Abstract
Objective: The study was conducted to investigate the after-effect of transcranial direct current stimulation (tDCS) applied over the human primary motor cortex (M1) on trigeminal and extracranial nociceptive processing.
Basic procedures: Nineteen healthy volunteers were stimulated using cathodal, anodal (both 1 mA) or sham tDCS for 20 minutes. Pain processing was assessed by recording trigeminal and extracranial pain-related evoked potentials (PREPs) following electrical stimulation of the contralateral forehead and hand at baseline, 0, 20 and 50 minutes post-tDCS.
Main findings: Cathodal tDCS resulted in decreased peak-to-peak amplitudes (PPAs) by 18% while anodal tDCS lead to increased PPAs of PREPs by 35% (p < .05).
Principal conclusions: The decreased PPAs suggest an inhibition and the increased PPAs of PREPs suggest an excitation of trigeminal and extracranial pain processing induced by tDCS of the M1. These results may provide evidence for the effectiveness of tDCS as a therapeutic instrument in treating headache disorders.
Keywords
Introduction
Transcranial direct current stimulation (tDCS) is a non-invasive method for selectively modulating cortical excitability in a polarity-dependent way. This method has recently received increased attention regarding potential therapeutic applications in the fields of neurology and psychiatry. It has been proposed that tDCS induces changes in electrical properties of the neuronal cellular membrane resulting in the hyperpolarization or depolarization of the stimulated area depending on the current direction chosen (1). tDCS has been shown to affect a range of motor (1,2), somatosensory (3,4), visual (5,6), affective and cognitive functions (7–9). Therapeutic effects have been demonstrated in clinical trials using tDCS for a variety of conditions characterized by alteration of cortical excitability including tinnitus, post-stroke motor deficits, fibromyalgia, depression, recognition memory in Alzheimer’s disease, epilepsy, Parkinson's disease and chronic back pain (4,10–20).
Recordings of pain-related evoked potentials (PREPs) and the nociceptive blink reflex (nBR) are well-established tools for evaluating the cranial (trigeminal PREP and nBR) and extracranial nociceptive systems (extracranial PREP) (21–23). Several techniques have been suggested for nociceptive stimulation of the skin (e.g. laser, heat). We favor nociceptive electrical stimulation by a custom-built concentric electrode, which is an affordable, non-invasive method easy to perform. By virtue of its concentric design and small anode-to-cathode distance, this electrode produces high current density at low current intensities. Thus depolarization is limited to the superficial layer of the skin mainly containing nociceptive A-δ fibers, but it does not reach the deeper layers, which contain predominantly A-β fibers (24). We demonstrated in previous experiments that both nBR and PREPs reflect trigeminal nociceptive processing quantitatively and that they are highly sensitive to changes in trigeminal nociception (23,25,26).
In this study, we have aimed to investigate modulatory effects of tDCS of the human primary motor cortex (M1) on trigeminal and extracranial nociceptive systems using electrophysiological recordings of PREPs and nBR. Analogously to studies reporting excitability changes in the M1 by tDCS (2,27), we hypothesized that cathodal tDCS inhibits, whereas anodal tDCS increases neuronal activity, and correspondingly decreases and increases pain perception within pain processing systems.
Methods
Subjects
Nineteen healthy volunteers participated in the study (mean age 29.5 ± 6.6 years; 8 women). None of them reported any history of headache or pain, nor took any medication regularly. Furthermore, they had no history or current signs or symptoms of cardiological, dermatological, neurological or psychiatric disorders. All subjects were interviewed by a neurologist prior to enrollment in this study. Informed written consent was obtained. The study was approved by local ethics committee of the Medical Faculty at the University of Duisburg-Essen.
Study design
A prospective cross-over design with three arms was used. Each participant was stimulated three times for 20 minutes (cathodal, anodal and sham) in a randomized sequence on different days to minimize carry-over effects (Figure 1). Randomization was performed using the order of recruitment of the subjects for the study. Stimulation here refers to the electrode’s position over the left M1. PREPs and nBR were recorded at four time points: baseline, 0, 20 and 50 minutes post-tDCS. We recorded PREPs until 50 minutes post-tDCS in order to detect the 60–90 minutes’ duration of previously described tDCS after-effects on motor cortex excitability (27–29). The time interval between cathodal, anodal and sham stimulation was at least one week to avoid interference effects. Study subjects and collaborators who analyzed data were blinded to the type of tDCS. The subjects were aware that they may experience three different stimulation conditions. The stimulation was done by two investigators, each of whom recorded PREPs for half of the subjects. Detection and pain threshold were determined before starting the experimental procedure.
Experimental protocol. The experimental protocol consists of 20 minutes of cathodal, anodal and sham transcranial direct current stimulation (tDCS) of the primary motor cortex (M1) following a randomized cross-over design. Before, immediately after, 20 and 50 minutes after tDCS pain-related evoked potentials (PREP), nociceptive blink reflex (nBR) and subjective pain perception on a verbal rating scale (VRS) were measured.
Experimental setting
Subjects were seated in a reclining chair with a mounted headrest throughout the experiments. The left motor cortex was identified by transcranial magnetic stimulation using the coil position leading to the largest motor-evoked potential of the right abductor digiti minimi muscle. The “stimulating” (cathodal, anodal or sham) electrode was placed at this position. The other, reference electrode was placed above the orbita on the contralateral side of the forehead. The electrodes were fixed by elastic bands.
Transcranial direct current stimulation
tDCS was delivered by a battery-driven constant current stimulator (NeuroConn GmbH, Ilmenau, Germany) with a maximum output of 5mA using a pair of surface rubber electrodes in a NaCl solution–soaked synthetic sponge with an extension of 4 × 4 cm over the M1 and 5 × 10 cm over the contralateral orbit. The use of non-metallic electrodes such as rubber electrodes avoids electrochemical polarization. The electrode for tDCS is large so that the stimulation encompassed a broad area of the motor cortex (upper limb and face). During tDCS, weak direct current (DC) penetrates the skull, entering the brain. Although there is a substantial current shunt within the scalp, enough residual current reaches the brain to modify the transmembrane neuronal potential (30) and thereby influence the level of excitability, thus modulating the firing rate of individual neurons. The direction of cortical excitability depends on current polarity, such that anodal polarization generally increases, whereas cathodal polarization reduces cortical excitability (2,27). We placed one electrode over the motor cortex to stimulate M1. The left motor cortex was identified by transcranial magnetic stimulation (see “Experimental setting”). The other electrode was placed above the right supraorbital area according to the international 10–20 system for EEG electrode placement (31). Stimulation electrodes were oriented parallel to the precentral sulcus and eyebrow, respectively. The type of stimulation (anodal or cathodal) refers to the electrode’s polarity above M1, whereas the two electrodes were placed in similar fashion for sham stimulation. The sham current stimulation was administered by fixing the electrodes at the same positions and switching on the current for less than five seconds at a current strength below 500 µA in order to cause a slightly itching or tingling sensation and simulate the real current stimulation. Thus the subjects felt the initial current sensation but received no current for the rest of the stimulation period. With this procedure, the subjects cannot distinguish between verum and placebo stimulation. Participants were blinded to the polarity of tDCS. The current was applied for 20 minutes at an intensity of 1.0 mA, according to current safety recommendations (32,33). The maximum current density was 62.5 µA/cm2 over M1 and 12 µA/cm2 at the reference electrode.
Electrophysiological settings
A custom-built planar concentric electrode (CE) consisting of a central metal cathode (D: 0.5 mm), an isolation insert (D: 5 mm) and an external anode ring (D: 6 mm) providing a stimulation area of 19.6 mm2 was used for nociceptive stimulation (25). Trigeminal stimulation took place contralateral to the tDCS in the first division of the trigeminal nerve with two electrodes were placed 10 mm above the supraorbital nerve. The individual pain threshold was identified by two ascending and descending stimulation sequences in 0.01 mA steps with successive increasing or decreasing current intensities. Stimulation with the CE using increasing intensities produced initially a sharp touching sensation, defined as the detection threshold, and later a pinprick or painful burning-like sensation defined as the pain threshold. Fifteen blocks of triple-pulse stimulation were applied (monopolar square wave, intensity: 1.5-fold of the individual pain threshold, duration: 0.5 ms, pulse interval: 5 ms, interstimulus interval: 12–18 seconds, pseudo-randomized).
nBR and PREPs were recorded simultaneously. PREPs were recorded with one subcutaneous needle electrode placed at Cz referring to linked earlobes according to the international 10–20 system. nBRs were recorded using bilateral Ag/AgCl surface electrodes placed infra-orbitally referring to the orbital rims. We used the recording parameters bandwidth 1 Hz to 1 kHz, sampling rate 2.5 kHz, sweep length 300 ms (1401plus, Signal, Cambridge Electronic Design, Cambridge, UK).
Extracranial PREPs were elicited following nociceptive stimulation of the second and third fingers of the hand contralateral to the tDCS side. The stimulation parameters were identical to those used for trigeminal stimulation.
An investigator blinded to the study design performed signal analysis. PREP negative peak (N) latencies and peak-to-peak amplitudes (PPAs) were analyzed as follows: the first sweep was rejected to avoid contamination by a startle response, and the remaining 14 sweeps were averaged. Analyzed were N2 latencies and PPAs. In the nBR analysis, onset latencies were identified for each sweep separately, and a mean 14-sweep block value was calculated. Areas under the curve (AUC) were calculated between 27 and 87 ms after rectifying and averaging the 14 sweeps of each block. The stimulation with the concentric electrode produces a polysynaptic R2 of the nBR mediated by wide–dynamic range neurons located bilaterally in the medulla. Mean onset latencies of the R2 response correspond to the Aδ-fiber nerve conduction (26).
In addition, we analyzed individual pain thresholds and subjective pain perception scores on a 0 to 10 verbal rating scale (VRS). Subjects were instructed to pay attention to PREP stimuli and asked to rate the perceived pain (from 0 = no pain to 10 = most intense pain imaginable). Mean values of stimulation blocks for each subject and subsequent mean values for each group were calculated. PC-based offline analysis was performed with custom-written software (Matlab 6.5, MathWorks, Natick, MA, USA).
Statistical analysis
Individual pain ratings (VRS), PREP PPAs and latencies, nBR onset latencies and AUC were individually averaged and entered into a multivariate repeated measures-analysis of variance (MANOVA) for both trigeminal and extracranial PREP separately (three different tDCS conditions [cathodal, anodal and sham] × 4 time points [baseline, immediately after tDCS, 20 minutes post-tDCS and 50 minutes post-tDCS]). Psychophysical or electrophysiological changes were only considered for further analysis if the time condition interaction was significant. In such cases, we used three ANOVAs for repetitive measurements, one per condition (factor time with four time points, namely at baseline, immediately after, 20 minutes post-tDCS and 50 minutes post-tDCS)—to investigate electrophysiological changes over time. Additionally, student t-tests (independent by group) were used to compare the changes in pain thresholds and stimulation intensities for the trigeminal and extracranial PREPs and nBR stimulations among the three tDCS conditions. Statistical analysis was performed using SPSS 16 (SPSS Inc., Chicago, IL, USA). The significance level was set at p < .05. Data are expressed as means ± standard error (SE).
Results
Subjects tolerated tDCS well. There were no adverse effects. At the initiation of cathodal, anodal or sham stimulation, all subjects felt a slight itching sensation at both electrodes. No motor symptoms were observed in association with any type of stimulation. Pain thresholds and stimulation intensities for the trigeminal and extracranial PREPs and nBR stimulations did not differ among the three tDCS conditions.
Trigeminal nociceptive system
Trigeminal PREPs and nBR were elicited using similar current intensities for the three tDCS conditions: 0.95 ± 0.14 mA during cathodal, 0.82 ± 0.1 mA for anodal and 0.72 ± 0.1 during sham stimulation.
Significant time × condition interactions in the trigeminal PREP amplitudes (degrees of freedom [df] = 6, F = 3.13, p < .05; Figure 2A, Table 1) were observed. Post-hoc ANOVAs for repeated measurements, and post-hoc t-tests showed that cathodal tDCS caused a significant decrease in trigeminal PPAs compared to baseline by 16.9 ± 5.8% (df = 3, F = 2.8, p < .05), and that anodal tDCS leads to a significant increase in the trigeminal PPAs compared to baseline by 30.8 ± 15.2 % (df = 3, F = 3.1, p < .05) and sham tDCS to insignificant (approximately 3%) deviations in the trigeminal PPAs compared to baseline (Table 1, Figure 2A).
Effects of transcranial direct current stimulation (tDCS) on pain-related evoked potentials (PREPs) and nociceptive blink reflex (nBR). Anodal tDCS results in increased trigeminal (A) and extracranial (C) peak-to-peak amplitudes (PPAs) compared to baseline (p < .05). Trigeminal (B) and extracranial N2 latencies (D) did not differ significantly between different stimulation paradigms (p > .8). Nociceptive blink reflex (nBR) area under the curve (AUC) (E) and onset latencies (F) were not modulated by tDCS (p > .4). Asterisks indicate significant changes in PREP PPAs 0, 20 or 50 minutes after cathodal or anodal tDCS compared to baseline. Circles and triangles represent means, bars represent standard error of the mean. *p < .05. Changes in trigeminal and extracranial pain-related evoked potentials and nociceptive blink reflex after tDCS tDCS = transcranial direct current stimulation. N = negative peak. NP = negative and positive peak. NRS = numerical rating scale. AUC = area under the curve. F = F-value. ANOVA = analysis of variance. df = degrees of freedom. Data are expressed as means ± standard error of the mean.
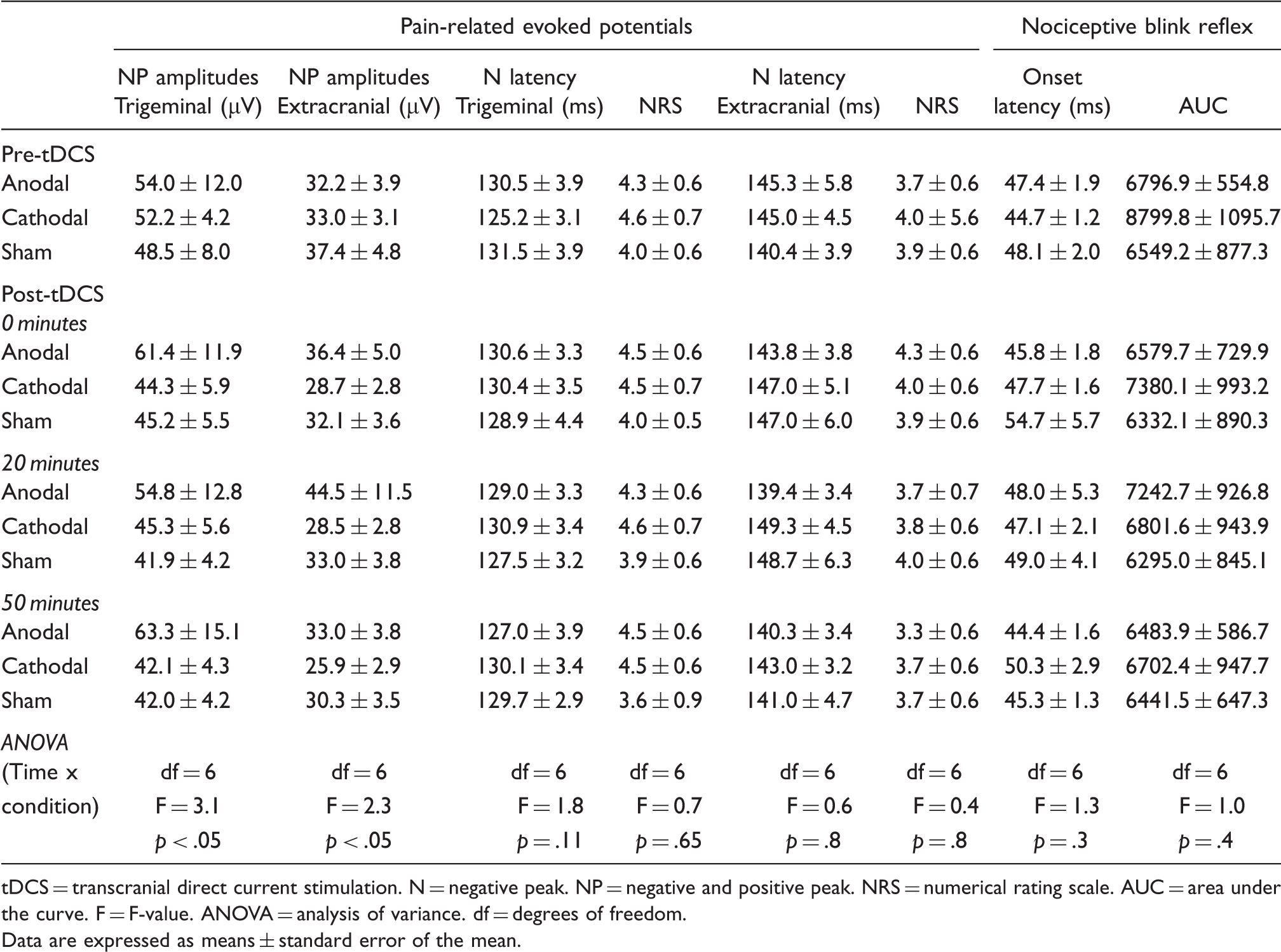
No significant time × condition interactions were apparent in trigeminal PREP N2 latencies (df = 6, F = 1.8, p = 0.11; Figure 2B, Table 1) and nBR onset latencies (df = 6, F = 1.3, p = .3; Figure 2F; Table1) and nBR AUC (df = 6, F = 1.0, p = 0.4; Figure 2E, Table 1). There were no significant time × condition interactions regarding individual pain ratings after tDCS (df = 6, F = 0.70; p = .65; Table 1).
Extracranial nociceptive system
Extracranial PREPs were elicited using stimulation intensities similar to those during cathodal (1.25 ± 0.19 mA), anodal (1.26 ± 0.20 mA) and sham (1.38 ± 0.22 mA) tDCS conditions.
Significant time × condition interactions were observed in the extracranial PREP PPAs (df = 6, F = 2.3, p < .05, Figure 2C, Table 1). Post-hoc analysis revealed that cathodal tDCS caused a significant decrease in extracranial PPAs compared to baseline by 18.7 ± 7.8% (df = 3, F = 2.5, p < .01), the anodal tDCS led to a significant increase in extracranial PPAs compared to baseline by 39.8% ± 16.6% (df = 3, F = 3.7, p < .05) and the sham tDCS to insignificant (approximately 10%) deviations compared to baseline (Table 1, Figure 2C).
No significant time × condition interactions appeared in extracranial PREP latencies (df = 6, F = 0.6, p = .8; Figure 2D, Table 1) and individual pain ratings after tDCS (df = 6, F = 0.4, p = .8; Table 1).
Discussion
Trigeminal and extracranial pain processing can be inhibited by cathodal tDCS and promoted by anodal tDCS. These results provide the physiological foundation for the effectiveness of tDCS in the treatment of pain and headache disorders.
We demonstrated that cathodal tDCS of M1 considerably reduced trigeminal and extracranial PREP PPAs by over 15%, while anodal tDCS of M1 increased trigeminal and extracranial PREP PPAs by over 30%. These changes are quite subtle, remaining below the individual pain perception threshold, as the pain perception ratings on VRS did not change accordingly. Nociceptive processing by tDCS remained unmodulated at the medullary-brainstem level, as we noted no changes in the R2 of nBR. Both the altered trigeminal and somatic PPAs of PREPs, together with the unmodulated nBR, suggested that tDCS modulates nociceptive processing of Aδ-fiber nociceptive input at a supraspinal level.
These results confirm previous investigations demonstrating cathodal tDCS to significantly reduce the N2, P2 and the N2–P2 amplitude of laser-evoked potentials (LEPs) (34–36). The decreased extracranial and trigeminal PPAs represent a general inhibition of pain processing that affects the trigeminal and extracranial nociceptive systems equally. LEPs more-pronounced pain inhibitory effect (reduced N2, P2 and N2–P2 LEP amplitudes) compared to merely reduced PREP PPAs after tDCS may be related to the fact that laser stimuli activate both C- and Aδ-fibers (37,38), whereas electrical stimuli with the concentric electrode activate predominantly Aδ- fibers (25). Moreover, the concentric electrode directly depolarizes cutaneous fibers by bypassing peripheral nociceptors, while laser stimuli activate the nociceptors primarily and may thus be influenced by an initial modulation at the receptor level (39).
The PPA of PREP has been shown to reflect human nociceptive processing quantitatively (21). In the same study we demonstrated that the effects of temporal and spatial summation caused an increase in PPAs of PREPs and subjective pain. No significant changes were observed after spatial and temporal summation in N latencies of somatic and trigeminal PREPs (21), supporting the possibility that the PPAs, not the N latencies of PREPs, may be an early and robust indicator of modulated pain processing in healthy human volunteers. Moreover, the amplitudes, not the latencies of evoked potentials in somatosensory evoked potential (SSEP) and visual evoked potential (VEP) tests, are changed by tDCS primarily (3,6,34,40). By contrast, in pathologic pain states such as trigeminal neuralgia, both the N latencies and PPAs are indicators of modulated pain processing due to damage to the nociceptive system (23). Thus it is not surprising that N latencies remained unchanged here in a non-pathologic pain condition. The strongest evidence for a specific response and against an incidential electrophysiological finding is the lack of response to sham stimulation. The fact that the cingulate cortex is the main generator of PREP (41) explains why the trigeminal and somatic PPA of PREP is an electrophysiological marker of cortical pain processing.
In conclusion, the changes in PPAs of PREPs in absence of other features (N2 latencies, pain perception) indicate an early and real difference in nociceptive processing, and do not reflect a non-specific electrophysiological phenomenon. Peripheral changes in conduction mechanisms are unlikely, as peripheral conduction of Aδ-fiber input is not modified by tDCS. Only the itching sensation at the beginning of tDCS application might be due to such peripheral nerve stimulation (33).
The effects of anodal tDCS on trigeminal and extracranial pain processing are in line with the results on motor cortex excitability measured by motor evoked potential (MEP) test (2,27). No study has thus far been able to demonstrate an acceleration in pain processing (reduced LEP latencies or increased LEP amplitudes) after anodal tDCS. Some studies provide indirect evidence of an increased pain perception threshold after laser stimulation of the hand contralateral to tDCS (7) or changed thermesthesia (35). Increased PPAs after anodal tDCS represents primarily the facilitation of central pain processing. Surprisingly, two randomized, sham-controlled clinical trials showed that anodal tDCS of M1 caused pain relief in spinal cord injury and fibromyalgic patients (12,16,42) indicating inhibition of pain processing. Those results are striking, as experimentally induced pain was significantly reduced by cathodal tDCS and not anodal tDCS in that case. This contradiction may involve potentially discordant tDCS effects between healthy people and patients. Future studies will have to clarify the different effects of tDCS on pain plasticity in the healthy and those with disease.
It is worth noting that we observed facilitation in both trigeminal and extracranial PREPs, indicating that facilitation of the nociceptive system is a general process not confined to the trigeminal system. A general nociceptive hypersensitivity has been demonstrated in many types of headache (i.e. those involving chronic and episodic tension-type headaches) (43,44). This was characterized by increased pain sensitivity in cephalic and extracephalic structures. Thus cathodal tDCS may prove beneficial in resolving headache disorders not involving the trigeminal nociceptive system alone in their pathophysiology. To the best of our knowledge, this is the first study to provide evidence that trigeminal pain processing can be modulated after tDCS, suggesting tDCS as a promising treatment option for headache disorders (45).
We used tDCS of the M1 because previous studies demonstrated an antinociceptive effect (34), and invasive stimulation of M1 has been used for years to alleviate chronic intractable pain. We are unable to state that the effect of tDCS is specific to the motor cortex as we did not use a control with stimulation in a different location. Indeed, functional MRI and MEP studies have revealed that although tDCS has the most activating effect on the underlying cortex (46,47), stimulation provokes sustained and widespread changes in many other subcortical and cortical brain regions (48).
Anodal tDCS modulates pain processing via the corticothalamic inhibition of epicritic and nociceptive sensation at thalamic nuclei (49). EEG studies have demonstrated that stimulation of a certain area (e.g. M1) induces changes in brain synchronization and topological functional organization (50). Moreover, the changes observed in pain processing occurred substantially in the anterior cingulate cortex, as it is probably the main generator of PREPs (41). The effect of tDCS on PREP was primarily mediated by M1 activity and secondarily by the somatosensory cortex, anterior cingulate cortex, thalamic nuclei and other pain-related structures modifiable via a network-driven effect (42,50) and interneuronal circuits (51).
The effects of tDCS are not specific to PREPs, as other evoked potentials, such as laser evoked potentials (LEP; 34), (SSEPs; 3,40), and (VEPs; 6,52) can be modified by tDCS. Cathodal tDCS is reported to diminish the N2 component of LEPs (34), resulting in reduced N20 median nerve SSEP up to 60 minutes after tDCS (3), producing no effects on SSEP (40) and decreasing the N100 amplitude and N70 amplitude of VEP (6,52). Anodal tDCS led to increased amplitudes of P25/N33, N33/P40 and P22/N30 amplitudes of SSEP following median nerve stimulation (40) and elevated N70 amplitude (6) and N100 amplitude of VEP (52). Thus, amplitudes of LEP, SSEP and VEP components are modifiable through tDCS such as PREP in a polarity-dependent manner.
At first glance, the lack of tDCS effect on subjective pain perception in our participants seems to contradict results from previous studies, that is, that anodal tDCS of S1 had no effect on subjective pain perception while cathodal tDCS of S1 significantly diminished pain perception (34); that cathodal tDCS reduced mild pain sensation, while anodal tDCS modified heat pain sensation (35); and that anodal M1 stimulation increased both the pain and perception thresholds (7). The small difference in tDCS duration (34) and the pain measurement scale (35) compared to our study are reasons for the non-modulatory effect of tDCS on subjective pain perception in our study.
tDCS induced an after-effect on pain processing that persisted over fifty minutes (53). Because after-effects of tDCS share similarities with long-term potentiation/long-term depression (LTP/LTD) and nociceptive LTP is believed to be a mechanism of central sensitization and pain memory (54,55), tDCS is a promising therapy, as are invasive motor cortex stimulation and repetitive transcranial magnetic stimulation (rTMS) (56,57) to modify pain perception. Although long-lasting after-effects of tDCS on the brain are thought to be mediated at the synaptic level by N-methyl-D-aspartate (NMDA) receptors (58–60), the mechanisms and time course underlying tDCS- induced changes remain unclear. The effect of anodal tDCS remained significant after three weeks of follow-up in patients (12), implying a form of LTP requiring de novo protein synthesis and the activation of transcriptional processes (61). Animal experiments have shown that tDCS can induce intracellular protein synthesis and alterations in cyclic adenosine monophosphate and calcium levels (62,63). The after-effects of cathodal tDCS in the human brain may arise through a non-synaptic mechanism involving alterations in transmembrane proteins and changes in pH (64). One potential mechanism of tDCS modulating cortical plasticity is a single nucleotide val66met brain-derived neurotrophic factor (BDNF) polymorphism (65). Studies have revealed the response to tDCS from met́ allele carriers to differ significantly from responses to val66val individuals. There is evidence that subjects with a val66met polymorphism showed a pronounced facilitation after anodal and a pronounced inhibition of motor-cortex excitability after cathodal tDCS (66). Thus the BDNF polymorphism shapes tDCS-induced cortical plasticity in humans.
We were unfortunately unable to examine the BDNF gene polymorphism in our participants; its influence on PREP modulation therefore remains unknown.
Although tDCS and rTMS revealed similarities in their effects, there are basic differences in their impact on neuronal function (1,56,67). Compared to rTMS, the application of tDCS is simpler, less expensive, and better tolerated by human patients and provides a far superior placebo stimulation condition (68).
Conclusions
In summary, we have demonstrated that tDCS is effective in the modulation of human trigeminal and extracranial pain processing. This modulation occurs via a network-driven effect at a supraspinal level. Our findings provide key basic insight into the physiological mechanisms underlying tDCS and support the implementation of tDCS in the treatment of different pain and headache disorders. Further clinical studies are necessary to prove the usefulness of tDCS in patients with primary headache and facial pain disorders.
