Abstract
Background
There are currently no intraoperative neurophysiological tools to assess the effectiveness of trigeminal nerve decompression during microvascular decompression surgery for drug-resistant trigeminal neuralgia. In microvascular decompression surgery for hemifacial spasm, an abnormal electromyographic activation of facial muscles after stimulation of the offending vessel was identified and named ‘Z-L response’.
Methods
We adapted a neurophysiological protocol to elicit a Z-L response during microvascular decompression surgery for trigeminal neuralgia and applied it to a prospective series of 18 surgical patients.
Results
Patients had suffered from trigeminal neuralgia for a median 9-year timeframe, and median preoperative Barrow Neurological Institute pain score was 4.5. Through monopolar stimulation, using rising amplitudes starting from 0.1 mA, we confirmed intraoperatively the true culprit vessel before decompression. In 4/18 cases, multiple offending vessels were identified (22 conflicts overall). After decompression, a significant increase in activation threshold (p < 0.0001) confirmed the effectiveness of the maneuver; in 10 cases, Z-L response was abolished. Using this technique, we obtained excellent or good outcome (Barrow Neurological Institute 1–3) in all patients, with a significant reduction in postoperative Barrow Neurological Institute score as compared with preoperative one (median Barrow Neurological Institute 1; p = 0.0002).
Conclusion
we provide the first evidence on the applicability and clinical usefulness of Z-L response during microvascular decompression surgery for trigeminal neuralgia.
Introduction
Microvascular decompression (MVD) is considered the only available etiological treatment for drug-resistant trigeminal neuralgia (TN) (1). This procedure has been traditionally indicated in young patients suffering from paroxysmal pain (‘classic’ TN), particularly when a neurovascular conflict (NVC) is evident on preoperative magnetic resonance (MR) (2) and discarded in the elderly or in patients with multiple sclerosis (MS). However, regarding the best treatment for patients with TN associated with MS, there is a wide debate in current literature, due to the lack of a full understanding of the exact pathogenesis of trigeminal pain in MS. While the current standard treatment in these cases rely on percutaneous neuroablative procedures (2–4), especially in patients with a demyelinating plaque on the trigeminal root entry zone (REZ) in the pons (5), it has been shown that at least in some MS patients a neurovascular compression can contribute to TN onset, underlying a possible dual mechanism in the pathophysiology of trigeminal pain in MS (6). Of note, a recent systematic literature review suggested a favorable outcome of MS TN patients undergoing MVD (7), particularly when there is no evidence of a demyelinating plaque on trigeminal nerve pathway (7) or when a NVC is evident on preoperative MR (8).
In parallel, advances in the operative room instrumentation have helped make MVD surgery for TN and other NVCs a safer procedure, including for elderly patients (9).
Notwithstanding the wider spectrum of indications and the technological refinements, MVD for TN still suffers from the lack of neurophysiological tools to identify intraoperatively the culprit vessel and predict the effectiveness of the decompression. This is a different scenario than MVD for hemifacial spasm (HFS), in which the disappearance of the abnormal muscle response (AMR), also known as ‘lateral spread’ response, heralds the success of the surgical maneuver (10). However, AMR response has limitations, since it is not detectable in 7–8% of HFS patients (11) and in some cases it lacks predictive value for symptom resolution (12). In the search for novel intraoperative neurophysiological tools in HFS MVD surgery, an abnormal direct electromyographical activation of the facial muscles through stimulation of the offending vessel has been described and named ‘Z-L response’ (ZLR) (12). Literature data, though limited, show the effectiveness and reliability of this novel tool in detecting the offending vessel and predicting surgery success during MVD for HFS (11–17).
In the present work, we aimed at translating the experience with ZLR monitoring in the setting of MVD surgery for TN. We describe our adapted technique for intraoperative ZLR implementation and present our results in a prospective series of 18 TN patients.
Methods
Patient enrollment and collection of clinical data
We prospectively enrolled patients undergoing MVD for TN between June 2023 and January 2024 at our Institution. The study was conducted according to the Declaration of Helsinki principles and was approved by our Institutional Ethics Committee (study no. 4759); all patients signed an informed consent form. All surgeries were performed by the senior author (N.M.). Indications for MVD and surgical technique have been described elsewhere (18). Patients suffering from MS were excluded. Demographic, surgical and follow-up data were collected. NVC was graded as follows: A: arterial conflict, encompassing two subgrades: A1, arterial contact, A2, TN arterial distortion/indentation; B, venous conflict (19). Acute pain relief was defined as a pain-free condition at discharge. Pain severity was scored using the Barrow Neurological Institute scale (BNI) (20) and was assessed preoperatively and at the end of follow-up. A BNI score of 1–2 was defined as excellent outcome, a BNI score of 3 was defined as good outcome. The report was drafted according with the STrengthening the Reporting of OBservational studies in Epidemiology (STROBE) guidelines.
Standard intraoperative monitoring
Our technique for intraoperative monitoring in cerebellopontine angle surgery has been detailed elsewhere (21). Briefly, corticobulbar motor evoked potentials (CbMEP), free-running EMG (FEMG) and triggered EMG (TEMG) were performed. Bipolar subdermal needles were used for EMG recording from Masseter (V cranial nerve, c.n.), Orbicularis Oculi and Orbicularis Oris (VII c.n.) muscles. For CbMEP, corkscrew electrodes were placed on the scalp on Cz and C3 or C4 (international 10–20 System) depending on the side of surgery. Fz corkscrews were used as reference for TEMG. Double train stimulation with 40 ms intertrain interval was used for CbMEP stimulation in order to avoid false negative response. Single pulse stimulation (300–500 µs, 1 Hz, 0.1–2 mA) delivered by monopolar handheld probe was used for cranial nerve mapping and for ZLR monitoring.
ZLR monitoring
The protocol was adapted from the original technique described by Zheng et al. (12), in which a concentric stimulating electrode was placed on the wall of the putative culprit artery close to the conflict site, a square impulse (2 mA × 0.2 ms) was delivered and facial muscle response was registered at the mentalis, orbicularis oculi and orbicularis oris muscles. In our patients, after exposure of the putative offending vessel, direct stimulation was performed both close to the conflict site (‘proximal’ stimulation) and far from the conflict site (‘distal’ stimulation), while electromyographical response was registered in the masseter muscle. Firstly, proximal stimulation was performed using the monopolar handheld probe, starting from 0.1 mA amplitude and gradually increasing up to 0.5 mA. If no trigeminal EMG response was observed, we concluded that the stimulated vessel was not the actual offending one. If response was observed, to confirm the finding and rule out false positive responses from direct activation of the nerve due to local current spread, the distal stimulation was delivered. For distal response, amplitude was progressively increased up to 1 mA, since the threshold for distal stimulation was generally higher than from proximal stimulation. After the vessel dissection, both before and after Teflon positioning, stimulation was repeated, with an amplitude starting from the proximal activation threshold and gradually increasing up to a double value than pre-dissection distal threshold. We considered satisfactory any increase in stimulation threshold as compared with proximal pre-operative one.
Statistical analysis
Comparison of categorical variables was performed using the chi-squared statistic and adopting the Fisher exact test when appropriate. Comparison of paired measurements was performed using the Wilcoxon test, while comparison of continuous variables between groups was performed using the Mann-Whitney U test. Differences were considered statistically significant at p < 0.05. MedCalc ver 22.017 software (MedCalc Software Ltd, Ostend, Belgium) was used for analyses.
Results
Eighteen patients were enrolled in the study (Table 1). Baseline patient characteristics are shown in Table 2. Mean pain duration was nine years, while median preoperative BNI score was 4.5. Four patients had already been treated using percutaneous surgery or radiosurgery.
Details on patient characteristics.
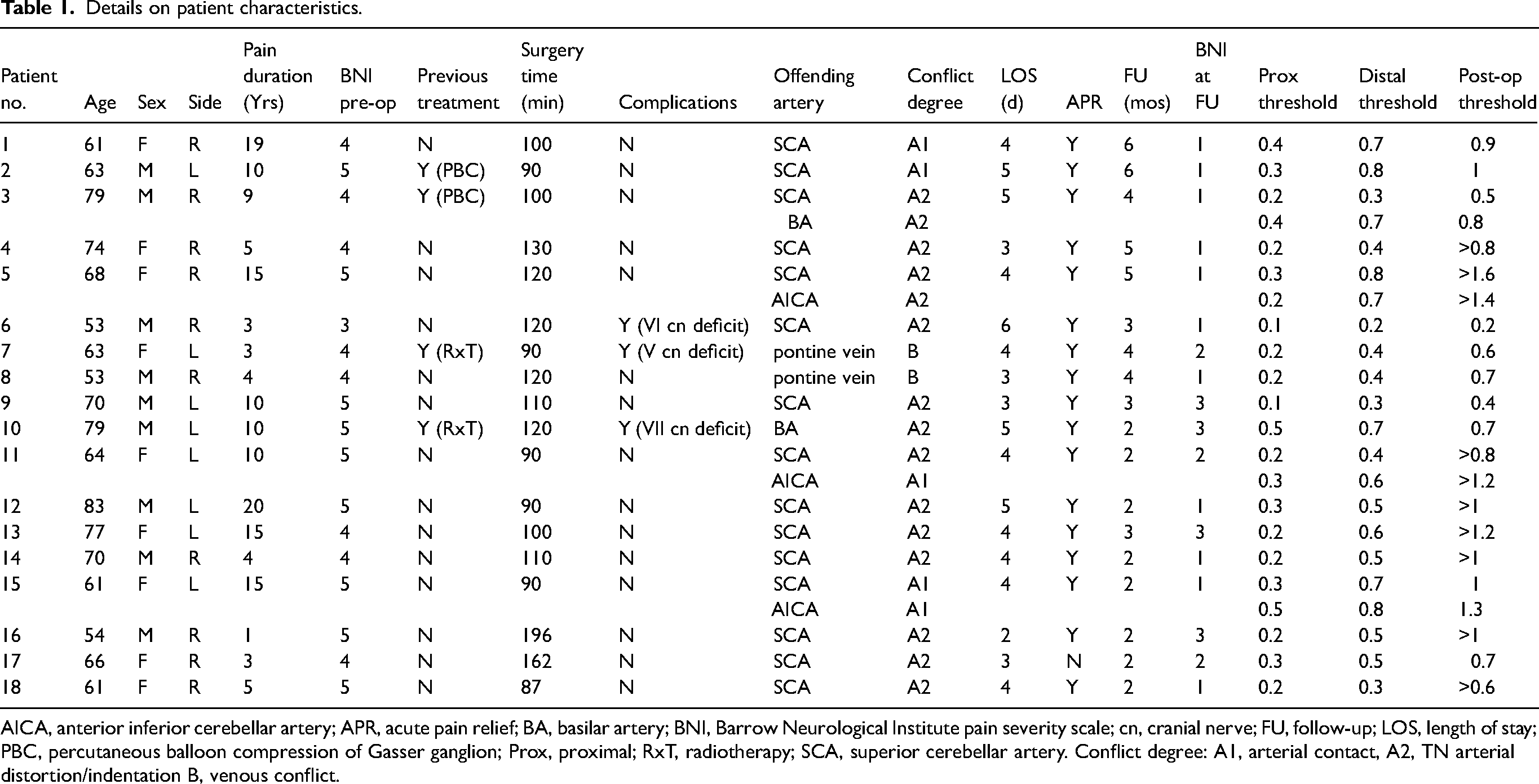
AICA, anterior inferior cerebellar artery; APR, acute pain relief; BA, basilar artery; BNI, Barrow Neurological Institute pain severity scale; cn, cranial nerve; FU, follow-up; LOS, length of stay; PBC, percutaneous balloon compression of Gasser ganglion; Prox, proximal; RxT, radiotherapy; SCA, superior cerebellar artery. Conflict degree: A1, arterial contact, A2, TN arterial distortion/indentation B, venous conflict.
Baseline patient characteristics.
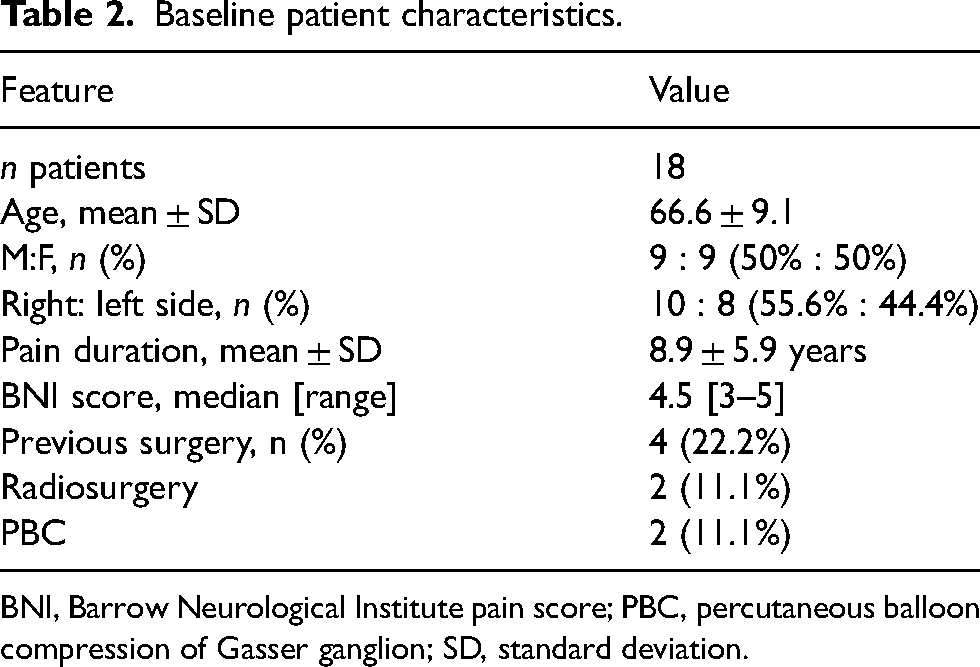
BNI, Barrow Neurological Institute pain score; PBC, percutaneous balloon compression of Gasser ganglion; SD, standard deviation.
At surgery, ZLR was used to confirm the anatomical identification of the offending vessel, that was the superior cerebellar artery in most cases (Table 3). In a non-negligible number of cases (4/18, 22.2%), multiple offending vessels were identified (22 offending vessels overall). Acute pain relief was achieved in 17/18 cases. After a three-month median follow-up, an excellent/good outcome (BNI score 1–3), was achieved in all cases (Table 3). In detail, an excellent outcome (BNI score 1–2) was achieved in 14/18 cases (77.8%), while a good outcome (BNI score 3) in 4/18 cases (22.2%). Notably, BNI at end of follow-up was significantly reduced as compared to pre-operative BNI (p = 0.0002, Wilcoxon Test). In three cases, a transient surgical complication was recorded: one case of severe facial hypoesthesia, one case of VI c.n. deficit and one case of VII c.n. deficit (House-Brackmann IV). In all three cases, clinical picture had resolved at the end of follow-up.
Surgical features and follow-up.
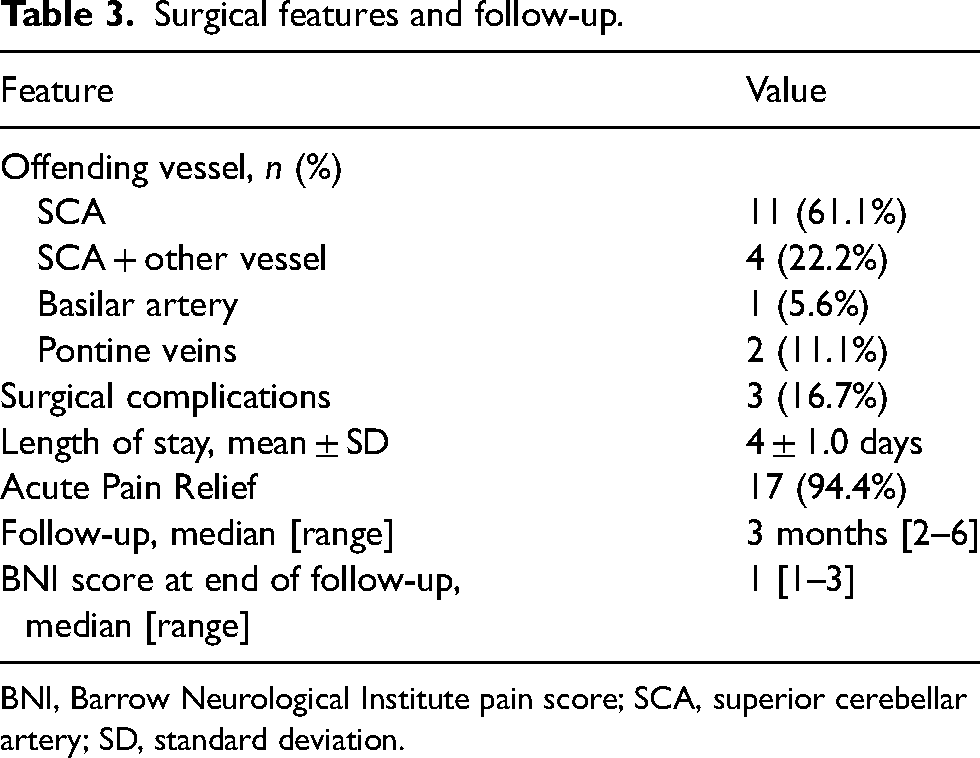
BNI, Barrow Neurological Institute pain score; SCA, superior cerebellar artery; SD, standard deviation.
Neurophysiological parameters for activation of the ZLR are shown in Tables 1 and 4. We defined as ‘proximal’ a stimulation administered in proximity to the conflict site (1–2 mm), and as ‘distal’ a stimulation administered far from the conflict site (>5 mm) (Figure 1). After conflict resolution, we observed a significant increase in the threshold for activation of the ZLR, as compared both to proximal and distal pre-decompression values (at least 234% and 65% increase, respectively; p < 0.0001 and p < 0.0001, respectively; Wilcoxon test). Notably, in 10 out of 22 conflicts the ZLR could not be recorded using a double stimulation amplitude than pre-decompression distal threshold. No correlations could be established between pre-decompression (proximal or distal) or post-decompression stimulation thresholds and post-operative outcome (BNI 1–2 vs 3) (Mann-Whitney test and Fisher exact test).
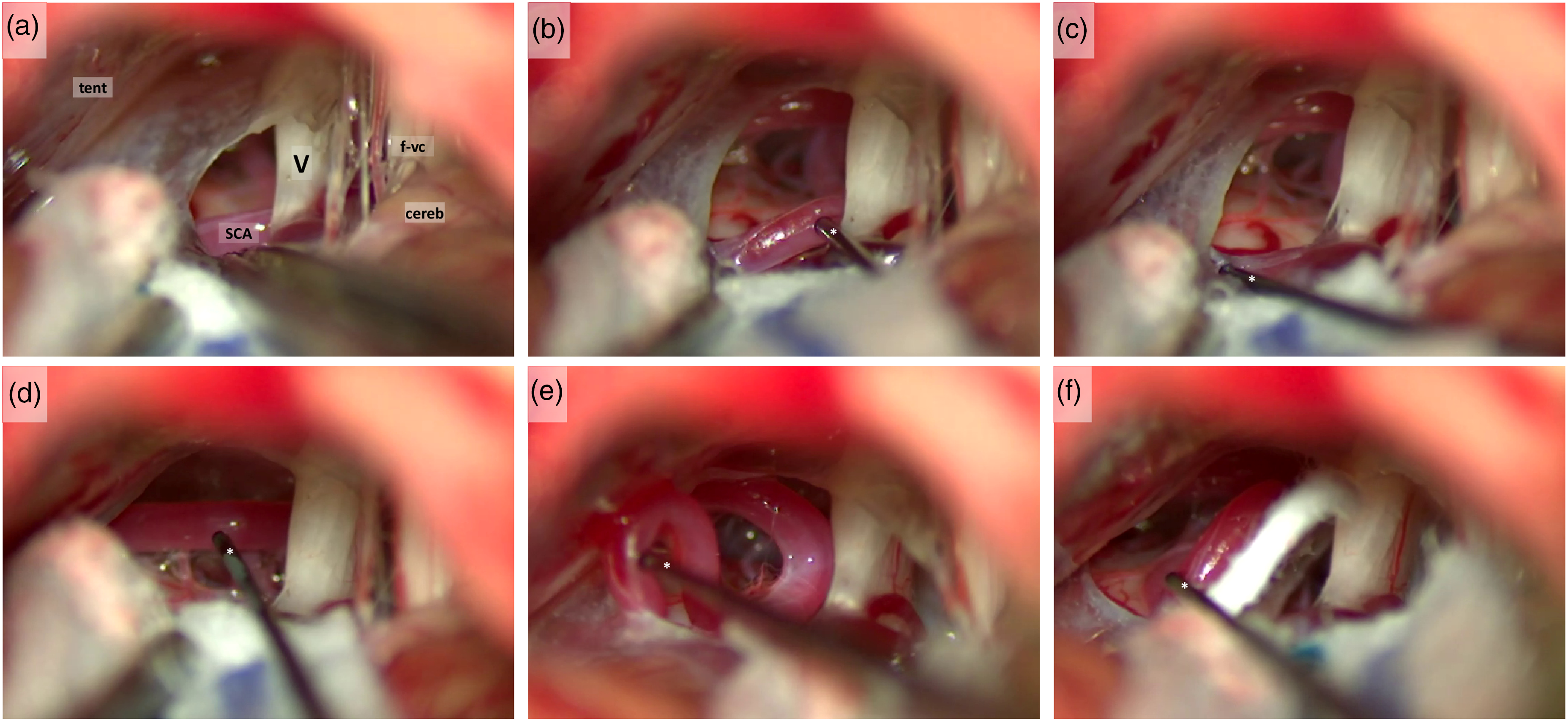
Intraoperative Z-L stimulation. (a) right cerebellopontine angle: a conflict between trigeminal nerve and superior cerebellar artery is present. cereb, cerebellum; f-vc, facial and vestibulocochlear nerves; SCA, superior cerebellar artery; tent, tentorium cerebelli; V, trigeminal nerve. (b), (c) stimulation of the SCA proximal (b) and distal (c) to the conflict site; (d)–(f), stimulation of the SCA after the resolution of the conflict, which failed at evoking a trigeminal response. *, stimulation probe.
Stimulation parameters for Z-L response.

Discussion
The so-named ‘Z-L response’ (ZLR) was described in 2012 by Drs. Zheng and Liat XinHua Hospital in Shanghai as an alternative method to identify the culprit vessel and monitor the effectiveness of decompression during MVD surgery for HFS (12). ZLR was evoked by electrically stimulating the wall of the compressing artery and registering facial EMG (12,13). ZLR is thus an orthodromic electromyographic potential elicited from current spread to facial nerve from electrically stimulated arterial wall that contacts the facial nerve within several millimeters. Therefore, the amplitude of ZLR differed in each point of electrical stimulation along the stimulated vessels, according to the distance between the stimulation site and the point of contact with the facial nerve (17). After decompression, a disappearance of the ZLR was described (12). This recording procedure was repeated for every offending vessel until the facial nerve was completely decompressed (16). Thus, in MVD surgery for HFS, ZLR has been proposed as a useful tool when AMR is absent before decompression, or persists after decompression of an identifiable offending artery, or in case of multiple suspected offending arteries around the REZ (13–15,17). Interestingly, a comparative study on nearly 1900 surgeries for HFS demonstrated that combining ZLR with AMR monitoring during MVD resulted in superior postoperative outcome than AMR monitoring only (11).
In the present work, we first adapted the original protocol for ZLR monitoring, shifting from facial to masticatory muscles and lowering the stimulation amplitude, in order to identify a stimulation threshold and not simply an on/off response. Using this technique, we could reliably identify the culprit vessel and obtain an excellent or good result (BNI 1–3) in all operated patients. As already demonstrated for HFS (17), ZLR was influenced by the distance between the stimulation and the conflict site: the threshold intensity to obtain a ZLR in the masticatory muscles was higher in a distal than in a proximal stimulation site. After decompression, the threshold for ZLR significantly increased as compared to pre-decompression values (Table 4).
From a surgical point of view, we think this is the most important finding of this paper. To date, the presence or not of an arterial conflict in cases of MVD for TN is based only on anatomical considerations that rely on the neurosurgeon's experience. So, the decision to perform the vessel dissection, to put or not put the Teflon pad (22), to do adjunctive maneuvers such as the partial sensory rhizotomy (23) or nerve combing (24) that significantly increase the risk of V c.n. dysfunction, are based solely on the anatomical judgment of neurosurgeon. The technique we are describing here provides, in our opinion, clear neurophysiological feedback about the vessel causing the NVC. Further, this neurophysiological feedback is important because it provides information on when the decompression of the fifth nerve can be considered satisfactory. In fact, in all cases the final threshold (measured when the anatomical conflict was considered resolved from the senior author performing the procedures) was at least doubled compared to the proximal threshold at the beginning of the operation (except the case of basilar artery compression; Case #10, see Table 1) and increased compared to the initial distal threshold (only in the Case #6 and in Case #10 the final threshold was equal to the distal threshold at the beginning, see Table 1). In this initial experience, we considered this neurophysiological feedback a valid confirmation of the anatomical intraoperative impressions on the NVC resolution.
The fact that ZLR could be evoked also after conflict resolution in the majority of cases, even though at increased amplitudes, can be explained by the persistence of a spatial proximity between the offending artery and the V c.n. even after decompression. In the present series we were not able to identify specific cut-offs for ZLR threshold increases that guarantee pain relief. This fact is due to the short follow-up of this series but we think that the elongation of follow-up will give more information about the ‘minimum’ threshold increases capable of affecting the long-term pain outcome.
Another interesting finding we were able to observe is that the ZLR could also be evoked from vein stimulation and that not all the stimulated veins provoked a response from the fifth nerve. This is very important because there is no agreement about the real existence of vein conflict (25,26) and even less about veins management if an arterial conflict is not evident (27,28). We had two cases in which we could not identify an arterial conflict, but the ZLR was evoked from a pontine vein that was dissected from the nerve, obtaining an increase of the final threshold in both cases (Cases #7 and #8, see Table 1). The vein conflict was resolved by putting a Teflon pad with no coagulation of these veins. Obviously more cases are needed to understand if this technique can be useful in venous conflicts too.
Limitations of the present work are the low numerosity and the short-term follow-up. These limitations probably played a role in hindering the identification of a cut-off value for the increase in the ZLR threshold able to predict outcome. Further, another limitation is that this is an initial non-comparative study, because our main goal here was to describe the technique. Thus, at this stage we cannot draw definitive conclusions on the actual clinical impact of ZLR monitoring. On the other hand, the uniformity of the surgical and neurophysiological protocols which have been consistently adopted, together with the prospective patient enrollment, are the strong points of the present study.
Conclusions
ZLR can be considered as a promising and simple neurophysiological tool to identify the culprit vessel during MVD for TN and to assess the effectiveness of decompression procedure. Further studies and longer follow-up are warranted to validate this technique and to establish reliable thresholds for satisfactory nerve decompression.
During microvascular decompression surgery for trigeminal neuralgia, an abnormal electromyographic activation of masseter after stimulation of the offending artery can be recorded (Z-L response). Z-L response allows to confirm that the identified vessel is the true culprit vessel. Threshold increase or Z-L response disappearance heralds a good outcome of surgery.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Italian Ministry of Health (Ricerca Corrente 2024).
