Abstract
Although myofascial tenderness is thought to play a key role in the pathophysiology of tension-type headache, very few studies have addressed neck muscle nociception. The neuronal activation pattern following local nerve growth factor (NGF) administration into semispinal neck muscles in anaesthetized mice was investigated using Fos protein immunohistochemistry. In order to differentiate between the effects of NGF administration on c-fos expression and the effects of surgical preparation, needle insertion and intramuscular injection, the experiments were conducted in three groups. In the sham group (n = 7) cannula needles were only inserted without any injection. In the saline (n = 7) and NGF groups (n = 7) 0.9% physiological saline solution or 0.8 µ
Introduction
Bilateral pericranial myofascial tenderness is a frequent and prominent clinical sign in tension-type headache (TTH) (1, 2). With manual palpation of head and neck muscles, pericranial tenderness is diagnosed in patients with episodic and chronic TTH. Pericranial tenderness is positively associated with both the intensity of headache and the frequency of TTH attacks (3, 4). Therefore, the International Classification of Headache Disorders stresses the importance of neck muscle tenderness and recommends its evaluation in patients (5). Although myofascial tenderness is supposed to play a key role in the pathophysiology of TTH (1–3), very few studies have addressed neck muscle nociception.
Only a very few studies have focused on the afferent innervation of neck muscles. Afferent projections of biventer cervicis, splenius, and occipitoscapularis neck muscles were electrophysiologically investigated in anaesthetized cats (6). Electrical stimulation of these muscle nerves evoked neuronal activity in the ipsilateral external cuneate nucleus. About 40% of biventer cervicis neurons received monosynaptic muscle input. Antidromic discharges were evoked in about 30% of all neurons by electrical stimulation of the ipsilateral inferior peduncle and anterior lobes of the cerebellum. No antidromic activity could be elicited by electrical stimulation of the contralateral thalamus. Primary afferent fibres from suboccipital muscles were transganglionically labelled with horseradish peroxidase in rats and cats (7–11). Retrogradely labelled cells were located in the ipsilateral C2 to C4 spinal ganglia, in C1 down to C6 spinal segments, and in the medulla oblongata. Many labelled fibres projected to the lateral and medial cuneate nuclei. Sensory neurons in laminae V/VI of the C2 spinal dorsal horn had convergent afferent input from deep paraspinal muscles evoked by noxious mechanical stimulation in rat (12). These studies collected some anatomical and physiological data on neck muscle innervation, but did not document the neuronal activation pattern in the central nervous system due to preferential activation of neck muscle nociceptors.
Nerve growth factor (NGF) seems an appropriate chemical stimulus in order to induce selective nociceptive input from single muscles. NGF applied into gastrocnemius-soleus muscle in anaesthetized rats excited more than 60% of high-threshold mechanosensitive afferent group IV fibres (13). NGF activated neither low-threshold mechanosensitive group IV fibres nor any other muscle afferents of groups I, II and III. Thus, NGF probably induced a selective activation of group IV nociceptors in skeletal muscles. Local NGF injection into neck muscles induced sustained central facilitation of orofacial sensorimotor processing in anaesthetized mice (14). This long-term facilitation by a singular injection possibly reflects plastic changes of nociceptive synaptic processing that may be involved in the pathophysiology of TTH.
c-Fos expression is an appropriate marker of neuronal activation following noxious stimulation. c-Fos is an immediate-early gene whose expression results in production of the protein Fos, detectable in cell nuclei through the presence of Fos-like immunoreactivity (15). It is well established that myofascial nociceptive input results in increased expression of c-fos in the central nervous system (16, 17). In the present study the neuronal activation pattern following local NGF administration into neck muscles in anaesthetized mice was investigated using Fos immunohistochemistry.
Methods
Fos expression following NGF injection in neck muscles was investigated in 21 adult male C57BL/6 mice (about 12 weeks old; 22–30 g; Charles River Laboratories, Sulzfeld, Germany). All procedures received institutional approval from the local ethics committee of the University of Aachen (ref. no. 50.203.2-AC 15, 16/03). The principles of laboratory animal care and use of laboratory animals [European Communities Council Directive of 24 November 1986 (86/609/EEC)] were followed. All efforts were made to minimize animal suffering and to use only the number of animals necessary to produce reliable and statistically justified scientific data.
The detailed description of anaesthesia and surgery has recently been published (18). The mice were anaesthetized by an initial intraperitoneal (i.p.) injection of a 0.5% pentobarbital sodium salt solution (Sigma-Aldrich Chemie GmbH, Taufkirchen, Germany) with a dose of 70 mg/kg. Depth of anaesthesia was checked by ensuring that noxious pinch stimulation (blunt forceps) of the hindpaw, the forepaw and the ear did not evoke any sensorimotor reflexes. When the mouse was sufficiently deeply anaesthetized the skin of the neck was carefully shaved and Lidocaine gel (AstraZeneca GmbH, Wedel, Germany) was applied to induce local anaesthesia. Dexpanthenol eye ointment (Roche AG, Grenzach-Wyhlen, Germany) was applied to the cornea of both eyes to protect them from drying. The core body temperature was maintained at 37.5°C with a heating blanket and a fine rectal thermal probe (FMI GmbH, Seeheim, Germany).
The semispinal neck muscle on both sides was carefully exposed via a small neck skin cut and blunt surgical preparation of the superficial trapezius muscle. A 27 G injection cannula needle (0.4 mm diameter) was successively inserted into both semispinal neck muscle bellies. The order of needle insertion (right, left) was balanced (right–left n = 11; left–right n = 10). In order to differentiate between the effects of NGF administration on Fos protein expression and the effects of surgical preparation, needle insertion, and intramuscular injection, the experiments were conducted in three groups. The following experimental procedure differed in three groups of seven mice each (sham, saline, NGF). In the sham group (n = 7) needles were only inserted without any injection. In the saline group (n = 7) 20 μl of 0.9% physiological saline solution were injected in both muscles. In the NGF group 20 μl of recombinant human β-nerve growth factor (0.8 μm; NGF; Calbiochem.com) was injected in both muscles. In two animals 20 μl of 2.4 μ
After 2 h of incubation, the animal was deeply anaesthetized by i.p. administration of 1 ml 0.5% pentobarbital sodium salt solution (Sigma-Aldrich). Transcardial perfusion was performed with 20 ml physiological saline solution (37°C) and cooled formalin (4%, 4°C, at least 30 ml) in phosphate-buffered saline (PBS) via a 20 G cannula in the left ventricle. Brain and cervical spinal cord (down to C4) were carefully exposed, removed, and postfixed in a 4% formalin PBS solution for 1 h at 4°C. After cryoprotection in 20% sucrose PBS solution overnight with repeated solution changes, mice brains were frozen on the cryostat (−20°C), and 30-μm cryostat sections were cut. After air-drying overnight the sections were rehydrated in PBS at room temperature for 30 min before incubation with a 5% normal goat serum diluted in PBS+ (0.1
The quantitative evaluation in the dorsal horn of the spinal cord segments C1 to C3 and in the brainstem was performed under light microscope (magnification ×100–160). Fos-immunoreactive cells were counted in three adjacent sections each at seven anatomical levels: spinal dorsal root entry zones at levels C3 (−12.2 mm distance from bregma), C2 (−11.7 mm), C1 (−10.3 mm), caudal and rostral medullary sections (−7.8 mm; −7.0 mm), pontine level (−5.9 mm), and the mesencephalic level (−4.8 mm). Arithmetic mean and standard error of cell counts were calculated (mean ± SEM). One-way analysis of variance (
Results
Fos positive cells were found in all experiments but not in all sections. Only those anatomical levels were included in the analysis where in all experiments of one group three adjacent sections in one animal showed at least one immunoreactive cell. Therefore, pontine and rostral medullary levels were excluded from further analysis. In the remaining five anatomical levels Fos-positive cells were located in the mesencephalic periaqueductal grey (PAG), in the medullary lateral reticular nucleus (LRN), and superficial laminae I and II of the spinal dorsal horns C1, C2, and C3 (Fig. 1). Cell counts in bilateral LRN and spinal dorsal horns C1 to C3 did not show significant lateral differences. Therefore, arithmetic means of left and right side values were calculated. Fos-positive cells in the PAG were counted without consideration of laterality.

Anatomical sections in brainstem and cervical spinal cord. Line drawings (left; modified from (30)) and histological sections (right; stained with cresyl violet) of brainstem and cervical spinal cord levels that were included in the quantitative analysis of Fos immunohistochemistry. Periaqueductal grey (PAG), lateral reticular nucleus (LRN), and superficial layers of spinal dorsal horns at segments C1, C2 and C3 are marked by lines. Distances of sections from bregma are given.
In two control mice without any surgery and without any intramuscular drug administration only a very few Fos-immunoreactive cells were detected at levels C3 (≤1), C2 (≤3), C1 (≤1), LRN (≤2), and PAG (≤3). Under sham, saline, and NGF conditions immunoreactivity occurred in all spinal segments, LRN, and to a lesser extent also in PAG (Figs 2 and 3). Cell counts in sham and saline groups did not statistically differ in any analysed level (
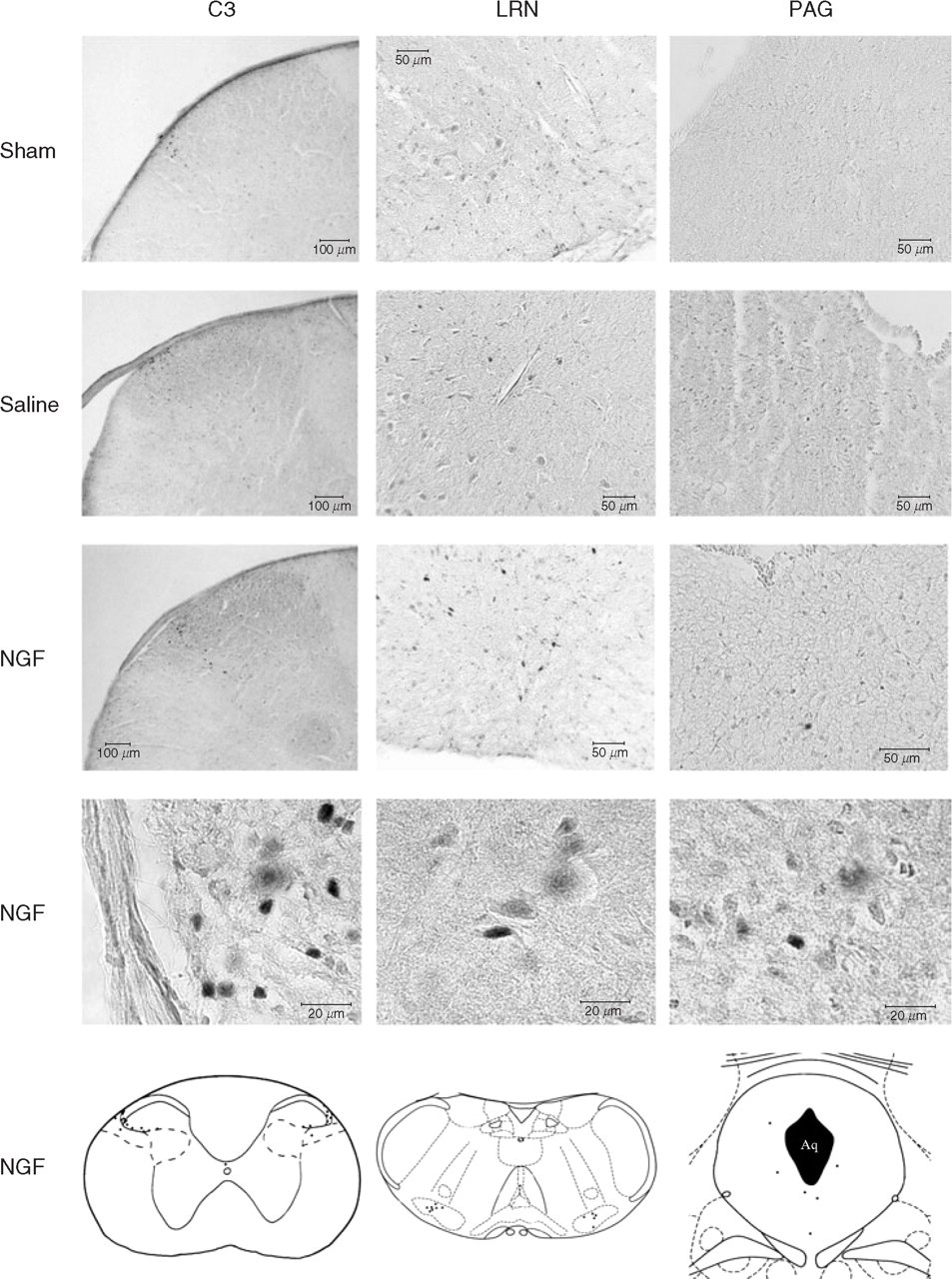
Fos immunoreactivity under sham, saline, and nerve growth factor (NGF) conditions. Photomicrographs with specimen histological sections at levels of periaqueductal grey (PAG), lateral reticular nucleus (LRN), and spinal dorsal horn at segment C3 show immunoreactivity under sham conditions, after intramuscular administration of saline and NGF in mice. Line drawings (bottom row) present manually marked Fos-immunoreactive cells after NGF administration.
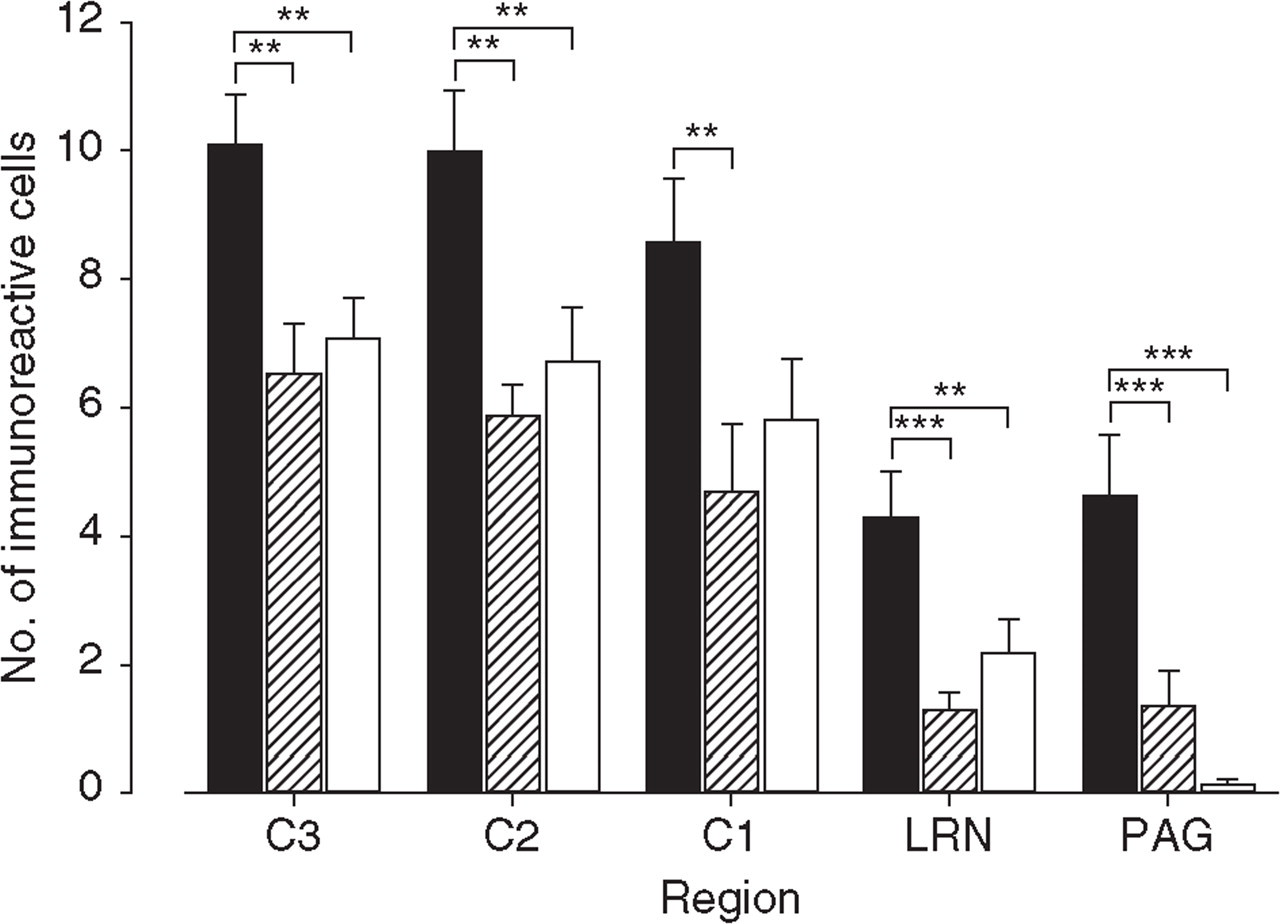
Fos immunoreactivity in brainstem and cervical spinal cord. Number of immunoreactive cells in superficial layers of spinal dorsal horns C3, C2, C1, medullary lateral reticular nucleus (LRN) and mesencephalic periaqueductal grey (PAG) under nerve growth factor (NGF) (▪, n = 7), saline (hatched, n = 7), and sham (□, n = 7) conditions in seven mice each. Cell counts are presented in arithmetic and standard error (mean ± SEM). Asterisks indicate levels of significance as calculated by the Holm–Sidak post hoc test: ∗∗P < 0.01; ∗∗∗P < 0.001.
In summary, NGF administration in neck muscles of anaesthetized mice induced significant Fos immunoreactivity in PAG, LRN and superficial layers of the cervical spinal dorsal horn levels C1–C3.
Discussion
Fos immunoreactivity was induced in all three different experimental groups, stressing the importance of control groups in these studies. The activation under sham and saline conditions is probably due to the invasive surgical procedure during exposure of the semispinal neck muscles (cutting skin and preparation of trapezius muscle). The number of Fos-immunoreactive cells did not differ between sham and saline groups. Thus, the volume effect of fluid injection into the semispinal neck muscle (saline vs. NGF) may not be involved in the NGF effect.
The absolute number of Fos-immunoreactive neurons following NGF administration was higher in spinal than in brainstem regions. In electrophysiological experiments eight neurons in the C2 spinal dorsal horn with input from the dura mater responded to noxious pressure applied to deep paraspinal muscles of the neck in anaesthetized rats (12). The lesion sites within the C2 dorsal horn indicating the recording positions corresponded to laminae V, VI and VII. No recordings in superficial layers were presented. Whereas the C2 location in electrophysiology is concordant with the detection of Fos-positive cells in C2, immunoreactivity in deep spinal dorsal horn layers was only exceptionally found under NGF. The apparent discrepancy between the electrophysiological study in rat and the present immunohistochemical study in mice may be due to the species, the anaesthesia protocols, the more invasive surgical procedure in rat, and the local chemical stimulation of the well-defined semispinal muscle in mice in contrast to mechanical stimulation of ‘deep paraspinal’ muscles in rat. Studies applying anterograde and transganglionic transport techniques detected labelled spinal structures in C2 to C4 dorsal root ganglia (7) and mainly in spinal dorsal horns of the segments C1–C3 (7, 8, 11). Group III and IV primary afferents from rat splenius muscle projected to the dorsal laminae of cervical dorsal horn and terminated in the lateral parts of these laminae (10). Following horseradish peroxidase application to the proximal stump of the rat major occipital nerve, labelled structures were found in the lateral parts of laminae I to III of the dorsal horn segments C2 and C3. In C1, primary afferent terminals were more sparsely distributed and restricted to laminae I and II (11). These anatomical data with labelled terminals in dorsolateral regions of the upper cervical spinal dorsal horns correspond to the localization of NGF-induced Fos immunoreactivity in superficial layers in the present study.
The most consistent immunoreactivity within the medulla oblongata was localized in the lateral reticular nucleus. Whereas anatomical studies reported on some labelled nuclei in the caudal brainstem such as the cuneate nucleus and the spinal trigeminal nucleus, there is no information about an involvement of the lateral reticular nucleus (7, 8, 10, 11). The lateral reticular nucleus (LRN) plays a significant role as an integrator for both ascending and descending nociceptive information (21–23). Focal electrical stimulation in the LRN inhibited the spinal nociceptive tail-flick reflex, indicating a role in the centrifugal modulation of spinal nociceptive transmission (23). Neuroanatomical studies showed that the nucleus raphe magnus sends direct projections to the LRN (22). Electrophysiological recordings of LRN neurons demonstrated that 95% of the LRN cells responded to nucleus raphe magnus stimulation and were also responsive to noxious peripheral stimulation, indicating that these cells are receiving ascending information from the spinal cord as well as receiving descending input from the endogenous pain control system (e.g. nucleus raphe magnus). Thus, Fos immunoreactivity of LRN after activation of neck muscle nociceptors by NGF is highly compatible with the anatomical connections known for these neurons.
The PAG plays a key role in nociceptive processing (24, 25). It receives afferent projections from a number of brainstem and spinal areas which are known to be involved in the modulation and conduction of nociception. The PAG is also a major component of the endogenous pain control system that is able to inhibit and facilitate pain processing. The PAG seems to play an important role in the pathophysiology of primary headaches (26, 27). Furthermore, the PAG is also involved in fear, anxiety and cardiovascular control (28, 29). Different representations of inescapable noxious stimuli in the PAG and upper cervical spinal cord were investigated in freely moving rats (17). After bilateral injection of 5% formalin solution (50 μl) in deep dorsal neck muscles, rats were subjected to a social interaction test. About 150 min later Fos immunohistochemistry was initiated. Very few Fos-immunoreactive cells were observed in the PAG of controls. In contrast, there was a significant increase in the number of immunoreactive cells in the PAG under formalin injection. Furthermore, immunoreactivity was detected in superficial layers I and II and deep layers IV and V of upper cervical spinal dorsal horns. The authors concluded, however, that in the upper cervical spinal cord noxious manipulation evoked a distinct pattern of Fos expression which reflected the pattern of primary afferent termination arising from the neck muscles. They suggested that the immunoreactivity pattern in the PAG reflected a quality more akin to the behavioural significance of the noxious event. There are some important methodological differences between the paper of Keay et al. (17) and the present study. Whereas Keay et al. applied formalin to an undefined number of ‘deep dorsal neck muscles’ in briefly anaesthetized rats, we administered NGF to the semispinal neck muscles in deeply anaesthetized mice. In the present study deeply anaesthetized mice did not wake up after intramuscular injection but remained under deep anaesthesia until the initiation of the perfusion procedure. The different anaesthetic procedures and the stronger chemical stimulus (formalin vs. NGF) probably caused a stronger degree of Fos expression in the PAG and a more widespread spinal activation in the study of Keay et al. (17). The absolute numbers of immunoreactive cells within laminae I and II of the upper cervical spinal dorsal horns correspond in both studies.
Finally, local NGF injection into semispinal neck muscles in mice induced Fos expression in superficial layers of upper cervical spinal cord, in the LRN, and the PAG. This activation pattern corresponds well to the anatomy of central nervous system processing of deep noxious input. The knowledge of central representation of neck muscle pain is an essential prerequisite for the investigation of neck muscle nociception in order to develop a future model of tension-type headache.
Footnotes
Acknowledgements
This research project was supported by grants of the German Headache Consortium (Federal Ministry of Education and Research, 01 EM 0117, project A3) and the Interdisciplinary Centre for Clinical Research BIOMAT of the Medical Faculty of the University of Aachen. The Fos immunohistochemistry was established in our laboratory with the friendly assistance of Priv.-Doz. Dr rer. nat. Ulrike Hanesch (Institute of Physiology, University of Würzburg) and Professor Dr Ruth Knüchel-Clarke (Institute of Pathology, University of Aachen).
