Abstract
Background
To investigate whether epidemiological characteristics, including comorbidities, income, and education, have changed over time in people with cluster headache compared to controls.
Methods
Trends in cardiopulmonary, neurological, and psychiatric comorbidities, and income and education categories, were assessed using linked data from Norwegian health registries for 2009–2022. Comorbidities were defined based on relevant diagnosis codes from both the primary and the specialist healthcare. Income was categorized based on the national median for each year. Education was assessed based on the International Classification Standard for Education. Each cluster headache case was matched with 20 controls without cluster headache. Prevalence rate ratios were calculated using generalized estimating equations.
Results
People with cluster headache had higher prevalence than matched controls without cluster headache in all examined comorbidities, particularly migraine (men: 9.8% versus 0.3%; women: 23.1% versus 1.6%), mood disorders (men: 6.7% versus 2.8%; women: 9.2% versus 4.9%), and pulmonary disorders (men: 3.0% versus 1.8%; women: 4.8% versus 2.3%). Hypertension, coronary heart disease, and psychiatric disorders in specific age- and sex groups increased more in people with cluster headache than in controls during the recording period. Low/medium income was more prevalent in people with cluster headache versus controls (men: 50.2% versus 42.2%; women: 51.4% versus 46.6%). Similarly, low/medium education was more prevalent in people with cluster headache than controls (men: 77.0% versus 65.1%; women: 65.0% versus 55.0%). The prevalence of low/medium education increased in young women during the recording period.
Conclusions
People with cluster headache have a higher prevalence of comorbidities compared to matched controls, particularly migraine, mood disorders, and cardiopulmonary disease. Multiple comorbidities have increased in prevalence over time. Annual income and years of education were lower in people with cluster headache compared to matched controls. Cluster headache is a complex disease that requires specialist follow-up and individualized therapy.
Introduction
Cluster headache is a primary headache characterized by excruciating unilateral pain around, or in, the eye that may be accompanied by either restlessness, ipsilateral cranial autonomic symptoms, or both. The disease can be categorized as episodic or chronic depending on the length of pain-free remission periods. Episodic cluster headache has remission periods of more than three months, whereas chronic cluster headache has less than three months of remission during a year. 1 Cluster headache is a chronic disease with an onset of around age 30 years. 2 Multiple treatment options exist, but the evidence base is limited and efficacy varies. 3 Given the substantial disease burden of cluster headache, many patients experience disability and comorbidities.4,5
Longitudinal data on socioeconomic factors such as income status and educational attainment in people with cluster headache are sparse. Consequently, it remains unclear how the socioeconomic and comorbidity profile of cluster headache has evolved. Understanding such trends can characterize the disease burden over time and guide healthcare planning and resource allocation. While psychiatric comorbidities like depression and anxiety are well-documented, knowledge about non-psychiatric comorbidities remains limited. This is concerning given that comorbidity patterns have implications for management and treatment algorithms.
Norway provides tax-funded universal health care and social security for all its residents. Patients with chronic diseases receive full reimbursement for their medications when a very low annual out-of-pocket payment cap has been reached. Practitioners in primary and specialist care use the International Classification of Primary Care (ICPC-2) and International Classification of Diseases (ICD-10) diagnostic coding systems, respectively. Such mandatory and unified registration of precise diagnoses at all consultations with long-term follow-up ensures that Norwegian health registries have nationwide and comprehensive coverage. Linkage between these registries give a unique opportunity to examine trends in disease and comorbid conditions, allowing hypothesis-free exploration. The objective of this study was to investigate the prevalence and recent time-trends of comorbidities, as well as income and educational attainment in people with cluster headache relative to the general population in Norway over a period of 14 years (2009–2022).
Methods
Study design
This is a registry-based nationwide matched cohort study including all adult (≥18 years) individuals diagnosed with cluster headache residing in Norway at any time point between 2009 and 2022. Both prevalent and incident cases of cluster headache were analyzed to ensure complete representation. Each individual with cluster headache was compared to 20 matched controls without cluster headache. The study adheres to the Reporting of Studies Conducted Using Observational Routinely-Collected Data (RECORD) guidelines. 6
Study material
The Norwegian Registry for Primary Healthcare, the Norwegian Control and Payment of Health Reimbursements Database, and the Norwegian Patient Registry provided diagnostic codes for everyone who has received treatment in the primary and specialist healthcare.7–9 Data on prescriptions, including the Anatomical Therapeutic Chemical (ATC) classification code for the prescribed drug, amount of drugs prescribed, date for prescription fills, and ICD-10 codes and ICPC-2 codes for drug indication, were obtained from the Norwegian Prescribed Drug Registry. 10 This registry captures all prescriptions filled at pharmacies in Norway, but does not include over-the-counter purchases or medications administered in nursing homes or during hospitalizations.
Statistics Norway provided individual-level data on age, sex, death, emigration, educational attainment, and annual adjusted household income with yearly updates. 11 Our analysis of educational attainment was restricted to those over the age of 25 years, to ensure that all the included individuals were old enough to have achieved higher education. Household income was adjusted based on the number of people in each household, with adults and children being weighted differently. 12
This study uses the categories for sex registered in the Norwegian National Population Registry (Folkeregisteret). Sex is given as male or female and assigned for everyone at birth, but may be changed later following an application to the registry. We recognize that gender diversity exists among people with and without cluster headache. This binary classification may therefore not align with how they identify themselves. When referring to sex and gender identity, we use the terms “men” and “women” instead of “male” and “female”, as these terms were considered more accurate.
All registries contained person-level information with yearly updates from 2008 to 2022. The income variable was available up to the year 2021. Data from the different registries were linked for each person using their national identity number.
Definition of cluster headache and selection of controls
A case of cluster headache in a given calendar year was defined as having at least two contacts for cluster headache within a 365-day period, with the last contact in the year in question. To include a minimum of 365 days of observation time for each individual, we defined cases in the time period 2009–2022. A contact was defined as either a consultation with the diagnosis of cluster headache in specialist care (ICD-10-code G44.0) or in primary care (ICPC2-code N90), or a prescription for a cluster headache-specific triptan (sumatriptan [ATC: N02CC01] or zolmitriptan [ATC: N02CC03]) with reimbursement specified for cluster headache. The case definition includes both incident cases with newly diagnosed cluster headache and individuals with more long-standing disease. Analyzing all cases allowed us to map the disease burden in the complete cluster headache population, reflecting the real-world situation more accurately.
For each case of cluster headache identified in a given calendar year, 20 controls were randomly selected among individuals within the total population of Norway who did not satisfy our criteria for cluster headache. The controls were matched on age, sex, and year of observation using exact matching with the MatchIt package in R. 13 We employed nearest neighbor matching with Mahalanobis distance estimation, specifying exact matching on three covariates; age (in years), sex, and calendar year. Matching order was randomized to prevent systematic bias in our selection of controls. Because exact matching was required on all covariates, we achieved perfect covariate balance by design. The selection of controls was done separately for each calendar year, allowing the possibility of the same individual being re-selected as a control for more than one calendar year. Matching within each calendar year was done without replacement, meaning that each control could only be matched to a single cluster headache case each year.
Outcome definitions
For a given calendar year, we measured the prevalence of comorbidities, income category, and educational attainment among all individuals with cluster headache that year, regardless of time since diagnosis of cluster headache.
Adjusted household income was categorized as low/medium or high based on the national median for each year, with low/medium income defined as ≤99% of the national median. Educational attainment was dichotomized based on the Norwegian Standard Classification of Education (NUS); the Norwegian version of the International Standard Classification for Education (ISCED). 14 Low/medium education was defined as compulsory education, high school, or pre-university education (≤14 years).
Comorbidities were defined as relevant diagnostic codes from primary health care (ICPC-2) and specialist healthcare (ICD-10), and selected based on existing knowledge, clinical observations, and data availability. For example, the association between pulmonary disorders and cluster headache was explored because of their shared association with tobacco smoking.15,16 Prescription data was not used to define comorbidities, as drugs for cardiovascular, neurological, and psychiatric disorders are often used off-label for cluster headache and could therefore inflate comorbidity estimates. Comorbidities were selected based on data availability and clinical relevance for cluster headache and categorized into ten groups: coronary heart disease, hypertension, pulmonary disorder, migraine, epilepsy, anxiety disorder, mood disorder, facial pain, sleep disorder, and substance abuse. Coronary heart disease included unstable angina and acute myocardial infarction. A comorbidity for a given person in a given calendar year was defined as two or more diagnostic codes from either primary or specialist health care. This definition was applied for all comorbidities.
Diagnostic codes for acute myocardial infarction and depressive disorders in Norwegian registry data have been validated. Acute myocardial infarction demonstrated high validity, and depressive disorders had high specificity and low sensitivity.17,18 The complete list of diagnostic codes for each category is provided in the supplementary materials (Online Appendix 1).
Statistical analysis
We estimated both relative differences (prevalence ratios) and absolute differences (prevalence differences) between people with cluster headache and matched controls for comorbidities, education categories, and income categories. Time trends were estimated as absolute and relative changes in prevalence per year.
Prevalence rate ratios were analyzed using the coefficients from Generalized Estimating Equations and complete-case analysis with a binomial distribution, log-link, and robust standard errors to account for repeated observations for the same individual. We used Poisson Generalized Estimating Equations where the models did not converge, as exponentiated coefficients from Poisson-models for binary outcomes can be used as estimates of risk ratios for common outcomes in observational studies. 19 The coefficients from the models were exponentiated and interpreted as prevalence ratios. Prevalence differences were analyzed using the coefficients from Generalized Estimating Equations with a Gaussian distribution, identity-link, and robust standard errors. For each approach, we constructed two models for every outcome – one with and one without an interaction term between year and a binary group variable for cluster headache versus controls. This made it possible to examine whether relative and absolute differences between cluster headache cases and controls changed over time. This was estimated by multiplying the coefficient for year by the coefficient for the interaction term between year and cluster headache prevalence. Age and year of observation were included as continuous covariates.
All statistical analyses were conducted in R version 4.3.0 and Stata version 18. We did not use any imputation methods as minimal data were missing. Missingness was reported for variables with any missing data. No adjustments for multiple comparisons were made.
Sensitivity analyses
Sensitivity analyses were performed to investigate how the prevalence of comorbidities changed when we required ≥1 instead of ≥2 diagnoses from either specialist or primary care. We also investigated differences in prevalence for comorbidities being recorded in the primary and specialist health care.
Results
We analyzed a total of 7,803 people with cluster headache and 387,042 matched controls. In both groups, 55% were men with a median age of 46 years with an interquartile range of 35–56 years. For each year in the study period, 60–70% of the included cases had cluster headache contacts in the previous four years and were defined as prevalent cases. The remaining 30–40% were defined as incident cases. People with cluster headache had a numerically higher prevalence of all comorbidities compared to controls (Table 1). Migraine was the most prevalent comorbidity, affecting 9.8% of men and 23.1% of women with cluster headache, compared to 0.3% and 1.6% in controls, respectively (Table 1). Other prevalent comorbidities included mood disorders, anxiety disorders, pulmonary disorders, and hypertension (Figures 1 and 2, Online Appendix 2). The prevalence of hypertension, coronary heart disease, acute myocardial infarction, mood disorders, and anxiety disorders increased more over time in people with cluster headache than in matched controls (Online Appendix 3).

Prevalence of migraine in cluster headache and matched controls (2009–2022). Lines represent locally weighted smoothed trends with individual data points showing annual prevalence. Migraine was defined as ≥2 diagnostic codes from primary or specialist healthcare within a calendar year. The y-axis shows percentage of individuals with migraine diagnosis. Each person with cluster headache was matched to 20 controls by age, sex, and year of observation.
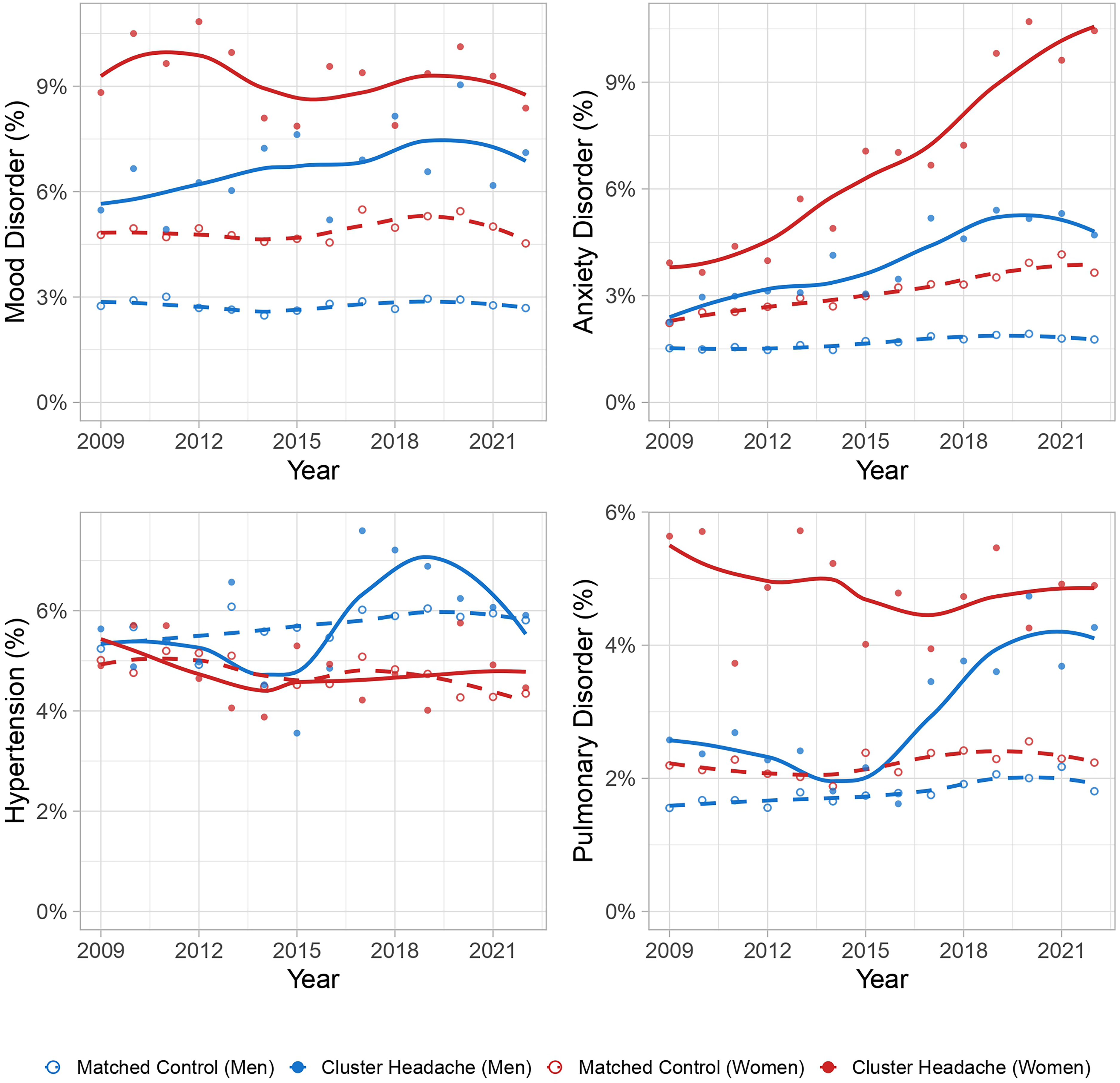
Prevalence of mood disorder, anxiety disorder, hypertension and pulmonary disorder in cluster headache and matched controls (2009–2022). Lines represent locally weighted smoothed trends, with individual data points showing annual prevalence. Comorbidities were defined as ≥2 diagnostic codes from primary or specialist healthcare within a calendar year. Each panel shows percentage of individuals with the specified comorbidity. Each person with cluster headache was matched to 20 controls by age, sex, and year of observation.
Prevalence of comorbidities.

This table demonstrates the overall prevalence of comorbidities for matched controls and people with cluster headache, respectively, during the whole study period 2009–2022. Prevalences are shown as absolute numbers and percentages (in parentheses). Some cases and controls contribute with more than one year, and therefore contribute with more than one observation. The number of unique cases and controls is 7,803 and 387,042, respectively.
People with cluster headache had higher rates of socioeconomic disadvantage compared to controls (Table 2). Low/medium income was present in 50.2% of men and 51.4% of women with cluster headache, compared to 42.2% and 46.6% in the controls, respectively. Low/medium education was recorded in 77.0% of men and 65.0% of women with cluster headache, compared to 65.1% and 55.0% in controls. The socioeconomic differences between cases and controls remained stable during our recording period, with the exception of low/medium education in young women, which increased.
Prevalence of income and education categories.
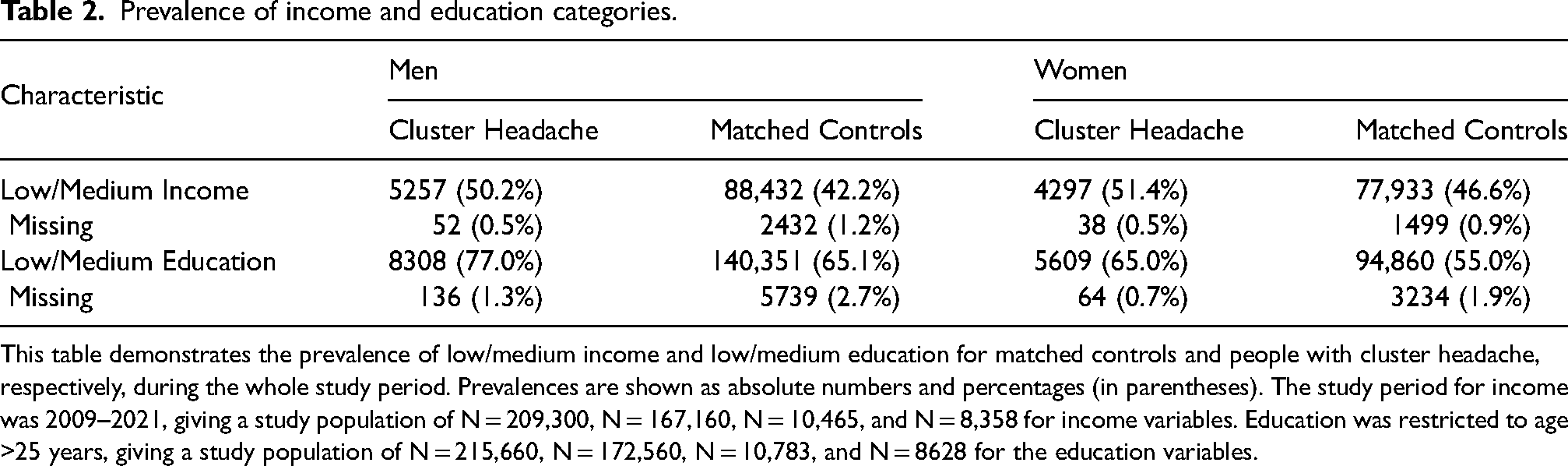
This table demonstrates the prevalence of low/medium income and low/medium education for matched controls and people with cluster headache, respectively, during the whole study period. Prevalences are shown as absolute numbers and percentages (in parentheses). The study period for income was 2009–2021, giving a study population of N = 209,300, N = 167,160, N = 10,465, and N = 8,358 for income variables. Education was restricted to age >25 years, giving a study population of N = 215,660, N = 172,560, N = 10,783, and N = 8628 for the education variables.
Cardiopulmonary comorbidities
Both men and women <40 years with cluster headache had a higher prevalence of hypertension than controls (PRR: 2.03; 95% CI: 1.56–2.64; PRR: 2.27; 95% CI: 1.66–3.12). Pulmonary disorders were more prevalent among people with cluster headache compared to controls in all groups, except for men <40 years. The difference was highest in women <40 years (PRR: 2.47, 95% CI: 2.00–3.06). For coronary heart disease, the prevalence was higher in people with cluster headache compared to controls in both men and women aged 40–59 years (PRR: 1.45, 95% CI: 1.16–1.81) and (PRR: 2.12, 95% CI: 1.37–3.27).
From 2009 to 2022, hypertension had an average yearly increase of 5.7% in men <40 years and of 11.2% in women <40 years with cluster headache. These increases were significantly different from the control groups, where hypertension decreased by 3% and 1% each year, respectively (p-value for interaction: 0.005; p-value for interaction: 0.011). Similarly, coronary heart disease increased 10.5% each year in men <40 years with cluster headache, while the disease decreased in the controls (p-value for interaction: <0.001). No significant changes over time in relative prevalence differences were found for pulmonary disorders.
Neurological comorbidities
Migraine, sleep disorders, and facial pain were more prevalent in people with cluster headache compared to controls in all groups. The relative difference for migraine was highest in men ≥60 years (PRR: 58.4, 95% CI: 44.4–76.9). For sleep disorders, the relative difference was highest in men <40 years (PRR: 3.81, 95% CI: 3.11–4.67). For facial pain, the relative difference was highest in women <40 years (PRR: 38.7, 95% CI: 24.9–60.1). For epilepsy, people with cluster headache had a higher prevalence compared to controls in all groups except men ≥60 years, with the highest relative difference observed in women <40 years (PRR: 3.35, 95% CI: 2.46–4.55) (Online Appendix 2).
The prevalence of migraine increased during our observation period in the cluster headache group, irrespective of sex and age. Except for women 40–59 years, no significant change in relative difference in prevalence over time between cluster headache cases and controls was found. However, when examining absolute numbers, significant increases in prevalence differences were found for all sex- and age groups, with a larger increase for people with cluster headache (Figure 1). No significant changes over time in relative prevalence differences were found for sleep disorders, facial pain, and epilepsy (Online Appendix 3).
Psychiatric comorbidities
People with cluster headache had a higher prevalence of mood disorders, anxiety disorders, and substance abuse compared to controls in all groups. For mood disorders, the relative difference was highest in men ≥60 years (PRR: 2.77, 95% CI: 2.29–3.35). For anxiety disorders, the relative difference was highest in women <40 years (PRR: 2.60, 95% CI: 2.34–2.89). For substance abuse, the relative difference was highest in women ≥60 years (PRR: 4.28, 95% CI: 2.86–6.39) (Online Appendix 2).
Mood disorders in people with cluster headache had an average yearly increase of 4.9% in men ≥60 years, but an average yearly decrease of 4.0% in women 40–59 years. Both time trends differed from the controls (p-value for interaction: 0.033 and 0.013, respectively). Anxiety disorders in the cluster headache group had an average yearly increase in women <40 years, men 40–59 years, and women 40–59 years of 11.3%, 6.1%, and 7.1% respectively. This was more than in the same sex- and age control groups (p-value for interaction: 0.004; 0.003, and 0.007, respectively). No significant time changes in relative differences were found for substance abuse (Online Appendix 3).
Income Status
Men with cluster headache had 8% – 22% higher prevalence rates of low/medium annual income compared to controls (men <40 years: [PRR: 1.17, 95% CI: 1.14–1.21], men 40–59 years: [PRR: 1.22, 95% CI: 1.18–1.25], men ≥60 years: [PRR: 1.08, 95% CI: 1.03–1.14]). Women with cluster headache had on average 5% – 13% higher prevalence of low/medium income compared to controls (women <40 years: [PRR: 1.10, 95% CI: 1.07–1.13], women 40–59 years: [PRR: 1.13, 95% CI: 1.09–1.18], women ≥60 years: [PRR: 1.00, 95% CI: 0.95–1.05]) (Online Appendix 2). No changes over time in relative differences were found for income status (Figure 3) (Online Appendix 3).

Prevalence of low/medium income and low/medium education in people with cluster headache and matched controls. The study period for income was 2009–2021. The study period for education was 2009–2022. Lines represent locally weighted smoothed trends (LOESS), with individual data points showing annual prevalence. Low/medium income was defined as ≤99% of the national median household income (adjusted for household composition). Low/medium education was defined as ≤14 years of education (up to high school or pre-university level). Education data was restricted to individuals ≥25 years. Each person with cluster headache was matched to 20 controls by age, sex, and year of observation.
Educational attainment
Men with cluster headache had 5% – 25% higher prevalence rates of low/medium education compared to matched controls (men <40 years: [PRR: 1.25, 95% CI: 1.22–1.29], men 40–59 years: [PRR: 1.17, 95% CI: 1.15–1.19], men ≥60 years: [PRR: 1.05, 95% CI: 1.03–1.08]). The relative difference was even higher for the women with cluster headache, with prevalence rates 5% – 45% higher than in controls, the highest difference observed for young people (women <40 years: [PRR: 1.45, 95% CI: 1.40–1.51], women 40–59 years: [PRR: 1.10, 95% CI: 1.08–1.13], women ≥60 years: [PRR: 1.05, 95% CI: 1.02–1.08] (Online Appendix 2). The prevalence of low/medium education in women <40 years with cluster headache had an average yearly increase of 1.0%, which was different from the observed decrease of 2% in controls (p-value for interaction: 0.006). No other changes over time in relative education differences were found (Figure 3) (Online Appendix 3).
Sensitivity analyses
The prevalence of comorbidities increased when we required only ≥1 diagnostic consultation codes in primary or specialist care, compared to ≥2 consultation codes (Online Appendix 4). However, the relative differences between people with cluster headache and matched controls remained relatively unchanged. We found a higher prevalence of comorbidities in primary care data than in specialist care data (Online Appendix 5).
Discussion
Summary of findings
Comorbidities were more common in people with cluster headache in nearly all sex and age groups when compared to matched controls. The strongest associations were seen for migraine, facial pain, substance abuse, and sleep disorders. Comorbidity prevalence increased more in people with cluster headache than in the matched controls during our registration period. People with cluster headache had a lower annual income and shorter education than matched controls. Young women with cluster headache had particularly low educational attainmentwith a worsening trajectory over time, which is concerning.
Comorbidities
The most prevalent comorbidity in people with cluster headache was migraine, which increased over time in both cluster headache cases and matched controls. The difference between cases and matched controls increased in both men and women ≥40 years. This may be explained by increased knowledge of the diagnostic criteria for headache disorders in the health service. Contributing factors may include more awareness of individuals with multiple headache conditions, leading to more precise classification of comorbid headaches.
The migraine prevalence in our matched controls was lower than the assumed migraine prevalence of around 12% in the general adult population.20,21 Migraine was defined as having ≥2 diagnostic codes recorded in either primary healthcare (ICPC-2) or specialist healthcare (ICD-10) in one year, which likely selects people with chronic and high frequent migraine, rather than episodic or mild migraine. Prescription data was not used to define migraine to maintain consistency with the definition of other comorbidities. Most individuals with episodic migraine manage their symptoms without frequent consultations in primary or specialist health care. This could explain the discrepancy between the assumed migraine prevalence in the Norwegian adult population and our recorded data in this study. In contrast, people with cluster headache and comorbid migraine will likely get both conditions recorded during a consultation.. Nevertheless, our findings illustrate that migraine is an important comorbidity in people with cluster headache, particularly in women. This warrants consideration in clinical management, as prophylactic and acute treatments may need adjustments to address both headaches effectively while avoiding side effects and medication overuse headache.
Mood disorders, anxiety disorders, and substance abuse were all more prevalent in people with cluster headache compared to controls. This aligns with previous findings in smaller cohorts.22–24 The prevalence of these psychiatric conditions increased more over time in people with cluster headache compared to controls. This may reflect a cumulative burden, where recurrent pain, cascading social consequences, and associated disability could lead to increasing psychological distress. Additionally, recent evidence suggests a functional overlap in neurobiological pathways between cluster headache and psychiatric disease, which may lead to a higher baseline risk. 25 Our findings underscore the importance of monitoring psychiatric comorbidities in people with cluster headache and prevent secondary drug addiction challenges.
Epilepsy was more prevalent in people with cluster headache than in controls in most sex and age groups. There is a known association between epilepsy and migraine, but a similar association has not previously been demonstrated for cluster headache. 26 This finding suggests shared pathophysiological mechanisms. Neuronal hyperexcitability, a hallmark component in epilepsy, has also been demonstrated in people with cluster headache.26,27 Several antiseizure drugs, including topiramate and valproate, have shown efficacy as preventive therapies in cluster headache. 28 The increased occurrence of epilepsy in individuals with cluster headache may in part be explained by the higher frequency of cardiovascular disorders, which are associated with focal epilepsy. 29
We found higher prevalence rates of coronary heart disease, hypertension, and pulmonary disorders in people with cluster headache compared to matched controls. The strongest associations were observed at age <40 years for both sexes, and for middle-aged men 40–59 years. Notably, the relative difference in prevalence for hypertension in young adults, and for acute myocardial infarction in young men increased during our 14 years study period. These findings may be explained by multiple risk factors converging in people with cluster headache, which may also contribute to reduced life expectancy. 30 Smoking is a joint risk factor for both cluster headache and cardiovascular disease.31,32 Unhealthy lifestyle factors, including high alcohol consumption and high body-mass index, are more prevalent in people with cluster headache, and increase the risk of cardiovascular diseases.33,34 Cardiovascular examination, risk management, preventive interventions, and a low threshold for examinations should be prioritized in cluster headache management.
Income and education
People with cluster headache had lower income and education compared to controls. The relative difference for income was more pronounced in men, while the relative difference for education was more pronounced in women. The relative differences between cluster headache and controls for both income and education remained stable over time apart from reduced educational trajectories in young women with cluster headache. Reduced income aligns with previous studies, where people with cluster headache were more likely to receive disability pension and had lower economic status compared to controls.4,5
Cluster headache typically manifests around age 30 years, an age at which most people have finished their education. 2 However, an international survey from 2021 found that cluster headache has a pediatric onset in 27.5% of all participants. 35 Lower educational attainment may therefore, in some individuals, be explained by early-onset cluster headache causing disability that made it difficult to pursue higher education. Shared socioeconomic factors may offer an alternative explanation. Socioeconomic disadvantage is associated with both higher smoking rates and lower educational attainment. 36 As tobacco smoking is a plausible risk factor for cluster headache, this could explain some of the observed association between cluster headache and lower education. 15 Causality cannot be established from our data, and the direction of these relationships remains uncertain.
Strengths and limitations
We used mandatory nationwide data from multiple sources, including data from both the primary care and specialist healthcare. These data have been recorded prospectively, eliminating recall bias and reducing selection bias. The inclusion of matched controls allowed us to compare comorbidities, income, and education between cluster headache cases and people without cluster headache. The large sample size secured detection of small effect sizes for less frequent comorbidities such as epilepsy, particularly for cluster headache sex and age subgroups. This also allowed exact matching and perfect covariate balance by design.
We required two diagnostic codes from primary or specialist care consultations per calendar year for the comorbidities. This represents a conservative approach that increases the positive predictive values, mitigating the risk for misclassification. The trade-off of this approach includes reduced sensitivity, particularly for comorbidities without frequent consultations, such as episodic migraine. Prevalence is therefore underestimated for some comorbidities. Since people with cluster headache have more frequent healthcare consultations, there is a risk of detection bias and higher sensitivity for comorbidity diagnoses in the cluster headache group. While our sensitivity analysis showed that relative differences remained similar when requiring one versus two diagnostic codes, this does not eliminate the potential for detection bias. Such bias is inherent in all diagnosis-based studies of comorbidities.
We did not adjust for multiple comparisons. Each comorbidity and socioeconomic category represents a distinct exploratory clinical question. We cannot exclude the possibility that some findings are due to chance, especially if only marginally significant. Such findings should therefore be interpreted with some caution.
Reporting routines for diagnostic codes may have changed during the study period. This could create selection bias. However, it would not affect the relative difference between cluster headache and controls. The lack of validation of diagnostic codes for primary care is another limitation. Our sensitivity analysis revealed that comorbidity prevalences were somewhat higher in primary care than specialist care, but differences varied widely and ranged from 0.03% (facial pain) to 5.9% (hypertension). Combining primary and specialist care data captures a more complete picture, though at the expense of the potentially lower validity in primary care data.
Conclusion
People with cluster headache have a higher burden of comorbidities and socioeconomic disadvantages compared to matched controls. The most prevalent comorbidity was migraine. Cardiovascular disease is more common in people with cluster headache. The relative differences in comorbidity prevalence between people with cluster headache and controls increased during our registration period. We found important socioeconomic disparities where people with cluster headache had lower income and less education than matched controls. These findings demonstrate that people with cluster headache comprise a complex and vulnerable patient group that requires individualized and complex therapy with a close follow-up both in the primary and specialist healthcare.
Article highlights
People with cluster headache had substantially higher prevalence of migraine, mood disorders, and cardiopulmonary disease versus matched controls.
Multiple comorbidities increased more in people with cluster headache than controls during the recording period.
People with cluster headache demonstrated notable social disadvantage with lower income and shorter education compared to controls.
Supplemental Material
sj-docx-1-cep-10.1177_03331024261428583 - Supplemental material for Cluster headache and comorbidities, income and education: A nationwide registry-based matched cohort study
Supplemental material, sj-docx-1-cep-10.1177_03331024261428583 for Cluster headache and comorbidities, income and education: A nationwide registry-based matched cohort study by Einar Naveen Møen, Marte-Helene Bjørk, Nils Erik Gilhus, Dagrun Slettebø Daltveit, Daniela Contreras, Knut Hagen, Mahendran Jayaraman, Andrej Netland Khanevski, Tore Wergeland, Anker Stubberud, Erling Andreas Tronvik and Jannicke Igland in Cephalalgia
Footnotes
Author note
Research Data
Data analyzed in this study will not be made publicly available as they include individual patient information. Corresponding datasets can be requested from Helsedataservice following appropriate regulatory approvals and registry data applications. 37 Programming code can be provided upon request.
Acknowledgements
Not applicable.
Author contributions
All authors contributed to the conceptualization, planning and design of the study, interpretation of analyses, and subsequent revision of the manuscript. The formal data analysis was conducted by ENM under the supervision by JI. The first and subsequent drafts were written by ENM. Data acquisition was done by MHB and JI. All authors read and approved the final manuscript.
Consent to participate
All patient information was de-identified, and patient consent was not required.
Consent for publishing
All authors consent to publication in Cephalalgia.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ENM reports no competing interests relevant to the manuscript. MHB reports research grants from the Research Council of Norway, the Research Council of the Nordic countries (Nordforsk), the Norwegian Health authorities (KLINBEFORSK), The Norwegian Epilepsy association and Trond Mohn Foundation. Institutional payment from the Marked authorization holders of valproate (Sanofi, Desitin, and more) for the conduction of a post-authorization safety study. Lecture honoraria from Eisai, AbbVie, Angelini Pharma, Novartis, Pfizer, BestPractice. Advisory board honoraria from Jazz Pharmaceuticals, Angelini Pharma, Lundbeck, Pfizer, and Eisai. NEG has received consultative, trial, and speaker’s honoraria from Argenx, Alexion, Amgen, UCB, Merck, Roche, Immunovant, NMD Pharma, Novartis, Janssen, Johnson & Johnson, Huma, Denka, Dianthus, Lundbeck, Grifols, and Takeda outside the topic for this study. DSD reports no competing interests relevant to the manuscript. DC reports no competing interests relevant to the manuscript. KH reports no competing interests relevant to the manuscript. MJ reports no competing interests relevant to the manuscript. ANK has received consultative, trial, and speaker’s honoraria from TEVA, Lundbeck, and Abbvie, outside the topic of this study. TW reports research grants from The Liaison Committee for Education, Research and Innovation in Central Norway (Samarbeidsorganet) and The DAM Foundation. He has received lecture honoraria from TEVA, Roche, LUNDBECK and Lilly, and is a shareholder in two medical device start-up companies (Vilje Bionics AS and Keimon Medical AS). AS reports no competing interest relevant to the manuscript. EAT has received personal fees for lectures and advisory boards from Novartis, Eli Lilly, Abbvie, TEVA, Roche, Lundbeck, Pfizer, Biogen, and Organon. He is a consultant for and holds stocks and intellectual property in Man & Science and Nordic Brain Tech, and holds shares in Keimon Medical. He has received non-personal research grants from the European Commission, Norwegian Research Council, KlinBeForsk, and Nordic Innovation, and has conducted commissioned (non-personal) research for Lundbeck. JI is the head of the Core Facility for Biostatistics and Data Analysis, which has received institutional payment for conducting post-authorization safety studies and drug utilization studies for Sanofi, Pfizer and Novartis, including a drug utilization study for migraine preventive treatments.
Data availability statement
The dataset analyzed in this study will not be made publicly available as it contains individual patient information. Similar dataset can be provided upon regulatory approvals and data applications to Helsedata (helsedata.no).
Ethical considerations
This study received ethical approval from the Regional Ethical Committee in Western Norway (ID: 198928). This is a registry-based study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The registry research program at the Norwegian Center for Headache Research (NorHead) is funded by the Research Council of Norway (grant number: 328615) and covers costs for data management and supervision, as well as other running costs. Einar Naveen Møen received funding through the Medical Student Research Programme (Forskerlinjen) at the University of Bergen.
Open practices
Not applicable.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
