Abstract
Objective
Cerebrospinal fluid-venous fistula is an uncommon cause of spontaneous spinal cerebrospinal fluid leak (SSCSFL). We aim to describe the clinical presentation, imaging evaluation, treatment and outcome of SSCSFL secondary to cerebrospinal fluid-venous fistula.
Methods
A retrospective review was undertaken of SSCSFL cases secondary to cerebrospinal fluid-venous fistula confirmed radiologically or intraoperatively, seen at our institution from January 1994 to March 2019. Cases with undetermined SSCSFL etiology, alternative etiology or unconfirmed fistula were excluded.
Results
Forty-four of 156 patients met the inclusion criteria (31 women, 13 men). Mean age of symptom onset was 52.6 years (SD 8.7, range 33–71 years). Headache was the presenting symptom in almost all, typically daily (69%), and most often in occipital/suboccipital regions. Headache character was most commonly pressure (38%), followed by throbbing/pulsing (21.4%). Orthostatic headache worsening occurred in 69% and an even greater percentage of patients (88%) reported Valsalva-induced headache exacerbation or precipitation. Headache occurred in isolation to Valsalva maneuvers in 12%.
Of 37 patients with documented cerebrospinal fluid opening pressure, 13% were <6 cmH2O; 84%, 7–20 cmH2O; and one, 25 cmH2O. Fistulas were almost exclusively thoracic (95.5%). Only one patient responded definitively to epidural blood patch (EBP). Forty-two patients underwent surgery. Most improved following surgery; 48.7% were completely headache free and 26.8% had at least 50% improvement.
Conclusion
In our series, cerebrospinal fluid-venous fistula was associated with a greater occurrence of Valsalva-induced headache exacerbation or precipitation than orthostatic headache and did not respond to EBP. Surgery provided significant improvement. Cerebrospinal fluid-venous fistula should be considered early in the differential diagnosis of Valsalva-induced (“cough”) headache.
Keywords
Introduction
An orthostatic headache, worsening with upright posture and improved in recumbence, raises clinical suspicion for a state of low cerebrospinal fluid (CSF) pressure/volume, most commonly related to trauma to the spinal thecal sac. Trauma may be iatrogenic (such as from a lumbar puncture or intraoperative dural tear) or may occur spontaneously, sometimes referred to as “spontaneous intracranial hypotension (SIH)” or “CSF hypovolemia.” Patients with the latter, however, often have normal opening pressure and evidence suggests hypovolemia alone may not fully explain this condition, suggesting other mechanisms are likely at play beyond simple loss of CSF pressure or volume (1,2). For the purpose of this manuscript, therefore, we refer to this disorder of spontaneous loss of CSF pressure/volume secondary to spinal CSF leak as “spontaneous spinal cerebrospinal fluid leak (SSCSFL)”. Despite increased clinical awareness of SSCSFL and improved neuroimaging techniques such as CT myelography, the etiology of spinal leak is not identified in 46–55% of patients (3,4).
Recognition of cerebrospinal fluid-venous fistula (CVF) as an elusive etiology of SSCSFL has improved considerably since the first published case series of three patients by Schievink and colleagues in 2014 (5). With current knowledge of CVF based on a handful of case series in the literature (5–12), however, there is still limited understanding of the varied clinical presentation and surgical outcome for this unique population. In this study, we have reviewed our clinical experience with these patients. As we frequently see these patients first in our subspecialty Headache Clinic, we were particularly interested in the clinical presentation and headache phenotype described by patients with SSCSFL due to CVF.
Methods
This investigation is a retrospective case series of patients with SSCSFL secondary to CVF confirmed by neuroimaging and/or surgery who presented to our institution (Mayo Clinic in Rochester, Minnesota), a tertiary care medical center, between January 1994 and March 2019. Cases were identified with Advanced Text Explorer using the search term “CSF-venous fistula.” One hundred and fifty-six patients were identified. Patients with an undetermined etiology of CSF leak (88), an alternative etiology (15), a possible but unconfirmed fistula (6), and those who did not consent to research participation (3) were excluded. The Mayo Clinic Institutional Review Board deemed the study exempt.
We collected demographic data including age at symptom onset, symptom duration, prior headache history, headache characteristics, associated symptoms, CSF opening pressure, neuroimaging studies, epidural blood patches (EBP) and response, fistula diagnostic method, fistula location, time to surgery and outcome.
Results
Clinical presentation
Demographic and baseline clinical characteristics for 44 subjects with CSF-venous fistula.
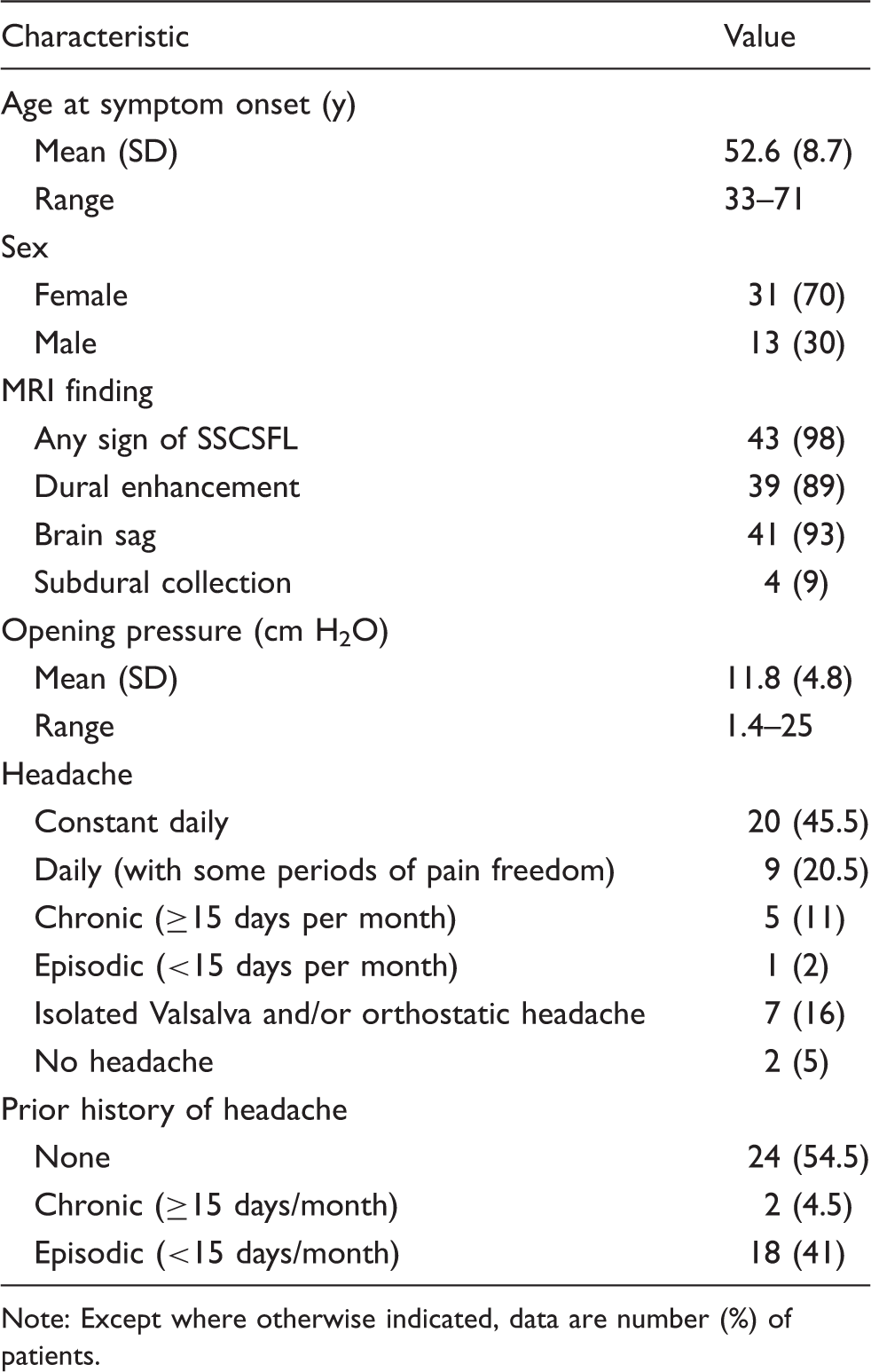
Note: Except where otherwise indicated, data are number (%) of patients.
Prior headache history
Of the 42 patients who presented with headache, 22 (52.4%) had no prior headache history. Of the 20 patients with a prior headache history, the majority (90%) had a history of episodic headaches (<15 days per month), including eight patients with migraine, seven with tension type-headache, one with transient headaches associated with meningitis, one with orthostatic headache following obstetric epidural anesthesia and one with undefined headache phenotype. One of the two patients with chronic headaches (≥15 days per month) had neuropathic pain and allodynia in the area of prior left frontal craniotomy for treatment for Langerhans cell histiocytosis also requiring radiation and chemotherapy. The other patient with a history of chronic headaches did not have the phenotype characterized.
Headache due to CVF
Of the 42 patients with headaches as part of their clinical presentation, 20 (45.5%) experienced constant daily headache, nine (20.5%) experienced daily headache with some periods of pain freedom, five (11.9%) experienced chronic but not daily headache (≥15 days/month), one (2.4%) experienced episodic headache (<15 days/month) and seven (16.7%) experienced isolated headache transiently exacerbated or precipitated by Valsalva and/or orthostatic changes.
The most common location of head pain was the occipital and suboccipital region in 15 patients (36.6%), followed by frontal in 10 patients (24.3%) and holocranial in six patients (14.6%). The most common description of pain was pressure in 16 patients (38%), followed by throbbing/pulsing in nine patients (21.4%), a combination of pressure and throbbing in three patients (7.1%), sharp in three patients (7.1%), “freezing” in one patient (2.3%) and aching in one patient (2.3%). A description of the headache was either not addressed or listed as “difficult to describe” in seven patients. Sixteen patients (38%) had associated migranious features including nausea/vomiting, photophobia or phonophobia.
The most common exacerbating or provoking factor was the Valsalva maneuver (e.g. coughing, sneezing, laughing, straining), associated with an abrupt transient headache exacerbation or provocation in 88% of patients. Interestingly, five patients (12%) experienced headaches only with the Valsalva maneuver and were otherwise headache free. Twenty-nine patients (69%) reported an orthostatic component to their pain that would worsen while upright and improve when recumbent, and two patients (4.8%) experienced headache precipitated by both Valsalva and orthostatic changes. One patient reported paradoxical worsening of her pain while lying down. Nine patients (21.4%) reported “second half of the day headaches” or head pain “worsening as the day goes on”.
Associated symptoms in CVF
Though headache was the most common complaint, all but four patients reported non-headache symptoms. The most common associated symptom was cognitive disturbance, often described as “mental fogginess” (45%). This was followed by tinnitus (32.5%), gait unsteadiness or imbalance (27.5%), alterations in hearing including muffled/reduced hearing, popping and abnormal sounds (27.5%), dizziness/vertigo (25%), movement disorders including tremor, choreiform movements, parkinsonism and ataxia (12.5%), alteration in vision including blurred vision, spots and diplopia (12.5%), and personality changes (12.5%) with one patient endorsing suicidal ideation. Less common associated symptoms included spells with altered awareness, numbness and paresthesias, fatigue, other body pain, sleep disturbance, choking, bruxism, anorexia and constipation.
Diagnostic evaluation
Thirty-five (79.5%) patients had a diagnosis of CSF leak or suspected CSF leak at the time of initial Mayo Clinic evaluation. Alternative initial diagnoses included “cough headache,” “Valsalva-induced headache,” “positional headache,” “chronic headache with Chiari malformation”, “transformed migraine”, “headache with migrainous components and mild descent of cerebellar tonsils”, “headache status-post radiotherapy,” and “chronic headache”.
Of 37 patients with documented CSF opening pressure, five had pressure <6 cm H2O (13%), 31 (84%) had pressure measuring 7–20 cmH2O, and one (2.7%) had a pressure of 25 cmH2O. Eight patients (18%) had a normal brain MRI scan at some point during the course of their illness prior to surgical treatment; one patient never had a documented abnormal brain MRI. At least one sign of SSCSFL was present in 97.8% of cases, though this was not necessarily present on initial imaging. Thirty-eight patients (86.4%) had undergone conventional CT myelogram without identification of fistula. Forty-one patients (93%) had undergone MRI of the cervical, thoracic and lumbar spine without identification of fistula. The average number of neuroimaging studies per patient completed from onset of symptoms to identification of fistula, including MRI brain, entire MRI spine, Nuclear Medicine (NM) cisternogram, conventional CT myelogram, positive pressure myelogram and digital subtraction myelogram, was 6.7 (SD 3.6, range 3–27). It should be noted that neuroimaging capability has expanded over the years included in our retrospective search, with digital subtraction myelogram not widely common in practice at our institution until 2018.
CSF-venous fistula was most commonly diagnosed by positive pressure myelogram in 21 patients (47.7%), followed by digital subtraction myelogram in nine patients (20%) and CT myelogram in three patients (6.8%). Eleven fistulas were surgically corrected following preoperative suspicion on neuroimaging, which included positive pressure myelogram in two cases, digital subtraction myelogram (Figure 1) in five cases and CT myelogram in three cases; one case was diagnosed intraoperatively. No fistulas were localized by classical MRI imaging of the spine, and this imaging modality is not felt to be sensitive to identification of CVF.
Fistulas were located between C7 and T12. All but two fistulas were located in the thoracic region, with the majority located in the lower half of the thoracic spine between T7–T12 (77%). No fistulas were identified in the lumbar spine. The vast majority were right-sided (76.7%), 13.9% were left-sided and 9.3% were possibly bilateral. One patient's fistula was confirmed intraoperatively at another institution, where laterality was not documented. Of those with suspected bilateral CVFs, three were suspected on positive pressure myelogram and one was suspected on digital subtraction myelogram. Three patients underwent bilateral CVF surgery. Only one of these patients (2.2%) responded completely to bilateral surgical correction of her CVFs. The remaining two patients with suspicious findings on positive pressure myelogram both had persistence of symptoms after surgery. Overall, although suspected on preoperative imaging, it was not possible to definitively confirm the presence of bilateral CVF in our series. The patient with suspected bilateral CVFs on digital subtraction myelogram did not elect to undergo surgery at our institution.
Response to epidural blood patch
Forty patients (90.1%) received an EBP; of those patients, 13 had addition of fibrin glue in at least one EBP. Five patients (12.5%) had documentation of no response to EBP. Eighteen patients (45%) had poor response (improvement lasting hours to weeks). Three patients (7.5%) had a modest response (1 month to less than 3 months). Four patients (10%) had good response that lasted for at least 3 months. Only one (2.5%) patient responded long term to EBP to the point that she required no further procedures or surgical intervention. Nine patients (22.5%) had variable responses to multiple blood patches that ranged from no response to greater than 3 months of improvement. There was no consistent difference between patients' response to blind or targeted EBP, or patches that included fibrin glue, with marked variability in responses even in individual patients.
Surgical correction of fistula and outcome
Forty-two of the 44 patients underwent surgical correction of their fistula. One patient had surgery offered but did not pursue this at our institution and was lost to follow-up. Another patient noted such a significant improvement in headaches after EBP that she did not feel surgical intervention was warranted. The average time from onset of symptoms to surgery was three years (SD 7.8, range 1–16 years). Surgical intervention included laminectomy, facetectomy and ligation of the nerve root and associated veins in all patients except for two patients with C7/T1 CVFs, in whom the nerve root was preserved. The average time to last follow-up after surgery was 5.6 months (SD 6.7, range 1–36 months). One patient is still pending follow-up from surgery; this patient did not have headache as a presenting symptom. Unlike a CSF leak through the dura and into the surrounding epidural fat, where a defect in the dura is visualized intraoperatively prior to repair, CVFs are not readily identifiable intraoperatively and a surgeon will more commonly visualize veins surrounding the root sleeve without obvious connection. Figure 2 is an intraoperative photograph of direct CVF visualization.
((a) and (b)) Digital subtraction myelogram: Digitally subtracted (a) and unsubtracted (b) images demonstrate linear irregularity (white arrow) going superiorly off a diverticulum (black arrow) with wispy loss of contrast typical of a CSF-venous fistula. ((c) and (d)) Post-contrast coronal T1 SPACE with fat saturation (c) performed at our institution at the time of initial evaluation demonstrates diffuse dural enhancement and thickening. Follow-up imaging (d) performed 3 months after surgery at the site of CSF-venous fistula shows resolution of the dural enhancement and thickening. (a) Intraoperative laminectomy and total facetectomy showing vein draped over the root. (b) Intraoperative laminectomy showing vein moved and a Pacchionian granulation (arachnoid villi under vein) connecting root sleeve to vein.

Forty-one of the patients who underwent surgical correction had headache as a presenting symptom. Of these 41 patients, 31 (75.6%) experienced at least a 50% improvement in overall symptoms; including 20 (48.7%) who achieved complete headache freedom and 11 (26.8%) who felt their headache improved by at least 50%. Of the patients who were completely headache free, the most common pre-operative headache phenotype was a constant daily headache provoked by both Valsalva and orthostatic changes (40%) followed by an isolated headache provoked only by Valsalva (15%). Eight patients (19.5%) reported no improvement in headache by their last follow-up.
Of patients who did not report improvement in headache by their last follow-up, three had transient improvement. One patient was completely headache free after surgery until a concussion later triggered exacerbation of migrainous headaches and persistent headache ever since. One patient had a 25% reduction in headache severity for 5 weeks, followed by a return to baseline. One patient continued to experience daily headaches that were less severe than before surgery but still provoked by Valsalva maneuvers; NM cisternogram suggested ongoing leak and the patient is still under clinical investigation. Attempted surgical correction of the fistula had been pursued in two patients based on high preoperative suspicion for fistula based on Digital Subtraction Myelogram (DSM) in one patient and Positive Pressure Myelogram in the other. Enlarged veins or dual-venous connections were not observed at surgery in these cases. Lack of response to surgery raises the question of whether a fistula was truly ever present at that level, although other reasons for surgical failure, including other potential sites of leak, are possible. Of the three remaining patients who were characterized as surgery non-responders, one patient had no change in headaches with continued abnormal NM cisternogram and is under ongoing clinical investigation. One patient experienced initial worsening of headaches before returning to baseline. Lastly, one patient had no change in symptomatology but has not undergone further work-up.
Thirty patients underwent post-operative MRI brain, 23 of these showed resolution of MRI findings. In the seven patients whose MRI findings had not resolved, five did not have clinical improvement in symptoms, one noted only a 50% improvement, and another reported resolution of headaches but continued cognitive disturbance.
Postoperative events occurred in 12 patients (28%) and were mostly mild. These included three patients (7%) with presumed rebound intracranial hypertension (headaches worse supine and improved when upright) lasting less than 2 weeks and one patient with continued rare, intermittent presumed high-pressure headaches. Four patients (9.5%) experienced numbness, paresthesias or burning involving various extremities. One patient who underwent surgical correction of left T6/7 CVF reported new mid-thoracic paraspinal pain. One patient reported transient urinary retention, postoperative delirium and thoracic neuropathic pain that eventually resolved. One patient reported hand weakness. One patient reported an episode of vasovagal syncope 2 weeks following surgery. With the exception of transient presumed rebound intracranial hypertension and rare thoracic back pain following surgery, other side effects following surgery could not be linked directly to the surgical procedure.
Discussion
Our results indicate that patients with SSCSFL secondary to CVF usually experience constant daily headaches but may also have episodic Valsalva- or posture-induced headaches, often in the absence of a prior headache history. Similar to prior descriptions of spontaneous spinal CSF leak (13), headaches in our patients tended to occur occipitally or suboccipitally and were commonly described as pressure-like or throbbing pain. Additionally, it was common for our patients to have associated non-headache symptoms, especially cognitive disturbance (often described as “mental fogginess”), tinnitus, gait imbalance, alterations in hearing or vision and personality change. Therefore, the presence of otherwise unexplained new daily occipital/suboccipital headaches accompanied by these associated symptoms should alert clinicians to the possibility of a spontaneous spinal CSF leak.
One of the most interesting aspects of this study is the finding that a large majority of patients (88%) noted cough/Valsalva maneuver could trigger or exacerbate their headache. Even this number may be an underestimate, as four of the five patients without report of Valsalva-induced headache did not have mention of the effect of this maneuver in the chart. In other words, the clinician may not have directly investigated or documented the effect of this maneuver in these patients. Only one patient was reported specifically to have no worsening with Valsalva. Furthermore, 12% of patients experienced headache only with Valsalva maneuvers, with two patients diagnosed initially with “primary cough headache”. Following the Valsalva maneuver, the next most common aggravating factor was orthostatic worsening (69%), as expected in the setting of an SSCSFL.
Orthostatic headaches or lingering headaches with postural aggravation in SSCSFL have been traditionally attributed to brain descent (“brain sag”) exerting traction on the pain-sensitive cerebral suspending structures while erect (1). As our patients reported exacerbation by Valsalva maneuver more commonly than by upright posture, there may be different headache mechanisms at play in SSCSFL secondary to CVF. Valsalva-induced headaches are well known to occur in congenital Chiari I malformations (CM1), and most of our patients had “brain sag” or brain descent (which may lead to “acquired Chiari”) secondary to CSF volume loss. It would therefore be attractive to think that at least in some of our cases, Valsalva-induced headaches could have occurred on the basis of mechanisms shared with CM1 Valsalva-induced headaches. As recently reviewed by Bezuidenhout and colleagues (14), a transient pressure dissociation between the head and spine may be an important underlying mechanism in CM1 Valsalva-induced headaches. During Valsalva, an initial spinal pressure elevation (from intra-thoracic pressure elevation and resultant epidural venous distension) displaces CSF to the head. Immediately after Valsalva in healthy subjects, the displaced CSF returns promptly to the spinal canal, but this does not happen in CM1 patients whose impacted cerebellar tonsils obstruct CSF flow at the foramen magnum. This creates pressure dissociation between head and spine, where intracranial pressure is at least temporarily higher than spinal pressure. The transient increase in intracranial pressure may cause headache by stimulating pressure-sensitive dural pain receptors. There may also be mechanistic overlap with the orthostatic headaches that have been reported in some patients after Chiari decompression surgery in the absence of CSF leak, which have been attributed to possible changes in dural compliance or to dural sensitization (15). Performing MRI in the upright position or with sequences that reveal CSF flow during Valsalva maneuvers may reveal other mechanisms of these headaches that, at present, seem to be possible in the absence of brain sag and dural tension.
Our retrospective review suggests there is a greater occurrence of Valsalva-induced headache exacerbation or precipitation than orthostatic headache in SSCSFL secondary to CVF; however, it is not yet known if this is more common in CVF compared with other etiologies of SSCSFL (e.g. dural tear, diverticulum). In the future, we hope to review our institutional experience, examining headache phenotypes across all etiologies of SSCSFL to determine if certain headache characteristics are more suggestive of specific SSCSFL types. It would be valuable to clinicians to know if there are key differentiating clinical features, such as exacerbation or precipitation of headache by Valsalva, an orthostatic headache component or a headache that worsens as the day progresses (i.e. “second half of the day” headache), that are associated with one etiology of SSCSFL versus another.
In our experience, CVF can be difficult to detect. Most of our patients underwent extensive diagnostic evaluation with multiple neuroimaging studies prior to identification of CVF. Similar to other authors' experience (5,6,8,11), fistulas often are not identified by a conventional CT myelogram. In our study, 79% of fistulas were not identified with this imaging modality. Consistent with prior studies, our results indicate that CSF opening pressure is most often normal in SSCSFL (2).
We identified one case of SSCSFL with a borderline elevated opening pressure of 25 cm H2O, which has also previously been reported (2).
Also similar to others' experience (6,8), SSCSFL secondary to CVF was generally refractory to EBP in our series. The vast majority of patients experienced variable responses that included no response or response less than 3 months, regardless of blind, targeted, multilevel or fibrin glue patching. Surgical intervention, in contrast, resulted in >50% headache reduction in the majority of patients. A recent prospective case series also found a high response rate to surgery, with most severe headache sufferers showing major improvement in severity and all subjects reporting the highest level of satisfaction with treatment (7). Most patients in our study who did not respond to surgical intervention had complicated cases with potential for re-leak or alternative site of leak.
Our investigation has several limitations. First, our practice at a tertiary care referral center introduces the potential for selection bias. Many of the patients were referred for suspicion for SSCSFL on neuroimaging, therefore our initial diagnosis and directed interventions may have been expedited by this preexisting knowledge. Because this was a retrospective review, we were also limited to the available documentation in the chart. One post-operative phenomenon that may be under-recognized in our review, for instance, is the temporary occurrence of postoperative headaches suspected to be associated with rebound intracranial hypertension, as this is thought to be transient and may not have been documented at the time of follow-up. In their recent prospective case series, Wang and colleagues found that 70% of subjects experienced transient postoperative headache consistent with symptoms of presumed rebound intracranial hypertension (7). Lastly, although fistulas were most commonly detected by PPM in our series, it should be noted that DSM has only recently been available at our institution (within the last year of our review). Even within this time period, our experience has been consistent with prior reports that the ability to detect fistulas is markedly improved with DSM (9,11).
Conclusions
This is the largest case series to date of SSCSFL secondary to CVF. This is a recently described and potentially treatable etiology of SSCSFL that may go unrecognized on commonly utilized imaging techniques, including conventional CT myelogram. Our results are helpful in guiding clinicians in cases of elusive etiologies of SSCSFL, where advanced neuroimaging should be considered. Our experience has revealed a higher occurrence of Valsalva exacerbation or precipitation of headache than orthostatic headache, a clinical phenotype which may be helpful for clinicians when encountering such difficult cases. In headache clinics, we recommend considering this condition when a patient presents with a new daily occipital/suboccipital headache, with marked worsening with cough/Valsalva maneuver, especially when combined with orthostatic worsening and associated non-headache symptoms (vision/hearing changes, gait imbalance, cognitive complaints). Because a significant number of patients present with isolated headaches precipitated by Valsalva maneuvers, CVF should also be considered early in the differential diagnosis of Valsalva-induced (“cough”) headache. In our experience, CVFs appear largely unresponsive to EBP, both blind and targeted. Advanced neuroimaging may be required, and surgical treatment likely provides the best clinical outcome, with limited complications.
Footnotes
Clinical implications
Results provide a better understanding of the typical clinical features of SSCSFL secondary to CSF-venous fistula.
Greater occurrence of Valsalva-induced headache exacerbation or precipitation than orthostatic headache; CVF should be considered early in the differential diagnosis of Valsalva-induced (“cough”) headache.
CVFs rarely respond to EBP, while surgical treatment generally provides significant improvement.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JRD reports no relevant disclosures. CER reports honoraria from UpToDate as author; advisory board for Amgen, Alder and Eli-Lilly. JKC-G reports honoraria from Oxford University Press for the textbook Mayo Clinic Medical Neurosciences. CMC and JLDA report no relevant disclosures. IG reports honoraria from UpToDate as author.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
