Abstract
Introduction
A demyelinating plaque and neurovascular contact with morphological changes have both been suggested to contribute to the etiology of trigeminal neuralgia secondary to multiple sclerosis (TN-MS). The aim of this study was to confirm or refute whether neurovascular contact with morphological changes is involved in the etiology of TN-MS.
Methods
We prospectively enrolled consecutive TN-MS patients from the Danish Headache Center. Clinical characteristics were collected systematically. MRI scans were done using a 3.0 Tesla imager and were evaluated by the same experienced blinded neuroradiologist.
Results
Sixty-three patients were included. Fifty-four patients were included in the MRI analysis. There was a low prevalence of neurovascular contact with morphological changes on both the symptomatic side (6 (14%)) and the asymptomatic side (4 (9%)), p = 0.157. Demyelinating brainstem plaques along the trigeminal afferents were more prevalent on the symptomatic side compared to the asymptomatic side (31 (58%) vs. 12 (22%), p < 0.001). A demyelinating plaque was highly associated with the symptomatic side (odds ratio = 10.6, p = 0.002).
Conclusion
The primary cause of TN-MS is demyelination along the intrapontine trigeminal afferents. As opposed to classical trigeminal neuralgia, neurovascular contact does not play a role in the etiology of TN-MS. Microvascular decompression should generally not be offered to patients with TN-MS.
The study was registered at ClinicalTrials.gov (number NCT04371575)
Keywords
Introduction
Trigeminal neuralgia (TN) is characterised by very severe attacks of unilateral facial pain that can be triggered by trivial sensory stimuli (1,2). TN and multiple sclerosis (MS) have been known to be associated since the description of demyelinating plaques, which were found at autopsies in the pontine area in patients with MS and TN (TN-MS) patients in 1894 (3). The prevalence of TN in patients with MS is up to 6% (4) and MS patients have a 20-fold increased risk of developing TN compared to the background population (4).
Imaging studies have shown that a neurovascular contact (NVC) with morphological changes of the trigeminal nerve is an important cause of primary TN (5,6). By microvascular decompression, the trigeminal nerve can be decompressed from offending vessels, and in primary TN the procedure is highly efficacious adding further support to the importance of NVC in primary TN (7–9).
A recent Italian study with 28 TN-MS patients recruited from an MS cohort reported that both NVC with morphological changes and pontine demyelinating plaques in the territory of the afferent trigeminal fibers contribute to the disease etiology of TN-MS in a dual concurrent mechanism (10). Proof of such a dual mechanism would be clinically highly important because it would confirm that microvascular decompression should more often be considered in medically refractory TN-MS patients.
This is a systematic prospective study cohort of consecutive TN-MS patients with blinded evaluation of 3.0 MRI scans. We aim to describe the clinical characteristics and neuroanatomical abnormalities in patients with TN-MS. We hypothesised that both pontine demyelinating plaques and NVC with morphological changes contribute to the disease etiology in TN-MS patients.
Materials and methods
This prospective, cross-sectional study was performed at the Danish Headache Center (DHC), Department of Neurology, Rigshospitalet – Glostrup, Denmark, from May 2012 to October 2019. DHC receives patients from all over Denmark, referred from general practitioners, private neurologists and other hospital departments.
Patient inclusion and collection of data
After referral to DHC, all the included patients underwent a clinical interview, an examination by experienced neurologists, and a 3.0 tesla MRI with a special protocol for the trigeminal nerve. The methodology of the data collection was largely similar to our previously published studies in primary TN (5,11–13). We prospectively and systematically collected data from consecutive patients with the diagnosis of TN-MS who attended out-patient visits at DHC using a purpose-built semi-structured interview, which among others included pain characteristics, duration of disease, onset of disease, previous and current medical treatment, previous surgical treatment, other diagnosis of headache, comorbidities and neurological abnormalities. To assess pain intensity, we used the verbal numerical rating scale (VNRS) which ranges from 0 = “no pain at all” to 10 = “worst possible pain”.
Facial sensory abnormalities were examined with routine neurological examination for touch with cotton swab both extra- and intraorally and for nociception using pinprick extraorally in the three branches of the trigeminal nerve. We did not include sensory abnormalities in the statistical analyses in patients who had undergone prior TN surgery, because sensory abnormalities in these patients could have been a complication to surgery (12).
The diagnosis of TN was based on the ICHD-2, ICHD-3 beta and ICHD-3 criteria, respectively, depending on time of inclusion, with the important exemption that we included patients who did not have a visible plaque on the symptomatic side but who otherwise fulfilled the diagnostic criteria to avoid circular reasoning (11,14,15). Exclusion criteria were communication barriers and no informed consent. Exclusion criteria regarding MRI analyses were a) microvascular decompression prior to MRI; b) no MRI due to, for example, claustrophobia or implanted magnetic devices; c) MRI not performed according to the pre-specified scanning protocol. MRI findings of patients with bilateral TN are presented separately because paired analyses are not possible in bilateral pain. The number of patients in the inclusion period determined the sample size.
Definitions of clinical characteristics
Pain onset and quality
TN onset was defined as the year of the first symptom of TN, while MS onset was the year of MS diagnosis. We defined two types of pain: a) Paroxysmal pain lasting from a split second to two minutes and described by patients as superficial, stabbing, or electrical lightning-like; b) concomitant persistent pain, which occurred in addition to the paroxysmal pain, lasting from 2 min up to being constantly present and described by patients as nagging, throbbing or dull.
Remission periods
Remission periods were defined as pain-free periods lasting at least a month without medication or with a low dosage of medication defined as carbamazepine 100 mg daily, oxcarbazepine 150 mg daily or corresponding doses of other relevant drugs. Some patients reported periods with almost no pain but only if they avoided triggering the pain; for example, by avoiding chewing hard foods or shaving. This was not deemed a remission period.
Subtypes of multiple sclerosis
Information regarding the subtype of MS was collected from journal notes from each patient. The subtypes were categorised as: a) Relapsing-remitting MS; b) primary progressive MS; c) secondary progressive MS. MS was diagnosed by a neurologist subspecialised in multiple sclerosis.
Treatment response
Patients were considered as responders to carbamazepine and oxcarbazepine if the patients reported that at least one of the drugs had relieved the pain to some extent. If patients stopped using the drugs due to side effects or allergic reactions before therapeutic dosages and possible pain relief were reached, the response was recorded as inconclusive.
MRI evaluation, protocol and definitions
All MRI scans were evaluated by an experienced neuroradiologist (ES) who was blinded to the symptomatic side. We used a 3.0 Tesla Phillips Achieva imager (Phillips Medical Systems, Best, the Netherlands) equipped with a 32-channel head coil with Multi Transmit parallel RF transmission. The MRI protocol included sagittal T2-weighted turbo-spin-echo sequences covering the whole brain, axial thin-section T2-weighted GRASE sequences encompassing the posterior fossa, 3D time of flight MR angiography (s3DI MC HR) and 3D high spatial resolution heavily T2-weighted sequences (3D balanced fast field echo (BFFE)). Multiplanar reconstructions were performed of the cisternal part of the NVC strongly parallel to the trigeminal nerve in the axial and sagittal plane and perpendicular to the trigeminal nerve in the coronal plane.
NVC was defined as a contact between a blood vessel and the trigeminal nerve without visible cerebrospinal fluid between the two structures (Figure 1(a)). If the neuroradiologist was uncertain whether there was a contact or not, it was considered as no NVC. An NVC was evaluated for a) degree of contact, b) localisation and c) type of blood vessel causing the contact. The degree of contact was graded on a three-point scale as simple contact, displacement or atrophy of the nerve. We distinguished between NVC with or without morphological changes. NVC with morphological changes was defined as an NVC with compression, displacement, distortion or atrophy of the trigeminal nerve. NVC was classified to be either at the root entry zone (REZ) or peripheral. The REZ was defined as the area from the site of entry of the trigeminal nerve into the pons to 7 mm peripherally along the nerve (16).

(a) T2 W 3.0 T MRI scan from a patient with right-sided primary trigeminal neuralgia. Balanced fast-field-echo sequence of the brainstem at the level of the pons in the axial plane showing a neurovascular contact (arrow) between an artery and the trigeminal nerve. (b) T2 W 3.0 T MRI scan from a patient with right-sided secondary trigeminal neuralgia attributed to multiple sclerosis. The scan shows brainstem at the level of the pons and a demyelinating plaque peripherally located at right side of pons.
Pontine demyelinating plaques were considered relevant when located along the course of the intrapontine trigeminal afferents in the ventrolateral part of the pons, including the pontine part of the trigeminal nuclei (Figure 1(b)). We did not include the trigeminal nuclei located at the medulla oblongata or the spinal medulla. To explore the possible differences between TN-MS patients with and without a visible demyelinating brainstem plaque on the symptomatic side, we compared the demographics and the clinical characteristics between the two groups.
Statistical analyses
Descriptive statistics were used to summarise continued and ranked data such as age and pain score by VNRS. Categorical variables are presented with frequency distributions (n, %) and with exact confidence limits. The chi-square test, Fisher’s test and McNemar’s test were used as appropriate to assess associations of categorical variables. Wilcoxon’s signed-rank test was used to compare non-parametric distributed data. We used multivariate conditional backward logistic regression to estimate the combined effect of any type of NVC and demyelinating plaque in predicting the symptomatic side. We included side of pain, sex, demyelinating plaque and NVC in this analysis. The sensitivity and specificity were calculated comparing the symptomatic (diseased) and asymptomatic side (healthy). Positive likelihood ratio was calculated as (sensitivity/[1-specificity]) and negative likelihood ratio as ([1-sensitivity]/specificity). If more than 5% of the data for an individual patient was missing, the patient was excluded from the analysis. Missing data from included patients were considered missing at random. p-values are reported as two-tailed and levels under 5% were considered significant. All analyses were carried out using SAS software 9.4 enterprise (SAS Institute Inc., Cary, NC, USA).
Results
There was a total of 841 patients with TN at the TN database at DHC (Figure 2). Eighty-seven percent (n = 736) of the patients were excluded due to primary TN. Of the remaining 105 secondary TN patients, 42 patients were excluded due to other secondary causes of TN than MS, missing data, no journal notes being available, and there being no informed consent to include data in a research paper (Figure 2). Finally, a total of 63 TN-MS patients were included to describe the clinical characteristics. Out of these, nine were excluded from MRI evaluation due to having bilateral TN (5) and there being no available MRI (4).

Flowchart of the inclusion process.
Demographics and pain localisation
The average age of TN onset was 49.3 years and the average age at MS diagnosis was 42.7 years (Table 1). The average current age was 56.5 years. The average interval between onset of MS and TN was 11.0 years. Eleven patients (19%) were diagnosed with TN prior to MS and five (8%) patients were diagnosed with TN at the same year as they were diagnosed with MS. Relapsing remitting MS was the most prevalent type of MS (55%) while primary progressive MS accounted for 25% and secondary progressive MS for 20%. There were more women (37 (59%)) than men (26 (41%)) (p = 0.166) in the cohort.
Demographics and diagnoses of 63 patients with trigeminal neuralgia secondary to multiple sclerosis.

CI: confidence interval; TN: trigeminal neuralgia; MS: multiple sclerosis.
aPatients with TN diagnosis before MS diagnosis (n = 11) were excluded from this analysis.
Note: We did not record when the patient had the first symptoms of multiple sclerosis.
More patients had left-sided pain (36 (57%)) compared to right-sided pain (22(35%)) but this was not statistically significant (p = 0.066). (Table 2). In five patients (8%), the pain was bilateral. Most of the patients had pain in the V3 branch (15 (24%)) and V2+V3 branches (16 (25%)). Pain in the V1 branch alone was rare (5 (8%)).
Pain localisation, quality and periodicity, family history, previous surgery and response to sodium channel blockers.
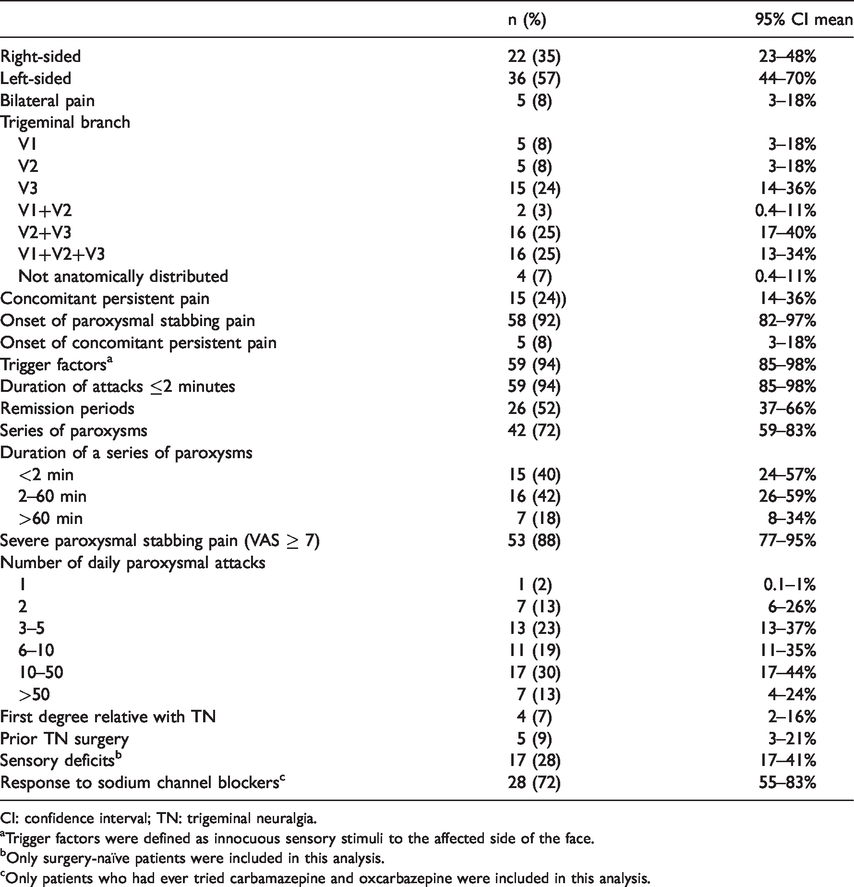
CI: confidence interval; TN: trigeminal neuralgia.
aTrigger factors were defined as innocuous sensory stimuli to the affected side of the face.
bOnly surgery-naïve patients were included in this analysis.
cOnly patients who had ever tried carbamazepine and oxcarbazepine were included in this analysis.
Fifty-eight (92%) patients experienced paroxysmal stabbing pain at TN onset (Table 2). In the rest, TN onset was with persistent concomitant pain and thereafter came the paroxysmal stabbing pain. Nineteen (33%) patients had concomitant persistent pain. Seventeen (30%) patients experienced 10–50 paroxysms daily and seven (13%) patients experienced > 50 paroxysms daily. In the patients with sensory deficits on the symptomatic side (17 (28%)), the most frequent sensory abnormalities were hypoesthesia (10 (16%)) and dysesthesia (2 (3%)). A small number of patients had undergone previous TN surgery (5 (9%)). The surgeries were glycerol rhizolysis (n = 4) and balloon compression (n = 1).
Neurovascular contacts and demyelinating plaques on the symptomatic and asymptomatic side
NVC of any type was found with the same prevalence on the symptomatic side and the asymptomatic side (29 (66%) vs. 30 (68%), p = 0.564) (Table 3). There was also no difference in the prevalence of NVC with morphological changes of the trigeminal nerve on the symptomatic (6 (14%)) vs. the asymptomatic side (4 (9%)) side (p = 0.157). The neuroanatomical findings of the three patients with bilateral TN and without any microvascular decompression prior to initial clinical examination are presented in Table 4.
Prevalence and characteristics of neurovascular contact and plaques from multiple sclerosis in 52 patients with trigeminal neuralgia attributed to multiple sclerosis.

CI: confidence interval; NVC: neurovascular contact; REZ: root entry zone; mc: morphological changes. Test for significant difference: Mc Nemar's test. a: The neuroradiologist was not able to evaluate NVC in 10 cases due to artefacts (n = 5) and poor image quality (n = 5). Only data on demyelinating plaques was included from these patients. b: One or more arterial contacts. c: One or more venous contacts. d: At least one artery and one venous contact. e: Defined as >7 mm from the site of entry of the trigeminal nerve into the pons. f: If there were two NVCs with equal degree of contact on the same side, the one in the REZ was favored. g: Morphological change is defined as compression, displacement and or atrophy of the trigeminal nerve. h: If there was more than one NVC, the one with the highest degree of contact was graded. i: Plaque was defined as neuroradiological signs of demyelination along the intrapontine trigeminal afferents or nuclei.
Characteristics of neurovascular contact and plaques from multiple sclerosis in three patients with bilateral trigeminal neuralgia attributed to multiple sclerosis.

NVC: Neurovascular contact; REZ: Root entry zone.
Note: The remaining two patients with bilateral TN were excluded due to MVD prior to the initial examination.
There were significantly more demyelinating plaques affecting the trigeminal intrapontine afferents or nuclei on the symptomatic side compared to the asymptomatic side (31 (58%) vs. 12 (22%), p < 0.001). The odds ratio (OR) for the association between a demyelinating plaque and the symptomatic side was 10.6 (95% CI 2.5–44.8, p = 0.002). After controlling for the possible confounders – sex, NVC and the interaction between NVC and demyelinating plaques – in a regression model, none of these variables affected the association between the symptomatic side and a demyelinating plaque. Considering demyelinating plaques as a diagnostic tool for patients with TN-MS, the sensitivity was 59% and specificity 77%.
Differences in the clinical characteristics in patients with and without a demyelinating plaque on the symptomatic side are illustrated in Table 4. There were significantly more men with visible plaques (18 (59%)) compared to women (13 (41%)), p = 0.021. There were no differences concerning the side of pain, other clinical characteristics, or the prevalence of NVC with morphological changes (Table 5).
Demographics, diagnoses and clinical characteristics of patients with TN secondary to MS with (n = 31) and without a demyelinating plaque affecting the trigeminal afferents or nuclei on the symptomatic side (n = 19).

CI: confidence interval; TN: trigeminal neuralgia; MS: multiple sclerosis; NVC: neurovascular contact.
Note: Test for significance: aChi-square test, bunpaired t-test, cFisher exact test, dWilcoxon signed rank test.
Discussion
This is the largest study to date evaluating the clinical characteristics and neuroanatomical abnormalities in patients with TN-MS. The main finding is that an NVC does not play a prominent role in the etiology of TN-MS. This is in direct opposition to the findings of a previous smaller study (9). We show that demyelinating plaques along the trigeminal nerve or nuclei are highly associated with the symptomatic side in patients with TN-MS. In line with previous studies, we show that TN-MS patients are younger at disease onset and more often have left-sided and bilateral pain compared to primary TN patients (16,17). Thus, although TN-MS patients in most aspects of the clinical characteristics are indistinguishable from primary TN patients, the underlying disease etiology is completely different. Our results carry major implications for understanding the etiology of pain and neurosurgical decision-making in patients with TN-MS. Based on our findings, it is unlikely that microvascular decompression is efficacious in TN-MS patients.
Etiology of trigeminal neuralgia secondary to multiple sclerosis
There was a strong association between a demyelinating brainstem plaque and the symptomatic side in TN-MS, indicating that a demyelinating brainstem plaque is the primary disease etiology in TN-MS. Conversely, we found no association between NVC and the symptomatic side in TN-MS as well as a very low prevalence of NVC with morphological changes. Our findings are comparable to the prevalence estimates of NVC in previous imaging studies on the asymptomatic side in primary TN, in persistent idiopathic facial pain, in healthy subjects and in cadaveric studies (5,6,18–20). This indicates that NVC is not involved in TN-MS etiology.
Interestingly, our findings directly contradict the results of a recent study that reported a dual concurrent mechanism with both a demyelinating brainstem plaque and NVC with morphological changes contributing to disease etiology in TN-MS (10). The MRI methodology was largely similar to the present study, but our study includes more patients. Finally, the patient cohort in the previous study was recruited from an MS cohort whereas our cohort was recruited from a tertiary headache center.
Notably, there was not a visible demyelinating plaque affecting the trigeminal afferents in all of our patients. The absence of a demyelinating plaque may be explained by the quality of the imager or to the fact that the MRI protocol did not include the trigeminal nuclei in the medulla oblongata and the medulla spinalis. Moreover, potential plaques located at or along the REZ are likely to be very small and difficult to detect using a 3.0 Tesla Imager. Finally, other yet unknown disease etiologies could contribute to the development of TN-MS.
To investigate the potential differences in patients with and without a demyelinating plaque, we compared the clinical characteristics and other neuroanatomical findings of the two groups. We found that the two groups are comparable, especially with respect to sidedness, age of onset, presence of trigger factors, response to sodium channel blockers and with respect to the lack of association between NVC and the symptomatic side. If the group of patients without a visible demyelinating plaque should rather be diagnosed with primary TN, it would be expected that they had a higher prevalence of right-sided pain, a higher age of onset, a better response to sodium channel blockers and the same significant association between a neurovascular contact with morphological changes at the symptomatic side as was demonstrated in primary TN (5,6,11,21).
In previous neurosurgical studies of TN-MS patients, the results are diverging as some report good efficacy from microvascular decompression (22,23), and others report poor efficacy (24,25). Importantly, there are no high quality prospective neurosurgical studies using independent evaluators of treatment outcome in patients with TN-MS. We found NVC with morphological changes in only a few patients. Our data do not exclude that microvascular decompression could be of benefit to this small subgroup.
Clinical characteristics in secondary trigeminal neuralgia attributed to multiple sclerosis
We show that patients with TN-MS more often have primary progressive MS compared to a comparable MS cohort, where the prevalence of primary progressive MS was reported to be 16.3% (26). In line with this, one study found that patients with primary progressive MS were more likely to have neuropathic pain in general and severe MS-related attacks (27). Compared to primary TN patients, TN-MS patients differed in the clinical characteristics with respect to a young age of onset of TN, and a higher prevalence of left-sided and bilateral pain (28).
The evidence on the efficacy of medical treatment in TN-MS is scarce (17), but it is general expert opinion that the response to medication is poorer compared to primary TN (8). Our findings support that the number of responders to carbamazepine and oxcarbazepine is somewhat lower compared to primary TN patients (11). In contrast to one previous study, we did not find that TN-MS patients have a lower prevalence and frequency of remission periods compared to primary TN (29). Also contrasting previous studies (10,28), we did not find a difference in the prevalence of sensory abnormalities when comparing to our own data collected using the same methodology in primary TN (30).
Our data confirm the widespread clinical notion that MRI is important early on in TN work-up to rule out secondary TN, since the clinical characteristics that are included in the TN diagnostic criteria; that is, strictly trigeminal pain that is short-lasting, stabbing, intense and triggered by innocuous stimuli, are highly prevalent in the current TN-MS cohort.
The diagnostic criteria should be changed
The diagnostic criteria for TN-MS were changed in 2018 with the 3rd version of the International Classification of Headache Disorders (ICHD-3) (14). The criteria state that not only should MS have been diagnosed, there should also be imaging proof of either an MS plaque at the trigeminal root entry zone or in the pons affecting the intrapontine primary afferents, or electrophysiological proof of impairment of the trigeminal pathways. We argue that if an MS patient also gets TN or vice versa, given the low incidence of both disorders and given their high co-prevalence, it is statistically highly unlikely that the two disorders should not be related; that is, that TN should not be a result of demyelination from MS. Moreover, previous studies showed few cases with normal responses from trigeminal reflex testing, thus testing does not significantly add to the diagnostic accuracy (10,31,32).
When time comes to revise ICHD-3, we suggest changing the diagnostic criteria of TN secondary to MS (13.1.1.2.1) (14). Firstly, we argue that an MS patient with TN should be diagnosed with TN-MS even without a visible demyelinating plaque on the symptomatic side on the MRI scan. Secondly, as the current study reflects, neurophysiological assessment like trigeminal reflex testing is not always used even in tertiary pain centres and it is safe to assume that it is rarely used in a typical neurological clinic, not to mention neurological practice in low-income countries. In our opinion, the diagnostic criteria must reflect the routine in the typical neurological clinic to be possible to use worldwide. We therefore suggest that trigeminal reflex testing can be performed as a supplementary test to differ primary TN from secondary TN in difficult cases.
Strengths and limitations of the study
Our study has the strength of a high volume of patients and prospective enrolment of consecutive broad-spectrum TN-MS patients recruited from a neurological setting, diagnosed by expert neurologists and using high quality MRI evaluated by an experienced neuroradiologist blinded to the symptomatic side.
Our MRI sequence did not include the medulla oblongata or the medulla spinalis and we did not systematically look for plaques in higher brain structures; for example, the insula (33).
Future perspectives
Imaging studies using 7.0 Tesla magnetic field strength and DTI sequences are warranted to find microstructural abnormalities of the trigeminal nerve in patients with TN-MS that may not be visible with conventional MRI.
High-quality, systematic and prospective neurosurgical studies using independent assessors of outcome are highly warranted in TN-MS as our findings indicate that microvascular decompression should generally not be performed in TN-MS patients. Prospective high-quality studies of the efficacy of balloon compression and glycerol rhizolysis are also much needed.
Conclusions
A demyelinating plaque along the intrapontine trigeminal afferents is the primary cause of TN-MS. NVC does not play a significant role in the etiology of TN secondary to MS. Only few TN-MS patients have NVC with morphological changes of the trigeminal nerve, and the prevalence is similar to previously reported prevalence estimates in asymptomatic nerves. Although primary TN and TN-MS largely display the very same clinical symptoms, the etiology is completely different and microvascular decompression should probably be reserved for classical TN patients. However, the final decision of treatment strategy should rely on individual assessment, as this study cannot give a final conclusion on the possible benefits of microvascular decompression in patients with TN-MS.
Clinical Implications
Neurovascular contact plays no role in trigeminal neuralgia secondary to multiple sclerosis Demyelinating plaque is the primary cause of trigeminal neuralgia secondary to multiple sclerosis Microvascular decompression should not be offered to patients with trigeminal neuralgia secondary to multiple sclerosis.
Footnotes
Acknowledgements
Professor Jes Olesen played a major part in initiating research into TN-MS at our center. We also thank Professor Jes Olesen for valuable contributions to the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
