Abstract
The aim of this study was to investigate the prevalence of patent foramen ovale (PFO) in a consecutive unselected cohort of migraine patients (with and without aura) and compare it with a group of ischaemic young and elderly stroke patients. One hundred and forty-one migraine patients were compared with 330 stroke patients (130 young patients; 200 elderly patients) selected from our hospital stroke data bank. PFO was assessed with transcranial Doppler sonography with i.v. injection of agitated saline. The prevalence of PFO was 51.7± in migraine with aura (MA) patients, 33.7± in migraine without aura (MoA) patients, 33.8± in young stroke patients and 20.5± in elderly stroke patients (P < 0.001). The prevalence of PFO in cryptogenic stroke in young and elderly stroke patients was, respectively, 41.1± and 25± (P = 0.04). The difference between MA and MoA patients was significant (odds ratio = 2.1). The prevalence of PFO in MA patients is higher than in MoA patients and in young cryptogenic stroke patients.
Introduction
Right-to-left shunt, usually due to patent foramen ovale (PFO), is a well-established risk factor for ischaemic stroke in young patients (1), while the role of PFO as an independent factor in migraine is still debated. Atrial septal aneurysm (ASA) has also been identified as a potential risk factor for stroke (2). Migraine has a high prevalence rate in the general population and accounts for significant morbidity, lost productivity and healthcare visits. Migraine with aura (MA) has been associated with a 1.5-fold increase in the risk of total and a 1.7-fold increase in the risk of ischaemic stroke (3).
Increasingly, emerging data show a link between migraine headache, especially MA, and PFO. Recent studies have reported an increased prevalence of PFO in caucasian MA patients (4–8). Whether the relationship between PFO and migraine is causal and, if so, the precise mechanisms are still a matter of debate. Potential mechanisms in stroke and migraine patients include paradoxical embolism from a venous source, direct embolization from thrombi formed within the PFO, and thrombus formation caused by atrial arrhythmias, such as paroxysmal atrial fibrillation (9, 10).
The prevalence of PFO in migraine and stroke patients has been poorly studied in South-America. We hypothesized that PFO could be a risk factor for aura in migraine with aura patients. The aim of our study was to investigate the prevalence of PFO in a consecutive unselected cohort of migraine patients with and without (MoA) aura in Brazil. The secondary objective was compare PFO prevalence data in migraine patients with young and elderly stroke patients.
Methods
Patients
A total of 141 migraine patients (115 women, 26 men), consecutively admitted to the neurology out-patient clinic between July 2004 and June 2005, were studied. Data regarding prevalence of PFO in migraine patients were compared with 330 stroke patients from an ongoing prospective hospital stroke registry. For the purpose of this study we included 130 young ischaemic stroke patients (mean age 33.8 years, range 15–45 years; 58.5% females) consecutively admitted to the Neurology Department between January 2002 and December 2003 (young stroke data bank) and a group of 200 stroke patients aged >45 years admitted during the same time (mean age 61.5 years; 46.5% females).
The aetiological classification of stroke followed the TOAST criteria (11). Cryptogenic stroke was defined by a normal carotid Doppler, normal magnetic resonance angiography, absence of cardioembolic pathology on transthoracic and/or transoesophageal echocardiography except a PFO, absence of arrhythmia on eletrocardiography and a normal blood work-up including coagulation tests. All the subjects underwent transcranial Doppler (TCD) with injection of contrast. All stroke patients with a positive TCD underwent a transoesophageal ecocardiography.
Assessment of migraine
Information about migraine was obtained by an interview using a structured face-to-face questionnaire before the TCD. Diagnosis of MA and MoA was established by at least two independent neurologists. Data regarding the following characteristics of migraine in each migraine patient were obtained: presence of aura, unilateral location of pain, pulsating quality, interference with daily activities, duration of 4–72 h, aggravation by routine physical activity, nausea or vomiting, sensitivity to light, sensitivity to sound, tingling or numbness, difficulty with speech, dizziness or vertigo, or unilateral weakness in face, arms or legs. Additional information was obtained about the type of drugs used to treat migraine, presence of triggering factors, family history, age at onset and frequency of the attacks in the last 4 weeks. Patients who reported the presence of aura were classified as having MA.
The diagnosis of MA or MoA was based on the criteria adopted by the International Headache Society. Diagnostic criteria for migraine with typical aura was done according to the International Classification of Headache Disorders, 2nd Edition (ICHD-2) (12): (i) at least two attacks fulfilling criteria B–C; (ii) aura consisting of at least one of the following but no motor weakness: (a) fully reversible visual symptoms including positive features (i.e. flickering lights, spots, lines) and/or negative features (scotoma); (b) fully reversible sensory symptoms including positive features (i.e. pins and needles) and/or negative features (numbness); (c) fully reversible dysphasic speech disturbance; (iii) at least two of the following: (a) homonymous visual symptoms and/or unilateral sensory symptoms; (b) at least one aura symptom developing gradually over ≥5 min and/or different symptoms occurring in succession over ≥5 min; (c) each symptom lasts ≥5 min and ≤60 min.
Patients with MoA who many years ago had once had a visual disturbance and were otherwise always MoA were classified as MoA patients. A non-aura reversible visual disturbance was considered in these patients. Migraine attacks had to be preceded by an aura on at least two occasions to be classified as MA. Isolated or single episodes of visual disturbance were not considered as MA.
Investigators were asked to differentiate between MA and MoA and were blinded to the presence of PFO. Informed consent was obtained from migraine patients. Investigators were not asked to differentiate MA from MoA in stroke patients in the hospital young stroke registry, so no data concerning migraine subtype were available.
Diagnosis of PFO
Diagnosis of PFO was done using a trancranial Doppler instrument (Nicolet Pioneer TC4040). The TCD-microbubble test technique consists of the injection of a contrast-enhancing agent into a peripheral vein while recording the flow velocity of the middle cerebral artery (MCA) at 56 mm depth with a hand-held transcranial Doppler probe placed on the temporal bone (13). Bubble contrast, shaken saline so as to generate air microbubbles, was produced by pushing 9 ml of sterile saline (0.9% NaCl) and 1 ml of air back and forth between two syringes connected by a three-way tap until there were no visible bubbles. This mixture was injected through an 18-G butterfly needle into an antecubital vein. The test was performed three times: the first contrast injection was performed with the patient resting and breathing normally; two subsequent injections were performed with sudden release of the Valsalva manoeuvre. A criterion for diagnosing PFO was the presence of at least three microbubbles within a 20-s interval after injection (13).
TCD for detecting microembolic signals after injection of air-mixed saline has been previously validated and provides 90% sensitivity and 100% specificity, with an overall diagnostic accuracy of 95%, in comparison with transoesophageal echocardiography (14, 15). The size of the shunt was graded according to the maximum number of microbubbles (MB) observed: small shunts 3–10
Statistical analysis was performed using the Statistical Package for Social Sciences (SPSS computer program, release 9.0; SPSS, Chicago, IL, USA). Univariate statistical analyses were performed with χ2 and odds ratio (OR) calculations. Values of P < 0.05 were considered to be statistically significant. The association between migraine and PFO was done through logistic regression analysis with migraine as a dependent variable and age, sex and PFO as independent variables.
Results
Migraine patients
One hundred and forty-one consecutive migraine patients (mean age 33.1 years; 81.6% females) were studied. Fifty-eight patients (41.1%) reported an aura preceding the migraine attack; auras were more common in men than in women (57.6% vs. 37.3%; P = 0.05). The most frequently observed aura was the visual type in 78.9%. Migraine onset was 3 years before in migraine with aura patients (15.8 vs. 18.8 years; P = 0.05). The clinical characteristics of the migraine population are shown in Table 1. A PFO was observed in 41% of migraineurs: 29 MoA patients (33.7%) and 30 MA patients (51.7%). This difference was statistically significant [OR = 2.1, 95% confidence interval [CI] 1.06, 4.18, χ2 = 9.52, P = 0.002).
Clinical characteristics of migraine population patients (n = 141)

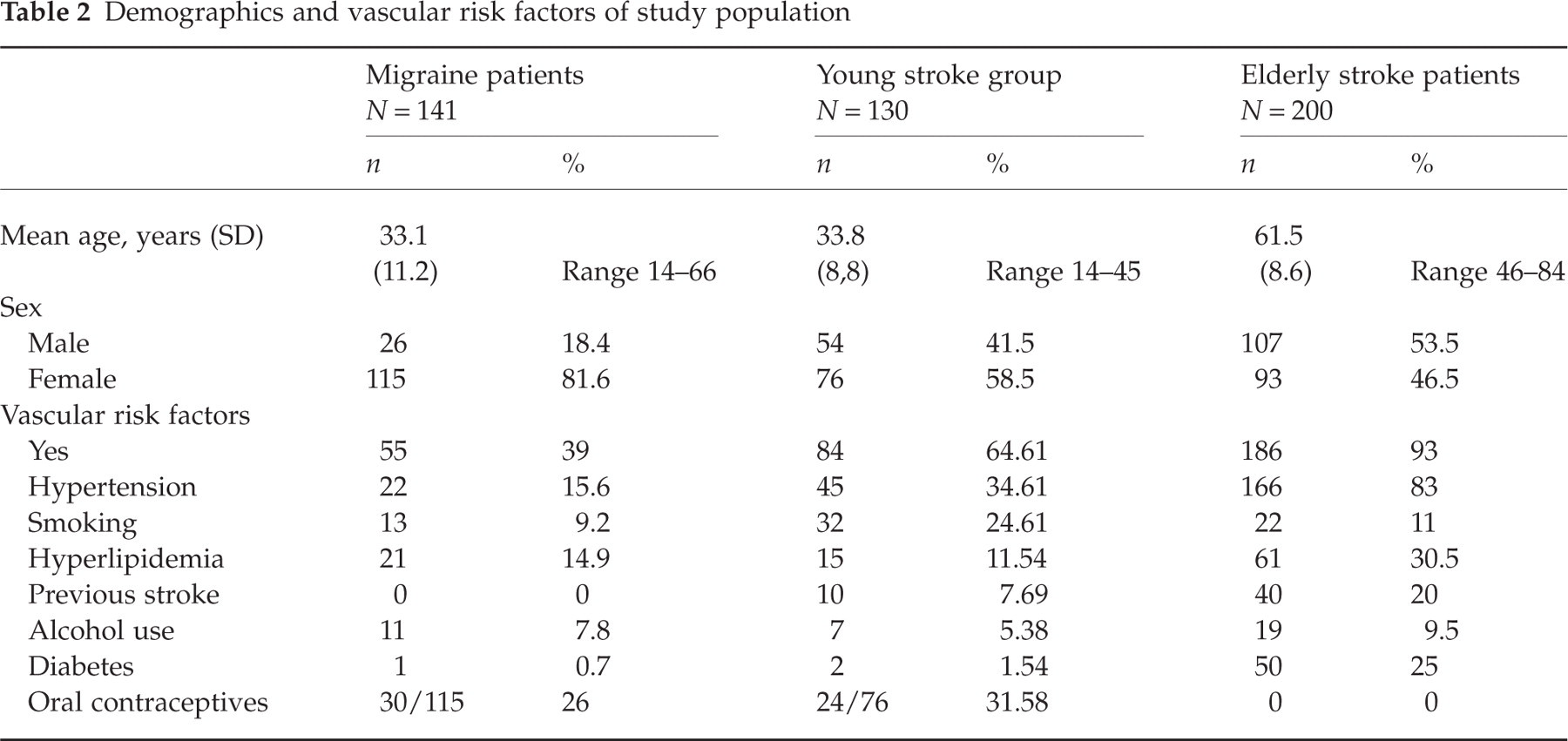
Thirty-eight percent of MA and 40% of MoA patients had at least one vascular risk factor (VRF). No differences were observed in VRF frequency between MA and MoA patients. VRFs in stroke and migraine patients are shown in Table 2. No association was found between the presence of a PFO and smoking or oral contraceptive use in MA patients. Logistic regression analysis showed that younger age onset, male sex and PFO were significantly associated with MA.
Demographics and vascular risk factors of study population
The pattern of microbubbles on TCD was analysed in migraine patients at rest and during two consecutive Valsalva manoeuvres. Of PFO+ migraine patients, 65.5% had a functioning PFO at rest. PFO was functioning at rest in a higher proportion of MA patients (73.3%) than of MoA patients (57.1%). However, this difference was not significant (χ2 P-value = 0.19). The frequencies of microbubble patterns during the Valsalva manoeuvre in MA and MoA patients were: (i) <10 MB (MA, seven patients, 23.3%; MoA, 10 patients, 35.7%); (ii) shower pattern (MA, 14 patients, 46.7%; MoA, 10 patients, 35.7%); (iii) massive or curtain pattern (MA, nine patients, 30%; MoA, eight patients, 28.6%). Differences in microbubble distribution during the Valsalva manoeuvre were not statistically significant (P = 0.552).
Stroke patients
Three hundred and thirty stroke patients underwent a TCD. The mean ages in young and old stroke patients were 33.8 and 61.5 years, respectively. Forty-two young stroke patients had a PFO (32.3%; 16 males and 26 females), whereas 41 patients in the elderly group (20.5%; 18 females and 23 males) had a PFO (P = 0.02; OR 1.85, 95% CI 1.09, 3.16). Stroke was classified as cryptogenic in 90 young stroke patients (69.2%) and in 64 elderly stroke patients (32%) (P = 0.001). PFO prevalence was higher in cryptogenic stroke: a PFO was observed in 41.1% of young stroke patients and in 25% of elderly patients with cryptogenic stroke (P = 0.04; OR 2.06, 95% CI 0.96, 4.59). A progressive decline in PFO prevalence was observed in stroke patients with age (from 46.7% at age 16–25 to 15.4% at age 76–85 years). The PFO prevalence in migraine and stroke groups is shown in Table 3. Stroke onset in young PFO patients was 3 years earlier than in young PFO– patients (31.4 vs. 34.7; P = 0.05).
Patent foramen ovale prevalence in migraine and stroke patients

Stroke subtypes in young PFO+ patients were: cardioembolism (n = 2, 4.76%), large artery atherosclerosis (n = 1, 2.38%), small vessel disease (n = 0), cryptogenic (n = 37, 88%) and other causes (n = 2, 4.76%). No statistically significant relationship was observed between the presence of traditional vascular risk factors (hypertension, diabetes, arrhythmia, dislipidaemia) and the presence of PFO in young or old stroke patients.
Of young stroke patients, 13.8% (17 females and one male; mean age 33.5 years) suffered from migraine; cryptogenic stroke was diagnosed in 17 young patients. Of young stroke patients with migraine, 55.6% had a PFO, whereas 28.6% of non-migrainous stroke patients had a PFO.
Of young cryptogenic stroke patients, 18.5% suffered from migraine; 23.8% of young PFO stroke patients (10/42) suffered from migraine while 9.1% of young non-PFO stroke patients had migraine (OR 3.13; 95% CI 1.02, 9.69; χ2 5.16; P = 0.02). Migraine, aetiology of stroke subtype (cryptogenic stroke) and absence of hypertension (P = 0.02; Fisher's 0.03) were more prevalent in the young stroke patient group.
Discussion
TCD is a very sensitive tool for detecting microembolic signals in the brain vessels after injection of air-mixed saline in case of right-to-left shunt. The major finding of the present study was that PFO can be found in more than half of MA patients in a Brazilian population. It is not known if the association between PFO, MA and stroke is causal or coincidental. The prevalence of PFO is high in the general population (a transoesophageal echocardiography study found a PFO in 25.6% of healthy subjects) (16), as is migraine (10%) (17). In a study of 965 autopsy patients, a PFO was found in 27.3% of the hearts with a progressive decline from 34.3% in the first three decades to 20.2% in the ninth and tenth decades of life (18). We have also observed a progressive decline in PFO prevalence in stroke patients with age. A hypothesis that could explain the decline of PFO frequency with increasing age might be a late anatomic closure of PFO related to chronic heart disease.
The magnitude of the observed association between PFO and MA is consistent with the results from previous studies in caucasian populations. Three studies have previously investigated the relationship between PFO and migraine using TCD with i.v. injection of agitated saline. Del Sette et al. (4) evaluated 44 MA patients compared with 73 patients under 50 years with focal cerebral ischaemia and 50 controls. The prevalence of PFO was significantly higher in MA patients (41%) and stroke (35%) than in controls (8%). Anzola et al. (5) performed a case–control study including 113 consecutive MA patients, 53 MoA patients and 25 age-matched non-migraine subjects. The prevalence of PFO was 48% in MA patients, 23% in MoA patients and 20% in controls (OR 3.13). Domitrz et al. (8) assessed 62 patients (48 females) suffering from MA, 60 from MoA and 65 normal controls. The presence of PFO was found in 53% of MA patients compared with 25% of MoA patients and 25% of control subjects. These findings are in accordance with our study reporting a significantly higher frequency of PFO in non-caucasian MA patients compared with MoA patients. We have found PFO in MA patients (51.7%) at a frequency higher than encountered in young (41.1%) and elderly cryptogenic stroke patients (25%).
More than half of the young stroke patients with migraine had a PFO in our non-caucasian population. The prevalence of migraine is higher in cryptogenic stroke patients with PFO than in the general population. Of cryptogenic stroke patients in our study, 18.5% suffered from migraine. Sztajzel et al. (7) reported a prevalence of migraine in cryptogenic stroke of 27% in Switzerland. The Patent Foramen Ovale and Atrial Septal Aneurism study (1) examined the prevalence of migraine in a cohort of 581 young patients with cryptogenic stroke. In this cohort the prevalence of migraine was nearly double among those with a PFO vs. those without (27% vs. 14%; OR 1.75, 95% CI 1.08, 2.82; P = 0.0001). These are similar results to those obtained in our South American study (23.8% vs. 9.1%; OR 3.13).
Recent studies have suggested that transcatheter closure of the PFO might reduce migrainous symptoms (19–22), whereas PFO closure seems to have no effect on headache frequency in patients with non-migrainous headache. All these studies, however, have major methodological limitations: (i) migraine improves spontaneously with age; (ii) none of the studies mentioned above had a control group; (iii) any kind of intervention has a high placebo response in migraine, which can reduce migraine frequency by up to 70% (23); (iv) aspirin has a modest migraine prophylactic activity, at least in men (24); (v) retrospective collection of headache data is highly unreliable; (vi) recall bias may have a major influence on the results.
Oral anticoagulation has been reported to improve migraine in case reports and observational studies (25), suggesting a potential role for paradoxical embolism. However, a recent randomized, open, cross-over study in migraine patients has shown that low-intensity acenocoumarol treatment has no prophylactic effect in migraine patients (26).
It has also been hypothesized that a particular genetic substrate might determine both atrial septal abnormalities and migraine. PFO and MA can be inherited and transmitted simultaneously (27). At present, three genes are related to atrial septal abnormalities (GATA4, NKZ2-5 and TBX5) and none of them has been linked to migraine (28, 29). Another hypotheses is that PFO might allow trigger substances of migraine (such as vasoactive chemicals) in the venous blood to bypass the pulmonary filter and to reach the systemic circulation in amounts large enough to induce a migraine attack.
An increased propensity to paradoxical cerebral embolism may explain the higher prevalence of PFO in MA and in young cryptogenic stroke patients compared with the normal population. The PFO–migraine connection is, at present, an epidemiological association without a clear physiological explanation. Further studies are needed to elucidate the relationship between MA, PFO and other septal abnormalities such as atrial septal aneurysm.
Conclusions
The prevalence of PFO in patients with MA is significantly higher than in young and elderly patients with stroke.
