Abstract
Background
Migraine with aura is independently associated with increased risk of ischemic stroke, especially in younger subjects. This association might be related to an impairment of cerebral autoregulation, which normally maintains cerebral blood flow independent of arterial blood pressure variations.
Methods
Patients aged 30–55, fulfilling ICHD-3 beta criteria for migraine with aura, were prospectively enrolled and compared with gender- and age-matched healthy controls without a history of migraine. Patients and controls with a history of stroke or any disease potentially impairing cerebral autoregulation were excluded. We assessed cerebral autoregulation with two different methods: Transfer function analysis, and the correlation coefficient index Mx. The transfer function phase and gain reflect responses of cerebral blood flow velocities to relatively fast fluctuations of arterial blood pressure, whereas Mx also reflects responses to slower arterial blood pressure fluctuations.
Results
A total of 22 migraine with aura patients (median age [IQR]: 39.5 [12.5] years) and 22 controls (39 [9.75] years) were included. Transfer function parameters and Mx were not different between patients and controls. However, Mx was inversely correlated with age in patients (ρ = −0.567, p = 0.006) and not in controls (ρ = −0.084, p = 0.509). Mx was also inversely correlated with migraine with aura duration (ρ = −0.617, p = 0.002), suggesting improvement of cerebral autoregulation efficiency with disease duration.
Conclusions
Cerebral autoregulation did not differ between patients and controls aged 30–55. However, cerebral autoregulation efficiency was strongly correlated with migraine with aura duration. Further studies in younger patients are needed to determine whether cerebral autoregulation is impaired early in the course of disease.
Trial Registration
NCT02708797.
Introduction
Migraine with aura (MA) is associated with increased risk of ischemic stroke, especially in women and under the age of 45 (1,2). The risk is more pronounced in smokers and in women currently using oral contraceptives. The mechanisms that might account for this association have been of considerable interest. They include, but are not limited to, cerebral hypoperfusion during cortical spreading depression, hypercoagulability, endothelial dysfunction, higher prevalence of patent foramen ovale and interatrial septal aneurysm, and higher incidence of cervical artery dissection (1,3–5). Impairment of cerebral autoregulation (CA) is another possible mechanism that might render MA patients more vulnerable to cerebral ischemia. CA has been the subject of only a few studies in MA patients, with variable findings that may possibly be explained by differences in age of studied populations and in methods used to assess CA (6–8). In the present study, we compared CA in MA patients with healthy volunteers. CA was assessed from spontaneous oscillations of arterial blood pressure (ABP) and cerebral blood flow velocities both in time and frequency domains. Furthermore, we explored the relationship of CA efficiency with age and duration of MA.
Methods
Population
MA patients aged 30–55 were prospectively recruited at the headache outpatient clinic of a university hospital from May 2016 to June 2017. Healthy volunteers without history of migraine, and matched with patients for age and sex, were recruited among the population of the same geographical area. MA, or the absence of migraine history, was confirmed by a trained neurologist using the ICHD III criteria (9).
Patients and controls were free from any previous cardiac or neurological disease (except MA in patients). Patients and controls with any condition potentially disturbing CA (e.g. uncontrolled hypertension, diabetes, sleep apnea syndrome, carotid stenosis) or taking any treatment interacting with cerebral autoregulation (e.g. calcium inhibitors) were not included. MA patients treated with beta blockers were included after a washout period of 72 hours. MA patients had to be free of headache and triptan consumption for at least 8 days before inclusion. All participants were asked to avoid coffee, tea and tobacco during the 24 hours before CA assessment.
Data collected in MA patients included age at migraine onset and aura onset, frequency of migraine attacks in the past 12 months, type of aura (visual, sensory, aphasic or other), duration of aura, and frequency of aura among all migraine attacks.
All patients and controls underwent a carotid and transcranial color-coded ultrasonography examination before CA assessment (IU 22, Philips). Patients without sufficient acoustic temporal bone window or with carotid stenosis of more than 50% were excluded.
Assessment of cerebral autoregulation (CA)
CA was assessed using transcranial Doppler (TCD, Looki 2 TC, Atys Medical) in the supine position, in a temperature-controlled, quiet room, at least one hour after the last meal.
Flow velocity (FV) of the right middle cerebral artery was continuously monitored for 30 minutes at a depth of 50–55 mm, using a 2 MHz probe maintained with a headframe.
Continuous monitoring of arterial blood pressure (ABP) was achieved using a finger plethysmograph (Nexfin, BNEYE). FV and ABP signals were synchronized.
The end-tidal carbon dioxide concentration was measured with a Capnostream 20 (Oribon Medical).
We assessed CA from spontaneous oscillations in FV and ABP using two methods: The mean flow index (Mx), and transfer function analysis (TFA).
Mx is a time-domain evaluation of CA, based on a measure of correlation between mean ABP (MABP) and mean FV (MFV) signals. With efficient CA, Mx will tend to be close to 0, whereas impaired CA will manifest as an increase of Mx toward Mx = 1. An Mx value of 0.3 has been proposed as a threshold for disturbed autoregulation (10,11).
Assessment of CA in the frequency domain with TFA is based on the assumption that CA can be modeled as a linear high-pass filter, allowing rapid changes in ABP to be transmitted to FV, but dampening lower-frequency changes. Three parameters are used to evaluate CA: Coherence, gain and phase. The coherence reflects the degree of linear correlation between the ABP and FV signals. If the coherence is low, there is a degree of non-linearity in the relationship between ABP and FV, and the estimations of gain and phase are not reliable (12). The gain reflects the quantity of ABP variation transmitted to FV. A low gain suggests an efficient CA, whereas impaired CA results in a high gain. The phase measures the delay between the ABP and FV signals. Intact CA is associated with a positive phase shift between FV and ABP. Impaired CA results in a reduced phase shift.
Mx was evaluated using the ICM + software (University of Cambridge, UK). MAP and MFV values were averaged at 10 second intervals. A Pearson’s correlation coefficient was computed using 30 consecutive 10-second intervals. Mx was obtained by averaging the results for each recording session. Sx and Dx were similarly extracted from the systolic and diastolic values of FV and ABP, respectively (13).
TFA was performed using the Notocord software (Notocord Systems, France) (14,15) over a period of 10 minutes with six consecutive 100-second data segments. A Hann window was used to correct the spectral leakage. A Fourier transform was carried out on six successive windows with Welch’s analysis. We assessed CA using the coherence, gain and phase between MFV and MABP signals in the very low frequency (VLF, 0.02 Hz to 0.07 Hz), low frequency (LF, 0.07 to 0.2 Hz), and high frequency (HF, 0.2 to 0.5 Hz) bandwidths. We considered that a coherence < 0.29 reflected an insufficient linear correlation of the signals to allow assessment of CA (16).
Assessment of cerebrovascular reactivity
Cerebrovascular reactivity was evaluated with a voluntary breath holding test of 30 seconds. Apnea was performed after a normal inspiration and was not preceded by hyperventilation or accompanied by a Valsalva maneuver. The quality of apnea was controlled by a sensor of carbon dioxide pressure in the exhaled air at the end of apnea (Capnostream device, Covidien – Medtronic). The cerebrovascular reactivity was calculated as the increase in percentage of basal FV using the value of FV immediately after rebreathing.
Statistical analysis
Given the paucity of previous data on CA in migraine, the present study was exploratory. Statistics were carried out with the R software 3.2.5 using non-parametric tests. Results were expressed as median with interquartile ranges (IQR) or percentages. Continuous variables were compared with the Wilcoxon test and qualitative variables with Fisher’s exact test. Correlations were tested using the Spearman rank correlation coefficient. Differences and correlations were considered as statistically significant when p < 0.05.
Ethics
The study was approved by the local institutional Ethics Committee (Comité de protection des personnes Sud-Ouest I). All participants gave written informed consent.
The study was registered on Clinicaltrials.gov (number of registration: NCT02708797).
Results
Population
Characteristics of patients and controls.

IQR: interquartile range; BMI: body mass index; EtCO2: end-tidal carbon dioxide; HR: heart rate; MABP: mean arterial blood pressure; MFV: mean flow velocity. Values in parentheses are IQR or percentages.
Characteristics of migraine.
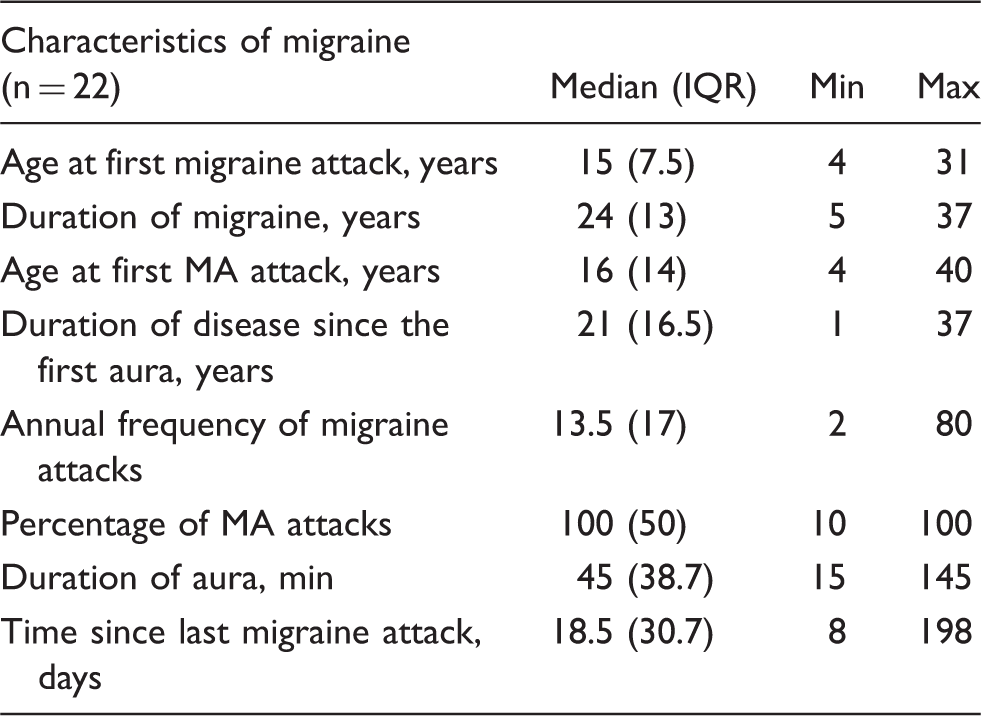
Six patients with migraine had preventive treatment: Beta blockers for two, interrupted for 4 days (propranolol) and 6 days (metoprolol) before inclusion; oxetorone, topiramate, sodium valproate and the valpromide-aspirin combination for the remaining four subjects.
Cerebral autoregulation
CA in MA patients and controls. Values are medians (IQR).

The TFA method did not show any difference in ABP power, FV power, coherence, gain, and phase between patients and controls in any frequency bandwidth.
Cerebrovascular reactivity
The breath holding test showed similar cerebrovascular reactivity in patients and in controls: 37.1 % (0.21) and 36.3% (0.13), respectively (p = 0.57).
Correlation of CA with age and migraine duration
A negative correlation was found between Mx and age in MA patients (ρ = −0.567, p = 0.006), but not in controls (ρ = −0.084, p = 0.509). There was also a strong negative correlation between Mx and the duration of MA (ρ = −0.617, p = 0.002) (Figure 1). A similar correlation was found with Dx (ρ = −0.491, p = 0.020) but not with Sx (ρ = −0.053, p = 0.814). Those correlations were not found with TFA.
Correlation of Mx with duration of MA.
The frequency of migraine attacks, and time since the last attack, were not correlated with CA (data not shown).
Discussion
This study did not show any difference between MA patients and healthy volunteers for CA efficiency both in time and frequency domains. We found, however, a correlation between the duration of MA and CA efficiency in the time domain.
Only a few previous case-control studies have analyzed CA in migraine with variable findings. In a study assessing CA in the frequency domain in 33 healthy controls (mean age ± SD: 36 ± 13 years) and in 22 patients with migraine (mean age: 39 ± 7 years), including 12 MA patients, large differences were found between patients and controls. The usual phase lead of cerebral blood flow velocity (CBFV) over ABP was absent in patients, indicating lack of CA; but, surprisingly, the gain between CBFV and ABP was reduced to almost zero in patients, suggesting efficient CA. These discrepant results may possibly be explained by some methodological reasons, because the spectral power density of ABP and CBFV were much higher in patients compared with controls (6). In another study assessing CA in time and frequency domains in middle-aged subjects, no differences were found between 19 migraineurs (mean age: 54 ± 16 years) and 75 non-migraineurs (mean age: 59 ± 13.1 years) (7). A subsequent study by the same group in younger subjects (mean age: 28.7 ± 7.2 years), including 17 MA patients, 17 patients with migraine without aura and 35 healthy volunteers, showed that MA was associated with less efficient CA in the time domain (Dx), but not in the frequency domain (8). CA of patients with migraine without aura was not different from controls. We could not confirm a less efficient CA in MA patients either in the time or frequency domain. However, we found a strong correlation between age and CA efficiency in MA patients and not in controls. There was also a highly significant correlation between MA duration and CA efficiency among patients. These correlations were found with CA assessed in the time but not in the frequency domain. It should be noted that the 22 MA patients from the present study (mean age: 39.5 ± 12.5 years) were 10 years older than those included in the previous study demonstrating less efficient CA in MA patients.
Taken together, these findings suggest a transient impairment of CA in MA with less efficient CA early in the course of disease or in younger patients, subsequently improving in older patients. We acknowledge that this hypothesis has to be confirmed by further studies in young subjects. It is, however, consistent with the observation that the risk of stroke associated with MA is greater in younger subjects (1) and during the short term after diagnosis of MA than the long term (17,18), as well as with the lack of additional silent brain infarcts among MA patients from the CAMERA study reevaluated 9 years after baseline MRI (19).
Interestingly, the previously reported reduced efficiency of CA in young MA patients and the correlation of CA with MA duration in the present study were noted only in the time domain and not in the frequency domain. It is likely that time-domain and frequency-domain methods reflect different aspects of CA. In particular, Mx and Dx describe slower components of CA than those assessed with TFA. In the present study, the frequency of slower waves analyzed by TFA was 0.02 Hz, corresponding with a period of 50 s, while Mx analyzed correlation of signals over 300 s. Previous work in the setting of traumatic brain injury showed that such differences were clinically relevant as MX, but not TFA, correlated with the patients’ outcome (20).
The strengths of this study are its prospective design, with the inclusion of MA patients and healthy volunteers matched for age and gender, and the use of two different validated methods for CA assessment. Patients were evaluated away from any headache period, making changes related to migraine attack unlikely. Its main limitations are the small sample size, although the number of included MA patients was greater than in previous similar studies, and the lack of inclusion of patients younger than 30. Although the medications possibly interfering with CA were interrupted several days before assessment, we cannot exclude a residual effect of beta blockers in two MA patients treated with these drugs.
Conclusion
CA did not differ between MA patients and controls aged 30–55. However, CA efficiency was strongly correlated with MA duration. Further studies in younger subjects are needed to determine whether CA is impaired early in the course of disease.
Clinical implications
This study showed no difference in CA between MA patients aged 30–55 and healthy controls. There was, however, a strong correlation between CA efficiency and MA duration. Further clinical research is warranted in younger MA patients to determine whether CA is impaired early in the course of disease.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
