductor is increased in chronic migraine patients with ongoing headache. We suggest that calcitonin gene-related peptide, besides being a migraine trigger also acts as a modulator of nociceptive transmission in the trigeminal system.
Keywords
Introduction
Chronic migraine (CM) has an estimated prevalence of ∼9% in patients with migraine (1) and is associated with high personal and societal costs (2). CM patients report higher attack frequencies, higher attack-related pain intensities and average attack durations double that of episodic migraine patients (EM) (2). Population-based studies report that EM transforms into CM and vice versa (3,4). Although the mechanisms driving these transformations are uncertain, central sensitisation (an increased responsiveness of nociceptors in the CNS) is widely believed to be involved (5). In this context, calcitonin gene-related peptide (CGRP) may play a role (6). CGRP is a well-established experimental trigger of migraine in patients with EM (7). Moreover, monoclonal antibodies targeting CGRP or its receptor have been proven efficacious in the preventive treatment of migraine in both EM and CM (8). Hypersensitivity to CGRP in CM patients has never been investigated before. Here we conducted an open-label, single arm CGRP provocation study to investigate CGRP sensitivity and mechanical pain thresholds in CM patients. We compared our findings to previous comparable data on CGRP provocation in EM patients.
Methods
We recruited patients aged 18–75 years from the outpatient clinic of the Danish Headache Center with a diagnosis of CM according to the International Classification of Headache Disorders, 3rd edition (ICHD-3) (9). Patients were migraine free for at least 24 h before the experimental day but were allowed to have a non-migrainous headache with a pain intensity of ≤3 on a 0–10 numeric verbal rating scale (NRS). Exclusion criteria were i) known cardiovascular or cerebrovascular disease; ii) any psychiatric conditions, unless well treated; iii) hypertension (>150 mmHg systolic or >100 mmHg diastolic blood pressure) or hypotension (<90 mmHg systolic or <50 mmHg diastolic blood pressure) on the experimental day; iv) intake of any analgesics or migraine-specific acute medications within 24 h of the experimental day and v) medication overuse headache. Women of childbearing age were excluded if currently breastfeeding or if testing positive for pregnancy on a urine-hCG level test on the experimental day. The study was approved by the Ethics Committee of the Copenhagen Capital Region (H-16014580, H-19015903) and the Danish Data Protection Agency, and was conducted according to the Declaration of Helsinki of 1964, as revised in 2008.
Experimental design
The study was conducted as a non-randomised, single-arm open-label study. All patients arrived non-fasting and free of common analgesics and migraine-specific acute medications for at least 24 h. Eligibility criteria were assessed for all patients, and females of childbearing age were required to perform a urine pregnancy test. Thresholds for mechanical pain were examined prior to the CGRP-infusion. All patients received a 20 min continuous intravenous infusion of 1.5 µg/min αCGRP (BIO-Techne, Minneapolis, USA). This total dosage of 30 µg αCGRP is similar to previous provocation studies in migraine analysis (13–15). CGRP infusion vials were produced at the central pharmacy in the capital region of Copenhagen. Patients were informed about the possible side effects of CGRP, and that CGRP might induce headache, but the timing and characteristics of CGRP-induced headache were not disclosed. All procedures were performed with the patients in the supine position. An 18G intravenous catheter was placed in a cubital vein for CGRP administration. Baseline assessments were performed after 15 min of bed rest and were immediately followed by start of the infusion (T = 0). CGRP was infused over 20 min using a time- and volume-controlled infusion pump. The patients were monitored for a total of 60 min from the start of the infusion, after which they were discharged home. Headache characteristics, vital signs (blood pressure and heart rate), adverse events and medication use were recorded every 10 minutes from infusion start until discharge (T = 0–60 min) using a standardised questionnaire. The patients were instructed to fill out a standardised home diary after hospital discharge, recording headache characteristics, adverse events and medication use once every hour until 12 h post infusion start.
Systematic review of open-label CGRP-provocation studies in episodic migraine
We compared our data with previously published studies of EM patients receiving a single intravenous infusion with CGRP. We searched Pubmed and Embase on 30 May 2020 for the following search string: “(Migraine and (calcitonin gene-related peptide or CGRP) and (Provocation or infusion) and Human)”.
Studies were eligible if they met the following criteria: a) human subjects with migraine without aura according to the at the time current version of the ICHD; b) open-label, non-randomised study; c) intravenous infusion with CGRP; d) study reports the number of participants that develop migraine-like attacks following CGRP infusion. Studies were excluded for the following reasons i) placebo-controlled studies; and ii) studies included participants with CM, migraine with aura or hemiplegic migraine. The following variables were extracted from all eligible articles: a) author and publication year; b) study site; c) dose and duration of CGRP infusion; d) headache on the experimental day; e) total number of participants and number of participants reporting migraine following CGRP infusion. One investigator (AI) performed the search, study selection and data extraction.
TTS, PPT and STPP
Mechanical pain assessments were performed either by a trained biomedical laboratory scientist (MBB) or a physician (AI). Pericranial muscle tenderness was examined using the total tenderness (TTS) score (10) using a validated approach (11). TTS was assessed with conventional palpation on eight pairs of muscles and tendon insertions: Masseter, lateral pterygoid, frontal muscle, temporal muscle, mastoid process, sternocleidomastoid, neck-muscle insertions on the occipital bone and trapezius. All patients were examined in a dental chair. Palpation was performed by applying a pressure of 140 arbitrary units with the second and third fingers, using small rotating movements for approximately 5 sec. Patients were then asked to grade the tenderness on each side on a 0–3 NRS, where 0 = no tenderness, 1 = mild tenderness with no visible discomfort, 2 = moderate tenderness with or without visible discomfort and 3 = marked tenderness and a visible expression of discomfort (11).
Pressure pain thresholds (PPT) were recorded using an electronic pressure algometer (Somedic AB, Sweden). A circular stimulation probe (0.5 cm2 contact area) with a pressure loading rate of 10 kPa/sec was used. Pain thresholds were examined bilaterally in the temporal muscle, trapezius muscle and on the dorsum of the second finger of the non-dominant hand (NDH). The temporal muscle was examined 3 cm above the midline of a line drawn between the lateral canthus and the external ear canal while m. trapezius was stimulated 3–5 cm lateral to the spinous process of vertebra C5. PPT was performed by placing the algometer perpendicular to the surface while steadily increasing the pressure. The patients were instructed to indicate the first sign of any pain by pressing a handheld button, at which point the value shown on the algometer display would freeze. Supra-threshold pressure pain (STPP) was recorded on the dorsum of the second finger of the NDH and bilaterally on the temporal muscles. A pressure 1.5 times the value recorded during PPT was applied at each point, after which the patient scored the pain level on a 0–100 visual-analog scale. All examinations for PPT and STPP were performed three times at least 30 sec apart, with the final score calculated as an average of the three scores on the left and right sides.
Data analysis
The primary endpoint was the incidence of migraine-like attacks after CGRP. Exploratory endpoints were the association between the incidence of migraine-like attacks and presence of headache on the experimental day, and headache frequency in the past month. Migraine-like attack data was compared to a historic cohort of 91 episodic migraine patients without headache on the experimental day. The differences in TTS, PPT and SSTP for CM patients with headache compared to CM patients without headache on the experimental day.
Categorical variables were presented as the total number and the proportion of the total. Numerical variables were presented as mean ± standard deviation if normally distributed and median with 25th to 75th centiles (IQR) if non-normally distributed. Migraine induction was derived from the standardised questionnaire and the headache diary. Migraine was defined as a headache meeting the following criteria for at least one time point: i) Headache fulfilling criteria C and D of the ICHD-3 definition of migraine without aura (9) or ii) a headache mimicking the patient’s usual migraine and attempted treatment with the patient’s usual rescue medication (9). A diary was deemed eligible if it had ≤25% missing data, in which case missing variables were imputed by last observation carried forward. Patients with diaries with >25% of missing data were not included in the analysis. The area under the curve (AUC) for the headache intensity was calculated using the trapezoid rule (12) and was adjusted for baseline headache, defined as the NRS at T = 0 carried forward. We performed a logistic regression analysis to assess the relationship between headache on the experimental day and headache frequency in the past month, and migraine induction. A significant positive association was observed between headache on the experimental day and number of headache days in the past month, so that patients with more frequent headache were more likely to participate with headache on the experimental day. We therefore performed univariate logistic regression for each variable but decided not to include both variables in the combined model. Differences in migraine induction rates between EM compared to the pooled sample of patients with CM, and EM compared to CM with and without headache on the experimental day were examined with the z test of proportions. Differences in TTS, PPT and STPP between patients with and without headache on the experimental day were analysed with the two-sample t-test or the Mann-Whitney test, depending on the distribution of the data, while Spearman’s Rank Correlation was used to assess the correlation between these variables and headache frequency in the past month. Statistical significance was defined as α ≤ 0.05. Data analysis was performed with Microsoft Excel for Office 365 and R (version 3.6.0).
Results
Fifty-eight patients (52 women and six men) completed the CGRP infusion (Figure 1). Mean age was 43.3 ± 13.1 years. Twenty patients were headache free on the experimental day while 38 patients reported headache with median intensity of NRS = 2 (range 1–3) (Table 1). None of the patients reported a headache fulfilling the criteria for migraine attack prior to the CGRP infusion. Study characteristics for each patient are presented in Table A in the supplemental material.
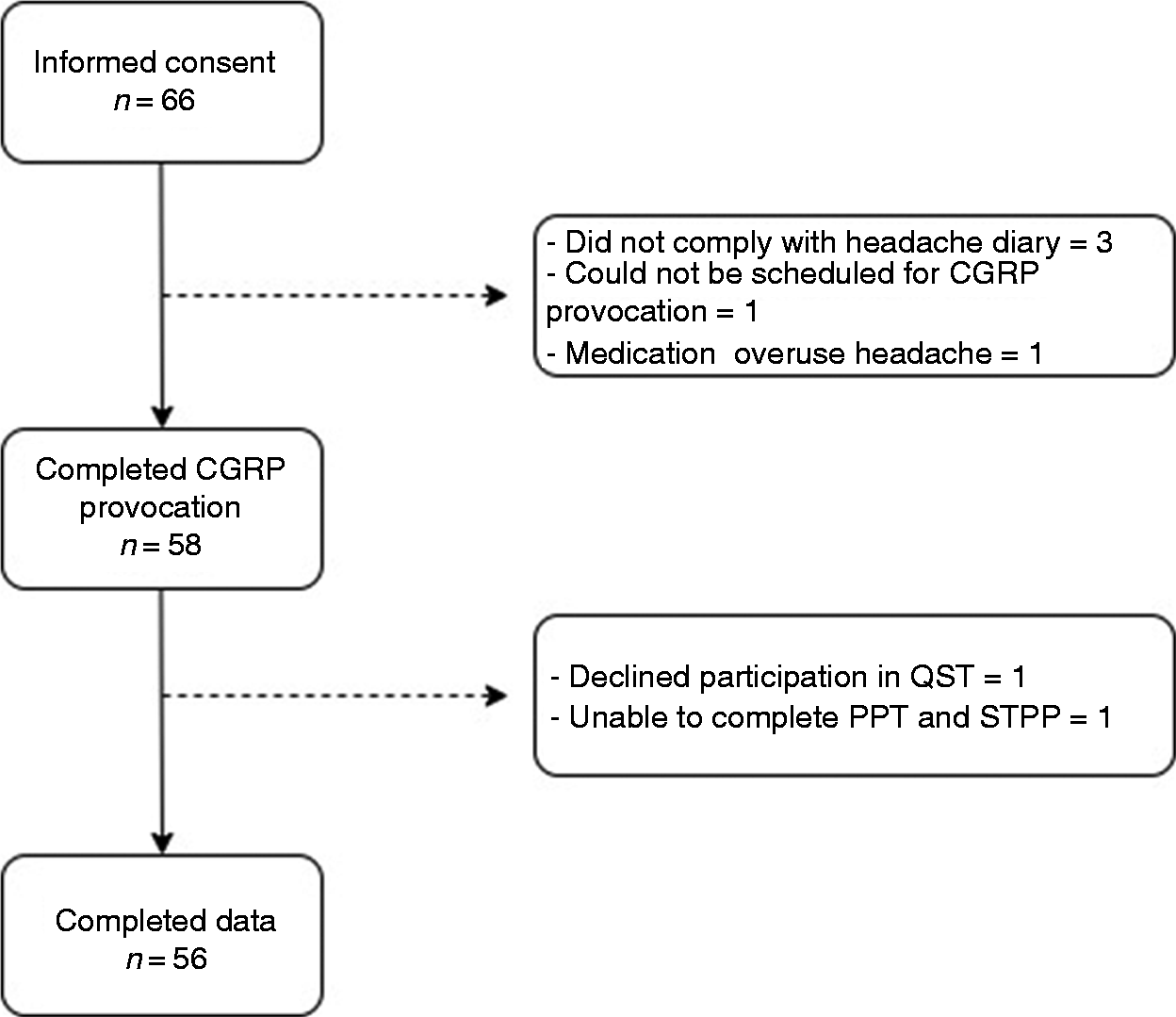
Flow chart.
Baseline characteristics.

*All patients, including patients not currently on preventive medications, were on a waiting list for treatment with erenumab.
CGRP-induced migraine in chronic migraine
Forty-eight of 58 patients (83%, 95% confidence interval (CI) = 71–90%) experienced migraine-like attacks after CGRP infusion (Table 2). Median time to onset was 40 min (IQR = 20–180 min). Headache intensity peaked after a median of 120 min (IQR = 40–360 min) with a mean increase in headache intensity from baseline to peak intensity of NRS = 4.9 ± 2.2 (Figure 2(a)). The exploratory analysis showed that 92% (95% CI = 84–100%) of patients with headache on the experimental day developed migraine-like attacks after CGRP infusion, whereas the corresponding result was 65% (95% CI = 44–86%) for patients without headache. The difference in proportions was statistically significant, χ2 (1, n = 58) = 6.6747, p < 0.01. The univariate logistic regression showed a significant association between headache on the experimental day and migraine induction (β = 1.84, 95% CI = 0.41–3.49, p = 0.016, AIC (Akaike Information Criterion) = 50.9). The association between headache frequency over the past month and migraine induction did not reach significance (β = 0.01, 95% CI = −0.002 to 0.020, p = 0.058, AIC=53.6). Following CGRP infusion, 98% experienced flushing, 97% a warm sensation and 78% palpitations within 10 min after the beginning of the infusion. The most common adverse events experienced by more than 10% of the patients were orbital, retroorbital or periorbital pain (21%), jaw pain (17%) and a freezing sensation (10%).
CGRP-induced migraine characteristics and headache characteristics in patients grouped by headache at baseline.
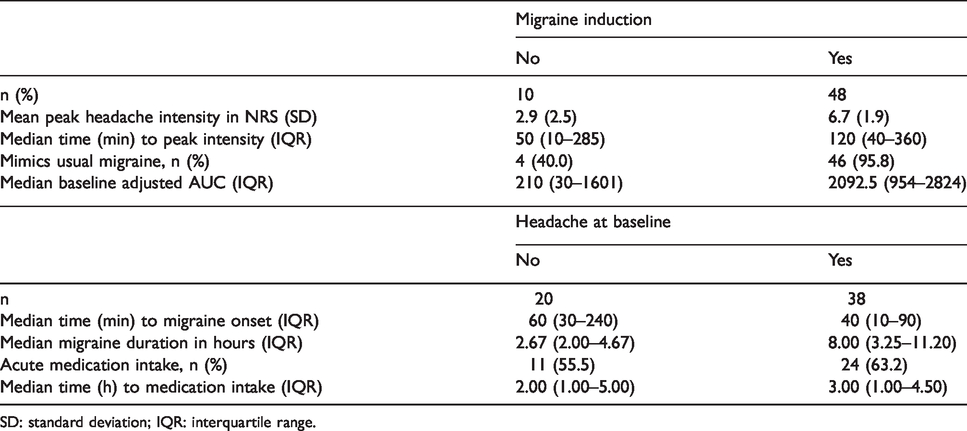
SD: standard deviation; IQR: interquartile range.

Headache intensity and hemodynamic variables.
Historic data on CGRP-provocation studies in episodic migraine
The electronic database searches yielded 223 hits. Following abstract and full-text screening, three studies were determined eligible and included in the analysis (13–15). The three studies included a pooled sample of 91 subjects. All studies were performed at the Danish Headache Center. The proportion of female patients was 89% for the pooled sample, while mean age was 35 years. All subjects had EM with migraine frequencies ranging from once every second month to 9 days/month. Two studies required that subjects were headache free for ≥2 days, while one study required subjects to be headache free for ≥5 days. All subjects received an intravenous infusion with CGRP 1.5 µg/min over 20 min. The incidence of CGRP-induced migraine was 67% (95% CI = 53–82%) for the pooled sample of subjects with EM.
Comparison of CGRP-induced migraine between episodic and chronic migraine
The incidence of CGRP-induced migraine was significantly higher in CM compared to EM (83% vs. 67%, p = 0.035). A post-hoc analysis stratifying CM patients by presence of headache on the experimental day showed no difference in the incidence of CGRP-induced migraine between CM without headache on the experimental day and EM (65% vs. 67%, p = 0.87), while a significant difference was found between patients with CM with headache on the experimental day and EM (92% vs. 67% p = 0.0029).
Quantitative sensory testing
Fifty-seven patients completed TTS and 56 patients completed PPT and STPP prior to CGRP infusion. Mean TTS was 19.6 ± 9.1. Mean PPT was 262 ± 88.4 kPa in the dorsum of the second finger of the non-dominant hand (NDH), 193 ± 114 kPa in the trapezius and 171 ± 58.4 kPa in the temporal muscle. Median STPP was 22.5 (range: 0–53) in the dorsum of the second finger of the NDH and 31 (range: 1–63) in the temporal muscle (Table 3). No differences were found for TTS, PPT and STPP in the finger (Table 3) between patients with and without headache on the experimental day, although patients with headache on the experimental day reported slightly higher levels of subjective pain for STPP in the temporal muscle compared to patients without headache (34/100 vs. 20/100, p = 0.041). There was no correlation between TTS, PPT and STPP and headache frequency in the past month.
Pressure pain threshold and supra-threshold pressure pain, overall results and results stratified by headache at baseline.

kPa: kilopascal; NDH: dorsum of second finger of non-dominant hand; PPT: pressure pain threshold; SD: standard deviation; STPP: supra-threshold pressure pain; TTS: total tenderness score.
Hemodynamic variables
Heart rate (HR) increased by 49.0% ± 22.6% on average, while MAP decreased by 12.3% ± 7.4% (Figure 2(b)). MAP returned to baseline before discharge, while average heart rate remained elevated at T = 60 by 15.0% ± 15.0%. No differences in hemodynamic responses were observed between patients with and without headache at baseline.
Adverse events
The most common adverse events were flushing (n = 57, 98.3%), warm sensation (n = 56, 96.6%), palpitations (n = 45, 77.6%), No differences in adverse events were observed between patients with and without headache at baseline.
Discussion
The present study showed that a larger proportion of CM patients with headache on the experimental day (92%) reported migraine-like attacks after CGRP compared to headache-free CM on the experimental day (65%). The proportion of headache-free CM on the experimental day (65%) was similar to historic data on headache-free headache EM patients (67%). We found no difference in mechanical pain detection thresholds in patients with and without headache on the experimental day.
The higher frequency of migraine-like attacks in CM patients with headache on the experimental day compared to patients without headache raises questions on CGRP’s role in nociceptive transmission in migraine. CGRP is localised in nerve fibres in the perivascular space surrounding cerebral vessels (24), in meningeal nociceptors (25) and in the trigeminal ganglion (26), and is released upon noxious stimuli (25,27,28). Human and animal studies have shown that CGRP is not directly algogenic (29–31), but that it most likely acts as a pain modulator. Systemic administration of CGRP antagonists inhibits inflammatory pain (32) and alleviates mechanical allodynia (33). In an animal model of peripheral pain, repeated subthreshold challenges with CGRP caused long-lasting sensitisation (34), even though direct CGRP application has not been shown to excite or sensitise meningeal nociceptors (35). Activation of the CGRP receptor initiates an intracellular cascade with cAMP accumulation and activation of protein kinase A. This, in turn, phosphorylates downstream targets such as Katp channels (36). Compounds affecting downstream signaling of the CGRP receptor, such as the cAMP phosphodiesterase inhibitor cilostazol (37) and the Katp channel opener levcromakalim (36), potently trigger migraine in 86% (37) and 100% (36) of patients with EM, respectively. Interestingly, CGRP induces migraine just as potently as compounds acting downstream of CGRP in patients with ongoing headache. A possible explanation could be activation of the trigeminovascular system in CM patients with mild non-migrainous headache, which lowers the threshold of CGRP to induce a migraine-like attack. Another explanation for pre-existing mild headache and increased CGRP sensitivity could be dysfunction of descending pain modulation. However, we found no difference in mechanical pain thresholds in the temple, trapezius and finger (PPT) between CM patients with and without headache at baseline. In addition, the thresholds were comparable to healthy controls with similar age- and sex in a previous study with an identical methodology (23). Subjective pain ratings to pressure (STPP) were equal in the finger but slightly elevated in the temple in patients with headache compared to those without headache. However, muscle tenderness in the head and neck region, assessed by TTS, was equal between the two groups. Our findings suggest that our patients with mild headache on the experimental day did not show signs of sensitisation of higher-order neurons at the time of examination, and that headache frequency in the past month or dysfunction of descending pain modulation are unlikely causative factors for CGRP sensitivity in CM patients.
It is debated whether EM and CM constitute a disease spectrum or two separate entities (16,17). EM can transform into CM (18), with an estimated 3% of EM patients developing CM over a 1-year period (18), but reversal from CM to EM migraine also occurs with 26% remitting from CM to EM over a 2-year period (19). Migraine attack frequency is a known risk factor for the transformation from EM to CM, with an odds ratio of 20 in EM patients with 10–14 headache-days/month compared to EM patients with 0–4 headache-days/month (3). Other known risk factors include ineffective acute treatment (4), stressful life events (4), depression (20) and obesity (20). It has been proposed that these risk factors and dysfunction of the descending pain-modulating network may lead to a decreased threshold for the generation of migraine attacks (21). Interestingly, two previous studies of CGRP-induced migraine-like attacks in EM patients reported that the median time until onset of attack was 165 min (13) and 180 min (15), which was longer than the median onset of 60 min found in CM patients without headache prior to provocation (Table 2). Thus, CM patients seem to have a faster development of induced migraine-like attacks than EM patients. In the present study, the CM patients that were headache free prior to the CGRP infusion were equally as likely to report CGRP-induced migraine as headache-free EM patients. Moreover, we found no correlation between headache frequency during the past month and the incidence of migraine-like attacks following CGRP infusion in CM. One previous study specifically investigated the extent to which attack frequency might increase the likelihood of migraine induction in a human provocation model (22). The major outcome was no difference in the incidence of migraine-like attacks following infusion with an NO donor between migraine patients with ≤4 attacks/year compared to patients with ≥12 attacks/year (22). Thus, most of our findings support that EM and CM are part of the same spectrum, with a shared underlying pathophysiology.
Limitations
The current study has some limitations that need to be addressed. First, headache and migraine frequency over the past month were assessed retrospectively. Some patients presented a headache diary; this, however, was not the case for all patients. Ideally, this should be collected prospectively leading up to the experimental day. Second, because all patients had chronic migraine, all reported a high number of monthly headache days. This confounds the interpretation of the regression analysis with monthly headache days as a predictor. Ideally, patients with a wider range of monthly headache days and migraine days should be recruited. However, this was not feasible in this study due to recruitment of chronic migraine patients from a specialised headache center, which receives the most severely affected patients. Third, we did not record the number of days since last migraine, days since last intake of migraine-specific medication, or the duration of headache prior to the CGRP infusion. Fourth, patients were allowed to use migraine-preventive medications. However, the proportion of patients using migraine-preventive medications was equally distributed between patients with and without migraine induction, and with and without headache on the experimental day. Furthermore, our results in headache-free CM patients were equal to previous findings in EM patients who were free of migraine preventive medications. Lastly, there is a limitation in the comparison of the present study with historic data from previous studies in episodic migraine, which ideally should have been appropriately studied in a prospective and patient-matched fashion.
Conclusion and future directions
The migraine inducing potency of CGRP was equal in headache-free patients with chronic and episodic migraine. However, CM patients with headache on the day of CGRP infusion reported significantly higher incidences of CGRP-induced migraine. We therefore suggest that CGRP, besides being a potent migraine trigger, also acts as a modulator of nociceptive processing in the trigeminal system. Future studies should investigate the underlying mechanisms that regulate sensitivity to CGRP infusion in migraine.
Key findings
Intravenous infusion with CGRP potently induces migraine in patients with chronic migraine. The migraine induction rate depends on the presence of headache on the experimental day, with 92% of patients with a mild headache developing migraine within 12 h of the CGRP infusion, while the corresponding result was 65% for headache-free patients.
Supplemental Material
sj-pdf-1-cep-10.1177_0333102420981666 - Supplemental material for Hypersensitivity to calcitonin gene-related peptide in chronic migraine
Supplemental material, sj-pdf-1-cep-10.1177_0333102420981666 for Hypersensitivity to calcitonin gene-related peptide in chronic migraine by Afrim Iljazi, Håkan Ashina, Zixuan Alice Zhuang, Cristina Lopez Lopez, Josefin Snellman, Messoud Ashina and Henrik Winther Schytz in Cephalalgia
Footnotes
Acknowledgements
We would like to thank biomedical laboratory scientist Merete Bak Bertelsen for her skilful help with subject recruitment and assistance with CGRP infusions.
Authors’ contributions
Concept and design: AI, HWS, MA.
Data acquisition: AI.
Data analysis: AI, ZAZ.
Interpretation of data: All authors.
Drafting of the manuscript: AI.
Critical revision of the manuscript for important intellectual content: All authors.
Supervision: HWS, HA, MA.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JS and CLL are employees of Novartis Pharma AG and have Novartis shares. HWS has received speaking fees from Novartis, Lilly and Teva and has received a grant from Novartis. MA has received personal fees from Alder BioPharmaceuticals, Allergan, Amgen, Eli Lilly, Lundbeck, Novartis, and Teva. MA also participated in clinical trials as the principal investigator for Alder, Amgen, Eli Lilly, Novartis, and Teva. MA also serves as an associate editor of Cephalalgia, associate editor of Headache and associate editor of the Journal of Headache and Pain. MA is President of the International Headache Society. MA reports research grants from The Lundbeck Foundation, The Novo Nordisk Foundation and Novartis. The other authors declare no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received funding from Novartis.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
