Abstract
Background
The present study aimed to investigate whether levcromakalim, a KATP channel opener, induces migraine attacks in people with migraine pre-treated with erenumab, a monoclonal CGRP receptor antibody.
Methods
In this double-blind, placebo-controlled, two-way cross-over study, adults with migraine without aura received a subcutaneous injection of 140 mg of erenumab on day 1. Subsequently, they were randomized to receive a 20-minute infusion of 0.05 mg/ml levcromakalim or placebo on two experimental days separated by at least one week (between days 8 and 21). The primary endpoint was the difference in the incidence of migraine attacks between levcromakalim and placebo during the 12-hour post-infusion period.
Results
In total, 16 participants completed the study. During the 12-hour observation period, 14 (88%) of 16 participants experienced migraine attacks after levcromakalim, compared to two (12%) after placebo (
Conclusions
The induction of migraine attacks via opening of KATP channels appears independent of CGRP receptor activation.
Introduction
Migraine is a disabling neurological disorder that afflicts approximately 14% of the global population (1). Its pathogenesis remains incompletely understood, but increasing evidence points towards the involvement of specific molecular and cellular mechanisms (2). Central to our understanding is calcitonin gene-related peptide (CGRP) (3), a potent endogenous vasodilator that is widely expressed in both the peripheral and central nervous system (4). Upon release from sensory afferents, CGRP binds to its G protein-coupled receptors (5). This, in turn, activates cAMP-dependent signaling pathways, leading to dilation of meningeal arteries and neurogenic inflammation (5).
Ample evidence from human experimental studies has shown that intravenous (IV) infusion of CGRP can induce migraine attacks in the majority of individuals with migraine (6). In addition, several medications against CGRP signaling have proven effective for migraine prevention (7). These include erenumab, a monoclonal antibody (mAb) against CGRP receptors. Findings from both randomized clinical trials and real-world studies have confirmed the effectiveness and tolerability of erenumab for the preventive treatment of migraine (8–11). However, no medication has provided an absolute therapeutic response, indicating the contribution of other mechanisms than CGRP in the pathogenesis of migraine.
One proposed mechanism by which CGRP induces migraine attacks involves downstream opening of ATP-sensitive potassium (KATP) channels on the vascular smooth muscle cells within the walls of the meningeal arteries (12,13). This assertion lends support from preclinical studies showing that CGRP elicits vasodilation via opening of KATP channels (14,15). Furthermore, IV infusion of the KATP channel opener levcromakalim is known to induce migraine attacks in people with migraine (16,17). A recent hypothesis posits that the resultant vasodilation and potassium efflux activates the meningeal nociceptors via mechanical and chemical stimulation, respectively (13). This line of reasoning implicates that KATP channels function downstream of CGRP in migraine pathogenesis. However, recent preclinical research has posed a challenge to this notion by suggesting that levcromakalim-induced hypersensitivity is dependent on the CGRP receptor (18). Consequently, a dedicated examination in humans emerges as critically important.
Understanding the intricate relationship between CGRP and KATP channels in migraine pathogenesis might carry implications for developing mechanism-based treatments. If CGRP blockade prevents levcromakalim-induced migraine attacks, this indicates a potential involvement of CGRP in the downstream signaling cascade. Conversely, if CGRP receptor blockade does not attenuate the migraine-provoking properties of levcromakalim, this suggests that alternative signaling pathways leading to migraine attacks might be able to bypass the CGRP receptor pathway. To our knowledge, the direct effect of blocking the CGRP pathway on levcromakalim-induced migraine has not been assessed in previous human studies.
In the present study, we investigated whether pre-treatment with erenumab, a mAb against the CGRP receptor, would prevent the induction of migraine attacks after IV infusion of levcromakalim, as compared to placebo.
Methods
Study oversight
The study protocol received approval from the Committee on Health Research Ethics in the Capital Region of Denmark (H-22031695) and the Danish Data Protection Agency. The study was furthermore registered with ClinicalTrials.gov (NCT05889442) and adhered to the principles outlined in the Declaration of Helsinki. All participants were required to provide their written informed consent before the commencement of study-related procedures or tasks.
Study population
The study population consisted of otherwise healthy individuals with migraine without aura according to the criteria of the International Headache Classification of Headache Disorders 3 (ICHD-3) (13). Participants were recruited through a Danish website for test subjects (https://forskningnu.dk) or advertisements at local hospitals. Inclusion criteria were age between 18 and 65 years, history of migraine for 12 months or more, migraine frequency of one to five migraine days per month before screening. Potential participants were excluded if they had a history of any other primary or secondary headache disorder (apart from tension-type headache with a frequency of ≤5 days/month) or any serious somatic or psychiatric disease. The intake of any prophylactic migraine medication within 30 days or five plasma half-lives (whichever longer) before screening or the prior intake of therapies targeting the CGRP signaling pathway were not permitted. Further exclusion criteria for female participants were pregnancy, breastfeeding or planning to become pregnant during the study.
Study design
This was a randomized, double-blind, placebo-controlled two-way crossover study. After an initial prescreening by phone, the study comprised the following on-site visits: a screening visit, a pre-treatment visit and two experimental visits (Figure 1).

Study design. ECG, electrocardiogram; s.c., subcutaneous.
At screening, study staff assessed the eligibility criteria, conducted a semi-structured headache interview, and performed a complete medical examination, including blood pressure, heart rate, a 12-lead electrocardiogram and a urine pregnancy test in female participants of childbearing potential. In case of eligibility, the pre-experimental visit was conducted after screening and included the administration of one subcutaneous injection of 140 mg of erenumab in the upper thigh or abdomen. Participants were then randomized into two treatment sequences: levcromakalim on the first experimental day and placebo on the second day, or vice versa. Participants were informed that levcromakalim could lead to headache or migraine attacks without specific details regarding the frequency, timing of onset or characteristics of such induction. Additionally, participants were informed that erenumab might potentially alleviate the effects of levcromakalim. Randomization was performed based on a manual list with a predetermined sequence of treatment for each participant. An unblinded team prepared the infusions according to this list. The unblinded team had no contact to study participants and was not involved in data analysis or interpretation. The randomization list was made available to the blinded investigators only once the study was completed.
The first experimental visit occurred between days 8 and 14 after erenumab administration, and the second was between days 15 and 21. Both visits were separated by at least seven days. Erenumab has an estimated time for maximum concentration (
During the in-hospital phase, research staff recorded headache characteristics using a standardized headache diary and vital signs (i.e. blood pressure and heart rate) every 10 minutes. The headache diary contained information about headache intensity, character, localization, accompanying symptoms, rescue medication and adverse events, as well as whether the headache was like their typical migraine. In addition, the diameter of the superficial temporal artery (STA) was measured every 20 minutes by high-frequency ultrasound (Dermascan C; Cortex Technology, Aalborg, Denmark).
Participants left the hospital one hour after the beginning of the infusion. They were asked to complete the headache diary hourly for further 11 hours (i.e. for a total of 12 hours) after infusion start.
Endpoints
The primary endpoint was the difference in incidence of migraine attacks during the 12-hour observation period after infusion of levcromakalim versus placebo. An attack was deemed present if it met the ICHD-3 criteria C and D for migraine without aura at any time point. Alternatively, the attack had to resemble the participant’s usual migraine attacks and prompting rescue medication use.
The secondary endpoints were the difference in headache incidence and the area under the curve (AUC) for median headache intensity scores over the 12-hour observation period between levcromakalim and placebo. Another secondary endpoint was the difference in AUC for the STA diameter within one-hour post-infusion between the two interventions. The AUCs were calculated according to the trapezium rule. The final secondary endpoint was the difference in incidence of adverse events during the 12-hour observation period between levcromakalim and placebo.
Statistical analysis
Sample size calculation was based on two paired proportions at a 5% significance level with ≥80% power using a one-sided McNemar’s test. We assumed off-diagonal probabilities of 0.1 (proportion of participants with migraine induction exclusively on the placebo day) and 0.7 (proportion of participants with migraine induction exclusively on the levcromakalim day). Upon computation that 10 and 70% of the pairs are discordant, a sample size of 16 participants completing both experimental study days would be required for a balanced distribution. Therefore, we planned to include 16 participants with complete study protocol.
Statistical analyses were performed with SPSS, version 27.0 (IBM Corp., Armonk, NY, USA). We used descriptive statistics to summarize demographic and clinical features. Categorical variables are presented as absolute frequencies and percentages, with numerical variables presented as mean or median values (range or SD), as appropriate by data distribution. The incidence of migraine attacks and headache attacks was analyzed using McNemar’s test as categorical paired data. The AUC values were compared using the Wilcoxon signed-rank test (headache intensity) or paired-sample
Results
Participants were screened from March through June 2023 (Figure 2). In total, 16 participants were enrolled and completed the study. The mean (range) age was 32.9 (range 1–61) years, and most of them were female (n = 14). The mean (range) body mass index was 24.8 (18.6–29.9) kg/m2 and, on average, the participants reported experiencing 3 migraine days (range 1–5) in the month prior to enrollment.

Flowchart of study progress.
Migraine attacks
During the 12-hour observation period, 14 (88%) of 16 participants developed migraine attacks following levcromakalim, compared to two (12%) after placebo (P < 0.001) (Table 1). Migraine attacks were reported by seven participants who received levcromakalim between days 8 and 14 after erenumab administration and seven who received levcromakalim between days 15 and 21. The median time to onset of migraine attacks was 150 minutes (interquartile range (IQR) 60–255) after levcromakalim and 510 minutes (IQR 420–510) after placebo.
Characteristics of spontaneous and provoked headache (0–12-hour observation period).

Localization (bilat, bilateral; unilat, unilateral)/intensity/quality (throb, throbbing; pres, pressing)/aggravation by cough or physical activity
Nausea/photophobia/phonophobia
Only participant without headache on levcromakalim day. STA dilation (from 1.31 mm at baseline to 1.58 mm after 20 minutes) and flushing (between 20 and 30 minutes) confirms the vascular action and thus the proper administration of levcromakalim.
Mild nausea was reported at one single time point (6 hours after administration of levcromakalim). At that time point, the other headache characteristics did not fulfill the ICHD-3 criteria of migraine and the headache was not similar to the participant’s usual migraine. Therefore, this attack was not classified as migraine-like attack. ASA, acetylsalicylic acid.
Headache responses
Among the 16 participants, 15 (94%) reported experiencing headache of any kind after levcromakalim, whereas six (38%) did so after placebo (

Headache intensity on a numerical rating scale (NRS) during the 12-hour observation period after infusion of (a) levcromakalim or (b) placebo. The thin black lines represent individual headache scores, and the thick red line represents the median value.
Hemodynamic variables
During the one-hour in-hospital observation period, the diameter of the STA increased significantly after levcromakalim, compared to placebo (AUC0–60min,

Mean diameter of the superficial temporal artery in the first hour after infusion of levcromakalim or placebo.

Mean (a) arterial blood pressure and (b) heart rate in the first hour after infusion of levcromakalim or placebo.
Adverse events
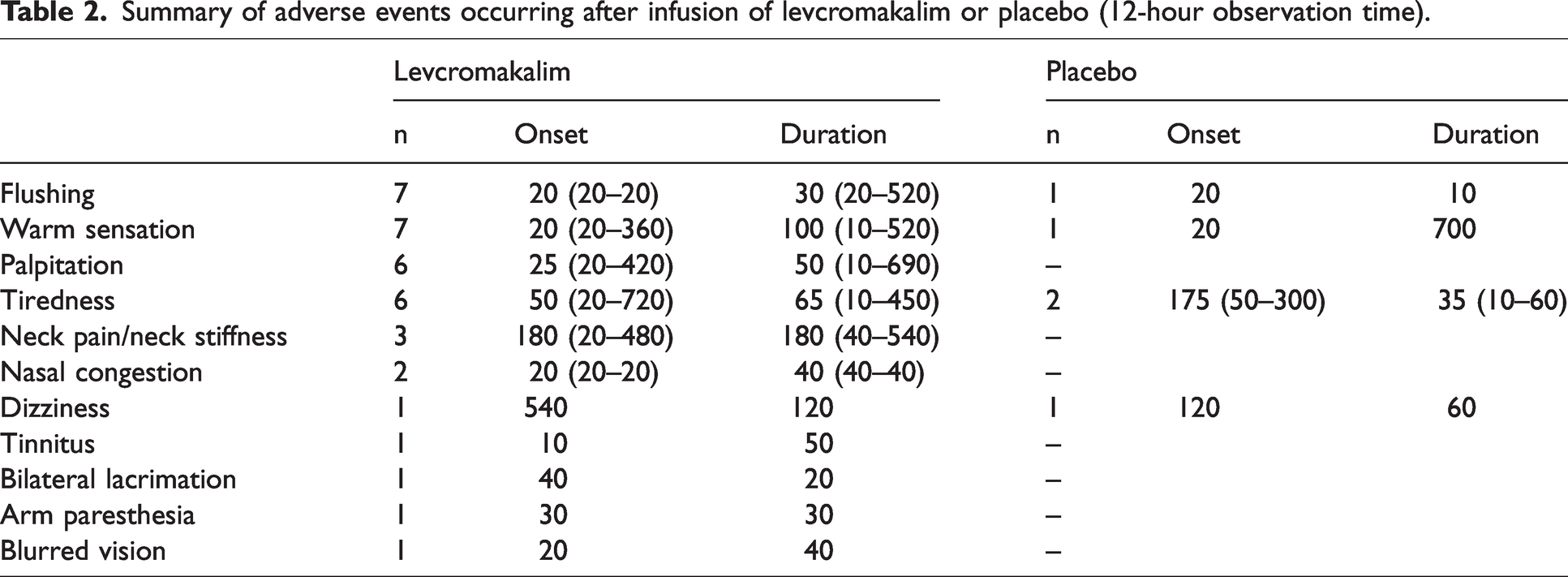
More participants reported adverse events after levcromakalim (n = 14; 88%) than after placebo (n = 2; 13%). The most common adverse events after levcromakalim were flushing, warm sensation, palpitation and unusual tiredness. Table 2 shows all the reported adverse events and their temporal dynamics.
Summary of adverse events occurring after infusion of levcromakalim or placebo (12-hour observation time).
Discussion
The present study demonstrates that the KATP channel opener levcromakalim can induce migraine attacks in participants pre-treated with the CGRP receptor antibody erenumab. These findings provide evidence that the initiation of migraine attacks through the opening of KATP channels does not require CGRP receptor activation. Migraine attacks can be triggered by direct stimulation of downstream elements in the signaling cascade. Hemodynamic changes and the occurrence of adverse events differed between levcromakalim and placebo infusions, consistent with the known profile of levcromakalim in human provocation models (16,17).
According to one proposed concept of migraine pathogenesis, the binding of CGRP to its receptor on vascular smooth muscle cells within the trigeminovascular system increases cAMP, leading to phosphorylation and subsequent opening of KATP channels (21). This process prompts potassium efflux and vasodilation, thereby sensitizing perivascular trigeminal afferents chemically and mechanically (12). The present study directly targets the final step of this proposed signaling cascade, demonstrating that KATP channel opening can effectively induce migraine attacks independently of CGRP receptor activation. Indeed, the use of erenumab at a dose of 140 mg s.c. is estimated to achieve complete saturation of all CGRP receptors (22), thereby preventing CGRP binding and subsequent initiation of the CGRP pathway. However, this does not exclude a residual activation of the CGRP pathway through other receptors of the calcitonin family (23). Accordingly, in a recent human provocation study, erenumab was effective in mitigating migraine induction rates and vascular response to IV infusion of CGRP, but did not reduce them completely (24). Moreover, it is essential to acknowledge that the migraine signaling cascade can be activated upstream by various peptides and neurotransmitters. This study specifically focuses on the blockade of the CGRP receptor and does not offer insights into the functioning of alternative upstream pathways.
Although the model presented here has provided crucial insights into the molecular underpinnings of migraine, it is important to recognize that migraine is a multifaceted disease, and its molecular pathways encompass a range of potential interaction sites. Given the widespread distribution of both CGRP receptors and KATP channels on trigeminal neurons, the possibility of a direct neuronal site of action cannot be dismissed. Nevertheless, our findings support a vascular site of action of levcromakalim (i.e. hemodynamic changes) and the current body of evidence underscores the primary involvement of neurovascular mechanisms in mediating the experimentally induced migraine attacks (13). To strengthen the argument for the downstream role of KATP channels in the proposed signaling cascade, a future study could employ a reverse design (i.e. migraine provocation via CGRP under KATP channel blockade). If IV CGRP failed to induce migraine attacks under KATP blockade, it would provide additional evidence supporting the downstream position of KATP channels.
Our findings align with previous studies on levcromakalim, which included participants with migraine without CGRP-blocking treatment (16,17). The induction rates of migraine attacks were similar to previous studies, along with the time to onset of migraine attacks, the proportion of headache attacks and the spectrum of adverse events (16,17). Changes in blood pressure, heart rate and STA diameter in the present study also match previous findings, suggesting no effect of the CGRP receptor blockade on levcromakalim-induced vasodilation. Although headache intensity on levcromakalim days in our study was somewhat lower than in previous investigations, this might be attributed to individual variability, demographic differences, and different baseline headache characteristics, such as lower average headache intensity during spontaneous attacks (median 6.5 vs. 7.5 NRS) (17) or earlier intake of rescue medication after levcromakalim (median 3.5 hours vs. 5 hours) (17). Nevertheless, it is important to note that the study was not powered to detect statistical differences from previous trials.
Our clinical findings contrast with data from preclinical models. In an
The ability of levcromakalim to induce migraine attacks despite CGRP receptor inactivation reinforces the strategic role of KATP channels in migraine pathophysiology and their potential as new targets for migraine treatment. In preclinical studies, the KATP channel blocker glibenclamide showed promising results in blocking trigeminal nociception (27,28). However, in human provocation models, glibenclamide only delayed headache onset after levcromakalim administration but did not affect headache occurrence (29,30). It should be noted that KATP channels have a complex structure consisting of eight subunits: four pore-forming subunits from the inward rectifier potassium 6 (Kir6) channel family and four regulatory sulfonylurea receptor (SUR) subunits from the ATP-binding cassette C transporter family (12). Different combinations of Kir6 and SUR subunits result in four KATP channel subtypes, expressed in distinctive tissues. Levcromakalim specifically targets KATP channels with SUR2B subunits, broadly expressed in the trigeminovascular system. Glibenclamide, on the other hand, has a higher affinity for the SUR1 subunit found primarily in the pancreas, explaining its antidiabetic properties (12). The low glibenclamide dose given in humans to avoid hypoglycemia might be insufficient to inhibit SUR2B and affect the trigeminovascular system (28). Currently, there is no selective SUR2B blocker available, but it represents a promising target for future development.
Strengths and limitations
The strengths of the present study include its placebo-controlled, randomized, double-blind design, strict inclusion and exclusion criteria and a cross-over design that minimizes clinical variability and enhances control over confounding factors. One possible limitation concerns the autonomic symptoms induced by levcromakalim, such as flushing or palpitations, which might have compromised the blinding and increased the nocebo effect. External factors beyond our control might have influenced the migraine induction, although standardized laboratory conditions were implemented. Moreover, it should be considered that migraine provocation in an experimental setting might not fully reflect the physiological migraine attack initiation during spontaneous attacks. Finally, the in-hospital observation period was limited to one hour, which is shorter than previous investigations. While longer observation periods could provide additional insights, the hemodynamic changes within the first observation hour closely resemble previous studies, making it unlikely that extended observations would yield significant new findings.
Conclusions
Our findings demonstrate that migraine provocation via the KATP channel opener levcromakalim does not require CGRP receptor activation. These findings support the central role of the KATP channels in migraine pathogenesis and highlight their potential as a novel target for migraine treatment.
Clinical implications
Levcromakalim, a KATP channel opener, induces migraine attacks in patients with migraine pre-treated with erenumab. The CGRP receptor blockade does not alter the hemodynamic effects typically induced by levcromakalim. Migraine provocation via KATP channel opening is independent from CGRP receptor activation, underscoring the central role of KATP channels in the downstream intracellular signaling cascade of migraine pathophysiology.
Footnotes
Acknowledgements
We thank the unblinded staff members (Anne Mette Autzen, Rune Häckert Christensen, Marianne Hestad and Susanne Leed) for their help in preparing the infusions. This study received funding from Novartis Pharma AG. BR was supported by a research grant of the German Research Foundation (GZ: RA 3907/1-1). MA was supported by the Lundbeck Foundation professor grant (R310-2018–3711).
Author contributions
BR and BAC were responsible for investigations. BR was responsible for data curation, formal analysis, visualization and writing the original draft. TPD, FMA, HA, JS, TMT and MA were responsible for conceptualization. TPD, FMA, HA and MA were responsible for methodology. TPD was responsible for project administration. TPD, BAC, FMA, HA, JS, TMT and MA were responsible for reviewing and editing. MA was responsible for project administration and supervision.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
BR reports personal fees from AbbVie, Eli Lilly, Lundbeck, Novartis and Teva, as well as research grants from Novartis, all outside of the submitted work. BR reports serving as junior associate editor of
