Abstract
Despite being an excruciating headache, little is known about the burden of cluster headache (CH) regarding its various subtypes. In a multicentre, prospective study, patients with chronic CH (n = 27), with episodic CH in the active (n = 26) and outside the active period (n = 22), migraine patients (n = 24) and healthy controls (n = 31) were included. Epidemiological data, the German version of the Headache Disability Inventory (HDI) and a screening for psychiatric complaints were applied. About 25% of chronic CH patients in our study received invalidity allowance due to CH. HDI scores (total and subscales emotion and function) indicated a severe headache-specific disability (one-way ANOVA: P < 0.01). Patients with chronic and active episodic CH were significantly more affected than patients with inactive CH and migraine. Healthy volunteers were significantly less affected than all headache patients. Symptoms suggestive of psychiatric co-morbidity were found predominantly in chronic CH: depressive symptoms (56%), signs of agoraphobia (33%) and suicidal tendencies (25%) were frequently reported. Patients with chronic and active episodic CH were severely impaired in non-economic and economic domains such as disability, working life and psychiatric complaints. Remarkably, psychiatric co-morbidity was highest in chronic CH. Thus, especially chronic CH warrants special medical and further supportive care.
Keywords
Introduction
The consequences of pain and specifically headache extend beyond pure discomfort caused by pain itself. Earlier studies have shown that isolated pain scores alone do not fully reflect these consequences on the affected patients’ life (1). Resulting disability has been identified as a major factor of health-related quality of life. Holroyd and colleagues (2) suggested that the impact of chronic headaches could be best described by the factors ‘headache density’ (representing headache activity), ‘affective distress’ (psychiatric co-morbidity) and ‘disability’ which comprises social functioning and efficacy at work. In numerous studies, the impact of headache has been determined with tools estimating the general impairment of health-related quality of life (such as the SF-36 Health Survey, SF-36) or disability (such as the Pain Disability Index, PDI). The number of headache-specific tools is limited and comprises the Migraine Disability Assessment (MIDAS) (3), the Headache Impact Test (HIT-6) (4), the Migraine-specific quality of life questionnaire (MSQ2.1) (5) and the Headache Disability Inventory (HDI) (6). These isolated aspects do not fully reflect the profound and diverse implications of headache. As a result of the Eurolight project (7), a more comprehensive approach covering economic (direct and indirect costs) and non-economic consequences of headache (disability as measured by, for example, MIDAS, impact outside attacks, quality of life, family impact and psychiatric complaints) was suggested.
While disability has been widely examined in migraine, relatively little is known about quality of life, disability, and socio-economic consequences in patients with cluster headache and its episodic and chronic subtypes. In addition, previous studies have focused either on episodic or chronic cluster headache and often lack appropriate control groups (8–13).
Therefore, we aimed to determine the extent of disability in cluster headache patients and its subtypes (chronic cluster headache, episodic in the active period and episodic outside the active period) in comparison to migraine patients and healthy controls. Apart from basic epidemiological factors, we examined disability with a validated German version of the HDI and screened for concomitant psychiatric symptoms.
Patients and methods
Patients
Epidemiology and clinical details of patients included
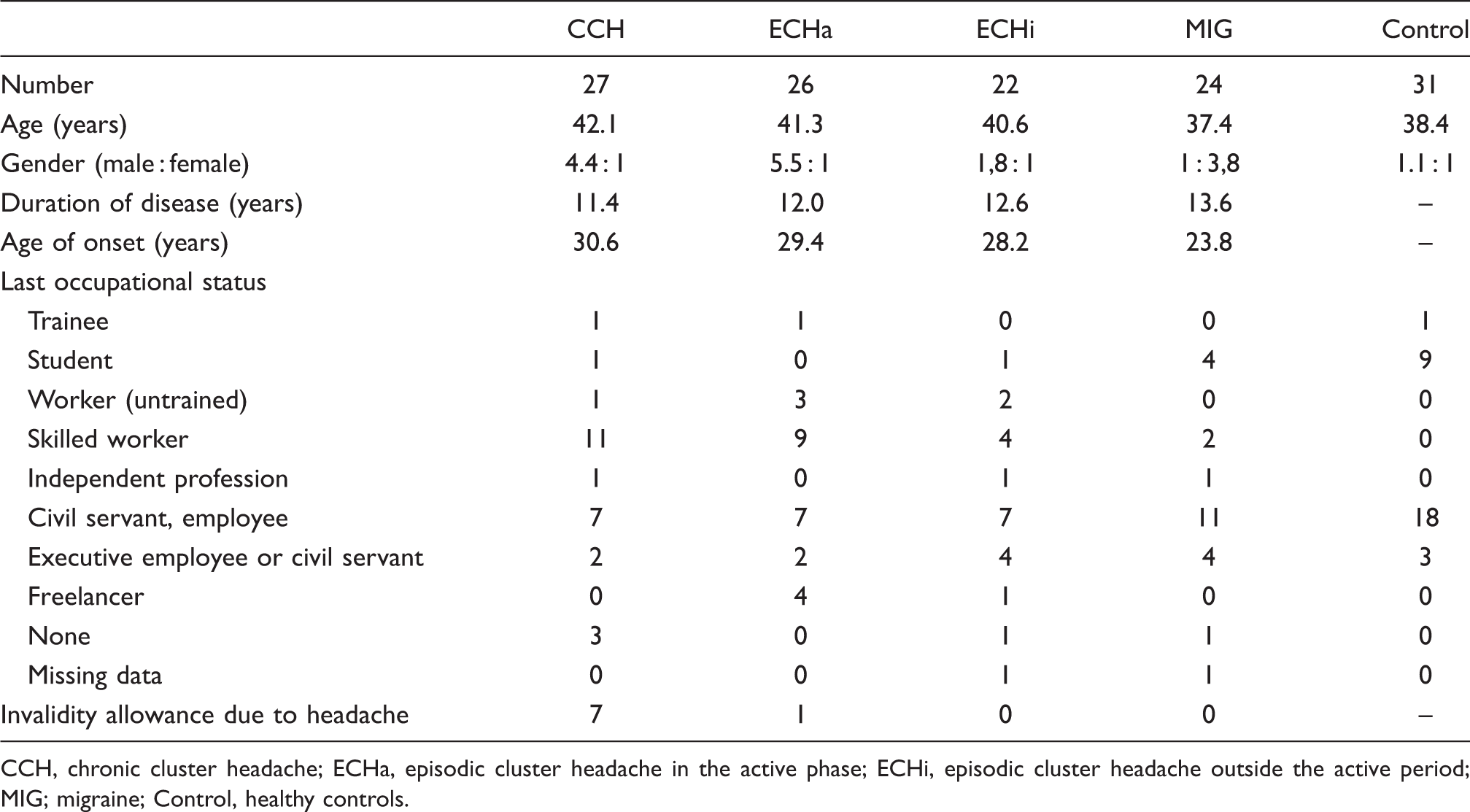
CCH, chronic cluster headache; ECHa, episodic cluster headache in the active phase; ECHi, episodic cluster headache outside the active period; MIG; migraine; Control, healthy controls.
Questionnaires
Demographic data including gender, age, current occupation and employment status and whether patients were incapacitated for work were collected using a custom-made questionnaire. In addition, basic data on headache characteristics were collected. All patients and the healthy volunteers completed the German version of the Henry Ford Hospital Headache Disability Inventory (HDI) (6,15). The HDI consists of 25 items (e.g. ‘Because of my headaches I feel handicapped’) that are answered on a rating scale with three response options (‘no’, ‘sometimes’, ‘yes’). Responses are coded 0, 2 and 4, respectively. Two sub scores can be calculated: EMOTION (13 items) measures the influence of the headache on mood and FUNCTION (12 items) measures restrictions concerning activities of daily living. A total score summing all items characterizes total impairment due to headaches.
Screening for life-time prevalence of psychiatric co-morbidity was done with elements from the Mini-DIPS, a validated structured clinical interview for the diagnosis of psychiatric disorders in the German language (16). The initial screening questions for the following disorders were presented to all participants: depressive syndrome, dysthymia, panic disorder, agoraphobia, social and specific phobia, generalized anxiety disorder (GAD) and obsessive-compulsive disorder (OCD). In addition, patients were asked whether they had thought about death or experienced suicidal tendencies.
Statistical analysis
Data were statistically analyzed with SPSS 16.0 for Windows (Chicago, IL, USA). To test for distribution differences of nominal data (i.e. gender, social, and educational status) chi squared statistics were calculated. Because sample sizes in particular cells were small with expected frequencies below 5, Fisher’s Exact probability test was applied and the Freeman–Halton extension was used whenever necessary (<http://faculty.vassar.edu/lowry/VassarStats.html>) (17). Continuous data were tested for Gaussian distribution by means of Kolmogorov–Smirnov statistics and entered into univariate ANOVAs. HDI data of the healthy volunteers were as expected not normally distributed and showed a reduced variance. However, results of the ANOVA with healthy volunteers excluded were fully comparable to the ANOVA with all groups. Therefore, only results of the latter ANOVA are presented. To disentangle significant main effects post hoc Student–Newman–Keuls (SNK) tests were applied. The SNK test extracts homogeneous subgroups that differ significantly from each other at a corrected P < 0.05, whereas groups within the same subgroup do not differ significantly. Where an a priori hypothesis was established, significant testing was one-tailed, otherwise two-tailed. Linear regression analyses were applied to explore the influence of demographic data (duration of headaches, onset of headaches, and number of attacks) on disability scores. Non-parametric tests (Mann–Whitney U-test) were applied within the CCH subgroup to test for the influence of disease activity as sample sizes differed substantially. Results with P-values < 0.05 were regarded as statistically significant.
Results
Patients’ characteristics
Table 1 summarizes data for epidemiological and educational status. As can be seen, the healthy volunteers differed in gender distribution from all the other headache groups (P < 0.01), except for the migraine patients (P > 0.10). Within the headache patients, there were no differences between the three cluster headache groups (P > 0.10), that all differed significantly from the MIG group. Gender in the healthy volunteers was equally distributed. All cluster headache groups consisted of more male than female patients. Whereas in the CCH and ECHa groups, this distribution difference in comparison to the healthy controls was significant (P < 0.01), it was only numerical in the ECHi group. In the migraine group, the majority of patients were females (P < 0.01).
Distribution of invalidity allowance was significantly different between the groups (P < 0.01) with the CCH group having the largest percentage of patients that were unable to work as a consequence of their headache (26%). This percentage was significantly higher than in the other groups (ECHa, 4%; ECHi and MIG, each 0%; P < 0.05).
All groups were comparable with respect to age (F (4,124) = 0.73; P = 0.57). For the four headache groups age of headache onset was comparable (F (3,92) = 1.96; P = 0.13), with MIG patients numerically displaying the earliest onset. Duration of headache was also comparable between the headache groups (F (3,92) = 0.32; P = 0.81).
In the MIG group, eight patients suffered from visual aura (33%), one from sensory aura (4%). In the CCH group 13 (54%) patients suffered from primary CCH, 11 (46%) from secondary CCH.
Headache specific disability (HDI) scores
Results of descriptive statistics for HDI scores are given in Table 2 and Figure 2. The ANOVAs (Table 2) revealed significant results for EMOTION and FUNCTION, as well as for the total score (F
(4,113) > 21.99; P < 0.01). Post-hoc SNK tests (Figure 2) on the three scales revealed three homogeneous groups that differed significantly from each other, with no differences within these groups. The first group comprised CCH and ECHa, the second ECHi and MIG and the third group the healthy volunteers. The first group was the most impaired one, the second showed significantly lower scores and the third group was the least impaired group.
Mean total scores of different patient groups in the Headache Disability Inventory (HDI). Whiskers represent standard error of the mean. CCH, chronic cluster headache; ECHa, episodic cluster headache in the active phase; ECHi, episodic cluster headache outside the active period; MIG; migraine; Control, healthy controls. Results of statistical analysis. Impairment measures on the German version of the Headache Disability Inventory (HDI): total score and scores of the subscales Emotion (13 items) and Function (12 items)

Regression analyses with the three HDI scales as dependent variables revealed a significant influence of first onset of headache on impairment values. The older the headache patients were at onset the higher was the reported impairment in the scales EMOTION (β = 0.22; P < 0.05; R2 = 0.05), FUNCTION (β = 0.22; P < 0.05; R2 = 0.05) and TOTAL (β = 0.23; P < 0.05; R2 = 0.06). Interestingly, further analyses revealed that these overall effects for EMOTION, FUNCTION and TOTAL were exclusively driven by the CCH and ECHa groups (all P < 0.05, all R2 > 0.18; only exception, for ECHa the influence of headache onset on FUNCTION was only marginally significant [P = 0.07, two-tailed testing]), but not by the ECHi and MIG group (all P > 0.15).
As the number of cluster headache attacks per day was not normally distributed across cluster headache patients, we calculated Spearman Rho rank correlations to investigate a possible influence on impairment. We found a significant positive association between number of attacks per day with the scales FUNCTION (ρ s = 0.20; P < 0.05) and TOTAL (ρs = 0.18; P < 0.05), thereby indicating a higher functional handicap especially with increasing number of daily attacks. For the MIG group, there was no significant relation between the number of monthly attacks and impairment scores. We did not find any correlations of age with number of attacks, neither for the cluster headache patients, nor for the MIG group.
In addition, we analyzed items measuring social impairment to get an impression on how cluster headache affects social activities. Two items of the German version of the HDI asked for such instances (item 7 ‘Because of my headaches I am less likely to socialize’; item 14 ‘My headaches place stress on my relationships with family and friends’). By means of the Chi squared test, we analyzed if the distribution of answer options (no versus partial or complete impairment) differed between the different headache groups (controls are not considered for always negating the items). For both items the test proved to be significant for (item 7 P < 0.01, item 14 P = 0.04). This was due to the increasing percentage of subjects supporting these items from migraine to chronic cluster headache patients (MIG 8%, ECHi 13%, ECHa 20%, and CCH 33%) as demonstrated in Figure 3. Thus, chronic headache seems to have an impact especially on socializing activities, whereas the impact on relationships with family and friends seems to be equal across the different headache groups
Within the CCH group, we exploratively investigated whether there was an influence of disease activity (ongoing attacks despite preventive medication). Mann–Whitney U-tests (CCH with ongoing attacks versus CCH without attacks) showed a significant effect on EMOTION (Z = –1.66; P < 0.05). Effects on TOTAL were statistically not significant but TOTAL scores increased numerically with increasing disease activity (Z = –1.40; P < 0.10). This suggests a somewhat higher handicap in patients that respond less to prophylactic pharmacotherapy (n = 17), compared with responders (n = 8, data not shown in Table 1).
Co-morbid psychiatric symptoms
Psychiatric symptoms

Symptoms were evaluated on the basis of the Mini-DIPS short structured interview. Total numbers of affected subjects in each group are given (percentage in parentheses). CCH, chronic cluster headache; ECHa, episodic cluster headache in the active phase; ECHi, episodic cluster headache outside the active period; MIG; migraine; CCH, chronic cluster headache; Control, healthy controls.
Discussion
To our knowledge, this is the first study in which patients with CCH and active and inactive ECH were compared to patients with migraine and healthy volunteers regarding the functional impact of CH and disability. In summary, we provide data that support the assumption of an increased functional impairment in cluster headache patients compared with migraine patients that is especially evident for the chronic cluster headache patients and patients in the active episode.
Patients’ characteristics
The gender distribution in the headache patients was comparable to recent reports. The gender ratio (male:female) of cluster headache patients has changed from 10 : 1 in early studies from the 1950s to 3 : 1 in current studies (18,19), while in migraine, a female preponderance is a well-established finding reaching up to 1 : 5 (20). Age of onset of ECH patients (28 years) was similar to that found in a large prospective study (19), while age of onset in the CCH group in our study was slightly lower (31 years) than in previous studies (37 years) (12,19). Another study reports onsets of 32 years and 37 years for ECH and CCH patients and a longer duration of disease (17 years for ECH, 12 years for CCH patients) (12). Taken together, demographic data of our headache patients were similar to data reported in other representative studies.
Non-economic burden
Disability
In our study, CCH and ECHa patients were more incapacitated than inactive ECH or migraine patients. Differences between CCH and ECHa patients were only minute and not statistically significant. This may be surprising as CCH patients suffer from attacks continuously. During the inactive period, attacks cease in ECH patients with significantly lower disability. No longitudinal studies with repeated evaluation of impairment over longer periods have been conducted, which would undoubtedly reveal a higher ‘cumulative’ impairment in CCH than in ECHa patients while currently disability at a given time is the same.
Findings of other studies addressing quality of life (QoL) and disability in cluster headache


CH, cluster headache; CCH, chronic cluster headache; Dis, Disability; ECH, episodic cluster headache; QoL, Quality of life; TTH, tension-type headache; NDPH, new daily persistent headache.
At least 3 months after the active period.
As headache specific QoL tool, the MSQ2.1 contains 14 items and has been validated to detect migraine-specific impairment in 3 dimensions: performance of normal daily activities, prevention of daily activities and emotional impact (frustration and helplessness).
Subtype of CH not specified.
No formal control group, but previously published normative data of 1636 Italian individuals were used.
Interestingly, in our study, CCH patients with little or no response to prophylactic medication had significantly increased disability scores in the mood domain of the HDI. This corresponds to findings from patients with intractable chronic migraine who received occipital nerve stimulation (21): all 25 patients showed severe disability on the MIDAS. Thus, refractoriness of headache should be regarded as a risk factor for high disability. Our findings are in agreement with the recent definition of refractoriness in migraine and chronic migraine patients, which defines disability in terms of MIDAS scores over 11 as ‘modifier’ (22). In contrast, in the Italian study, SF-36 scores did not change significantly between patients on sumatriptan and those without it (8). However, intake of sumatriptan and refractoriness are not necessarily identical. The SF-36 questionnaire is a quality of life (QoL) instrument, while the HDI detects disability which could also explain the discordant results.
The number of daily attacks correlated significantly with TOTAL and FUNCTION scores in the HDI, which suggests a higher functional handicap in severely affected patients. These findings suggest that disability seems to increase with the extent of clinical severity in CH patients. In addition, older age of onset correlated with higher impairment in all scores. We can only speculate that younger patients adapt more easily as they are in a period of fundamental changes anyway (i.e. in their twenties). Once, career and family planning have been settled, impact of CH with reduced work efficiency may be experienced as much more incapacitating.
Methodologically, both HIT-64 and MIDAS (3,23) have the advantage of being shorter than the HDI (only 6 and 5 items, respectively, instead of 25) and allowing the grading of severity into four categories. They refer to the last 4 (respectively 12) weeks. As a major disadvantage, both tests were not validated for the use in patients with cluster headache. If a combination of HDI with either MIDAS or HIT-6 is superior valid to a combination of both HIT-6 and MIDAS (24) remains to be elucidated in future studies.
Impact outside attacks
In our cohort, the headache–specific impairment measured by all HDI scores was surprisingly high in the inactive ECH patients showing high disability despite the absence of pain. Statistical analyses revealed no significant differences to the migraine group (Figure 2) while scores of active ECH and CCH patients were significantly higher. In contrast, Ertsey and colleagues (11) reported lowered MSQ2.1 scores during active periods in ECH indicating decreased quality of life. Outside the bout, values normalized to a level comparable to that of healthy volunteers. These differences could partly be explained by the use of different questionnaires. In particular, it is not specified in the German version of the HDI whether the questions address current pain or pain recently experienced in an active period. Use of tests referring to a limited period of time, such as the MIDAS (last 3 months), could have yielded different results. However, similar to our headache-specific findings, in ECHi patients, general QoL was reduced to levels comparable to that of migraine patients in most scores of the SF-36 in the sample by Ertsey and co-workers (11). The observation of Jensen and co-workers that 13% of the CH patients suffered from impairment of their daily life outside of active periods further corroborates our findings that disability was still substantial outside the active (and thus painful) period. We can only speculate that, even in the absence of headache, repercussions of the experienced headaches and the fear of new attacks may be present in the patients. A deranged social and emotional functioning generated within active periods could persist beyond the end of a bout.
General and headache-specific quality of life
First studies date back to Solomon and colleagues (13), who could show a higher reduction on QoL in a group of CH patients than in migraine. However, the sample was small (n = 13) and a control group was missing. In a larger study on chronic and ECH patients in the active period, a massively reduced QoL in various domains could be corroborated (8), although the study was limited by the use of normative data from a large population-based study instead of a specific control group. The same group examined SF-36 and MIDAS scores in 22 CCH patients and 150 patients with chronic migraine and medication overuse. Both were severely affected according to SF-36 and MIDAS scores with the chronic migraine patients even stronger (9), although differences between the two groups were not ascertained statistically. Again, normative data were used instead of an own control group. Ertsey and co-workers could show that general QoL in ECH patients is significantly impaired during the active period compared to a control group (except for the domains physical functioning and role emotional functioning of the SF-36). Differences between ECHa and migraine patients reached statistical significance only in two of eight domains (bodily pain and social functioning), although numerically the active ECH was more severely affected. Likewise, headache specific QoL measured by MSQ2.1 scores was significantly lower in the active ECH compared to the control group (the same held true for the migraine group compared to the control group) (11). This supports our findings that disability was higher in active ECH patients than migraine patients and controls. However, differences between ECH and migraine patients were only numerically higher in the study by Ertsey and colleagues. This could be explained by the large variability in the clinical presentations of migraine, especially as the number of attacks was relatively high in the Hungarian study (1.5 attacks per week). In addition, different modalities of impairment (QoL versus disability) were tested. According to the WHO, disability ‘is a complex phenomenon reflecting an interaction between features of a person’s body and features of the society in which he or she lives’ (25). QoL addresses the patient’s ability to succeed in the ordinary tasks of daily living and covers physical, emotional and social aspects. Although partly overlapping, both modalities emphasize different aspects of headache burden. It is nevertheless noteworthy that both the Hungarian and the Italian study found impairment in similar domains of the SF-36 in active ECH or CCH patients. In addition, the extent of impairment in the Hungarian study exceeded that of patients with preceding myocardial infarction (11).
Family impact
Data on the impact of CH on familial life are scarce. Although CH still shows a male preponderance the effects in women are severe, especially in those with onset of attacks before having entered reproductive life. They had fewer children than women who developed CH after their first child or onset of menopause (26). In another study, one-third of the female CH patients who intentionally did not have any children reported that their decision could be directly contributed to CH (27).
D’Amico (9) reported that 59% of the 22 CCH patients had lost family, social and leisure activities due to CH and had lost (28%) or reduced (32%) household work by at least 50% or more. Jensen found that only 27% of 85 patients with CH had no impairment of social activities and some 61% an impact on their family life. Eleven percent reported a changed family pattern due to CH (12). Likewise, we could show that mean scores for item 14 from the HDI (‘My headaches place stress on my relationships with family and friends’) are increased in all headache subgroups with highest impairment in the CCH and ECHa group. Similarly, item 7 of the HDI (‘Because of my headaches I am less likely to socialize’) reaches highest scores among CCH and ECHa patients. For both items, the percentage of subjects with partial or complete consent was highest in the CCH and ECHa group reaching values well above 50% (Figure 3).
Social impairment: Item 7 (grey bars) and item 14 (black bars) from the Headache Disability Inventory (HDI). Percentage of partial to complete impairment is given for item 7 (“Because of my headaches I am less likely to socialize”) and item 14 (“My headaches place stress on my relationships with family and friends”). CCH, chronic cluster headache; ECHa, episodic cluster headache in the active phase; ECHi, episodic cluster headache outside the active period; MIG; migraine; pCCH, primary chronic cluster headache; sCCH, secondary cluster headache; Control, healthy controls.
Psychiatric complaints
Psychiatric co-morbidity is well known in headache patients, first and foremost in migraine. In addition to a high proportion of concomitant diseases like generalized anxiety disorder, panic disorder and obsessive compulsive disorder a bilateral connection between depression and migraine headache has been well-established (28).
In CH, knowledge of co-morbid psychiatric disease is limited. Personality questionnaires have not yielded a substantial gain of information. Most did not find relevant differences in profiles of the Minnesota Multiphasic Personality Inventory MMPI among patients with various primary headaches including cluster headache (29–31). Others found increased levels of anxiety, but contributed it to the syndrome headache instead of a specific entity like cluster headache (32,33).
However, some recent studies have corroborated substantial psychiatric co-morbidity in CH using other tools. In the French study on patients with CCH (10), 81 of 102 (76%) patients had scores of at least 8 or more on the anxiety subscale of the Hospital Anxiety and Depression scale (HAD), 46 (43%) had scores of 8 or more on depression subscale of the HAD scale. Similarly, in another small study (n = 21), 24% met DSM-IV criteria for an anxiety disorder, mainly generalized anxiety and panic disorder (34). In our study, the percentage of patients reporting symptoms of anxiety was highest in the CCH group except for specific phobias. The proportion of symptoms suggesting panic disorder and agoraphobia was particularly high. In addition, the rate of patients with symptoms suggestive of depression and dysthymia was even higher in our sample of CH patients, especially in the CCH group (Table 3). Other studies, however, did not find higher rates of depression (34,35). Likewise, Ertsey and co-workers (11) could not confirm an increased rate of depression among CH patients (8.6%) compared to controls (11.3%). In a small sample (n = 13), Seifert and colleagues (36) provided evidence for increased depression scores (above the cut-off in all but three patients) inside the active period – together with moderate PDI scores. These divergent findings can be partly explained by the different group sizes, samples (including cultural differences), screening instruments (self-report inventories versus structured interviews), other methodological aspects (life-time prevalence versus shorter reference periods) and by the nature of disorders (mood disorders often tend to be periodic and thus tend to fluctuate).
Remarkably, thoughts about death and suicidal tendencies were most frequent in the CCH group (22%) followed by both ECH groups (15% and 14%, respectively).
Thus, we could show for the first time that disability outside the active period is still substantial according to HDI scores. As the question relates to previous thoughts, it underlines that even today patients with cluster headache have had these thoughts and tendencies in their headache career despite our current armamentarium of drugs. The old description of CH as ‘suicide headache’ (37) unfortunately still holds true today. Therefore, one should monitor especially CCH patients closely to register and to intervene into such tendencies early. In addition, these findings add further evidence to a substantial emotional impairment outside active periods which have been demonstrated for disability as well.
In conclusion, psychiatric co-morbidity is highest in CCH patients for nearly all screened psychiatric disturbances. ECH and migraine patients were similarly but in comparison to CCH patients less affected.
We are fully aware that the use of screening questions from the Mini-DIPS cannot replace a full diagnostic interview conducted by a trained psychiatrist or psychologist to establish a diagnosis according to ICD-10 or DSM-IV criteria. As seen in ‘specific phobias’ and ‘depressive syndrome’, a substantially higher degree of healthy volunteers reported symptoms suggestive of these disorders than one would expect in the general population. This could be due to a tendency to over-rate symptoms because of this practice. However, we felt that this approach was more suitable to screen for a range of common psychiatric symptoms rather than adding other self-reporting tools limited to one disorder such as Beck Depression Inventory. Future studies should be based upon structured interviews like the Mini-DIPS or structured interviews based on DSM-IV criteria.
Economic burden
Indirect costs
It is striking that in our study, seven (25%) patients from the CCH group received invalidity allowance, whereas in the active ECH patients there was only one (4%). The rate of skilled and unskilled workers was numerically higher in CCH and ECH patients than in migraine patients and healthy controls. In addition, the proportion of skilled workers was highest in the CCH group, while the rate of civil servants, employees and executive employees was highest in the migraine and control groups. Thus, career seemed to be especially limited in the CCH group, which could be contributed to CH itself. One might argue that our healthy controls were recruited mainly from the university sector; however, this may not explain the findings for the migraine group. In accordance with these findings Jensen and coworkers (12) reported similar observations in their sample of 85 patients with CH. Work absence due to CH was significantly higher in gainfully employed CH patients than in the general population and nearly 40% were convinced that their headache had limited their career. Only 38% of the patients reported no or minute reduction in work efficiency. Early retirement was observed in 8% of the patients and loss of job in 16%. D’Amico and co-workers (9) found that 36% of the examined 22 CCH patients had lost their job and that half of the patients had reduced their work time by at least 50%. These findings illustrate that CH has serious implications on patients’ working life and career planning.
Direct costs
Not being the main focus of our study, we did not gather data on CH-related healthcare costs – an area in which available information is rather limited. One Italian study with CCH patients who underwent deep brain stimulation of the posterior hypothalamus showed that the annual costs of acute medication with sumatriptan prior to the implantation amounted up to €37,175 (38). For one German patient with CCH, annual costs of €4,703 have been reported – again with acute medication representing the lion’s share (39).
General aspects
Some restrictions apply to our data. A referral bias cannot be ruled out as the patients were seen at specialized third level centres. In addition, this study was conducted in an academic environment which could account for the high proportion of students in the control group as they were partly recruited from local staff. Due to local restrictions for anonymity and data protection, the clinical details were not cross-checked by the physician which could represent a potential source of error. In addition, we cannot exclude a recall bias; however, as stated elsewhere, the high intensity renders it likely that at least CH patients reliably remember details like year of onset. In future studies, another group of chronic headaches, like chronic tension-type headache or chronic migraine (9), could be helpful to differentiate pain-related from headache-specific effects.
One strength of our study is the fact that all patients were seen by a headache specialist prior to inclusion who confirmed the diagnosis. In addition, this is the first study in which all subtypes of CH (CCH, inactive and active ECH) were examined and compared to patients with migraine and a control group.
Which instruments are best suited for further studies is currently not certain. As indicated above, concomitant use of QoL and disability scales is reasonable. A combination of MIDAS (7 items) and HIT-6 (6 items) is shorter than the HDI. In fact, a recent study additionally suggested that simultaneous use of both tests is favourable as both scales complement one another (24). The HDI has the slight disadvantage that severity categories are missing in contrast to MIDAS or HIT-6, although numerical rating scales are mostly added in a clinical constellation.
Conclusions
We have shown a massive impact especially of chronic and episodic cluster headache in the active period in various economic and non-economic domains. More detailed studies are needed especially with regard to socio-economic consequences which are largely unknown. Future studies on CH burden should cover a broader field of domains (both economic and non-economic) to elucidate the full consequences of cluster headache impairment (7).
Results of Student–Newman–Keuls (SNK) post hoc tests for variables : TOTAL, FUNCTION, EMOTION. Data was grouped into homogeneous subgroups that differ from each other with P < 0.05. Within subgroups no significant differences could be found. CCH, chronic cluster headache; ECHa, episodic cluster headache in the active phase; ECHi, episodic cluster headache outside the active period; MIG; migraine; Control, healthy controls.
Footnotes
Acknowledgements
This study was conducted by members of the DMKG (German Migraine and Headache Society) network of young researchers. Thomas Dresler was supported by the Deutsche Forschungsgemeinschaft (SFB TRR 58 C4).
