Abstract
Introduction: Data on clinical differences between episodic (eCH) and chronic cluster headache (cCH) and accompanying migraine features are limited.
Methods: History and clinical features of 209 consecutive cluster headache patients (144 eCH, 65 cCH; male:female ratio 3.4 : 1) were obtained in a tertiary headache centre by face-to-face interviews. Relationship between occurrence of accompanying symptoms, pain intensity, comorbid migraine, and circannual and circadian rhythmicity was analysed.
Results: 99.5% of patients reported a minimum of one ipsilateral cranial autonomic symptom (CAS); 80% showed at least three CAS. A seasonal rhythmicity was observed in both eCH and cCH. A comorbid headache disorder occurred in 25%. No significant difference was detected between patients with comorbid migraine and without regarding occurrence of phonophobia, photophobia or nausea during cluster attacks. Patients with comorbid migraine reported allodynia significantly (p = 0.022) more often during cluster attacks than patients without comorbid migraine.
Conclusion: Occurrence of CAS and attack frequency, as well as periodic patterns of attacks, are relatively uniform in eCH and cCH. Multiple CAS are not related to pain intensity. Allodynia during cluster attacks is a frequent symptom. The unexpectedly high rate of accompanying migrainous features during cluster attacks cannot be explained by comorbid migraine.
Introduction
Cluster headache (CH) is the most frequent trigeminal autonomic cephalalgia (TAC). TACs also include short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing (SUNCT), paroxysmal hemicrania, and probable trigeminal autonomic cephalalgia. TACs are characterized by stereotyped, unilateral pain attacks accompanied by various cranial autonomic symptoms (CAS). TACs are distinguished by the duration and frequency of headache attacks. Diagnostic criteria for CH have been established by the International Headache Society (IHS) (1). CH occurs with a lifetime prevalence of 124 per 100,000 and is more common in men than in women (2). In the majority of patients, the attacks appear in episodes for weeks or months separated by pain-free remission periods (episodic CH). In about 10–15% of patients, however, pain attacks occur without remission periods or with remissions of less than 1 month (chronic CH). Findings from neuroimaging studies, as well as the treatment response of deep brain stimulation (DBS) in patients refractory to medication, strongly suggest the involvement of the ipsilateral hypothalamus, particularly in the active headache period (3–5). Despite its characteristic features, diagnostic delay and misdiagnosis are common in CH, which might be related to reporting of uncommon accompanying symptoms, symptom duration, pain localization, or overlap with other headache diagnosis (i.e. migraine) (6,7). Furthermore, there are only limited data on whether clinical characteristics and circannual or circadian rhythmicity vary between eCH and cCH.
Frequently, cluster patients report uncommon localization of pain or different accompanying symptoms not mentioned in the IHCD-II criteria, which sometimes makes the diagnosis difficult. Therefore this study was performed to address the following items:
Frequency and lateralization of accompanying autonomous symptoms typically occurring in eCH and cCH attacks. Frequency of accompanying migrainous symptoms (phonophobia, photophobia, nausea, vomiting, allodynia). Association between comorbid migraine diagnosis and occurrence of accompanying migrainous symptoms in CH attacks. Differences in pain intensity and pain localization between eCH and cCH. Association between pain intensity and number and lateralization of reported autonomic symptoms. Comparison of circadian and circannual rhythmicity in eCH and cCH.
Methods
All patients diagnosed with CH were consecutively recruited between August 2009 and September 2010 from the Essen Headache Centre, a tertiary referral headache centre in Germany. Data were obtained with a standardized semi-structured questionnaire. The questionnaire, developed by the authors, consists of 44 questions in the following sections:
Demography (age, gender). Headache diagnoses: diagnosis of CH and concomitant headaches were made by an experienced neurologist according to the criteria established by ICHD-II (1). Only patients with primary CH were included; all patients underwent neurological examination and MRI to exclude secondary CH manifestation. Headache characteristics and course of disease (age of onset, length and frequency of cluster periods, average number of attacks per day, usual attack duration in minutes: untreated/treated, episodic or chronic pattern of CH, circannual and circadian rhythmicity, side and localization of headache, headache intensity on a 0–10 verbal numerical rating scale (NRS), behaviour during attacks, character of the headache) and accompanying CAS (conjunctival injection, lacrimation, nasal congestion, rhinorrhoe, eyelid oedema, forehead sweating, facial sweating, reddening of the face, miosis, ptosis). Information on trigeminal autonomic symptoms was obtained separately for the headache side and the contralateral side. Features typically associated with migraine (phonophobia, photophobia, nausea and vomiting), and allodynia. Side change of headache or accompanying symptoms.
We did not provide headache diaries to the patients supplementary as a study document. However, the headache centre in Essen provides very structured patient education and treatment, assisted by information on the web page, given by the headache nurses by telephone and some written material including headache diaries. The majority of the patients treated in the headache centre use headache diaries to document attack frequency. These are used to verify the information during interview if available. The study was approved by the local Ethics Committee of the University of Duisburg-Essen. Written consent was obtained from all included patients.
Statistical analysis
Descriptive statistics were used to report mean ± standard deviation (SD) as well as proportions. T-tests for an independent sample were used to compare means of different groups of the sample. Therefore continuous dependent variables were postulated. A chi-square test was used to study the relation between two categorical dependent variables. Correlation according to Pearson was used to study the relation between two continuous variables. We considered p ≤ 0.05 to be statistically significant. All analyses were performed using the Statistical Package for the Social Sciences (SPSS) for Windows software v18.
Results
Demographic and clinical characteristics
Demographic and clinical characteristics

eCH: episodic cluster headache; cCH: chronic cluster headache; SD: standard deviation.
not applicable in cCH.
There were no significant differences regarding age, age of onset, attacks per day or attack duration (not treated or treated), and smoking habits between male and female patients, and between eCH and cCH. Age of onset showed no significant difference between eCH and cCH (p = 0.094). Duration of cluster bouts and number of bouts per year were also not statistically different in males and females.
Accompanying autonomic and other symptoms
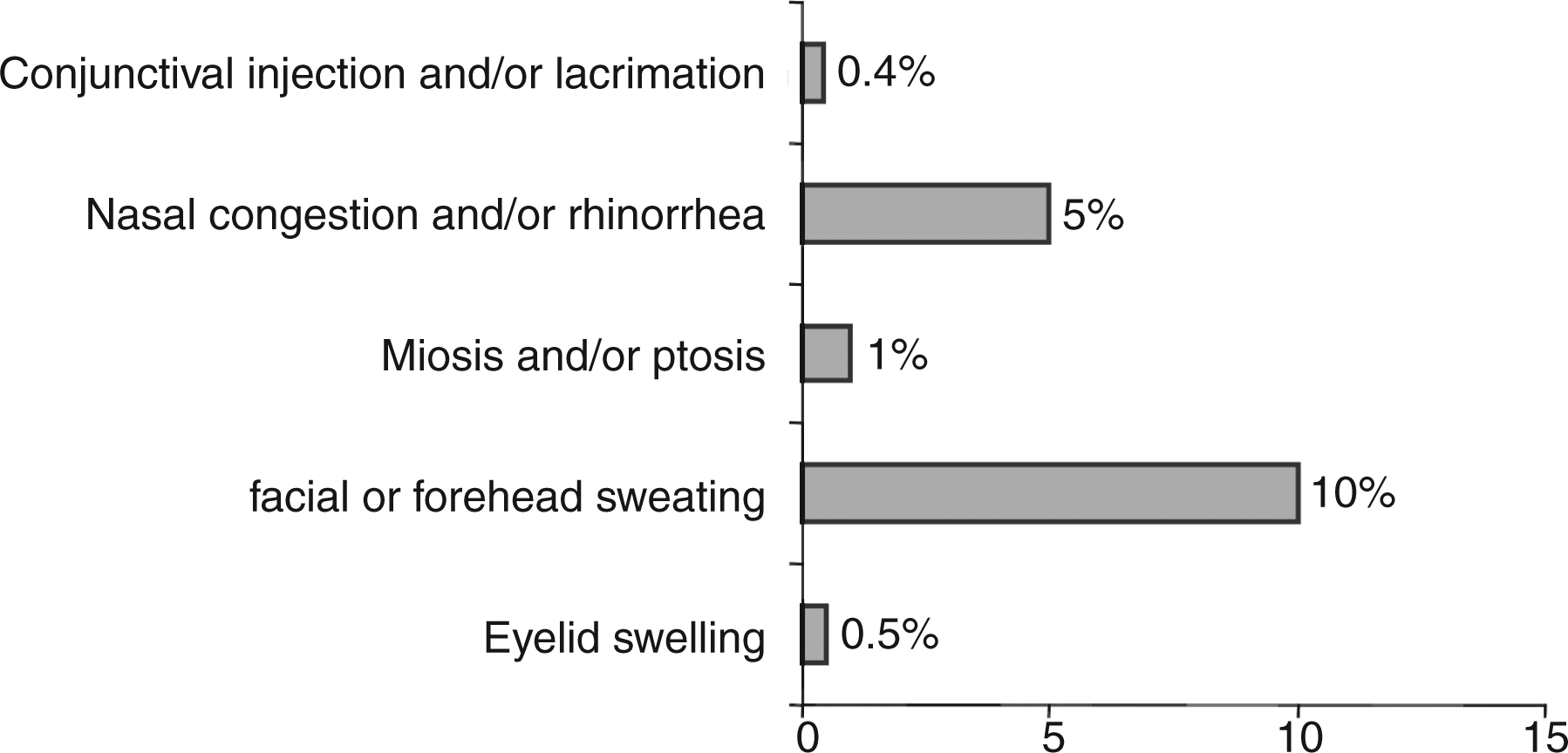
99.5% of patients reported a minimum of one ipsilateral CAS during cluster attacks (Figure 1). Patients with cCH reported eyelid swelling (p = 0.047) and miosis (p = 0.014) significantly more often than patients with eCH; subsuming both, the difference is not significant (p = 0.82). 80% of CH patients showed at least three CAS (Figure 2). CCH patients had at least three CAS significantly more often than patients with eCH (88% vs. 73%, p = 0.020). 93% of CAS were found to occur uni- and ipsilateral to the side of the headache. Facial or forehead sweating more often occurred bilaterally (Figure 3). No patient experienced CAS exclusively contralateral to the headache side. Bilateral CAS (n = 45) could not be explained by more intense pain when correlated with pain intensity measured with NRS (t(201) = 1.423, p = 0.156). In addition, higher pain intensity did not correlate with a higher number of ipsilateral CAS (r = −0.098, p = 0.165). It has to be considered that the overall pain intensity in CH attacks was high (mean NRS = 9, SD = 1). A higher number of attacks per day was not associated with the occurrence of bilateral CAS (total: t(197) = 1.429, p = 0.154, eCH: t(133) = 0.453, p = 0.652, cCH: t(62) = 1.532, p = 0.131) and the number of attacks per day did not correlate with the number of ipsilateral CAS (total: r = −0.105, p = 0.138, eCH: r = −0.148, p = 0.087, cCH: r = −0.063, p = 0.623).
Frequency of reported ipsilateral autonomic features in episodic and chronic cluster headache patients (% of patients, multiple answers were possible). eCH: episodic cluster headache; cCH: chronic cluster headache; *significant difference. Number of cranial autonomic symptoms in cluster headache. Bilateral cranial autonomic symptoms in cluster headache.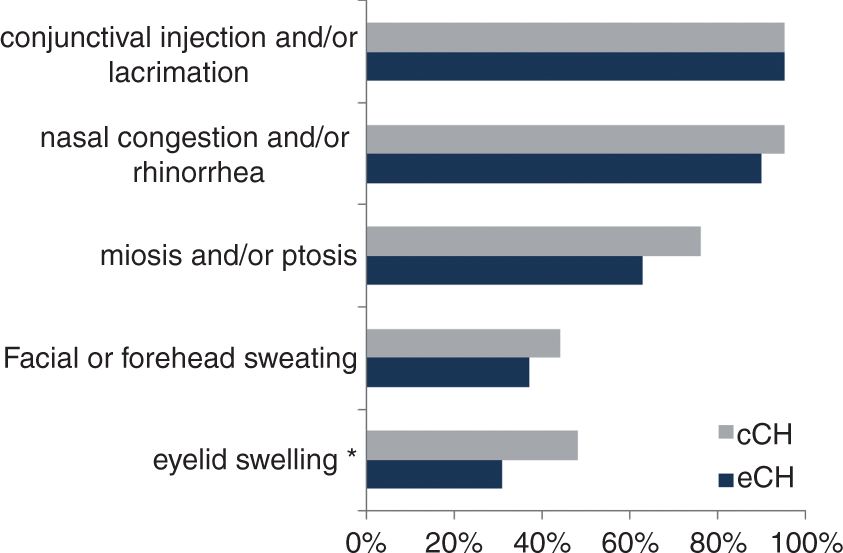

Restlessness was reported by 83% of all patients. Phonophobia or photophobia was reported by 73.2% of the patients. Patients with phonophobia more often reported allodynia than patients without (χ2(1,N = 209) = 3.775, p = 0.052). Vomiting or nausea was reported less frequently (Figure 4). Except for allodynia (67% vs. 41%; p = 0.02), no significant difference regarding occurrence of these accompanying symptoms was found between CH patients with and without comorbid migraine. No significant difference was found between eCH and cCH (p = 0.654) in occurrence of allodynia.
Allodynia, photophobia, phonophobia, vomiting and nausea in cluster headache with and without comorbid migraine (% of patients). CH: cluster headache; *significant difference.
Pain localization, side shift and pain characteristics
Headache attacks were most often localized periorbital including retro-ocular. The areas of maximal pain intensity marked by the patients are summarized in Figure 5. Thirty-three of 209 (15.8%) patients reported a change of side during the course of the disease. Details were available from 30 of those patients: 18 (8.6%) reported a side change for some attacks during a cluster bout or during the chronic course, 8 (3.8%) reported having one complete cluster bout contralateral to their usual dominant pain side and 4 patients (1.9%) reported side shift during an attack. Among 209 only one patient report side shift of autonomic symptoms and therefore dissociation between autonomic symptoms and pain. In all other patients with change of side autonomic symptoms shifted together.
Localization of maximal pain intensity (exemplary for the right side of face). Periorbital including retro-ocular: C1–4/D1–4/E1–4/F1–4; orofacial: C5–8/D5–8/E5–8/F5–8/G3–8/H3–8; maxilla: C5/D5/E5/F5; mandible: C7/D7/E7/F7/G7/H7; neck: A6–8/B6–8; occipital: A1–5/B1–5.
The most frequently reported CH pain character (Figure 6) was stabbing, followed by a ‘toothache-like’ pain description. There were no differences in pain characteristics between eCH and cCH.
Character of pain in episodic and chronic cluster headache (% of patients, multiple answers were possible). eCH: episodic cluster headache; cCH: chronic cluster headache.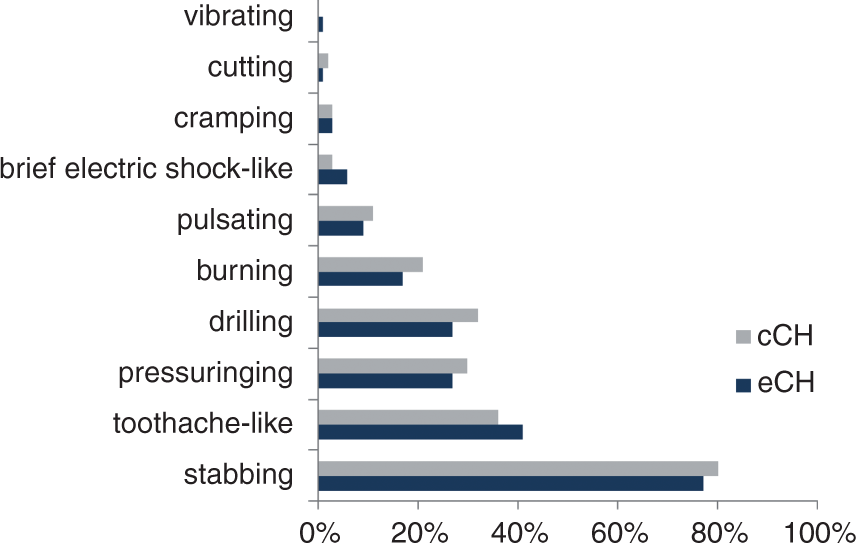
Additionally, neither a pressure-like pain character was significantly more often reported by patients with comorbid tension-type headache (p = 0.314), nor a pulsating pain character by patients with comorbid migraine (p = 0.124).
Comorbid headache diagnoses
Twenty-five per cent of CH patients reported at least one additional primary headache according to the ICHD-II classification (Figure 7).
Comorbid headache diagnoses in patients with cluster headache (comorbid medication overuse headache in one patient was related to diagnosis of migraine).
Circannual and circadian rhythmicity
A seasonal propensity was found both in eCH and cCH patients, with attacks occurring more often in spring and fall equinox (Figure 8).
Circannual rhythmicity of attacks in episodic and chronic cluster headache (% of patients, multiple answers were possible). eCH: episodic cluster headache; cCH: chronic cluster headache.
The majority of cluster attacks occurred between 13.00 and 18.00 hours in both eCH and cCH (Figure 9). Men showed cluster attacks between 19.00 and 24.00 hours significantly (p = 0.043) more often than women.
Circadian rhythmicity of attacks in episodic and chronic cluster headache (% of patients, multiple answers were possible). eCH: episodic cluster headache; cCH: chronic cluster headache; am: ante-meridian; pm: post-meridian.
Discussion
We present the clinical features of 209 consecutively recruited CH patients (144 episodic CH, 65 chronic CH) in a tertiary headache centre with special focus on autonomic and other accompanying symptoms. Our data are in line with previous data from other headache centres regarding mean age, male-to-female ratio, and clinical characteristics such as number and duration of attacks (8,9). The higher rate of cCH (31%; eCH:cCH ratio = 2.2 : 1) compared to 10–20% in other studies might be explained due to a selection bias; all patients in the presented study were recruited in a tertiary headache centre (8–10).
CAS were predominantly located ipsilateral to the headache side, which is in line with previous data by Lai and co-workers, who reported that 72–85% of their CH patients had uni- and ipsilateral CAS. Only facial or forehead sweating seems to occur more often bilaterally. Bilateral CAS are a common observation in migraineurs in 67–95% (11). However, patients with migraine mainly report one CAS, whereas CH patients seem more often to have multiple CAS. Ninety-six per cent of our patients reported more than one CAS, while 15% of patients with migraine reported more than one CAS (12). More distinct CAS could not be explained by increased activation of the autonomous nervous system through higher pain intensity or higher number of attacks in our study. The significant difference in number of CAS between eCH and cCH may be in part a result of a recall bias. Patients in a bout of eCH or ongoing cCH might quote a higher number of CAS compared to attack-free patients.
So far, CH has been regarded as an almost exclusively side-locked headache entity, typically without side shift of pain attacks. Within our cohort an unexpectedly high rate of side shift (15.8%) was reported. A side shift most often occurred between single attacks during a bout in eCH and cCH and very rarely during a single pain attack. The underlying pathomechanism of the side shift is unclear. Side shift may occur spontaneously as well as due to invasive treatment and has been reported due to unilateral occipital nerve stimulation (13).
Pain localization and pain characteristics were similar to previous studies (8,9). Periorbital pain localization was most frequently reported by more than three-quarters of the patients, followed by the occipital neck region and orofacial pain. Orofacial pain localization in particular, as well as the high rate of cluster patients reporting toothache-like pain, may lead to dental treatment resulting in unnecessary tooth extraction in some CH patients (14). Consulting a dentist prior to diagnosis has been reported by 45% of CH patients, resulting in 18% undergoing tooth extraction or maxillofacial surgery (6).
A high proportion of CH patients reported at least one accompanying symptom (phonophobia, photophobia, nausea or vomiting) typically described in patients with migraine attacks, but migrainous features were not more frequently reported by patients with CH who fulfilled the ICHD-II criteria of migraine.
Allodynia has been reported in up to 62% of migraine patients (15). In our patient cohort, allodynia was reported more frequently in CH patients with comorbid migraine. There was no difference between eCH and cCH in occurrence of allodynia, which confirms the data of Marmura et al. (16). Allodynia was detected with a testing for brush allodynia in 40–49% of CH patients (16,17). Allodynia might be a clinical feature of peripheral or central sensitization. Diagnosis of a comorbid migraine may, therefore, influence the clinical presentation of CH in this regard. In summary, accompanying migraine features are common in CH patients and may lead to misdiagnosis.
Cluster-tic syndrome was diagnosed in one patient of our cohort. Despite different pathophysiology of trigeminal neuralgia and CH, occurrence of both on the same side in one patient is possible (18).
Distinct circannual and circadian periodicity of cluster bouts have been described in several studies investigating patients with eCH. We found the same periodicity in the chronic course of the disease. Attacks in migraine with aura only peak once a year in May (19).
The high rate of current and former smokers in our patient cohort is in line with a former cohort of CH patients (9). Smoking habits were similar in eCH and cCH. In contrast, migraine patients are much less likely to smoke (41.8% non-smokers, 22.5% former smokers and 35.7% current smokers) (20).
The strength of our study is the large cohort of included CH patients who were thoroughly evaluated in face-to-face interviews to obtain valid and detailed information. Diagnosis of CH and other headaches was based on history and physical examination performed by an experienced neurologist using the ICHD-II classification. This might be an advantage compared to previous studies obtaining data by telephone interviews or the internet (8,9). However, there is of course a selection bias in our patient cohort, as all patients had been recruited in a tertiary headache centre.
In summary, occurrence of CAS, attack frequency, as well as the periodic pattern of cluster attacks, are relatively uniform in eCH and cCH, despite comorbid headache diagnosis. Occurrence of multiple CAS was not associated with higher pain intensity. Allodynia during headache attacks is a frequent symptom in CH, especially in patients with comorbid migraine. The unexpectedly high rate of accompanying migrainous features was not associated with comorbid migraine. A seasonal rhythmicity was significantly pronounced in eCH but occurs in cCH, too.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
The authors declare that there is no conflict of interest.
