Abstract
Aim
Migraine with aura is a risk factor for ischemic stroke. To further assess this risk factor in relation to ischemic stroke, along with other risk factors, we created the migraine associated risk of stroke score (MARS+), making it applicable to migraineurs. The risk score includes vascular risk factors, migraine characteristics and medications used in migraine patients.
Methods
We prospectively evaluated participants in Atherosclerosis Risk in Communities Cohort (ARIC) with a history of migraine. In this population, we tested the association of potential risk factors for ischemic stroke using a Cox proportional hazards model. The coefficient of each variable was divided by the lowest β value and rounded to the nearest integer. The sum of the weighted score of the reported risk factors was found and categorized into two prognostic groups.
Results
We assessed migraine characteristics (aura, migraine frequency and duration) and medications that were in current use by participants, mean ± SD age 58 ± 5.5 years, 86% white, 14% black and 77% women (ergot alkaloids, triptans, hormone replacement therapy, sympathomimetics, steroids, selective serotonin reuptake inhibitors and opioids), in addition to traditional risk factors. Based on the points derived from the significant factors we assigned age ≥65 years = 1, non-white race = 2, hypertension = 2, diabetes = 3, body mass index ≥30 = 2, atrial fibrillation = 2, use of steroid medications = 3, use of selective serotonin reuptake inhibitor medications = 1, opioids = 2, presence of aura = 2 and duration <5 years = 1 to total 21 points. A cut-off of MARS+ ≥5 was considered as a lifetime high risk for ischemic stroke based on receiver operating characteristic curve and Youden's index. Of the 1485 participants with migraine, 112 had an ischemic stroke. MARS+ ≥5 revealed a hazard ratio of 4.09 (95% confidence interval = 2.67–6.26).
Conclusions
The MARS+ score is used to predict ischemic stroke in middle-aged migraine sufferers. This risk score, in addition to generalizability, includes factors such as migraine characteristics and medications that may increase stroke risk.
This is a visual representation of the abstract.
Introduction
Migraine is a common, chronic neurologic disorder characterized by episodic attacks that can significantly impact a patient's quality of life. Approximately 6% of men and 18% of women in the USA suffer from migraine (1–3). There are two common subtypes of migraine: migraine with aura and migraine without aura. Aura is commonly characterized by visual symptoms, but can include motor, sensory or language symptoms. Aura is experienced by up to one third of patients who suffer with migraine before or during the migraine (4). The estimated population suffering from migraine with aura in the USA is 7.4–11 million people (5), with women disproportionately affected (6).
Individuals with migraine are known to also have significant comorbidities, including chronic pain syndromes, depression and anxiety, and vascular risk factors, such as hypertension (HTN), hyperlipidemia and diabetes mellitus (DM) (7). Additionally, migraine with visual aura has been associated with an increased risk of ischemic stroke (8,9). In the USA, approximately 800,000 strokes occur each year, 87% of which are ischemic. Stroke is the leading cause of disability and the fifth leading cause of death (10). In the Atherosclerosis Risk in Community (ARIC) study population, there was an association between migraine with visual aura and risk of ischemic stroke; however, it is unclear how migraine with aura increases risk for ischemic stroke (6). There is a clinical need to risk stratify migraine with aura patients, as well as patients with migraine without aura, to better identify their stroke risk.
To address this need, we created the Migraine Associated Risk of Stroke score (MARS+), which used a cohort of well-characterized patients (ARIC) who have migraine with and without aura to design a risk score to predict ischemic stroke in these patients. We sought to identify whether specific risk factors, including migraine characteristics, placed those who suffer from migraine at an increased risk of ischemic stroke. We also investigated whether certain medications taken by those with migraine with or without aura present a significantly increased risk of ischemic stroke given that certain medications have been proposed to cause an increased risk of ischemic stroke in previous studies (11,12). Our risk score, Migraine Associated Risk of Stroke score (MARS+), could be used by clinicians not only to identify migraine patients at an increased risk of stroke, but also to aid in guidance for stroke prophylaxis. Additionally, this risk score could be utilized by policymakers for allocating health care resources to assess stroke risk in the population of patients who have migraine.
Methods
Study population
The cohort of the ARIC study began in 1987 to analyze atherosclerosis causes and clinical outcomes. In total, 15,792 participants ages 45–64 years old were enrolled in the study. Participants came from four different US communities (Forsyth County, North Carolina; Jackson, Mississippi; suburbs of Minneapolis, Minnesota; and Washington County, Maryland). The first visits occurred from 1987–1989 to collect participant baseline information. The follow-up visits were scheduled as follows: visit 3 (1993–1995), visit 4 (1996–1998), visit 5 (2011–2013), visit 6 (2016–2017), visit 7 (2018–2019), visit 8 (2019–2020), visit 9 (2020–2022), visit 10 (2023) and visit 11 (2024–2025), with visit 12 planned for 2026. Participants were assessed for vascular risk factors and other health issues during each visit (6,13). Hospital medical records were obtained if vascular hospitalizations or stroke occurred.
All participants who completed third clinic visit procedures (1993–1995) in the ARIC study and assessed with headache history questionnaire were included in our study. Subjects with prior history of ischemic stroke at visit 3 were excluded. The study was approved by the institutional review boards of all participating institutions, and a written informed consent was obtained from all participants for the ARIC study.
Assessment of migraine
A headache history questionnaire was administered to ARIC study participants during visit 3 procedures. Modified International Classification of Headache Disorders, 3rd edition (ICHD-3) (beta version) diagnostic criteria were used to differentiate migraine with visual aura and migraine without visual aura subtypes based on this questionnaire (6). Migraine was defined by participant responses endorsing headaches lasting at least four hours or longer in duration, accompanied by symptoms of throbbing or pulsating pain, and were predominantly on one side. Additionally, headaches were characterized by photophobia and phonophobia or nausea or vomiting. Headache was separated into two categories: migraine with visual aura and migraine without visual aura. Migraine with aura participants reported seeing spots, jagged lines, or heat waves in one or both eyes lasting for at least five minutes and preceding onset of headache. Participants with migraine that did not report migraine with aura criteria were categorized into the migraine without visual aura category. Participants with headaches that lasted more than four hours that did not meet requirements for either category were classified as non-migraine headaches. All participants who experienced migraine, with or without aura, were included in this study. The number of years of recurrent migraine headaches, as well as, the number of such headaches over the past year were assessed at the third visit based by means of a questionnaire (9). A duration of onset of less than five years was chosen because our prior studies support recent onset migraine as a risk factor for stroke.
Ascertainment of ischemic stroke
Reports of a computed tomography (CT) scan or magnetic resonance imaging (MRI) of the brain were reviewed for stroke diagnosis and exclusion of non-vascular reasons for neurologic symptoms. Stroke diagnosis was based on computer-derived diagnosis and physician medical record review with differences adjudicated by a second physician reviewer. Verified incident ischemic strokes included all the events occurring between the date of the third clinic visit (1993–1995) and December 2019. Ischemic stroke diagnosis required evidence of sudden or rapid onset of neurologic symptoms lasting 24 hours or leading to death in the absence of evidence for a non-stroke cause or hemorrhage in neuroimaging studies (CT or MRI) (6).
Other variables of interest
We considered the following traditional cardiovascular risk factors as potential risk factors for ischemic stroke among participants with migraine: age, sex, race (categorized as white, black, or other), smoking status, alcohol use, atrial fibrillation, coronary artery disease (CAD) and congestive heart failure (CHF). These were assessed by self-report. Body mass index (BMI) was calculated as weight (kg)/height (m2). Hypertension was defined as a systolic blood pressure of 140 mmHg or higher, a diastolic blood pressure higher than 90 mmHg, or use of medications to treat HTN. Diabetes was defined as a fasting glucose ≥126 mg/dL or non-fasting glucose ≥200 mg/dL, self-report of a diagnosis of diabetes by a physician or current diabetes medication use (14).
History of smoking included both former and current smokers. Alcohol use includes current alcohol users. Prevalent CAD was defined by electrocardiographic evidence of previous myocardial infarction (MI), history of physician diagnosed MI or previous coronary revascularization procedure (bypass, angioplasty). Atrial fibrillation was determined on ECG using prior defined criteria (15). Medications obtained via questionnaire during visit 3 were also evaluated to determine whether certain groups increased risk of ischemic stroke in those with migraine. These medication groups included triptans, ergots, sympathomimetics, steroids (low dose, chronic use), hormone replacement therapy (HRT), opioids and selective serotonin reuptake inhibitors (SSRIs) (11). HRT included estrogen and progesterone therapies. Sympathomimetics included medications containing pseudoephedrine, phenylpropanolamine, phenylephrine and phentermine.
Statistical analysis
We prospectively evaluated participants in the ARIC cohort with history of migraine with and without visual aura (n = 1485). In this population, the association of potential risk factors for ischemic stroke was tested using a multivariate Cox proportional hazards model. This analysis created a prevalence statistic, hazards ratio (HR), β regression coefficient for each risk factor. For this analysis, those with 95% confidence intervals >1.0 were used (16). Assignment of points to risk factors was based on a linear transformation of the corresponding β regression coefficient. The coefficient of each variable was divided by 0.46 (the lowest β value, corresponding to SSRI use) and rounded to the nearest integer. For each participant, the sum of the weighted score of their reported risk factors was found and they were categorized into two prognostic index groups. Discriminative value to ascertain these two groups was determined via HR increment and discriminant analysis using area under the curve (AUC). The prognostic index contained two groups based on sum risk factor points: low risk (0–4 points) and lifetime high risk (≥5 points). We then performed survival analyses by Kaplan–Meier survival curves for ischemic stroke in subjects categorized according to MARS dichotomized 0–4 and ≥5.
Cox proportional hazards models were used to assess the HR and 95% confidence interval (CI) of incident ischemic stroke. Because death from any cause would be a competing risk for stroke, we used the cause specific HR to estimate sub-distribution relative hazards of ischemic stroke. All the data analyses for this study were conducted utilizing SAS, version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Participant selection
All ARIC participants who completed the third clinic visit procedure (1993–1995) were included in our study (n = 12,882). Within this cohort, 1545 had migraine with or without aura. We excluded 60 of these participants secondary to missing data thus we evaluated 1485 participants in our study (Figure 1). The participants had a mean ± SD age of 58 ± 5.5 years, 86% were white, 14% were black and 77% were women.

Strobe diagram.
Clinical and demographic characteristics of study participants
The prevalence of twenty-three independent stroke risk factors was assessed: age ≥65 years, sex (female), race (non-white), HTN, DM, BMI ≥30, CAD, atrial fibrillation (AFib), CHF, smoking history, alcohol use disorder, hypercholesterolemia, HRT, current use of orally administered sympathomimetics, use of ergots, use of triptans, use of steroids, use of selective SSRIs, use of opioids, presence of visual aura, number of migraine attacks per year (≥12), duration of presence of migraine (onset <5 years), and heart rate variability measured as the standard deviation of normal-normal intervals less than 50 milliseconds.
Results of the Cox proportional hazard analyses
The HR was found to be significantly high for eleven of these risk factors, including age ≥65 years, race (non-white), HTN, DM, BMI ≥30, AFib, opioid use, use of steroid medications, use of SSRI medications, presence of visual aura, and duration of migraine <5 years as shown in Table 1. The prevalence, HR and beta regression coefficient of these risk factors can be found in Table 2. Hypertension was found to have the highest prevalence of 39%. The use of steroid medication was found to have the lowest prevalence of 2%. Diabetes mellitus was found to have the highest HR of 3.93. Age ≥65 years had a HR of 1.76. Race (non-white) had a HR of 2.11. Hypertension had a HR of 2.77. BMI ≥30 was found to have a HR of 2.19. AFib was found to have a HR of 2.65. The use of steroid medications and opioid medications had HR of 3.42 and 2.14, respectively. The use of SSRI medications had a HR of 1.59. The presence of visual aura had a HR of 2.25. Finally, duration of migraine less than 5 years had a HR of 0.91.
Population characteristics of the development cohort and crude association of potential prognostic determinants with stroke.
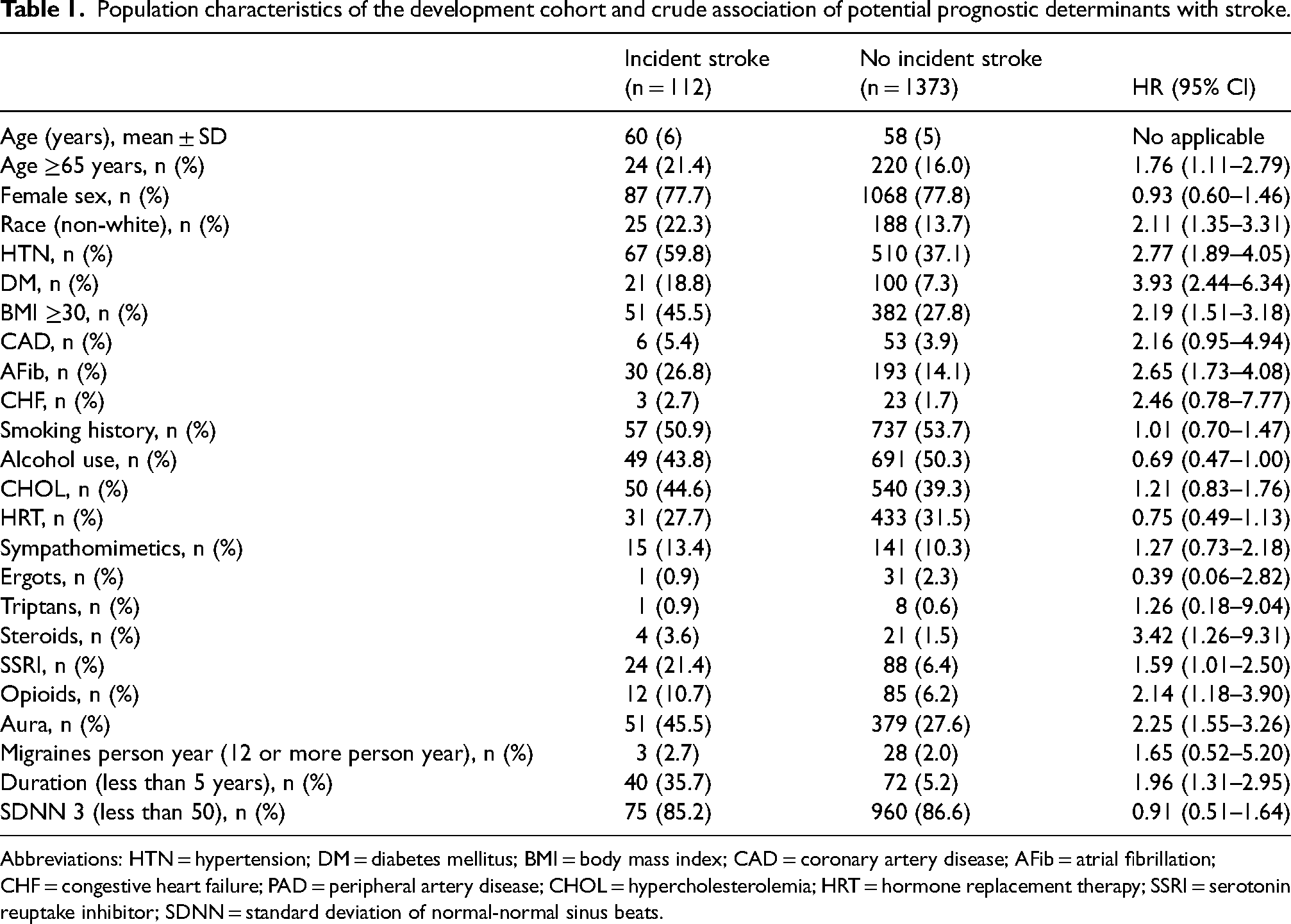
Abbreviations: HTN = hypertension; DM = diabetes mellitus; BMI = body mass index; CAD = coronary artery disease; AFib = atrial fibrillation; CHF = congestive heart failure; PAD = peripheral artery disease; CHOL = hypercholesterolemia; HRT = hormone replacement therapy; SSRI = serotonin reuptake inhibitor; SDNN = standard deviation of normal-normal sinus beats.
Multivariate cox proportional-hazards analysis of the development cohort and scoring system.

Assignment of points to risk factors was based on a linear transformation of the corresponding β regression coefficient. The coefficient of each variable was divided by 0.46 (the lowest β value, corresponding to SSRI use), multiplied by a constant (1), and rounded to the nearest integer.
Abbreviations: HTN = hypertension; DM = diabetes mellitus; BMI = body mass index; AFib = atrial fibrillation; SSRI = selective serotonin reuptake inhibitor; SEM = standard error of mean.
Clinical score development and outcomes
From these beta regression coefficients, a weighted score for the risk factors was created. The assignment of points to these risk factors can be seen in Table 2. Diabetes mellitus had the highest β regression coefficient of 1.37 and was given the risk score of 3. Age (≥65 years) had a β regression coefficient of 0.56 and was given a risk score of 1. Race (non-white) had a β regression coefficient of 0.75 and was given a weighted score of 2. Hypertension had a β regression coefficient of 1.02 and was given a weighted score of 2. BMI ≥30 had a β regression coefficient of 0.78 and was given a weighted score of 2. AFib had a β regression coefficient of 0.98 and was given a weighted score of 2. The use of steroid medications had a β regression coefficient of 1.23 and was given a weighted score of 3. The use of SSRI medications had a β regression coefficient of 0.46 and was given a weighted score of 1. The use of opioid medications had a β regression coefficient of 0.76 and was given a weighted score of 2. The presence of visual aura had a β regression coefficient of 0.81 and was given a weighted score of 2. Duration of migraine (<5 years) had a β regression coefficient of 0.67 and was given a weighted score of 1. The proposed MARS+ score for each participant was calculated combining these weighted scores and ranged from 0–21. Based on the deduced MARS+ score the risk of ischemic stroke was 3.7% in the low-risk group (MARS+ = 0–4) and 11.7% in the high-risk group (MARS+ ≥5) over a period of 25 years.
Model assessment
Analyses of MARS+ risk groups revealed a steep increment of HR from 2.70 (third MARS+ quartile) to 6.19 (fourth MARS+ quartile) (Figure 2). Discrimination analysis of MARS+ risk groups via AUC analysis revealed an AUC of 0.285 confirming cutoff at ∼5 (Table 3). Using the MARS+ score, a prognostic index was developed. This index placed participants into two groups according to which risk factors they report. The weighted score range of these groups can be seen in Table 2. The high-risk group includes participants with a risk score of ≥5 points, whereas the low-risk group participants have a risk score of 0–4 points. Further discriminative indicators such as sensitivity and specificity can be seen in Table 3. MARS+ performance was compared to the Framingham risk score (3). Receiver operating characteristics analyses compared sensitivity, specificity and model performance of the two risk scores. Our risk score, MARS+, demonstrated an AUC value of 0.69 with a model performance of 0.64 compared to the Framingham model, which demonstrated an AUC of 0.67 and a model performance of 0.62.

MARS+ hazards ratio (HR) forest plot. Quartile 1 (MARS+ 0–2): Reference; Quartile 2 (MARS+ 3–4): HR = 1.00, 95% confidence interval (CI) = 0.48–2.08; Quartile 3 (MARS+5–7): HR = 2.70, 95% CI = 1.50–4.87; Quartile 4: (MARS+ 8–21): HR = 6.19, 95% CI = 3.55–10.80.

(a) Receiver operating characteristic (ROC) curve for MARS+. (b) Receiver operating characteristic curve for the Framingham risk score.
Youden's index.

The test result variable(s): MARS+ has at least one tie between the positive actual state group and the negative actual state group.
The smallest cutoff value is the minimum observed test value minus 1, and the largest cutoff value is the maximum observed test value plus 1. All the other cutoff values are the averages of two consecutive ordered observed test values.
The participants from the migraine with or without aura cohort were categorized into the two risk groups for a survival analysis of ischemic stroke. The number of participants in the high-risk group is 709 and the number in the low-risk group is 776. The number of participants in the high-risk group who suffered an ischemic stroke was 83 and the number of participants in the low-risk group who suffered an ischemic stroke was 29. The HR for ischemic stroke in high risk (vs. low risk) was 4.09. The Kaplan–Meier survival curves of the two risk groups are shown in Figure 4. These curves showed significant separation of the survival curves based on the risk stratification groups (log rank p ≤ 0.001). Because death from any cause would be a competing risk event for ischemic stroke, we report a cause specific HR that included death as an additional event type in Table 4,

Kaplan–Meier survival curves for the development cohort according to the prognostic classification.
Hazard ratio (HR) and 95% confidence interval (CI) for the association of high risk (MARS+ ≥5), incident ischemic stroke and death (all-cause mortality).

Discussion
In the present study, we used a large prospective cohort to investigate the relationship between a multitude of risk factors, migraine with and without visual aura, and ischemic stroke. We then used risk analysis to create weighted scores for each of the risk factors to create the MARS+ risk score for patients with migraine with and without aura. The score developed had a range from 0–21, with a higher score translating to a higher risk of ischemic stroke risk, and a higher HR compared to MARS+ = 0. Finally, we determined that patients with a MARS+ risk score of ≥5 had a higher 25-year risk of ischemic stroke compared to patients with a MARS+ risk score of 0-4. We were also able to show that visual aura was a significant risk factor contributing 2 points to the weighted score.
Previous studies have shown migraine with aura is associated with increased risk of ischemic stroke (4,17). The presence of aura has been shown to be strongly associated with stroke/transient ischemic attack symptoms and ischemic stroke events for both migraine and other headaches in the ARIC cohort (8). Meta-analysis shows that the pooled relative risk of ischemic stroke for people with migraine with aura is significantly increased compared to people with migraine without aura and our study further shows visual aura as a significant risk factor (4).
Although the increased risk of stroke with migraine with aura has been established, the etiology remains unclear. Recent ARIC studies have shown an increased risk of AFib and cardioembolic ischemic stroke among those with migraine (with aura), which may play an important role to link this association (15). Additionally, autonomic dysfunction could increase risk of stroke (18,19). Associations have been found between low heart rate variability, a marker of cardiac autonomic dysfunction, and AFib and stroke risk (20). It is possible that autonomic dysfunction predisposes to AFib (19), which predisposes one to cardioembolic ischemic stroke. Additionally, studies have shown that those with migraine have a higher prevalence of patent foramen ovale (PFO), and it has been hypothesized that this could link migraine with stroke (21). Lastly, there are also vascular components of migraine that could impact patients. Endothelial dysfunction, hypercoagulability and pathological vascular reactivity are found in patients with migraine. These factors could provide the link between migraine with aura and ischemic stroke.
In addition to these factors, medications used by those who have migraine with or without aura may contribute to stroke risk reflected in MARS+. In particular, SSRIs, steroid use, and opioid use were each associated with increased ischemic stroke risk in our analysis. For example, SSRI are vasoactive agents and have been linked to reversible cerebral vasoconstrictive syndrome (22). Although their independent contribution to stroke risk remains debated, their use in combination with migraine or other vascular risk factors may warrant further investigation.
Steroid use was also found to be a significant risk factor contributing to our risk score. Chronic steroid use has been associated with increased risk of vascular risk factors such as HTN and DM (23). While this alone may largely be the reason use of steroid medications is a significant risk factor in our score, there may be underlying confounders such as why the patient is on steroid medication. For example, if the patient is taking these medications for underlying autoimmune disease, this could also contribute to increased risk of ischemic stroke.
Another interesting medication found to be a significant risk factor in our risk score is opioids. Opioid use may increase the risk of ischemic stroke, potentially through opioid receptor activity in the cardiovascular system and disruption of the blood–brain barrier, with one study finding more than twice the risk compared to non-steroidal anti-inflammatory drug use (12).
Prior studies have not evaluated risk factors specific to migraine with and without aura patients that could increase their risk for ischemic stroke. In the present study, we focused on the increased risk associated with specific risk factors within a migraine cohort, such as visual aura, duration and frequency of migraine headaches. Specifically, we found that migraine duration <5 years is a significant risk factor for incident stroke. Risk factors that predispose patients for migraine with aura can also be risk factors for ischemic stroke such as DM, HTN and PFO. Through the use of the risk analysis, our study provided insight into which risk factors create a more substantial risk for migraine patients. By analyzing the comorbidities of migraine patients, our study goes beyond determining the association between migraine and ischemic stroke. Additionally, the use of a weighted risk score gave us the ability to determine how the presence of multiple risk factors can create an increased risk of ischemic stroke. This can be employed to determine the specific risk of each migraine patient based on their combination of risk factors. The design of our study gave us the ability to determine how specific risk factors affect the migraine population and how a combination of studies affect an individual migraine patient.
There are several risk scores available for ischemic stroke. The CHA2DS2-VASc score (24) is useful for assessing the risk of ischemic stroke in patients with AFib. Similarly to our risk score, CHA2DS2-VASc assigns points based on various stroke risk factors and patients are placed into a “low” or “high” risk group. This risk score employs a similar method to our study as it assesses the stroke risk within a patient population. Additionally, it places the patients into different risk groups, allowing the risk score to be individualized for each patient. The Framingham stroke score is used to determine a patient's probability of stroke within 10 years based on several stroke predictors. Points are assigned based on the stroke predictors the patient reports, and each score is correlated with a percentage risk of stroke within 5 or 10 years. Both risk scores are very useful predictors for stroke risk. Their approach and use of risk factors share similarities with our study. However, there is no risk score for evaluating the risk of stroke in patients suffering from migraine. Our model is unique in that it assigns points to various risk factors based on how these risk factors predispose migraine patients to ischemic stroke. Although our study uses similar techniques, such as similar stroke risk factors or the use of a weighted score and risk groups, the use of the migraine patient population distinguishes our study from other stroke risk scores. Our study shows the first implementation of a risk model for migraine patients.
Additionally, a study titled “Self-Reported Stroke Risk Stratification” (25), compared self-reported, non-invasive/blood-less risk factors for stroke and compared their risk score to the Framingham risk score. Per this study, the data collected with this self-reported survey was able to identify those in the population without prior stroke but who were at risk for stroke with reported reliability better than that of the Framingham risk score (25). This further adds to the validity that a self-reported risk score, such as MARS+, could provide reliable data to assess for stroke risk. This could be a distinct contribution over the Framingham risk score, which continues to use laboratory and clinical measures.
There are limitations to our study. First, the risk score found in this study has not been validated with a different cohort for external validation. Validation with another cohort presents challenges due to other cohorts lacking covariate data found in the ARIC study. However, we have performed internal validation by comparing MARS+ with the widely used Framingham risk score used to identify stroke risk in the general population. Unlike MARS+, the Framingham risk score is based on laboratory testing and clinical assessment. MARS+ is able to predict stroke risk in this specific cohort by asking clinical questions as done in the ARIC study. These results lend to the validity of MARS+, as well as its generalizability to migraine patients.
Also, regarding limitations, given the nature of this longitudinal study that started in 1987, the medical context regarding migraine medications has changed overtime. This includes increased use of medications such as triptans, as well as the addition of calcitonin gene-related peptide medications, which is not reflected in our study.
In addition, the ARIC database lacks the availability of data on PFO, which has been attributed to be a covariate that may predispose migraine and ischemic stroke (21). A recent observational study reported that migraine with aura had a strong association with cryptogenic ischemic stroke in young patients with the presence of PFO and independent of vascular risk factors. In the routine clinical setting, assessment of PFO is not routinely advocated for in the general population without a history of stroke. Thus, incorporation of PFO would require additional routine testing not performed in the migraine population. Nevertheless, PFO may be an important covariate to factor into a migraine and stroke risk score. Another limitation of the study is how the migraine classification used in the ARIC study is susceptible to recalling bias. This is because the classification is based on a questionnaire and was administered to participants in middle life. Additionally, migraine with aura symptoms only included visual aura and may have inadvertently excluded migraine with sensory, motor (hemiplegic migraine) or brainstem aura due to the 1995 design of the headache questionnaire. We do not expect this to significantly alter our findings as between 82% and 90% of patients who have migraine with aura experience visual aura (26). Also, the modified ICHD-3 (beta version) criteria used to be more stringent and it is possible that we may have classified a few patients with migraine who presented with bilateral headache or lasted less than one year as the non-migraine headache group. We do not expect this to be biased in respect to association with stroke, and thus it would not necessarily change results. Also, unlike stroke diagnosis, headache diagnosis in the ARIC cohort has not been adjudicated by a headache specialist. The ARIC cohort utilized in the development of MARS+ score is limited to middle-aged white and African-American residing in the USA, and hence will need to be tested in other races and region to see if it is applicable worldwide.
Our mean age of 58 years could also be considered a limitation. While migraine with aura is an established risk factor for ischemic stroke in young adults, particularly women <45 years of age, the association in middle-aged and elderly populations remains controversial and requires further investigation (27). While some recent analyses suggest migraine with aura may also confer elevated risk into middle age, the evidence remains inconclusive and continues to be debated. Furthermore, our previous analysis in the ARIC cohort revealed a paradoxical finding where late-onset migraine with aura (≥50 years) with shorter duration was associated with higher stroke risk compared to early-onset migraine with longer cumulative exposure (9). The lack of association with longer duration may reflect several factors, including survival bias, evolving migraine characteristics with age and the predominance of traditional cardiovascular risk factors over migraine-related mechanisms in older populations (28). These findings underscore the complexity of migraine-stroke associations across the lifespan and suggest that age of onset and current age may be more important determinants of stroke risk than cumulative migraine exposure in middle-aged and elderly patients. The medical landscape, diagnostic criteria and treatment modalities for both migraine and stroke have evolved significantly since the 1990s. Consequently, the risk factors and patient profiles relevant at that time may not entirely align with the current understanding and characteristics of the conditions.
Despite the above limitations, the present study shows strong promise for the use of an ischemic stroke risk score in clinical practice. This study is one of the first to use a risk score to assess stroke risk of patients suffering from migraine. The implementation of this risk score in a clinical setting would be beneficial for clinicians counseling patients suffering from migraine on their individual stroke risk. It would allow clinicians to prescribe preventative treatment or lifestyle changes to patients based on their risk score, thus reducing the risk of ischemic stroke in these patients. For the future of this study, repetition with another population cohort that includes migraine with and without aura and incident ischemic stroke is an essential step before implementing this risk score in clinical practices. The strengths of the study include the prospective study design, long follow-up period, large population cohort and the use of generally available clinical information that allows easy risk stratification. Additionally, migraine and ischemic stroke were ascertained with current diagnostic criteria.
In conclusion, we report a newly designed simple risk score based on simple clinical criteria to predict stroke risk in those with migraine with and without aura. Once validated, our findings can be useful to clinicians for predicting stroke risk in patients suffering from migraine and directing preventive therapy. Migraine patients with a MARS+ score of ≥5 may be counseled by physicians about the additional stroke risk and measures that may be used to reduce the risk.
Article highlights
MARS+ score predicts ischemic stroke risk in migraine patients using vascular factors, migraine characteristics and medications.
Score includes age ≥65 years, non-white race, HTN, DM, BMI ≥30, AFib, opioids, steroids, SSRIs and aura.
MARS+ ≥5 indicates high lifetime stroke risk with HR = 4.09 (95% CI = 2.67–6.26).
Validated in 1485 ARIC cohort participants with migraine; 112 experienced ischemic stroke.
Footnotes
Data availability
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: No potential conflict of interest to be reported by the authors.
Ethical statement
Written informed consent was obtained from all participants for the ARIC study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Atherosclerosis Risk in Communities study has been funded in whole or in part with Federal funds from the National Heart, Lung and Blood Institute, National Institutes of Health, Department of Health and Human Services, under Contract nos. (HHSN268201700001I, HHSN268201700002I, HHSN268201700003I, HHSN268201700005I, HHSN268201700004I). Test role of heart rate variability (HRV) in predicting atrial fibrillation and ischemic stroke in migraine. This was funded by AHA 23MRFSCD1077176.
