Abstract
Objective
To investigate suicidality related to cluster headache and factors associated with increased suicidality in cluster headache patients.
Methods
In this multicenter study, 193 cluster headache patients were recruited between September 2016 and August 2018. Patients were asked about their suicidality during and between attacks, specifically about passive suicidal ideation, active suicidal ideation, suicide plan, and suicide attempt. Univariable and multivariable logistic regression analyses were performed to evaluate the factors associated with high ictal suicidality (sum of positive response ≥ 2). Patients were followed up when they were in the between-bouts period.
Results
A total of 175 cluster headache patients in the in-bout period were included in this study. Passive suicidal ideation, active suicidal ideation, suicidal planning, and suicidal attempt were reported by 111 (64.2%), 62 (35.8%), 10 (5.8%), and four (2.3%) patients during attacks; seven (4.0%), six (3.5%), five (2.9%) and two (1.2%) patients interictally; and none (0%), one (1.9%), one (1.9%), and none (0%) among patients in the between-bouts period. Factors associated with high ictal suicidality were longer disease duration, the Headache Impact Test score, and the Patient Health Question-9 score (multivariable OR = 1.90 per 10-year increase in disease duration, 95% CI = 1.18–3.05, p = 0.008; multivariable OR = 3.19 per 10-point increase in HIT-6, 95% CI = 1.73–5.87, p < 0.001; multivariable OR = 2.11 per 10-point increase in PHQ-9, 95% CI = 1.13–3.95, p = 0.020, respectively).
Conclusions
Cluster headache attack carries a high suicidality compared to the interictal or between-bouts state. An intensive treatment to reduce cluster headache burden may be helpful to alleviate suicide risk in cluster headache patients.
Introduction
Cluster headache (CH) is characterized by strictly unilateral, severe headaches, which accompany cranial autonomic symptoms and typical recurrence patterns (1). CH is often considered as one of the most painful disorders (2). Functional impairments from the burden of CH seriously impact the patients’ lives, most of whom are in the most productive age for social and family life (3).
Due to the excruciating attacks and highly-frequent recurrence during the bout, CH has also been referred to as the “suicide headache” (4). However, a limited number of studies investigating suicidality in CH patients exist to date (5,6). One prospective study, using a web-based survey, examined the suicidality of CH patients (6). Based on this study, the rate of suicidal attempt was considered similar to that of general population (7). However, this study result has not been replicated by studies recruiting CH patients with a verified diagnosis. Furthermore, suicidality might change according to phases of CH (i.e. during attack, within-bout interictal, and between-bouts periods). In addition, questions regarding the factors influencing suicide risk are yet to be answered.
To comprehensively investigate the suicidality related to CH and the factors associated with increased suicidality, we conducted a prospective multicenter study of CH. Patients with CH within their active periods were assessed regarding their suicidality during and outside the CH attacks and were followed up during their between-bouts periods. Both the risk of suicidality during attacks and the clinical factors associated with active suicidality were analyzed.
Methods
Study design
This study was designed as a part of the Korean Cluster Headache Registry Study (KCHR), which is a prospective, longitudinal, multicenter study that enrolled consecutive patients with CH from 15 hospitals (13 university hospitals, including eight tertiary and five secondary referral hospitals; and two general hospitals) in Korea. The suicidality study was conceived as a major axis of the KCHR and implemented prior to the first patient recruitment.
The study protocol was reviewed and approved by the institutional review board (IRB) of each participating hospital and written informed consent was obtained from every patient prior to the recruitment. Patient recruitment was conducted following the IRB approval in each hospital, mostly between September 2016 and February 2017. Only patients who visited the hospital and were diagnosed with CH by investigators were recruited at the clinic for the KCHR study. The diagnosis of CH was performed by each investigator based on the criteria from the International Classification of Headache Disorders (ICHD), 3rd edition, beta version. Patients with episodic, chronic, and probable CHs were included in this study (8).
Details about the KCHR
The KCHR comprises several sub-studies, including (but not restricted to) clinical manifestations, quality of life, suicidality, treatment response, validation of ICHD diagnostic criteria, and longitudinal follow-up for prognosis. All the participants were seen and interviewed by investigators, who determined the type, onset, clinical course, characteristics of CH, and treatment response. Comorbid depression and anxiety were assessed using the Korean version of the Patient Health Questionnaire-9 (PHQ-9) and the General Anxiety Disorder-7 item scale (GAD-7) (9,10). Patients were followed up during their between-bouts periods.
Assessment of suicidality
Suicidality was assessed through the four structured questions according to three (i.e. ictal, interictal, and between-bout) clinical situations. For ictal suicidality assessment, we asked patients if they experienced a passive suicidal ideation (“Have you thought that it was better to die while you experienced CH attacks in the active period?”), active suicidal ideation (“Have you thought of killing yourself while you experienced CH attacks in the active period?”), suicidal planning (“Have you planned suicide while you experienced CH attacks in the active period?”), and suicidal attempt (“Have you attempted suicide while you experienced CH attacks in the active period?”). Interictal suicidality was assessed by using the same questions about those experiences when they were between attacks but within the active cluster period (“Have you… when you were between attacks but in the active period?). The ictal and interictal suicidality scores were defined as the sum of the positive responses to the four questions. The between-bout suicidality was followed up in patients who visited the clinic during their between-bouts periods using same questions: “Have you… during the remission period?”.
Validation of questionnaires
We validated the suicidality questionnaire with the Korean version of the Columbia suicide severity rating scale (C-SSRS) for screening during bouts in 26 CH patients (11,12). Comparing the sum of positive response from five questions regarding suicidal ideation (wish to be dead; non-specific active suicidal thoughts; active suicidal ideation with any methods without intent to act; active suicidal ideation with some intent to act, without specific plan; active suicidal ideation with specific plan and intent) and actual suicidal attempt, the ictal suicidality score showed a good agreement (intra-class correlation coefficient = 0.854, 95% CI = 0.673–0.934, p < 0.001).
Statistical analysis
Categorical data were presented as numbers (%), and continuous variables were summarized as means (SD). The intra-subject odds ratio for suicidality during the attack compared to the interictal and between-bouts periods was calculated using the generalized estimating equation (GEE). To identify whether the ictal suicidal scores predicted interictal suicidal risk, the receiver operating characteristic (ROC) curve was analyzed. The area under the curve (AUC) and the best cutoff score were identified using the Youden’s index. The cutoff score of the ictal suicidality score was used as the surrogate outcome variable of regression analysis, due to the occurrence rate of the hard outcome; that is, the interictal suicidal attempt, was low. A logistic regression analysis was performed to assess the factors associated with high suicidality. Clinical factors were tested as explanatory variables and were selected in the multivariable logistic regression analysis using the forward stepwise method. Interictal suicidality scores were analyzed using the same method.
All the statistical analysis was performed using the Stata 15.0 software (StataCorp 2017. Stata Statistical Software: Release 15; StataCorp LLC, College Station, TX, USA). A p-value < 0.05 was considered statistically significant.
Results
Patients
Demographics and characteristics of study participants.

BMI: body mass index; ICHD-3b: International Classification of Headache Disorders 3rd edition beta version; NRS: numeric rating scale; HIT-6: headache impact test-6; GAD7: general anxiety disorder-7; PHQ9: patient health questionnaire-9.
In the case of first-onset CH with active periods that last without 1 month of attack-free periods, but with an observation period of less than 1 year.
Thirty-six patients with first-onset bout and one who could not recall the season of the previous bout (which was 13 years before the current bout) were excluded.
Data are presented as means (SD) or numbers (%).
Ictal vs. interictal suicidality during active periods
Ictal passive and active suicidal ideation were reported in 111 (64.2%) and 62 (35.8%) patients, respectively. A total of 10 (5.8%) patients had a suicidal plan, whereas four (2.3%) patients attempted suicide during the CH attack. In contrast, only seven (4.0%) and six (3.5%) patients reported passive and active suicidal ideation, respectively, interictally. Additionally, interictal suicidal planning and attempt were reported by five (2.9%) and two (1.2%) patients, respectively (Figure 1). There was no difference in suicidality between patients with definite CH and those with probable CH (definite vs. probable CH: 64.0% vs. 65.2%, p = 0.910 for passive suicidal ideation; 36.7% vs. 30.4%, p = 0.562 for active suicidal ideation; 6.0% vs. 4.4%, p > 0.999 for suicidal planning; and 2.7% vs. 0%, p > 0.999 for suicidal attempt).
Ictal and interictal suicidality in patients with cluster headache in their in-bout periods.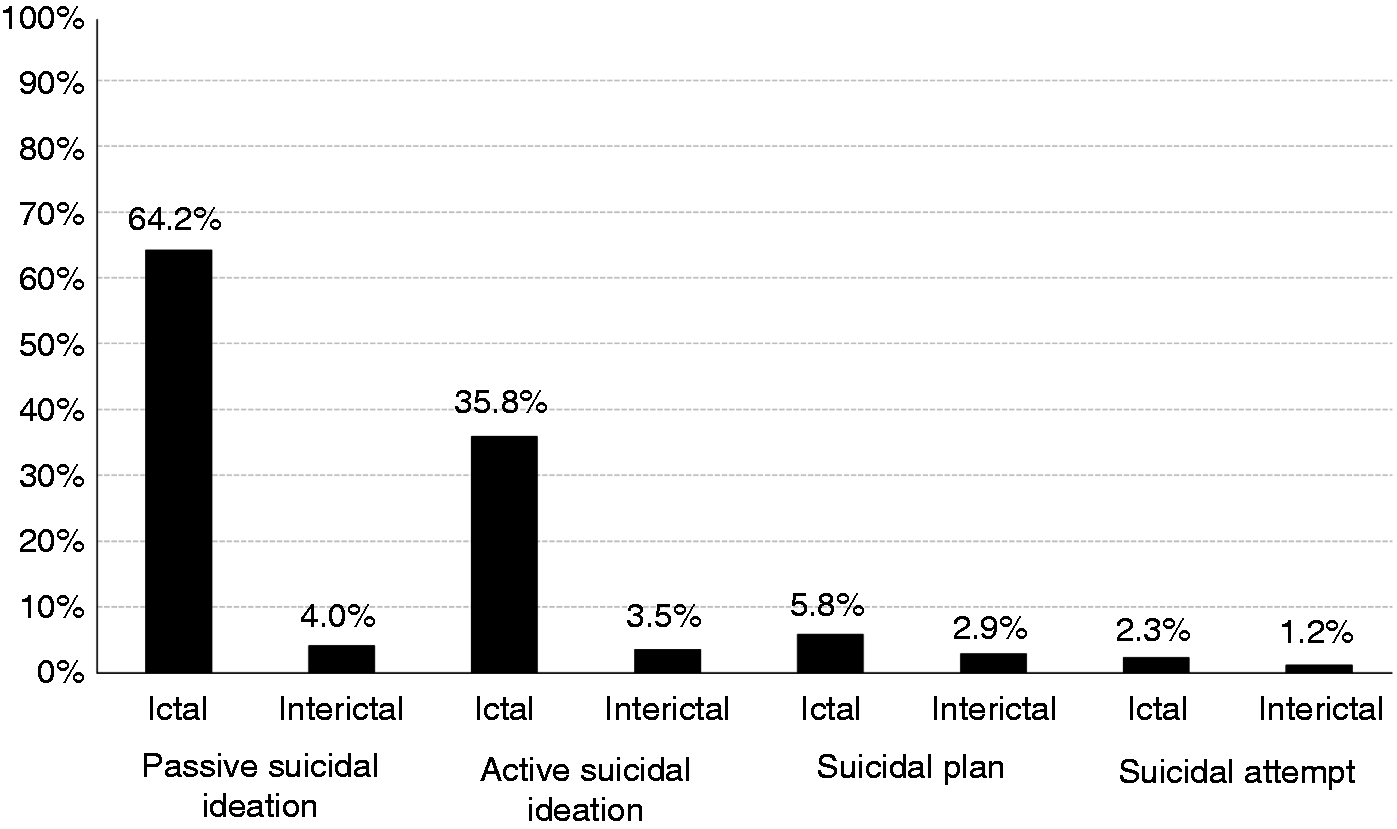
Cluster attacks increased the odds of passive suicidal ideation (OR = 42.46, 95% CI = 19.61–91.90, p < 0.001), active suicidal ideation (OR = 15.55, 95% CI = 7.02–34.41, p < 0.001), suicidal planning (OR = 2.06, 95% CI = 0.87–4.88, p = 0.100), and suicidal attempt (OR = 2.02, 95% CI = 0.5–8.26, p = 0.326) when compared to the interictal state, although the latter two were not statistically significant due to low event occurrence.
Ictal suicidality score and interictal suicidality
Using the ROC analysis, the ictal suicidality score was predictable of interictal passive suicidal ideation (AUC = 0.857, 95% CI = 0.769–0.945, p = 0.001), active suicidal ideation (AUC = 0.864, 95% CI = 0.769–0.959, p = 0.002), and suicidal planning (AUC = 0.875, 95% CI = 0.772–0.978, p = 0.004). Although not statistically significant, the ictal suicidality score yielded a high AUC to predict the interictal suicidal attempt (AUC = 0.895, 95% CI = 0.728–1.000, p = 0.055). In all four analyses, a cutoff of ictal suicidality score ≥ 2 provided the best discriminating accuracy (sensitivity and specificity: Respectively, 100% and 66.3% for interictal passive suicidal ideation; 100% and 65.9% for interictal active suicidal ideation; 100% and 65.5% for interictal suicidal planning; and 100% and 64.3% for interictal active suicidal ideation). None of the 62 patients without any (passive or active) ictal suicidal ideation planned or attempted suicide either ictally or interictally.
Factors associated with ictal suicidality
Factors associated with a high (≥2) ictal suicidality score.

CH: cluster headache, HIT-6: headache impact test-6; GAD7: general anxiety disorder-7; PHQ9: patient health questionnaire-9.
Factors associated with interictal suicidality
Factors associated with a high (≥2) interictal suicidality score.

No patients with chronic CH had interictal suicidal scores ≥ 2.
CH: cluster headache, HIT-6: headache impact test-6; GAD7: general anxiety disorder-7; PHQ9: patient health questionnaire-9.
In-bout vs. between-bouts suicidality
Of the 175 patients included in this study, 54 were followed up during their between-bouts periods (median 81 days [IQR 34–121] after the baseline visit). The in-bout (ictal and interictal) and between-bouts suicidality of these 54 patients are shown in Figure 2. Suicidality was overall very low outside the bout; specifically, none of the patients reported having passive suicidal ideation or suicidal attempt, whereas only one (1.9%) patient had both active suicidal ideation and suicidal planning.
Ictal, interictal, and between-bouts suicidality in the 54 patients who completed the follow-up evaluation in the between-bouts period.
When compared to the between-bouts period, the CH attacks were associated with significantly higher odds of active suicidal ideation (OR = 37.32, 95% CI = 3.22–433.01, p = 0.004) and increased suicidal planning, although not statistically significant (OR = 3.20, 95% CI = 0.62–16.42, p = 0.163). In contrast, given that passive suicidal ideation and suicidal attempt in the between-bouts period were not reported, the corresponding odds ratios could not be calculated.
Discussion
In this prospective multicenter study, we found a significant increase in passive and active suicidal ideation, suicidal planning, and suicidal attempt during attacks in patients with CH. Furthermore, increased ictal suicidality was suggested to predict interictal suicidality. Interictal suicidality was overall low, although still higher than between-bouts suicidality. In addition to depressive symptoms, longer disease duration and greater headache-related impact were associated with suicidality in CH patients, highlighting the importance of proper management of CH to reduce its burden.
The effect of CH on actual suicidal risk was believed to be low based on a previous study’s finding that the overall risk of suicidal attempt was not higher in CH patients when compared to the general population (6). However, our within-subject comparison demonstrated a significant increase in suicidality during attacks when compared to the interictal and between-bouts periods. In addition, both suicidal planning and attempt showed a twofold increase during attacks. Such an increase in ictal suicidality predicted interictal suicidality. Given that greater headache-related impact was independently associated with ictal suicidality, an intensive treatment to reduce the headache-related impact might be beneficial to prevent suicide in CH patients.
Our study showed the long-term disease burden (i.e. longer disease duration and number of lifetime bouts) was associated with increased suicidality. Specifically, longer disease duration and the number of lifetime bouts were associated with increased ictal and interictal suicidality, respectively. This association was not confounded by chronic CH, probably due to the small number of chronic CH patients in our data, similarly to other Asian studies (13–16). Rather, our data showed episodic CH with long-term disease burden might be as significant as chronic CH. In addition to reducing the short-term disease burden; that is, headache-related impact and headache intensity, a disease-modifying treatment to prevent further bouts is warranted to decrease suicidality in CH patients.
Our prospective evaluation showed that the between-bouts suicidality further decreased compared to interictal suicidality. Given that the lifetime prevalence of suicidal ideation, planning, and attempt was reported to be 15.2%, 3.3%, and 3.2% in Korean adults and 12.9%, 2.2%, and 2.5% in men, respectively, our CH patients had lower suicidality compared to the general population when they were in the between-bouts periods (17). As psychiatric disorders or chronic pain disorders are not considered as risk factors for CH, our study results suggest that patients remain mentally healthy when the bouts are effectively ended. As patients had relatively increased suicidality during bouts even when they were attack-free (interictally), a strategy to shorten the length of bout is warranted.
In our study, depressive symptoms measured by the PHQ-9 are associated with both ictal and interictal suicidality. Recently, a national claims data-based study showed an association between depression and CH (18). Given that depression is not considered as a risk factor for CH, the increased odds of depression could be a result, rather than a cause, of CH. More attention to depressive symptoms is warranted as they are associated with increased suicidality during and between attacks. For this reason, a further study on depressive symptoms in CH patients is currently ongoing in the KCHR.
Interestingly, suicidality did not differ between patients with definite versus probable CHs. This prompts clinicians for an increased identification and intensive treatment strategy for probable CH, as it carries a suicidality rate similar to that of definite CH. Therefore, the burden of probable CH should not be underestimated. In fact, probable CH might be either a milder (e.g. lower attack frequencies) or more severe (e.g. longer attack durations) end of CH spectrum. It should also be noticed that Asian patients may have more atypical features; that is, a lower mean number of attacks per day and less agitation/restlessness as an accompanying symptom, compared to study results from Europe, which may result in an increased diagnosis of probable CH (13–15,19–21).
The strengths of our study are as follows. To our knowledge, this is the first prospective study of detailed suicidality in different phases of CH. Although we collected data based on patients’ retrospective recall, we asked patients only about their active period when they were in active bouts, whereas suicidality of the recall period was followed up during the remission period. The latter allowed patients to focus on their recent remission period and to be less influenced by positive events (i.e. pain experience in CH), which affect the memory more powerfully than do negative events. In addition, an accurate diagnosis of CH was ensured by experienced neurologists who interviewed all the patients. Furthermore, a comprehensive evaluation was performed to fully elucidate our study questions. Our study also has limitations. First, the prevalence of chronic CH was low in our patients. Second, not all patients underwent follow-up visits in the between-bouts period. In fact, it was extremely hard to achieve an optimal follow-up rate in this investigator-initiated study, especially when patients were asymptomatic (i.e. in the between-bouts period). Improvements in the follow-up procedures were made for another sub-study on CH recurrence and its clinical course, for which the follow-up was conducted via telephone calls and e-mails. Third, although we used our best efforts to minimize recall bias and sampling error, as mentioned above, data obtained from recall can be less accurate than prospective recording. Fourth, the questions regarding suicidality were not quantitative: Frequencies of suicidal ideation, plan, or attempt were not taken into consideration. Lastly, we did not collect data on personality disorders, insomnia, or substance abuse or addiction, all of which can influence the suicidality in patients with chronic pain (22).
In conclusion, CH is associated with an increased suicidality during attacks and in within-bout periods. Furthermore, short- and long-term disease burden from CH as well as depressive symptoms contribute to the ictal and interictal suicidality in CH patients. Early diagnosis and effective treatment aimed at both reducing the headache-related burden and preventing further bouts are warranted.
Clinical implications
Cluster headache is associated with an increased suicidality during attacks and within the active cluster period. Increased suicidality during CH attack is associated with increased suicidality when patients do not have headaches. Short- and long-term disease burden from CH as well as depressive symptoms contribute to the ictal and interictal suicidality in CH patients.
Footnotes
Acknowledgements
We thank Ms Miran Jung and Ms Jinju Lee for aiding in data management. Sook-Young Woo helped with the statistical analyses used in this study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the Korean Neurological Association (KNA-16-MI-09).
