Abstract
Introduction
Levcromakalim opens ATP-sensitive potassium channels (KATP channel) and induces head pain in healthy volunteers and migraine headache in migraine patients, but no pain in other parts of the body. KATP channels are expressed in C- and Aδ-fibers, and these channels might directly activate nociceptors and thereby evoke pain in humans.
Methods
To assess the local effect of KATP channel opening in trigeminal and extra-trigeminal regions, we performed a crossover, double-blind, placebo-controlled study in healthy volunteers. Participants received intradermal and intramuscular injections of levcromakalim and placebo in the forehead and the forearms.
Results
Intradermal and intramuscular injections of levcromakalim did not evoke more pain compared to placebo in the forehead (p > 0.05) and the forearms (p > 0.05). Intradermal injection of levcromakalim caused more flare (p < 0.001), skin temperature increase (p < 0.001), and skin blood flow increase (p < 0.001) compared to placebo in the forehead and the forearms.
Conclusion
These findings suggest that it is unlikely that levcromakalim induces head pain by direct activation of peripheral neurons.
Introduction
ATP-sensitive potassium channels (KATP channels) are widely expressed in the peripheral and central nervous tissues, including sensory afferents (1), trigeminal ganglion (2), dorsal root ganglia (DRG) (3,4), as well as at the spinal and supra-spinal level of the CNS (5). These channels modulate nociceptive processing (2,4), and regulate membrane excitability and neurotransmitter release (6,7). Furthermore, KATP channel activation (opening) in central neurons mediates anti-nociception and suppresses neuronal hyperexcitability (8,9). Potassium channel openers decrease neuronal excitability (10), leading to the approval of Ezogabine as the first neuronal potassium channel opener for the treatment of epilepsy (11). In rats, KATP channel openers potentiate the anti-nociceptive effect of morphine (12,13), whereas it can be antagonized by the KATP channel blocker glibenclamide (1).
Clinical trials of KATP channel openers (KCOs) pinacidil and levcromakalim reported headache as a frequent adverse event (14,15). This effect was confirmed in a controlled study in healthy volunteers (16), and in migraine patients, intravenous infusion of levcromakalim induced migraine attacks (17). The migraine-inducing effect may be due to KATP channel activation in trigeminal afferents or a vasodilatory effect, but KCOs may also directly activate nociceptors and thereby evoke pain. It is still unknown whether any evoked pain might differ between trigeminal and extra-trigeminal regions.
In this double-blind, placebo-controlled, crossover study, we therefore investigated whether intradermal and intramuscular injections of levcromakalim evoke pain in the forehead and the forearms. We hypothesized that local levcromakalim injection would induce pain in the trigeminal but not in extra-trigeminal regions in healthy volunteers.
Materials and methods
We recruited 20 healthy volunteers through the Danish website www.forsøgsperson.dk for enrolment in health research. All participants gave written informed consent before inclusion. The female participants were requested to have sufficient contraception. Exclusion criteria were any type of primary headache (except episodic tension-type headache no more than one day per month), previous serious somatic or psychiatric diseases, or intake of daily medication, except oral contraceptives. A full medical examination and ECG were performed on the day of recruitment.
The study was approved by the Ethics Committee in the Capital Region of Denmark (H-18008792) and the Danish Data Protection Agency and was conducted according to the Declaration of Helsinki of 1964, as revised in 2008. The study was also registered at ClinicalTrials.gov (NCT03726242).
Pilot study
The purpose of the pilot study was to determine the dose of levcromakalim that significantly changes the skin blood flow compared with placebo. In a double-blind, placebo-controlled, four-way crossover design, three healthy volunteers were randomly allocated to receive 0.05 ml intradermal injection in the forehead and the forearms of placebo and one of four doses of levcromakalim (50, 100, 150 and 200 µg/ml) on four different days with a minimum interval of one day. Changes in skin blood flow were measured every 5 min from 0 to 30 min.
Main study
Twenty participants were randomly allocated to receive one of two different orderings of an intramuscular or intradermal injection of levcromakalim and placebo (isotonic saline) on two study days separated by at least 1 day. Each participant received four injections each day; first, levcromakalim in one and placebo in the other forearm; second, levcromakalim in one and placebo in the other side of the forehead. Before injection, participants rested for at least 30 min and the experiment was conducted in a quiet room (at a temperature of 23oC). The participants were seated on a stable and comfortable chair during the injections in the forearms and were then rested in a supine position on a comfortable bed during the injections in the forehead. The injection sites on the forearms and the forehead were delineated with tape (Paper Surgical Tape, Latex-Free) and disinfected. Pain intensity, itching, skin blood flow, flare, skin temperature, and wheal reaction were measured at baseline −5 and 0 min and every 5 min over a period of 45 min after the injection.
Pain intensity and itching were measured by numerical rating scale (NRS) 0–10, where 0 was equal to no pain/itching, 1 was a changed sensation and 10 was the maximum pain/itching. Flare (erythema/redness around the injection site) was identified visually and the area was measured by a predetermined, transparent acetate sheet in mm2. The wheal reaction (the area of edema caused by plasma extravasation) was inspected visually and by light palpation, and then the area of wheal reaction was measured by a predetermined transparent acetate sheet in mm2. Changes in skin temperature were measured with an infrared camera (Thermovision), while changes in skin blood flow were measured with laser speckle.
Levcromakalim was manufactured by Sigma-Aldrich under conditions and practices required by the good manufacturing practice regulations, reference number (CSQ-23637-1EA). Randomization and preparation of the study drug was performed by the Capital Region Central Pharmacy. The randomization code remained in the hospital during the study and was not available to the investigators until the study was completed.
Intradermal injection
We injected 0.05 ml of 150 µg/ml levcromakalim and 0.05 ml placebo with an angle of 0 to 15 degrees with a 29-gauge needle (Monoject ½ mL insulin syringe, Permanent Needle 29 G × ½). The injection was performed on the volar side of the forearms approximately 1/3 distal to the cubital fossa and within 1 cm from the midline to avoid visible veins. The injection in the forehead was performed approximately 3 cm above the eyebrow and 3 cm away from the midline. The intradermal injections of levcromakalim and placebo were performed simultaneously, first in the forearms and then in the forehead.
Intramuscular injection
We injected 0.2 ml of 150 µg/ml levcromakalim and 0.2 ml placebo at an angle of approximately 90 degrees with a 27-gauge needle (BD Safety Glide TB Syringe 1 mL 27 G × ½ (0.4 mm × 13 mm)). The injection was performed on the volar side of the forearms approximately 1/3 distal to the cubital fossa and within 1 cm from the midline, to avoid visible veins. The injection in the forehead was performed on the hairless region of the anterior temporal muscle, about 3–4 cm in front of the coronal suture. The intramuscular injections of levcromakalim and placebo were performed simultaneously, first in the forearms and then in the forehead.
Skin blood flow
Changes in skin blood flow were measured by laser speckle contrast imager (Moor Instruments speckle contrast blood assessment, moorFLPI Full Laser Perfusion Imager). The injection sites were scanned with laser speckle from a distance of 30 cm from the skin. The laser speckle was set with an exposure time of 8.3 ms/pixel with a time interval of 5 seconds and the number of images was fixed to1000 images. The bandwidth was set at 25 Hz. The laser speckle contrast imager was calibrated using a reference flux signal, in this case a suspension of polystyrene microspheres in water undergoing thermal or Brownian motion. The images were marked manually to obtain a scaled color-coded live flux image, as the participants were instructed to close their eyes and remain still. The red color is equal to high perfusion, while the blue color is equal to low perfusion. To analyse the data, the mean perfusion rate was calculated before and every 5 min after the injection. Laser speckle contrast imaging has been previously described (18).
Thermovision camera
The Thermovision 900 SWTE scanner is designed to function in the 2–5.4 µm band of the infrared spectrum using a 2 thermo-electrically cooled SPRITE (Signal Processing in The Element) detector. It gives a scan rate of 20 Hz and an image resolution of 140 elements per line, which provides an image of 204 pixels by 128 lines. The camera was fixed in a vertical position close to the injection sites to avoid irrelevant images in the background. The participants were instructed to sit completely still during measurements. The images were performed manually to ensure that the participants did not move. Images were taken before the injection and every 5 min after the injection for a period of 45 min.
Statistical analysis
The pain and itching are given as median values with quartiles. Other variables are given as mean values with standard deviation (± SD). The primary endpoint was the difference between the areas under the NRS-time curve (NRSAUC) after levcromakalim versus placebo. The secondary endpoints were the difference between the area under the curve (AUC) in flare, wheal, temperature, and change in skin blood flow after levcromakalim versus placebo. AUC was calculated according to the trapezium rule (19) to gain a summary measure to analyse the differences in response between levcromakalim and placebo. Sample size calculation was based on the difference between two paired groups reporting pain after levcromakalim and placebo, at 5% significance with 80% power. We assumed that 70% of participants would report pain after levcromakalim and 20% after placebo, and estimated that inclusion of at least 12 participants would be sufficient in a crossover study (20). To compensate for potential drop-outs, we decided to include 20 participants. To test the difference in AUC between levcromakalim and placebo we used a paired two-way t-test. The baseline was subtracted to compare changes in skin blood flow, flare and skin temperature after levcromakalim injection in the forehead with the forearms.
SPSS Statistics version 19 for Windows was used for all analyses and a p-value <0·05 was accepted as the level of significance.
Results
In the pilot study, three healthy volunteers (two women, age 21 and 22 years and one man, age 21) completed all four study days. The concentration used for systemic administration was 50 µg/ml (17). To find the proper concentration for local administration we performed dose titration with increase from 50 to 100, 150 and 200 µg/ml. AUC for skin blood flow change was significantly higher after l50 µg/ml (p = 0.03 for the forearms and p = 0.04 for the forehead) and 200 µg/ml (p = 0.04 for the forearms and p = 0.04 for the forehead) levcromakalim compared with placebo, without difference between these two doses. Thus, the main experiment was performed with 150 µg/ml levcromakalim (Figure 1).
Pilot experiment; placebo and four different doses of levcromakalim. Intradermal injection in the forehead (a) and the forearms (b).
Twenty healthy volunteers (13 women and seven men) completed the main study. The mean age was 22 years (ranged from 18 to 27), the mean weight was 69 kg, and the mean height was 174 cm.
Intradermal injection
We found no difference in cutaneous pain induction between levcromakalim and placebo in the forehead (p = 0.054) and the forearms (p = 0.9) (Figure 2). There was no difference in itching after levcromakalim injection compared to placebo in the forehead (p = 0.7) or the forearms (p = 0.9). Flare responses were significantly greater after levcromakalim compared to placebo in the forehead (p < 0.001) and the forearms (p < 0.001) (Figure 3). There was no difference in wheal reaction after levcromakalim injection compared to placebo in the forehead (p = 0.07) and the forearms (p = 0.4). We found a significant increase in skin temperature after levcromakalim compared to placebo in the forehead (p < 0.001) and the forearms (p < 0.001) (Figure 4).
Pain intensity after intradermal injection of levcromakalim versus placebo. Flare after intradermal injection of levcromakalim versus placebo. (a) Laser speckle showed that levcromakalim increases the blood flow of the skin in the forehead and the forearms. The circle on the right forehead indicates the side of placebo injection and the square on the left forehead indicates the side of levcromakalim injection. (b) Thermovision camera showed that levcromakalim increases the temperature of the skin in the forehead and the forearms. (c) Skin blood flow changes after intradermal injection of levcromakalim compared to placebo. (d) Skin temperature after intradermal levcromakalim injection compared to placebo.
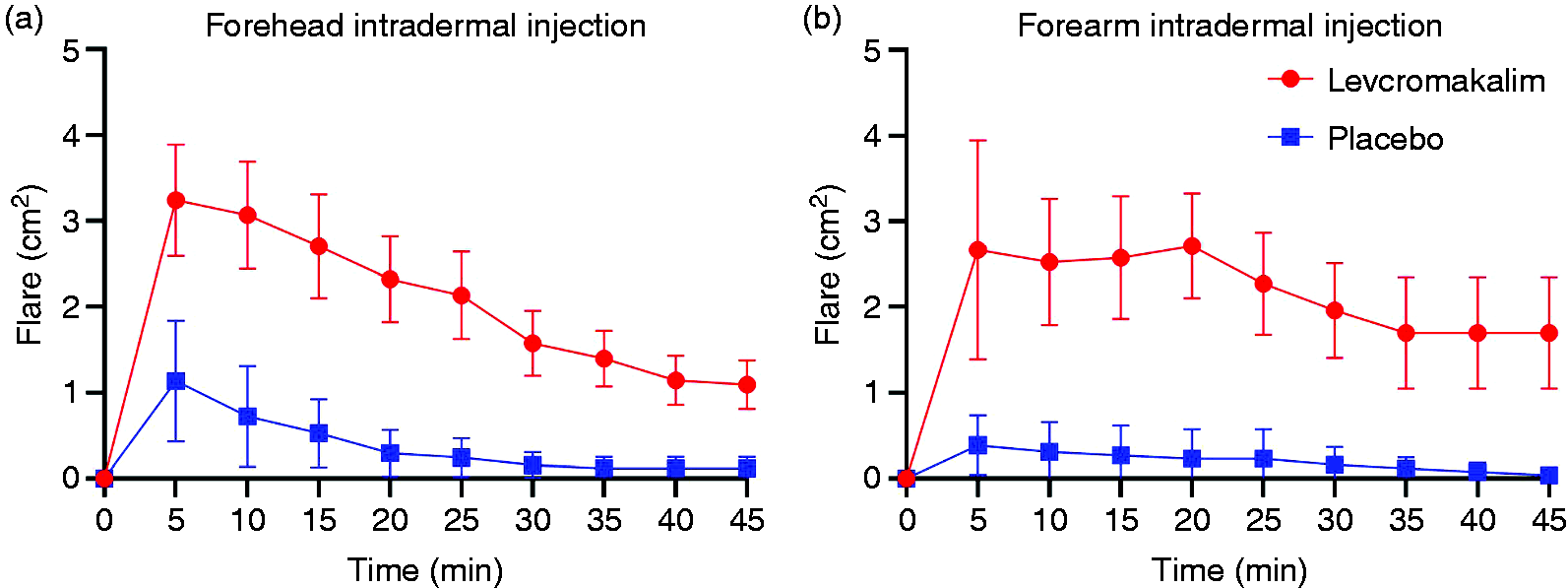

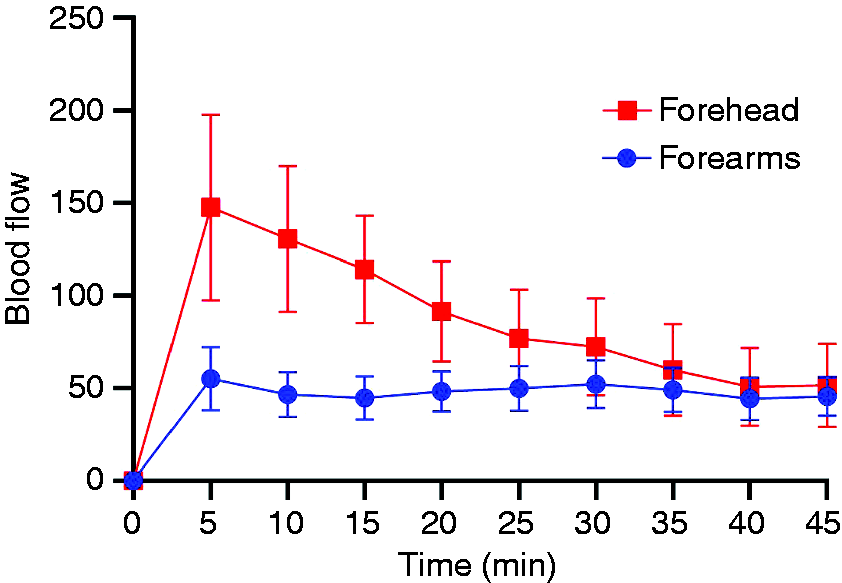
There was a significant change in skin blood flow after levcromakalim injection compared to placebo in the forehead (p < 0.001) and the forearms (p < 0.001) (Figure 4). After subtracting the baseline from AUC, the explorative analysis showed a significant increase in skin blood flow (p < 0.01), but not in flare nor in skin temperature (p > 0.05) after levcromakalim injection in the forehead compared to the forearms. An exploratory point-to-point comparison showed significant differences in flare (p = 0.012) and skin temperature (p < 0.001) after levcromakalim injection in the forehead compared to the forearms (Figures 5–7).
Flare (p = 0.58 between AUC) after levcromakalim injection between the forehead and the forearms. However, the point-to-point comparison showed significant differences in flare (p = 0.012). Changes in skin temperature (p = 0.36 between AUC) after levcromakalim injection between the forehead and the forearms, when the baseline was subtracted. The point-to-point comparison showed significant changes in skin temperature (p < 0.001) after levcromakalim injection in the forehead compared to the forearms. Skin blood flow changes (p = 0.003) after levcromakalim injection between the forehead and the forearms, when the baseline was subtracted.

Intramuscular injection
We found no difference in pain induction after levcromakalim compared to placebo in the forehead (p = 0.1) and the forearms (p = 0.7). There was no difference in itching after levcromakalim compared to placebo in the forehead (p = 0.9) and the forearms (p = 0.8). We found no differences in flare, wheal, temperature, and skin blood flow responses after levcromakalim versus placebo in the forehead and the forearms (p > 0.05).
Study safety
The volunteers tolerated the intradermal and intramuscular experimental drug injections in the forehead and forearms well and no adverse events or complaints were reported.
Discussion
The main finding of this double-blind, placebo-controlled, crossover study was that intradermal and intramuscular injections of levcromakalim did not evoke more pain compared to placebo. Intradermal injection of levcromakalim caused more flare, skin temperature increase, and skin blood flow increase compared to placebo. KATP channels are expressed in C- and Aδ-fibers (1), and their activation increases the K+ permeability and leads to hyperpolarization (3). It is known that hyperpolarization activates hyperpolarization-activated cyclic nucleotide-gated cation channels (HCN channels) and this results in a generation of an inward current (21). In rats, modulation of HCN channels in sensory fibers plays a significant role in sensory processing and contributes to spontaneous and persistent pain (22). Interestingly, KATP channel activation in nigral dopaminergic neurons stimulated HCN channels and thus increased the firing rate of these neurons (23). It is, however, unknown whether KATP channel-induced hyperpolarization activates HCN channels in sensory fibers. In the present study, injection of KCO levcromakalim did not evoke skin or muscle pain in either trigeminal or extra-trigeminal regions. In rats, extracellular application of KCOs cromakalim (a less potent form of levcromakalim) and diazoxide caused hyperpolarization in neurons isolated from the midbrain and sensory neurons, respectively (3,24). This hyperpolarizing effect can be reversed by the KATP channel blocker tolbutamide (25). We used one of the most potent KCOs (26) with a dose of 150 µg/ml (524 µM), which should be sufficient to induce hyperpolarization in the surrounding tissue. The lack of cutaneous and muscle pain in our experiment suggests that levcromakalim-induced hyperpolarization does not lead to activation of HCN channels in sensory fibres.
The loss of KATP channel currents after nerve injury has been proposed to contribute to neuropathic pain (4). Intradermal injection of capsaicin evokes pain, induces allodynia and produces flare response (27) by binding to the transient receptor potential calcium channel, subfamily V, member 1 (TRPV1) (28). In rats, KATP channel activation mediates anti-nociception (9), attenuates capsaicin-induced masseter hypersensitivity and, more importantly, the anti-hyperalgesic response is reversed by the KATP channel blocker glibenclamide (2). Interestingly, direct activation of the KATP channel in DRG neurons in vitro suppresses neuronal hyperexcitability induced by prostaglandins (8). KATP channel activation seems also to be decisive in µ-opioid-induced anti-nociception (5). In rodents, the intracerebroventricular (icv) and intraplantar administration of glibenclamide antagonized the antinociception induced by subcutaneous (sc) morphine and diclofenac (29–31). Furthermore, icv administration of the KCOs pinacidil and cromakalim potentiated the anti-nociception induced by sc morphine in the tail flick test (12,13) and the antinociceptive effect KCOs were antagonized by glibenclamide administration (30). Collectively, these findings are consistent with the present results and explain the lack of pain induction after levcromakalim injection.
It is well established that KATP channels contribute to the regulation of vascular tone, including in cranial arteries (32). Reduction of intracellular ATP causes KATP channel activation; K+ efflux hyperpolarizes the membrane and close voltage-operated Ca2+-channels (VOCC) (33). This leads to a decrease in cytosolic Ca2+ concentration followed by relaxation of vascular smooth muscle cells and an increase in blood flow (34). Several studies demonstrated that application of levcromakalim produced hyperpolarization and dilation of smooth muscle cells (35). In cardiac muscle tissue, KCOs prevent intracellular Ca2+ overload, as opening of plasma membrane KATP channels shortens the action potential duration (APD), limiting cellular injury by preserving cellular energetics and ultimately cell survival (36). Due to combined cardioprotective and vasodilatory properties, KATP channels are considered as a target for several cardiac conditions. The systemic administration of non-neuronal KCO leads to systemic vasodilation accompanied by increase in cardiac output, heart rate and circulating levels of norepinephrine and plasma renin activity (37). The present data showed that levcromakalim increased skin blood flow in humans and further support the previous findings. Skin blood flow may also explain the increased flare and skin temperature. At present, it is unknown whether KATP channel activation affects mast cells and histamine release. However, the lack of wheal reaction in the present study speaks against histamine release. The explorative analysis showed a significant increase in skin blood flow and skin temperature after levcromakalim injection in the forehead compared to the forearms. These finding are expected because of the quantity of vascularization in the forehead. However, intradermal injection of levcromakalim caused more flare in the forearms compared to the forehead. A possible explanation could be by color difference at baseline between the forehead compared to the forearms; thus, minor changes in the forearms can be easily detected, which is not the case for the forehead.
We measured no change in blood flow, flare, and temperature after intramuscular levcromakalim injection. These data might be explained by the increased vascularization and the large and deep space intramuscularly.
The ability of systemic levcromakalim to evoke only cephalic pain suggests that opening of KATP channels in trigeminal and extra-trigeminal regions may differ. Based on our findings a direct sensory neuronal activation is unlikely to be responsible for levcromakalim-induced headache and migraine. The induced headache could be explained merely by vasodilation, but this is not the case for migraine attacks (38,39). Sur1-regulated channels, including KATP channels, are also expressed in astrocytes, oligodendrocytes, endothelial cells, and microglial cells (40–42). Microglial cells’ upregulation of inducible nitric oxide synthase (iNOS) and production of proinflammatory cytokines TNF-α and IL-6 are inhibited by KATP channel blocker glibenclamide (42–44). Moreover, glibenclamide reduces plasma protein extravasation (45,46) and inhibits neutrophil migration and fluid leakage during acute inflammatory response (47). These studies indicate that levcromakalim-induced headache and migraine could be explained by different sequences of events secondary or tertiary to the dilation of the meningeal arteries. Although opening of KATP channels in CNS central neurons leads to hyperpolarization and thus inactivation (24), we cannot rule out activation of neurons in the hypothalamus and brainstem. As mentioned previously, one in vitro study reported that KATP channel activation increased the neuronal firing rate in nigral dopaminergic neurons (23).
Conclusion
The local use of levcromakalim does not evoke more cutaneous or muscle pain in trigeminal and extra-trigeminal regions in humans compared to placebo. Thus, KATP channels’ activation is not associated with pain, but increases skin blood flow. These findings suggest that a direct activation of peripheral neurons in levcromakalim-induced headache and migraine is unlikely.
Key findings
The local use of levcromakalim does not evoke more cutaneous or muscle pain in trigeminal and extra-trigeminal regions in humans compared to placebo. KATP channels activation is not associated with pain but increases skin blood flow. We suggest that a direct activation of peripheral neurons in levcromakalim-induced headache and migraine is unlikely.
Footnotes
Acknowledgements
The authors thank all participating healthy volunteers and our colleagues Ditte Georgina Zhang and Nita Wienholtz for expert assistance. Further thanks to Lundbeck Foundation and The Candys Foundation.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MA reports personal fees from Allergan, Amgen, Alder, Eli Lilly, Novartis and Teva. MA participated in clinical trials as the principal investigator for Alder, Amgen, ElectroCore, Novartis and Teva trials. MA has no ownership interest and does not own stocks of any pharmaceutical company. MA serves as associated editor of Cephalalgia, Headache, and co-editor of the Journal of Headache and Pain. MA is the President of the International Headache Society and General Secretary of the European Headache Federation. MMK, HG, JMH, SG, LTS and JO declare no conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
