Abstract
Aim
To compare the prevalence of unhealthy lifestyle factors and comorbid disorders in cluster headache patients with headache-free controls, in order to discuss pathophysiology and possible consequences.
Methods
Cluster headache patients from the Danish cluster headache survey aged 18–65 years, diagnosed according to ICHD-II, were compared to sex- and age-matched headache-free controls. Participants completed questionnaires and structured interviews.
Results
A total of 400 cluster headache patients and 200 controls participated. Patients had a more unhealthy lifestyle compared with controls in the form of current and current/former smoking (48.3% vs. 9.0%, p < 0.001 and 74.5% vs. 30.0%, p < 0.001, respectively), higher average alcohol intake per week (98.2 grams vs. 77.9 grams, p = 0.033) and BMI (26.1 vs. 24.2 kg/m2, p < 0.001), whereas coffee and energy drink consumption was equally distributed. Further, lifestyle-related, psychiatric and pain-related diseases were much more prevalent in patients compared with controls, except for diabetes. Sub-group analyses revealed that current/former smokers had a worse clinical presentation than never smokers.
Conclusion
Unhealthy lifestyle factors and lifestyle-related diseases were more prevalent in cluster headache patients compared to controls. As lifestyle-related diseases might have serious consequences in the management of cluster headache, it is key to inform patients at an early time point about the possible risks of their lifestyle choices.
Introduction
Cluster headache (CH) is one of the most painful conditions known to mankind. The severe, unilateral attacks of pain are located in the periorbital area and are accompanied by cranial autonomic symptoms and agitation. Observational studies have found a high prevalence of unhealthy lifestyle factors among CH patients. Most consistently reported are the high frequency of smoking (1–6) and unhealthy alcohol habits (1–3). The strong connection between CH and smoking has led to the hypothesis that smoking could be causally related to the development of CH (7,8). In contrast, it has been suggested that patients with CH have certain personality traits which makes them more prone to overindulge in certain unhealthy lifestyle habits (9–11). A higher use of illegal substances, such as cannabis and cocaine, has been reported in men with CH compared with controls, while no such difference was observed in women (12).
Unhealthy lifestyle factors such as smoking, having a high alcohol intake and unhealthy eating habits are all risk factors for the development of cerebro- and cardiovascular diseases such as atherosclerosis, stroke and cardiac infarction (13). Such lifestyle-related diseases can have negative consequences for the medical management of CH.
To our knowledge, lifestyle factors and the presence of comorbid lifestyle-related diseases have not previously been investigated in the same cohort. Therefore, we aimed to compare the prevalence of unhealthy lifestyle factors and comorbid disorders in a large cohort of well-characterized CH patients with headache-free controls, in order to discuss pathophysiology and possible consequences.
Methods
The data was obtained from the Danish Cluster Headache Survey, an observational study performed from 2012 to 2017. The methodology has previously been published in detail (14) but will be summarized in the following sections.
Participants
Patients diagnosed with episodic CH (eCH) or chronic CH (cCH) according to ICHD-II criteria (15) were mainly recruited from the Danish Headache Center, a tertiary headache clinic. Patients were identified from an existing database of present and former patients who had consented to be contacted for research purposes. A minority of patients were recruited through advertisements on the webpage of the Danish Cluster Headache Association and through neurologist practitioners. All patients were diagnosed by a headache specialist. If a patient suffered from a comorbid primary headache, they were only included if they could reliably distinguish between the different types of headache. We chose not to exclude patients with self-reported attacks lasting longer than 180 minutes who otherwise met the ICHD-II criteria. Patients with secondary CH were excluded. Headache-free controls were recruited through the internet (www.forsøgsperson.dk), work places and clubs for different leisure-time activities in the capital region of Denmark. Controls were excluded if they suffered from primary or secondary headache for more than 1 day a month. All participants had to be 18–65 years of age and had to understand and write Danish. All participants gave their informed consent according to the Helsinki Declaration. The study was first approved in a protocol along with two other studies (H-2-2012-016) but after these were finalized, this questionnaire-based study did not need formal approval according to Danish law. The local ethics committee was informed of the continued data collection and had no objections to it (file number: 17008910).
The questionnaire
After written consent was obtained, participants filled out the detailed questionnaire, which consisted of 362 questions concerning headache diagnosis, treatment, sleep, headache burden, work, lifestyle, and physical activity (14). Upon returning the questionnaire, all underwent a structured interview either in person or by telephone. This was performed by a physician, trained medical student, or a study nurse (MB, NL, AP, MF). For patients, it took roughly 1 hour to complete the questionnaire and 5–10 minutes to complete the interview, and for controls 30 minutes and 5 minutes, respectively.
Statistical analysis
To assess the burden of smoking for all participants with a smoking history (current and former smokers), the average number of pack-years was calculated from the number of cigarettes per day divided by 20 and multiplied by the number of years of smoking. Current and former smokers were compared with never smokers to assess the clinical phenotype in relation to smoking status. Former smokers were grouped with smokers, as previous findings indicate that these two groups are similar (16,17). Body Mass Index (BMI) was calculated from self-reported height and weight. Alcohol intake was positive if participants had reported any intake of alcohol per week during the past year. An unhealthy alcohol intake was defined according to the Danish Health Authority as >168 grams/week for men and >84 grams/week for women. A damaging alcohol intake was defined as >252 grams/week and >168 grams/week, accordingly (18). Energy drinks were defined as caffeine-containing cold beverages.
SAS 9.3 was used for all statistical analysis. When comparing means between two groups, the two-sample t-test and Mann-Whitney U-test were used. When comparing percent distributions, the chi-square test was used but Fisher’s exact test was used if the expected number in a category was < 5. Multiple logistic regression analyses were performed to determine odds ratios for those lifestyle-related diseases that occurred more frequently in patients than in controls. Model 1 was adjusted for age >49 years (yes/no) and sex (men/women); model 2 was additionally adjusted for the possible confounder smoking (current smoker or history of smoking/never smoker); model 3 was additionally adjusted further for the possible effect modifiers BMI >25 (yes/no) and unhealthy alcohol intake (yes/no). With the background of a report from the National Institution of Public Health, age >49 years was chosen since the prevalence of most cardiovascular diseases increases substantially from age 49 years (19). Two-tailed p-values were calculated and p < 0.05 was considered statistically significant. Because of the explorative nature of the study, no corrections for multiple comparisons were made.
Results
During the study period, 644 CH patients and 326 controls were invited to participate in the study. In total, 400 patients (269 men and 131 women) and 200 controls (133 men and 67 women) returned the questionnaire and completed the structured interview, resulting in a response rate of 62.1% for patients and 61.3% for controls (Figure 1).
Flowchart for participation in the questionnaire and interview for patients with cluster headache and headache-free controls.
Demographic factors for cluster headache (CH) patients and headache-free controls.

Data are reported as means (SD) or as percent distributions. T-tests comparing patients and controls were not significant for age (p = 0.17) and sex (p = 0.85), but chi-square test comparing educational level was (p < 0.001).
Lifestyle factors for cluster headache (CH) patients and controls.

Data are presented as means (SD) and percent distributions. CH: cluster headache; BMI: Body mass index.
P-values highlighted in bold are significant.
T-test or chi-2 test comparing CH patients with controls. Alcohol, coffee and energy drink consumption was calculated for those participants that reported any consumption, while number of pack-years was calculated for both present and former smokers. An unhealthy alcohol intake was defined according to the Danish Health Authority as >168 grams/week for men and >84 grams/week for women. A damaging alcohol intake was defined as >252 grams/week and >168 grams/week, accordingly. Energy drinks were defined as caffeine containing cold beverages.
Smoking
Current smoking and a history of smoking (current and former) were much more prevalent in patients compared with controls (48.3% vs. 9.0%, p < 0.001 and 74.5% vs. 30.0%, p < 0.001, respectively), as was the number of pack-years (23.3 years vs. 13.7 years, p < 0.001). The differences between patients and controls stayed highly significant when studying the two sexes separately for all three smoking variables (Table 2).
Clinical phenotype for cluster headache (CH) patients with and without a history of smoking.

Data are presented as means (SD) and percent distributions. CH: cluster headache; eCH: Episodic cluster headache; cCH: Chronic cluster headache.
P-values highlighted in bold are significant.
Pain intensity was rated from 0–4 with increasing intensity on a Likert scale.
Cluster duration and clusters/year was only assessed for ECH.
Of those with a history of smoking, 79.9% started smoking prior to CH debut, on average 15.9 years (SD 9.98) before. In CH patients, more men tended to have smoking onset prior to CH debut (men: 83.0% vs. women: 72.8%, p = 0.052) but no difference was identified in relation to phenotype (eCH: 81.4% vs. cCH: 78.4%, p = 0.55).
Alcohol
Alcohol consumption was assessed within the last year for both patients and controls. Far fewer patients than controls reported consuming alcohol (60.8% vs. 89.5%, p < 0.001) but the prevalence of an unhealthy alcohol intake was equally distributed (10.8% vs. 13.0%, p = 0.42) and the prevalence of a more severe, damaging alcohol intake was more frequent in patients (5.0% vs. 1.5%, p = 0.035). Overall, patients reported a higher average alcohol intake per week (98.2 vs. 77.9 grams, p = 0.033). In patients, more men than women reported alcohol consumption (79.6% vs. 69.5%, p < 0.001) and men consumed more per week (111.5 vs. 53.6 grams, p < 0.001). Further, more men than women had an unhealthy intake (13.8% vs. 4.6%, p = 0.005), whereas a damaging intake was equally distributed and rare (6.3% vs. 2.3%, p = 0.083). Alcohol consumption was more prevalent in eCH than in cCH (71.2% vs. 42.5%, p < 0.001), but fewer patients with eCH than cCH had an unhealthy or damaging alcohol intake (3.4% vs. 15.0%, p < 0.001 and 1.37% vs. 6.72%, p = 0.016, respectively). In contrast, the average number of grams per week did not differ between phenotype (eCH: 100.8 vs. cCH: 84.0, p = 0.41). Patients with a history of smoking more frequently had an unhealthy alcohol intake (12.8% vs. 4.9%, p = 0.027) whereas a damaging intake was not significantly different, probably due to low numbers (6.0% vs. 2.0%, p = 0.10).
A total of 14.3% reported drinking alcohol in attack periods. This was higher in men than in women (17.5% vs. 7.6%, p = 0.008), but was similar in eCH and cCH (12.7% vs. 17.1%, p = 0.22). Among patients consuming alcohol in the attack periods, fewer patients reported alcohol triggering CH attacks compared with those not drinking (22.8 vs. 39.1, p < 0.05).
Eating habits
Patients had significantly higher BMI than controls, an observation that remained strong when studying men and women separately (Table 3). No significant differences in BMI were found between men vs. women or episodic vs. chronic patients.
Appetite changes in attack periods were reported by half of the patients (53.3%) and was more common in women than in men (61.1% vs. 50.6%, p = 0.048) and in cCH than in eCH (59.6 % vs. 47.0%, p = 0.016). Appetite decreased in 80.1% of those that reported it to change, increased in 16.2% and changed in other ways in 3.7% (most reported that it was not decreased or increased in general but that they craved food, sweets or salty food items just after CH attacks). Food cravings in attack periods were reported by 19.8% of patients, of which 89.9% had cravings for sweets or unhealthy food items. There were no differences between sex and phenotype.
Caffeine consumption
There were no differences observed in relation to coffee consumption when comparing patients with controls. There were also no differences when comparing cCH with eCH, but CH men consumed more cups of coffee than CH women (5.74 vs. 3.83, p < 0.001).
Energy drink consumption was most prevalent among CH patients, but when comparing the number of units consumed there were no differences between the groups (Table 2). In patients, consumption was equally prevalent in men and women (23.1% vs. 30.5%, p = 0.11) but women consumed more units per day (2.7 vs. 1.8, p = 0.013) and craved it more frequently (37.3% vs 15.7%, p = 0.003). Patients with cCH were more often energy drink consumers than eCH patients (31.5% vs. 22.1%, p = 0.039), they consumed more units per day (2.65 vs. 1.77, p = 0.01) and they tended to crave it more often (30.0% vs. 16.9%, p = 0.054).
Illegal substances
The use of cannabis was rarely reported by both patients and controls (Table 2), but significantly more patients used it regularly (2.25% vs. 0%, p = 0.033). A regular or occasional use of cocaine, amphetamine, gamma-hydroxybutyric acid or other illegal substances was only reported by four patients and one control. Since this was reported by so few, no sub-group analyses were performed.
Comorbid diseases
Risk of lifestyle-related diseases in patients with cluster headache compared with headache-free controls.
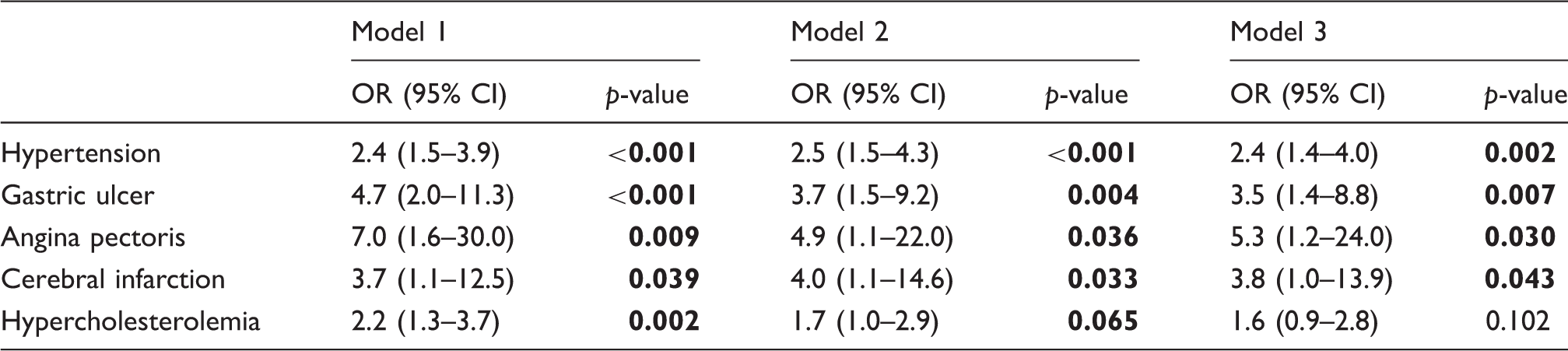
Model 1: Was adjusted for the most basic confounders: Age (±49 years of age), sex (M/F) and group (CH/controls).
Model 2: Was additionally adjusted for potential confounders: Smoking (current or former smoker/never smoker) and educational level (no education obtained/still a student or education obtained).
Model 3: Was additionally adjusted for possible effect modifiers: BMI (±25) and alcohol (±7/14 units/week).
CH: Cluster headache; OR: Odds ratio.
P-values highlighted in bold are significant.
Prevalence of comorbid-diseases in patients with cluster headache and headache-free controls.

CH: Cluster headache.
P-values highlighted in bold are significant.
Discussion
In this large, observational study of 400 well-characterized CH patients and 200 age- and sex-matched controls, we found that unhealthy lifestyle factors were much more prevalent among CH patients than controls. Further, CH was independently associated with several comorbid lifestyle-related diseases.
Smoking
In accordance with previous studies, we found a very high prevalence of current smoking and a history of smoking in CH patients (1–6,8,20,21). Although declining, the prevalence of current smoking is still very high compared with the general population in Denmark (15.9%) (22). Different hypotheses have emerged on the relationship between smoking and CH. Smoking has been suggested to be a risk factor for the development of CH (7,8) because smoking onset usually precedes CH debut (1,23). The fact that nicotine affected trigeminal nociception in healthy people (24) supports this hypothesis. On the other hand, we found that the latency between smoking onset and CH debut was long (15.9 years), smoking cessation has not been found to improve CH (16), and recently a large cohort study found that current and previous tobacco users had a later onset of CH than non-tobacco users (6). Also, not all smokers develop CH and people never exposed to smoking do. On this basis, it seems unlikely for smoking to have a direct causal relationship with CH, but indirect causality cannot be excluded. Pain has been shown to increase the urge to smoke in smokers without pain disorders (25). This resembles the behavior of CH patients in this study, who were found to smoke in close relation to their attacks, typically just after, suggesting that smoking could be a pain-related coping behavior. Possible reward mechanisms triggered by dopamine may alleviate the pain (26) acutely. The long-term effects of smoking may be opposite, since smokers in several studies have a more severe clinical phenotype than non-smokers (16,17,27). Overall, this is in agreement with this study; however, treated attack duration was shorter for patients with a history of smoking compared with never smokers, indicating a better treatment effect in patients exposed to smoking. Altogether, studies exploring the pathophysiological mechanisms of CH and phenotypes are necessary in order to establish the true connection between smoking and CH.
Alcohol
The average alcohol intake was unexpectedly higher in patients compared with controls; however, this seems to be due to a higher frequency of heavy drinkers in the CH population (especially in men and cCH patients). It was also surprising that 14.3% of patients consumed alcohol during their attack periods, as alcohol is a known trigger of attacks (6,28). This counter-intuitive observation has previously been reported in Sweden, where 21% of patients increased or did not change their alcohol consumption during the bout (10). Our post hoc analysis clarifies that alcohol is less often a trigger for those that continue to consume alcohol in the attack periods. A Danish epidemiologic study has also reported that people with sleep problems consume more alcohol than those without sleep problems (29). CH patients have significantly poorer sleep quality compared with controls even a year after the last cluster attack (14), and alcohol might therefore be perceived as an easily available hypnotic.
Eating habits
We found a higher BMI in patients, a consistent finding also when studying men and women separately. To our knowledge, BMI has not previously been assessed in studies of CH patients. The relation of appetite and pain is very complex. Painful stimulation of the dura has been found to suppress appetite, partly mediated through the hypothalamus (30). Hypocretin, secreted from the lateral hypothalamus, has many functions and is involved in sleep-wake regulation, addiction, psychiatric disorders and also in appetite regulation (involved in the reward and motivational aspects of appetite) (31). Patients with narcolepsy type 1, who have low levels of hypocretin-1, have been reported to be more prone to develop obesity and other metabolic diseases (32). Interestingly, patients with CH in the attack phase have been reported to have a marginally lower concentration of hypocretin-1 compared with matched healthy controls (33). Therefore, hypocretin-1 could be a potential link between CH and the observed high BMI in our cohort. In our study, almost 1/5 had food cravings, mostly for sweets and fast food. Eating unhealthy food and perhaps overindulging in food after CH attacks and during bouts could offer another possible explanation for the observed findings.
Caffeine consumption
As in a previous study, overall coffee consumption was similar in patients and controls (9), whereas our finding of equal intake in eCH and cCH was inconsistent with previous reports (1,27). The consumption of energy drinks was higher among patients, especially in male patients. Some patients report that energy drinks consumed in the pre-attack phase could offer some pain relief, but this needs to be confirmed in a formal setup.
Illegal substances
Very few patients and controls reported a regular or occasional use of illegal substances. Most likely, this is due to underreporting.
Comorbid diseases
The presence of comorbid diseases has not been consistently reported in previous studies, but gastric ulcer, depression, chronic fatigue, vertigo, arthritis and back pain have been more frequently reported in CH patients (4,20,34). Other studies, though, did not find an increased prevalence of cardiovascular diseases and gastric ulcer (20,23), which is surprising as patients in these cohorts also had a very poor lifestyle. Unhealthy lifestyle factors are consistent risk factors for the development of cerebro- and cardiovascular diseases, which, in this current study, were more prevalent among patients compared with controls. This might constitute a problem for CH patients because sumatriptan, one of the most effective abortive medications, is theoretically contraindicated if the patient develops a cerebro- or cardiovascular disorder or has a high risk thereof. Verapamil, the first-choice prophylactic therapy, is contraindicated in patients with heart failure, certain arrhythmias and recent myocardial infarction. Lifestyle-related diseases could therefore have serious consequences in the management of the disorder. A strong effort should be made to obtain and act on these unhealthy lifestyle factors among CH patients, the changing of which is always complicated. The primary sector is responsible for the improvement of unhealthy lifestyle factors; however, the motivation may be higher if the patients are informed of possible consequences when seen in the secondary/tertiary sector.
After adjusting for potential confounders and effect modifiers, CH was still associated with lifestyle-related comorbid diseases. The findings are interesting but should be interpreted with some caution, since causality cannot be concluded based upon these data. It is likely that the elevated risks reflect an incomplete adjustment for lifestyle factors, including physical activity and further sociodemographic factors than educational level, which was not possible in this current study. The significant differences in educational level between patients and controls supports that the two populations are not socioeconomically equally distributed.
Overall, the results of this study could be explained by social causation, in which case CH would be more prevalent in people predisposed to an unhealthy lifestyle and lifestyle-related diseases. Hypothetically, patients could have a more addictive personality trait, resulting in a predisposition to overindulge in unhealthy lifestyle factors (35,36), which is supported by the fact that current or former smokers were also more likely to have an unhealthy alcohol intake. Another explanation could be social drifting, where CH is the determinant for a life with unhealthy lifestyle and lifestyle-related disease.
Strengths and limitations
When investigating lifestyle, recall bias is important to consider. People often tend to underestimate or underreport information that makes them appear unhealthy. Participation was not anonymous, which may have biased some participants’ answers in sensitive areas, for instance the use of illegal substances. Participants were matched for age and sex but controlling for socio-economic factors would have been optimal. Another limitation was the length of the questionnaire, since it took patients roughly one hour to complete it. It was decided to include all questions in one large questionnaire to assess all questions of interest. Participation rate was in line with other epidemiological headache studies (37). CH patients were mainly recruited from the Danish Headache Center and participants could reflect a group of more severely affected patients compared to the total CH population. The major strengths of the Danish CH survey are the large number of participants, the diversity of the questions assessed without specific focus on lifestyle factors, that patients were correctly diagnosed, and the fact that all participants underwent a structured interview to resolve ambiguities.
Conclusion
We studied lifestyle factors and comorbid diseases in a large cohort of well-characterized CH patients compared with age and sex matched headache-free controls. The findings were thought-provoking: Unhealthy lifestyle factors in the form of smoking, alcohol overuse and high BMI were significantly more prevalent among patients with CH compared with controls, and CH was associated with several lifestyle-related diseases. As lifestyle-related diseases might have serious consequences in the management of CH, it is key to inform patients at an early time point about the possible risks of their lifestyle choices.
Clinical implications
Unhealthy lifestyle factors such as smoking, drinking alcohol and having a high BMI were more prevalent in cluster headache patients compared with controls. Lifestyle-related diseases were also found to be more prevalent in patients compared with controls. As lifestyle-related diseases might have serious consequences in the management of CH, it is key to inform patients at an early time point about the possible risks of their lifestyle choices.
Author contributions
NL undertook acquisition of data, data analyses and interpretation; AP undertook acquisition of data, and critical revision of the manuscript for intellectual content; AS undertook acquisition of data and critical revision of the manuscript for intellectual content; RJ created the study concept and design, and undertook interpretation of results and critical revision of the manuscript for intellectual content; MB created the study concept and design, and undertook acquisition of data, interpretation of results, and critical revision of manuscript for intellectual content.
Footnotes
Acknowledgements
The authors would like to thank all study participants. Also, special thanks to study nurse Mette Frank Fisker for skilful data acquisition and recruitment and to medical student Rasmus P Beske for recruitment.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NL has received a grant from Tryg Foundation, travel grants from Candy Foundation and iHEAD, and is sub-investigator for studies funded by Autonomic Technologies and Xenon Pharmaceuticals. AP has been sub-investigator for a study funded by Autonomic Technologies. AS has received a grant from Tryg Foundation and travel grants from Candy Foundation and iHEAD, has given lectures for AstraZeneca, and is a sub-investigator on clinical trials sponsored by Eli Lilly. RJ has given lectures for Pfizer, Berlin-Chemie, Norspan, Merck and Autonomic Technologies (ATI); has been a member of the advisory boards of ATI, Medotech and ElectroCore; and a principal investigator on clinical trials sponsored by Eli Lilly. MB has received honoraria from ATI and an unrestricted research grant from AGA, Linde-Gas. RJ has also been a principal investigator for “ATI and Xenon Pharmaceuticals” as well.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Danish Tryg Foundation.
