Abstract
Aim
This study aimed to investigate the extent of autonomic nervous system dysfunction in patients with chronic migraine using heart rate variability analysis. In addition, we explored the potential association between heart rate variability and treatment outcomes in patients receiving preventive treatment.
Methods
In this cross-sectional and prospective study, we compared heart rate variability profiles in 81 preventive-naïve chronic migraine patients and 58 healthy controls. In addition, treatment responses of patients, who received a 12-week treatment with flunarizine, were assessed in relation to baseline heart rate variability.
Results
We observed that chronic migraine patients had a reduced heart rate variability, signifying autonomic dysfunction in comparison to healthy controls. Furthermore, patients presenting normal heart rate variability, characterized by a standard deviation exceeding 30 milliseconds in normal-to-normal RR intervals, experienced a superior response to flunarizine treatment. This improvement was exemplified by a significantly larger reduction in monthly headache days for patients with higher heart rate variability compared to those with lower heart rate variability: −9.7 (5.9) vs. −6.2 (6.0) days (p = .026).
Conclusions
Autonomic dysfunction occurs in chronic migraine as evaluated by heart rate variability. A preserved function is associated with a better treatment outcome to flunarizine.
Introduction
Patients with migraine experience headaches and other constitutional symptoms such as nausea, vomiting, fatigue, loss of appetite, and palpitation. Due to the presence of these associated autonomic symptoms during or even between migraine attacks, dysfunction of the autonomic nervous system has long been regarded as a contributing factor to migraine pathogenesis. However, while most studies (1–12) have focused on episodic migraine, research on autonomic nervous system dysfunction in patients with chronic migraine, characterized by headaches on more than 15 days per month, of which at least eight fulfill the criteria for migraine, for ≥3 months (13), remains limited. Given that chronic migraine affects around 2% of the global population (14), it is crucial to conduct further research to determine the potential involvement of autonomic nervous system in its pathogenesis.
Heart rate variability (HRV) serves as a well-established, non-invasive measure of autonomic nervous system activity. By examining the fluctuations between successive R-peaks in the electrocardiographic (ECG) signal, HRV can be analyzed through time-domain, frequency-domain, and nonlinear approaches, capturing both sympathetic and parasympathetic components of autonomic nervous system function (15). However, recent studies (16,17) have suggested that HRV derived from resting ECG recordings mainly reflects vagally-mediated parasympathetic function. For instance, an animal study has shown that the power of low frequency (LF) band, traditionally regarded as a parameter for sympathetic activity, is not altered by a sympathetic blocking agent (i.e., beta-blocker) but rather is markedly (>90%) reduced by a parasympathetic blocking agent (16). This indicates that LF might embody a combination of both sympathetic and parasympathetic functions, with the latter predominant at rest. Thus, overall, the HRVs derived from resting ECG recordings are more representative of parasympathetic/cardiovagal function.
Numerous studies have investigated HRV abnormalities in various painful conditions and disorders, including fibromyalgia (18), neuropathic pain (19), chronic pain (20), and headache (21), as well as episodic migraine (1–12). As summarized in Table 1, these studies have consistently demonstrated reduced HRV in patients with episodic migraine compared to healthy controls, indicating that HRV may hold promise as an informative measure of autonomic nervous system dysfunction in patients with chronic migraine.
HRV of controls and patients with episodic migraine.
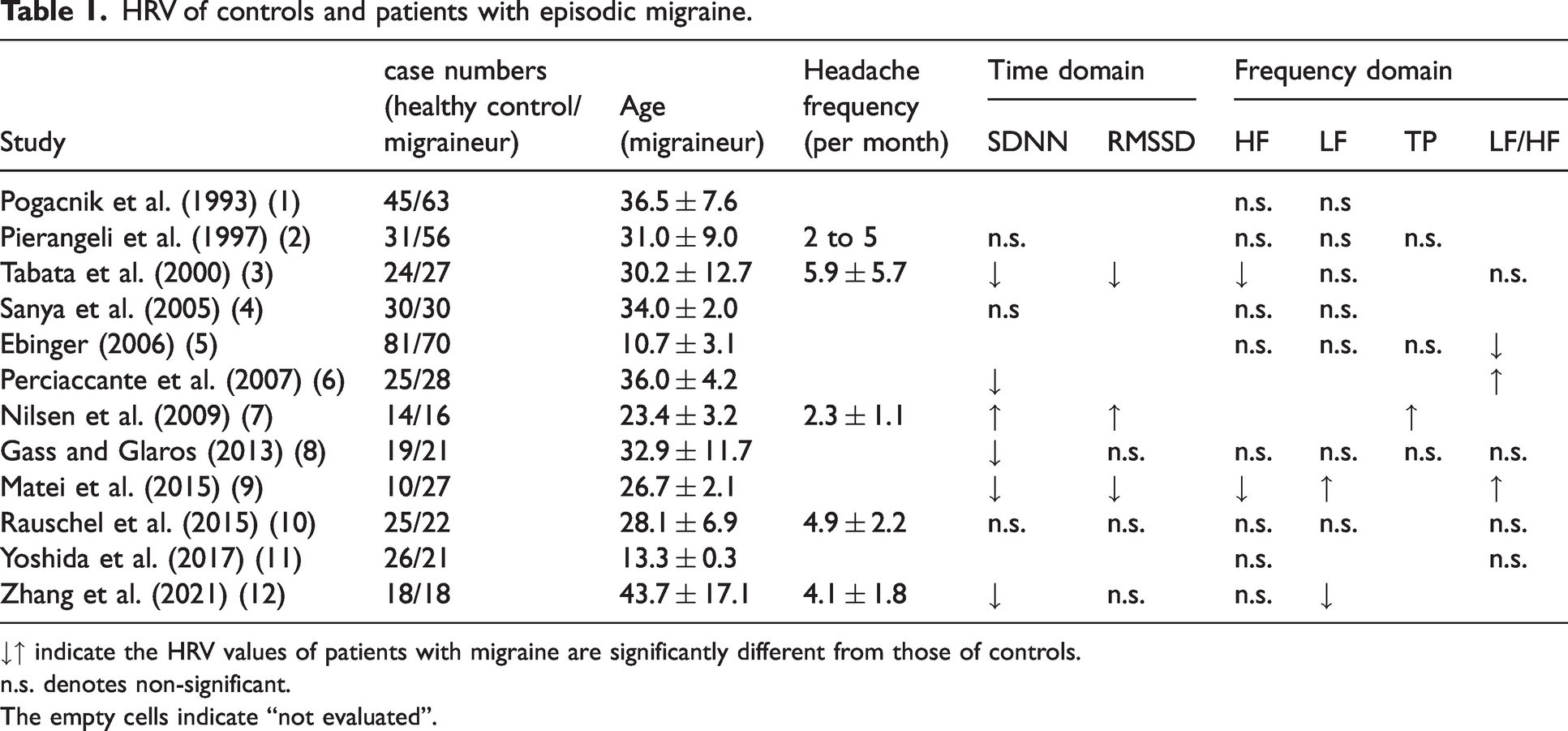
↓↑ indicate the HRV values of patients with migraine are significantly different from those of controls.
n.s. denotes non-significant.
The empty cells indicate “not evaluated”.
HRV has also emerged as a potential biomarker for painful disorders and as a surrogate marker or an outcome predictor of clinical improvement. Two studies (22,23) demonstrated that, following yoga exercise, the power and ratio of the high-frequency band of HRV increased, correlating with clinical improvement. On the other hand, several studies have demonstrated that higher parasympathetic function at baseline is associated with a more favorable outcome in different disease entities (24–26). For example, a preserved parasympathetic function predicted a better prognosis in pain symptoms in patients with pain-predominant multi-somatoform disease following psychotherapy (26). While the exact mechanism linking preserved parasympathetic activity to a better prognosis remains not fully understood, it might be attributed to two crucial functions of the cardiovagal/parasympathetic system. Firstly, due to its unique anatomical localization, the vagus nerve can modulate pain sensitivity through its central connection to the somato- and visceral-sensation relay center in the brainstem (27). Secondly, the vagus nerve could exert an anti-inflammatory effect via the cholinergic anti-inflammatory pathway (28,29). Both mechanisms play a broad role in various disease entities, including pain disorders.
This cross-sectional and prospective study was built upon two hypotheses. Firstly, previous studies (20,21) have shown that patients with episodic migraine and various chronic pain disorders often experience autonomic nervous system dysfunction, particularly in the parasympathetic branch. Drawing on these findings, this study hypothesized that a similar phenomenon may also occur in patients with chronic migraine. We aimed to investigate the presence of autonomic nervous system dysfunction in patients with chronic migraine by examining their resting HRV, thereby addressing this knowledge gap. Secondly, this study further hypothesized that a preserved baseline (pre-treatment) parasympathetic function might be associated with better clinical outcomes after a three-month preventive treatment. Additionally, the variation in HRV between post-treatment and pre-treatment phases might correlate with clinical improvement in these patients. Although such hypotheses align with findings from studies on numerous other disorders (22–26), these connections have yet to be confirmed explicitly for chronic migraine. Therefore, we sought to evaluate whether baseline HRV dysfunction and HRV shift during treatment could act as an indicator for clinical outcomes or serve as a surrogate marker for clinical improvement to preventive treatment, respectively.
Materials and methods
Ethics statement and study design
The study protocol (Figure 1a) was approved by the Institutional Review Board (IRB) of Taipei Veterans General Hospital, Taiwan (VGHTPE-IRB: 2015-10-001BC; ClinicalTrials.gov Identifier: NCT02747940). All participants provided their informed consent for inclusion before participating in the study.

Experimental design and data collection. (a) Study protocol. In this prospective trial, the baseline, 4 weeks (M0) before the treatment, was followed by a 12-week (M1-M3) treatment. A baseline (pre-treatment) resting-state ECG examination was conducted during the first visit. The treatment started after the first follow-up visit. Another (post-treatment) ECG was conducted after the 12-week treatment. Throughout the experiment, each patient was instructed to maintain a headache diary and rate their pain from no, mild, moderate, to severe pain. (b) ECG examination. The ECG signal was recorded for 10 minutes through the NuAmps system in the bipolar lead II configuration (negative and positive, RA-LL). The RR, the interval between successive R peaks in an ECG wave, was used for heart rate variability analysis and (c) Flow chart of recruitment for controls and patients with chronic migraine.
The study consisted of a prospective baseline period of four weeks (Week −4 to Week 0) to record the baseline headache profile, followed by a 12-week treatment period after enrollment (Weeks 0–12, as shown in Figure 1a). We targeted those patients with a disease duration exceeding three months. Eligible patients who met the ICHD-3β criteria for chronic migraine (30) were asked to maintain headache diaries from the prospective baseline period. This practice adhered to the current guidelines set by the International Headache Society for recruiting chronic migraine patients (31,32).
Subjects
To be enrolled in this study, subjects aged 20–60 had to meet the ICHD-3β criteria for chronic migraine (30) at the first visit and fulfill the following additional criteria. They had to experience ≥14 headache days lasting at least four hours during the baseline period, among which at least eight fulfilled migraine criteria, defined as moderate to severe headache with one or more of the following features: unilateral pain or pain that is worse on one side of the head, pulsatile nature, photophobia and phonophobia, nausea or vomiting, or pain that worsens with routine physical activity or is responsive to acute migraine treatment (33). To avoid the interference or lasting effects of other preventive medications, we recruited prophylaxis-naïve patients who had not received any preventive medications (such as beta-blockers, antidepressants, antiepileptic drugs, or calcium channel blockers), botulinum toxin injection, or acupuncture therapy before participating in the study.
The study excluded patients who had any of the following conditions: i) headache type other than chronic migraine, ii) migraine onset after the age of 50 years, iii) chronic migraine onset after the age of 60 years, iv) pregnancy or nursing status, v) history of the hepatic, renal disorder or other severe systemic diseases, vi) psychiatric diagnosis of major psychiatric disorders or severe depression (BDI score ≥30 at Visit 1), and vii) use of medications that may interfere with autonomic function (e.g., drugs with antiarrhythmic properties).
Age- and gender-matched healthy controls were recruited from a nearby neighborhood through an advertisement approved by the IRB. The healthy controls were excluded if they had the following conditions: i) headache disorder other than infrequent tension-type headache (less than once per month), ii) pregnancy or nursing status, iii) systemic diseases or major psychiatric disorders, or iv) use of medications that may interfere with autonomic functions.
Intervention and primary outcome measures
During the first visit, potential participants completed a structured headache questionnaire to collect information on their headache profile and assess disease severity (using the Migraine Disability Assessment [MIDAS]), psychological disturbance (using the Beck Depression Inventory [BDI] and Hospital Anxiety and Depression Scale [HADS-A & HADS-D]), and stress level (using the Perceived Stress Scale [PSS]).
Patients who met the inclusion criteria were started on flunarizine as a preventive treatment for 12 weeks. The initial dosage of flunarizine was 5 mg/day, which could be titrated up to 10 mg/day depending on the patient’s tolerance. The choice of flunarizine as a preventive medication was based on our previous study (33), which demonstrated comparable efficacy with fewer adverse effects than topiramate in patients with chronic migraine.
Throughout the study period, patients were instructed to maintain a headache diary, which was used to calculate the number of monthly headache days (MHD) as the primary outcome measure. Both the mean absolute change (
ECG recording and heart rate variability analysis
Two ECG examinations were performed for patients: one during the baseline period and the other after 12 weeks of treatment (as shown in Figure 1a). On the other hand, each healthy control only underwent a single ECG examination.
As shown in Figure 1b, a single-lead electrode pair with one electrode placed under the right clavicle at the mid-clavicular line and the other placed on the left upper abdomen was used to record resting-state ECG through the NuAmps system (Compumedics Limited, Australia) in the bipolar lead II configuration (negative and positive, RA-LL). The sampling frequency was 500 Hz. Subjects were informed not to drink coffee within 24 hours before each ECG recording.
Figure 2 presents the signal processing and HRV analysis flowchart used in this study. Initially, the Pan-Tompkins algorithm (34) was used to detect the R-wave peak of the QRS complexes in the ECG. The time elapsed between two successive R-waves, known as RR intervals, was then calculated for HRV analysis. Next, each RR interval time series was trimmed to a 5-minute epoch using Kubios HRV Premium (version 3.3.0), and an automatic artifact correction algorithm was employed to detect and correct ectopic, missed, and extra beats. Finally, the HRV profiles were evaluated through time-domain, frequency-domain, and nonlinear analyses. In total, eight HRV parameters were derived. Specifically, the time-domain metric included the standard deviation of normal-to-normal RR interval (SDNN, in millisecond [ms]) and the root mean square of successive RR interval difference (RMSSD, in ms). The frequency-domain metric was decomposed using fast Fourier transform, leading to absolute powers of the low-frequency band (LF = 0.04–0.14 Hz, in ms2), high-frequency band (HF = 0.14–0.4 Hz, in ms2), LF/HF ratio, and total power (TP < 0.4 Hz, in ms2). The nonlinear HRV metric derived by analyzing the geometry of consecutive RR intervals on the Poincaré plot included SD1 (in ms) and SD2 (in ms).

Flow diagram of signal processing and data analysis.
To investigate whether a preserved parasympathetic function might be linked with better clinical outcomes in patients, this study categorized patients based on their SDNN values. SDNN was selected due to its robust representation of overall parasympathetic (cardiovagal) function at rest, simplicity in the calculation, and widespread use in routine clinical settings. Following previous studies (35,36), a cutoff value of 30 ms was used to dichotomize patients into a “low-HRV” group (baseline SDNN < 30 ms) and a “normal-HRV” group (baseline SDNN ≥ 30 ms).
Sample size estimation
Studies (3,8,9,12) have shown that patients with episodic migraine typically exhibit HRV value at approximately 80% of those of healthy controls (meaning roughly a 20% difference), with a standard deviation of about 40%. Given these findings, we expected a moderate difference in HRVs between our subject groups, with an effect size estimated to be 0.5 (i.e., 20/40). We also planned for an allocation ratio of 2:1, aiming to recruit two patients (one each with normal and low HRV) for each healthy control, leading to equal group sizes. Utilizing G*Power (37), we applied a two-tailed test with a Type I error level of 0.05 and a Type II error level of 0.2 to determine the necessary sample size. This yielded a recommended total of 96 patients and 48 healthy controls. Ultimately, our collected sample sizes were 81 patients and 58 healthy controls.
Statistical analysis
To test the hypothesis concerning autonomic nervous system dysfunction in patients with chronic migraine, we commenced statistical analysis by comparing demographic and HRV profiles between patients and healthy controls. This was achieved by employing either a Chi-square test or an independent t-test as dictated by the nature of the data. Welch’s test was applied if an unequal variance was detected through the Brown-Forsythe test. Note that the HRV data for patients in this analysis were from the baseline period.
For the patient group, we further analyzed their HRV profiles in relation to a set of clinical parameters. These parameters encompassed baseline monthly headache days, MIDAS, BDI, HADS-A, HADS-D, and PSS. This analysis was conducted using Pearson’s correlation coefficient method.
To evaluate the hypothesis that maintained parasympathetic function could be linked to improved clinical outcomes, we contrasted the data between patients with normal and low HRV levels. Firstly, differences between patients with normal- and low-HRV levels in demographic characteristics, questionnaire scores, and baseline HRV profiles were analyzed using either a Chi-square test or an independent t-test as appropriate. analysis of covariance (ANCOVA) was utilized to accommodate potential covariates which might impact dependent variables. Secondly, a mixed factorial analysis of variance (ANOVA) was conducted to investigate the interaction effect between the HRV group (categorized as normal and low) and time point (including baseline, one month, two months, and three months of treatment) on the metric of monthly headache days. In this design, the HRV group functioned as the between-subjects factor, while the time point was the within-subjects factor. A mixed factorial ANCOVA was employed if there was a need to incorporate any potential covariates. The Bonferroni method was utilized to conduct post hoc tests.
Furthermore, the study calculated the relative risks to compare whether the risk of not attaining a 50% reduction in monthly headache days were equivalent between the two patient groups.
Lastly, this study assessed the changes in HRV profiles before and after the three-month treatment using paired t-tests. The pre-treatment and post-treatment HRV profiles were compared with the HRV profiles of healthy controls using independent t-tests to assess whether they were significantly different. Furthermore, a correlation analysis was undertaken to explore the relationship between HRV changes and clinical improvements.
All statistical analyses were performed utilizing JASP 0.17.1 (University of Amsterdam, Netherlands) and MedCalc 22.003 (Ostend, Belgium).
Results
Demographics
A total of 81 patients with chronic migraine (92.6% female, mean age: 38.1 years, SD = 11.4) and 58 age- and gender-matched healthy controls (84.5% female, mean age: 40.6 years, SD = 9.2) completed the study after excluding individuals with ineligible conditions (Figure 1c). The demographic and clinical characteristics of the participants are presented in Table 2, which indicates significant differences in the scores derived from BDI, HADS-A, HADS-D, and PSS when contrasting the group of healthy controls and the patients (p < .001).
Demographic and clinical characteristics of healthy controls and patients with chronic migraine.

The Welch’s test was performed due to unequal variance (by Brown-Forsythe test).
Comparison of 5-min HRV between healthy controls and patients at baseline
There were statistically significant differences between patients and healthy controls in most HRV parameters at baseline (independent t-tests, p < .001), except for LF and LF/HF ratio (Figure 3a, p > .05). Specifically, the SDNN (Figure 3b), RMSSD, TP, HF (Figure 3c), SD1, and SD2 (Figure 3d) of patients were significantly lower compared to those of healthy controls. The analysis of correlations between these HRV parameters and clinical characteristics did not reveal any specific associations (Online Supplemental Table 1). A summary of the baseline HRV data is provided in Table 2.

Comparisons of HRV between controls and patients with chronic migraine. (a) The p-values of independent t-tests comparing HRV. The p-value located in the outer circle is smaller than that located in the inner circle. The p-value located in the innermost circle indicates a nonsignificant difference in HRV between the two groups. Three HRV parameters, namely (b) SDNN (ms), (c) HF (×103 ms2), and (d) SD2 (ms), of patients with chronic migraine were compared with those of healthy controls, where three asterisks denote significant differences at p < .001 of independent t-tests. Dark and light boxes indicate the ranges of plus or minus one standard deviation and 95% confidence interval, respectively.
Given the significant differences observed in the scores of the four questionnaires between healthy controls and patients (Table 2), we employed ANCOVA to reassess the discrepancies in the eight HRV metrics between the two groups (Online Supplemental Table 2). This analysis was adjusted to consider potential influences from depression, anxiety, and stress factors. The results echoed our initial observations, demonstrating that group differences persistently manifested a substantial effect on most HRV parameters (ANCOVA with BDI, HADS-A, HADS-D, and PSS as covariates, p < .05), with the sole exception of LF/HF.
Difference in treatment efficacy between patients with normal-HRV and low-HRV
Figure 4a displays that 81 patients were separated into two groups based on the baseline SDNN value of 30 ms. The low-HRV group consisted of 50 patients (48 female) with an average age of 41.0 (SD = 10.4) years, while the normal-HRV group consisted of 31 patients (27 female) with an average age of 33.4 (SD = 11.6) years. As summarized in Table 3, age exhibited a statistically significant difference between the two groups (independent t-test, p=.003), while gender (Chi-squared test, p = .137) and questionnaire scores (p > .05) did not. In terms of HRV, except for LF/HF (p = .475), most parameters of the normal-HRV group were significantly higher than those of the low-HRV group (ANCOVA with age as the covariate or Kruskal–Wallis test, p < .001).

Chronic migraine subgroups and treatment efficacy. (a) Patients were assigned to two disjoint groups, thresholded by the SDNN value of 30 ms (b) The changes in
Demographic and baseline HRV of patients with normal- and low-HRV.

Owing to the significant difference in Age between the two groups, comparisons of the baseline HRV profiles were conducted using ANCOVA, with Age serving as a covariate. However, the analysis revealed that Age did not have a significant effect on the HRV profiles (p > 0.05), with the exception of SD2 (p = .027).
Kruskal–Wallis test was performed as the assumption of equal variances was violated (by Levene’s Test).
The normal- and low-HRV groups exhibited different responses to treatment in terms of pain relief, despite having similar baseline monthly headache days (M = 21.8, SD = 5.4 vs. M = 21.5, SD = 6.4; ANCOVA with age as the covariate, p = .442) (Figure 4b). The Mixed Factorial ANCOVA results in Table 4 demonstrate significant interaction effects between the HRV group and time point on both metrics of monthly headache day reduction: the mean absolute reduction (
Reductions in monthly headache days in patients with normal- and low-HRV.
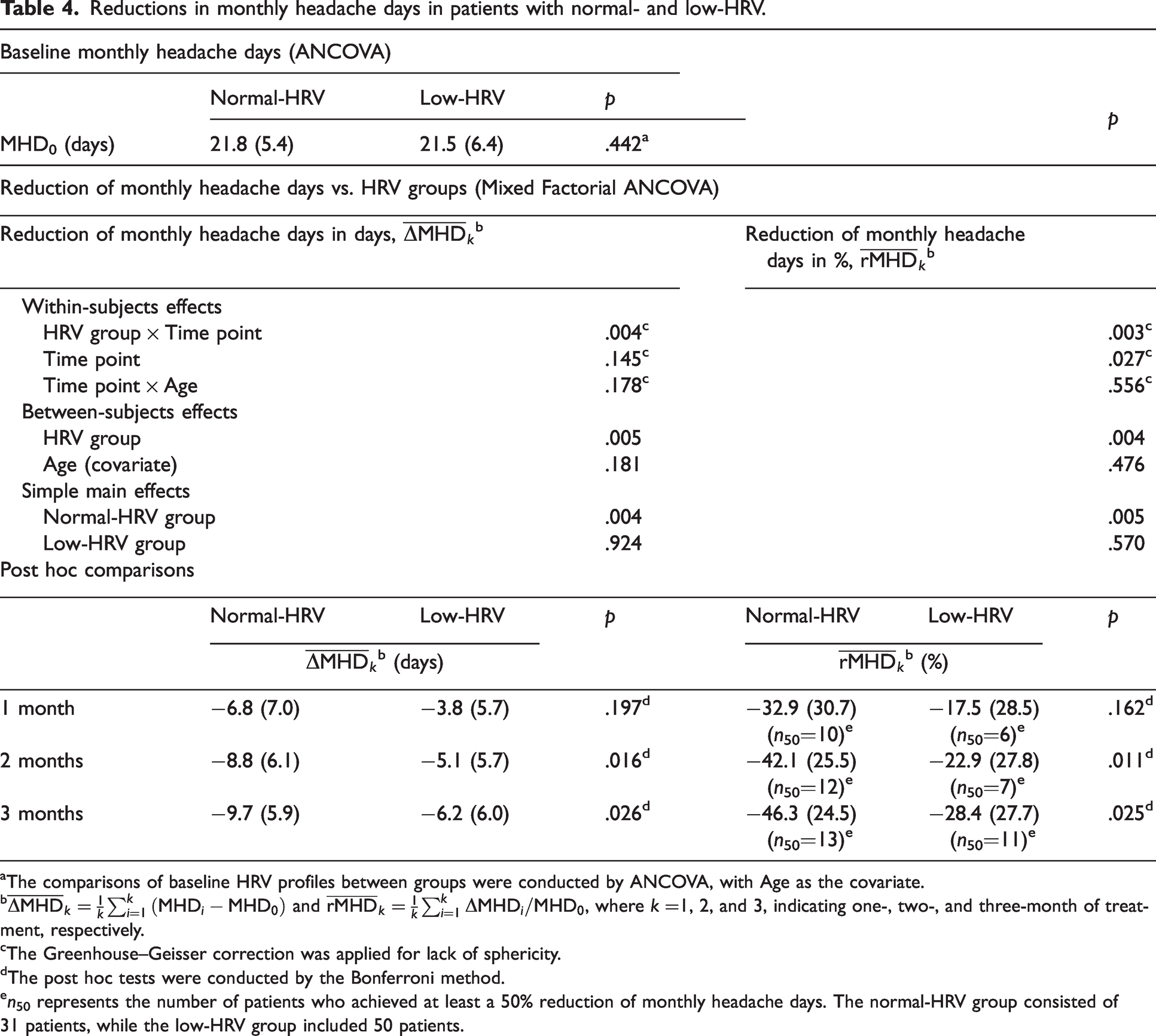
The comparisons of baseline HRV profiles between groups were conducted by ANCOVA, with Age as the covariate.
The Greenhouse–Geisser correction was applied for lack of sphericity.
The post hoc tests were conducted by the Bonferroni method.
n50 represents the number of patients who achieved at least a 50% reduction of monthly headache days. The normal-HRV group consisted of 31 patients, while the low-HRV group included 50 patients.
Further post hoc analysis showed that, following two and three months of treatment, the normal-HRV group experienced a greater reduction in monthly headache days compared to the low-HRV group (Table 4). Specifically, the mean absolute reduction of monthly headache days of two groups after one month of treatment was −6.8 days (SD = 7.0) for the normal-HRV group versus −3.8 days (SD = 5.7) for the low-HRV group (p = .197). This trend persisted after two months of treatment, with mean absolute reductions of −8.8 days (SD = 6.1) and −5.1 days (SD = 5.7) for the normal-HRV and low-HRV groups, respectively (p = .016). After three months of treatment, the normal-HRV group saw a mean absolute reduction of −9.7 days (SD = 5.9), compared to −6.2 days (SD = 6.0) days for the low-HRV group (p = .026). Comparable trends were also seen in the mean percentage reduction of monthly headache days (Table 4).
The number of patients achieving a minimum of 50% reduction in monthly headache days, referenced as n50 in Table 4, varied between the two groups. Specifically, 10 of 31 patients in the normal-HRV group achieved this reduction after one month of treatment, compared to six of 50 patients in the low-HRV group. By the second month, this number increased to 12 out of 31 patients for the normal-HRV group and seven out of 50 for the low-HRV group. After three months of treatment, 13 of the 31 patients in the normal-HRV group and 11 of the 50 patients in the low-HRV group achieved this milestone. As for the relative risk, the low-HRV group exhibited an increased risk of not attaining a 50% reduction of monthly headache days with relative risks of 1.299 (95% CI [0.998, 1.691], p = .051), 1.403 (95% CI [1.038, 1.897], p = .028), and 1.343 (95% CI [0.963, 1.875], p = .083) at one, two, and three months of treatment, respectively (Figure 4c).
Difference in HRV profiles after receiving treatment
Figure 5 shows the changes in HRV before and after the three-month treatment. The low-HRV group showed significant increases in most HRV parameters, except for HF and LF/HF, after treatment (p < .01, paired t-test or Wilcoxon signed-rank test if the normality assumption was violated). However, their HRVs after treatment, including SDNN, LF, HF, TP, and SD2, were still significantly lower than healthy controls (p < .05). On the other hand, for the normal-HRV group, all HRV parameters remained comparable to those of the healthy controls (p > .05) both before and after treatment. Correlation analysis, as presented in Online Supplemental Table 3, indicated that HRV changes did not have an apparent correlation with clinical improvements.

Comparison of HRV among controls and two patient subgroups with chronic migraine before and after prophylaxis.
Discussion
The present study provides evidence that the majority of HRV parameters derived from five-minute resting ECG recordings, which predominantly reflect the parasympathetic/cardiovagal modulatory capacity, were significantly lower in patients than in those with healthy controls. These findings support the notion that parasympathetic dysfunction is a common feature of both episodic and chronic migraine, broadening the current understanding of the autonomic dysfunction present in migraine spectrum. Given the lack of observed correlation between each HRV parameter and headache characteristic, it could be inferred that impaired parasympathetic function might be an intrinsic property in patients with chronic migraines. Additionally, our study demonstrated that patients with a normal parasympathetic/cardiovagal function, as indicated by a higher SDNN before flunarizine prophylaxis, had a more favorable treatment outcome than those with a lower parasympathetic/cardiovagal function (a lower SDNN). These findings suggest that pre-treatment assessment of parasympathetic/cardiovagal function may help predict treatment response in patients.
HRV has been widely used in clinical settings to assess autonomic nervous system function (38) by evaluating different parameters that deduce both parasympathetic and sympathetic nervous system functions (15). However, as mentioned in the Introduction, recent studies (16,17) have suggested that HRV derived from resting ECG recording mainly reflects vagally-mediated parasympathetic function. Accordingly, HRV studies (39,40) derived from resting ECG have challenged the notion that LF power and its related parameter (e.g., LF/HF ratio) could be used as indicators of sympathetic function or sympathetic-parasympathetic balance. The globally reduced HRV parameters, including SDNN, RMSSD, TP, HF power, SD1, and SD2 (except for LF power and LF/HF ratio) from resting ECG in this study lend support to our hypothesis that patients had a reduced parasympathetic/cardiovagal function. Such findings are consistent with recent meta-analysis studies, which demonstrated reduced parasympathetic/cardiovagal function in patients with headache disorders (21) as well as chronic pain (20). This suggests that reduced parasympathetic/cardiovagal function could be a universal epiphenomenon.
The SDNN is a commonly used time-domain metric to assess overall parasympathetic/cardiovagal function. Due to its ease of calculation, the SDNN has been widely used as a prognostic predictor in various diseases. Originally used to predict the outcome of coronary artery disease (24), the SDNN has also been applied in cerebrovascular disease (41), sepsis (42), and COVID-19 infection (25), with all studies demonstrating that a preserved (normal) SDNN predicts a better outcome. The exact mechanisms underlying this predictive power are not yet fully understood, but they may be related to the anti-inflammatory and pain modulation properties mediated by the vagus nerve (28,29), which are involved in the pathophysiology of various diseases, as mentioned in the Introduction.
Although the underlying pathomechanism of chronic migraines is not fully understood, it’s widely accepted that numerous factors may contribute, including systemic and neural inflammation, altered sensory processing, as well as functional and structural changes in the brain, particularly in areas involved in pain processing, among others (43). Indeed, inflammation plays a critical role in the pathophysiology of migraine (44) and may contribute to its chronification. During a migraine attack, it has been shown that levels of certain inflammatory mediators, such as calcitonin gene-related peptide (CGRP) (45), tumor necrosis factor, interleukin-1beta, and interleukin-6, are elevated (44). In recent years, therapies targeting CGRP antagonism, either through monoclonal antibodies or direct molecular blocking, have shown tremendous therapeutic effects in episodic and chronic migraines (46). Thus, a stronger vagal function, as indicated by SDNN, may have a relatively preserved anti-inflammatory function, which could explain the better response to prophylaxis observed in patients with a normal SDNN.
The vagus nerve’s anti-migraine effect may also be related to its ability to modulate pain sensitivity, a critical factor in migraine chronification (47) and treatment response (48). Altered sensory processing plays a fundamental role in the pathogenesis of migraines and could be one of the most upstream phenomena (43), making patients more susceptible to both external and internal sensory stimuli (49,50). When these sensory stimuli surpass the lowered threshold in patients, areas involved in pain processing may be activated, setting off subsequent cascades that lead to a migraine attack or chronification. About 80% of vagus nerve fibers are afferent, terminating in the nucleus of the solitary tract or the spinal nucleus of trigeminal nerves (27). In this way, the vagus nerve takes part in pain modulation, not only to external stimuli via its connection to the spinal nucleus of the trigeminal nerve but also to internal homeostasis via the nucleus of the solitary tract. This unique property has led to the use of transcutaneous noninvasive vagus nerve stimulation as an acute or chronic migraine treatment (49,50). Taken together, a robust vagal function may indicate better anti-inflammatory and pain modulation properties, resulting in a more favorable prophylaxis response.
In this study, we did not identify any correlation between the differences in HRV (post-treatment vs. pre-treatment) and clinical improvement over the course of a 12-week treatment (Online Supplemental Table 3). Thus, our results did not support the idea that HRV could serve as a clinical surrogate biomarker in treating patients with chronic migraine using flunarizine. Although previous studies (22,23) have demonstrated an improved HRV profile coinciding with clinical improvement in patients with episodic migraine, this improvement was exclusive to the group receiving yoga treatment and was not observed in those receiving conventional therapy (i.e., medication). The alteration in HRV profile in these studies may likely be associated with the known effects of yoga exercise on the parasympathetic system, as the HRV profile remained unchanged in the group receiving conventional therapy despite experiencing clinical improvement. This is consistent with the observations in our study (Figure 5). Interestingly, our study revealed that patients with a low baseline HRV experienced partial normalization following preventive treatment (Figure 5). Since acute pain is known to reduce parasympathetic activity and/or increase sympathetic activity, thereby affecting HRV, the observed improvement in headache symptoms within the low-HRV group could have contributed to this partial normalization. Conversely, the normal-HRV group might already possess robust parasympathetic/cardiovagal function, which could not be further improved, possibly due to a ceiling effect.
While our study provides important insights into the potential use of HRV as a predictor of treatment response in patients with chronic migraines, several limitations must be acknowledged. Firstly, we did not control patients’ headache status during ECG recording. This could lead to a potential confounding effect on the HRV measurements, as HRV may be affected by the presence or absence of pain. However, given that our patients had monthly headache days of 21.6 at baseline, they were most likely at the ictal period or between two close attacks, making it impossible to examine at the inter-ictal period. Therefore, the observed differences in HRV between healthy controls and patients could be a combination of state and trait effects, with the latter being predominant. Secondly, patients often have multiple comorbidities and may be taking multiple preventive medications. Our study recruited relatively homogeneous participants, such as prophylactics naïve and without depression. Therefore, our findings may not be generalizable to other populations with chronic migraine who have different comorbidities or are on different preventive medications. Lastly, our study only investigated the effect of flunarizine as a prophylactic regimen. Further research is necessary to examine whether the baseline HRV profiles can predict treatment response to other preventive strategies in patients with chronic migraine.
Conclusions
In conclusion, our study provides evidence that patients with chronic migraine have reduced parasympathetic/cardiovagal modulation and that a normal baseline parasympathetic/cardiovagal modulatory function, as indicated by normal SDNN, is associated with a better therapeutic response to flunarizine prevention. These findings suggest that the baseline HRV could be used as a predictor of treatment outcomes in patients. Further research is needed to validate these findings and to investigate the potential of HRV-based predictive models for individualized treatment selection in patients.
Article highlights
The HRVs in patients with chronic migraine were significantly lower than those in healthy controls. After preventive treatment with flunarizine, patients with chronic migraine with normal HRV showed more significant reductions in monthly headache days than those with low HRV. HRV owns the potential as an outcome predictor in patients with chronic migraine receiving preventive treatment.
Contributions
CHC: study design, data acquisition, data analysis, drafting initial manuscript, and manuscript revision; KLL: study design, data acquisition, data analysis, and manuscript revision; JYL: drafting initial manuscript and data analysis; JTK: data acquisition; WTC: study design and data acquisition; SPC: study design and data acquisition; YFW: study design and data acquisition; HYL: data acquisition; FJH: study design; LLHP: data acquisition and data analysis; SJW: study design, data acquisition, and manuscript revision. The authors participated in the discussion, provided comments, read, and approved the final manuscript.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231206781 - Supplemental material for Abnormal heart rate variability and its application in predicting treatment efficacy in patients with chronic migraine: An exploratory study
Supplemental material, sj-pdf-1-cep-10.1177_03331024231206781 for Abnormal heart rate variability and its application in predicting treatment efficacy in patients with chronic migraine: An exploratory study by Chun-Hsiang Chuang, Jhe-Yu Li, Jung-Tai King, Wei-Ta Chen, Shih-Pin Chen, Yen-Feng Wang, Hung-Yu Liu, Fu-Jung Hsiao, Li-Ling Hope Pan, Shuu-Jiun Wang and Kuan-Lin Lai in Cephalalgia
Footnotes
Acknowledgments
We would like to thank the study participants and the research assistants for actively participating. We are also profoundly grateful to Professor Ben-Chang Shia of Fu Jen Catholic University, Taipei, Taiwan, for his expert consultation and guidance on statistical analysis.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CH Chuang, KL Lai, JY Li, JT King, WT Chen, SP Chen, YF Wang, HY Liu, FJ Hsiao, and LLH Pan have no conflict of interest to declare. Dr. SJ Wang is the PI of clinical trials for Novartis, Lundbeck, Teva, and AbbVie. Dr. SJ Wang has received personal fees as an advisor or speaker from AbbVie, Pfizer, Orient EuroPharma, and Percept.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Science and Technology Council and the Ministry of Science and Technology of Taiwan (NSTC 112-2636-E-007-009, MOST 111-2636-E-007-020, and MOST 110-2636-E-007-018 to CH Chuang; MOST 109-2314-B-075-049 and NSTC 112-2314-B-075-035-MY3 to KL Lai; MOST 106-2321-B-010-009, MOST 107-2321-B-010-001, MOST 108-2321-B-010-014-MY2, MOST 108-2321-B-010-001, MOST 108-2314-B-010-023-MY3, MOST 109-2321-B-010-006, MOST 110-2321-B-010-005, MOST 110-2321-B-A49A-502, MOST 111-2321-B-A49-011, MOST 111-2321-B-A49-004, MOST 111-2314-B-A49-069-MY3, MOST 111-2314-B-075-086-MY3, and NSTC 112-2321-B-075-007 to SJ Wang), Taipei Veterans General Hospital and VGHUST Joint Research Program (VGHUST 108C-129 to WT Chen; VGHUST 112-G1-2-3 to KL Lai; VGHUST 112-G1-2-1, V112C-113 and V112E-004-1 to SJ Wang), Ministry of Health and Welfare, Taiwan (MOHW 109-TDU-B-211-114001, MOHW 110-TDU-B-211-124001, MOHW 111-TDU-B-211-134001, MOHW 112-TDU-B-211-144001), and the Brain Research Center, National Yang Ming Chiao Tung University, from the Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education of Taiwan.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
