Abstract
Objective
To investigate the intranetwork resting state fMRI connectivity within the Salience Network of chronic migraine with and without medication overuse headache.
Methods
We compared 351 pairs of intranetwork connectivity in chronic migraine (n = 13) and chronic migraine with medication overuse headache (n = 16) compared to matched controls, and between each chronic migraine subgroup.
Results
Compared to controls, 17 pairs of intranetwork connections in chronic migraine and 27 pairs in chronic migraine with medication overuse headache were decreased. When comparing chronic migraine with medication overuse headache versus chronic migraine, connectivity between bilateral extended amygdala, and between paracingulate to right ventral tegmental area/substantia nigra were decreased in chronic migraine (chronic migraine < chronic migraine with medication overuse headache). Connectivity between left dorsolateral prefrontal cortex to bilateral ventral striatum/pallidum, to bilateral dorsal anterior cingulate cortex; left anterior prefrontal cortex to contralateral orbitofrontal insula; and left ventral striatum/pallidum to ipsilateral supplementary motor area (SMA)/preSMA were decreased in chronic migraine with medication overuse headache (chronic migraine with medication overuse headache < chronic migraine).
Conclusion
Both chronic migraine subgroups had shared intranetwork connectivity abnormality, however, each subgroup had unique pattern of disruption within the salience network. The results suggest that the aberrant assignment of salience to external and internal stimuli plays an important role in chronic migraine and chronic migraine with medication overuse headache interictally, mostly involving mesolimbic pathways (especially bilateral extended amygdala) in chronic migraine, and prefrontal-subcortical limbic pathways in chronic migraine with medication overuse headache.
Introduction
Despite the prevalence and comorbidities of chronic migraine (CM) and CM with medication overuse headaches (MOH), the underlying mechanisms remain poorly understood (1–5). With recent advances in fMRI, changes in the functional connectivity of brain networks can be investigated. Many of the current neuroimaging studies in CM versus CM with MOH (CMMOH) were performed using task activated fMRI (6–8). During a risk decision task, decreased activation in the ventral tegmental area and substantia nigra (VTA/SN) was demonstrated in CMMOH compared to CM (9). Two recent studies found altered regional functional connectivity in episodic migraine and CM, focused on the amygdala and marginal division of the neostriatum (10,11). However, there is a paucity of research on how the functional connectivity of intrinsic networks is affected in CM and CMMOH. Given that the brain is a closely integrated system, and chronic pain is a multi-dimensional dynamic process, using established brain networks to evaluate patterns of functional neural disruption may be a unique approach in understanding the pathogenesis of CM and CMMOH.
Our recent investigation revealed that, within the salience network, there was an overall decrease in network connectivity strength in both CM and CMMOH; however, we found no difference between each group (16). The salience network is a well-defined, intrinsically correlated baseline brain network important for regulating salience-related information via integrating sensory, emotional, and cognitive inputs (12,13). It is considered to be a filtration and amplification network responsible for evaluating specific external/internal stimuli, and assigning the relevance of the stimuli for continuous processing and modulation of pain, as well as guiding goal-directed behavior (14,15). Given migraine is a complex neurological disorder affecting not only the sensory (painful sensation) but also the emotional, cognitive aspect of pain awareness, evaluation of the salience intranetwork connectivity may shed light on the key brain regions critically dysregulated in CM versus CMMOH.Currently, there is no report on the salience network intranetwork resting state functional connectivity comparing CM versus CMMOH.
The purpose of this study is to investigate the intranetwork interictal resting state functional connectivity within the salience network in CM and CMMOH. We hypothesized that the diminished overall connectivity of this network would be attributed to particular node to node connectivity (NTNC) disruptions within the mesocorticolimbic system, deferentially affected in CM and CMMOH. Additionally, we hypothesized that there would be a more widespread NTNC disruption in CMMOH than CM.
Methods
Participants
Women were eligible for the study if they were 18 years old or older, met diagnostic criteria fulfilling International Classification of Headache Disorders III beta for CM as determined by a headache specialist, or were non-pain, non-headache controls. CM subgroups were characterized based on medication overuse status (with or without) as per International Classification of Headache Disorders III beta diagnostic criteria (17). We have carefully selected our CMMOH patients to ensure that they started with episodic migraine decades ago, then developed CM due to overuse of acute pain medications based on our clinical assessment by a certified headache specialist. Participants were excluded if they had MRI contraindication, neurological or pain disorders other than CM, any chronic illness (i.e. hypertension, diabetes, hepatic, renal, chronic inflammatory, or infectious disease), or inability to follow study protocol while completing assessments. The healthy controls were selected to match the age and gender of the CM participants. Controls were excluded if they had a family history of migraine or used over the counter/prescription pain medication for more than 5 days per month. CM participants were scanned at their baseline level of pain, at least 24 hours outside of their acute pain exacerbation period; any participant who came in within 24 hours of acute pain exacerbation was rescheduled.
All participants underwent vital sign evaluation including BMI, a neurological examination, and completed a standardized questionnaire to ascertain demographics including age, sex, race, and educational level as well as clinical characteristics including: (a) duration of migraine history; (b) duration of CM history; (c) family history of migraine; (d) current medications; (e) number of moderate to severe headache days per month; (f) location of migraine; (g) presence of aura; (h) headache-related disability, as determined by Headache Impact Test (HIT-6); (i) depression, as determined by the Patient Health Questionnaire (PHQ-9); (j) allodynia, as measured by the allodynia symptom checklist (ASC 12) (18).
HIT-6 and PHQ-9 scale
The HIT-6 is a validated questionnaire that consists of six items reflecting quality of life. Higher scores (range of 36–78) indicate an increasing impact of headaches on daily functioning (19). The PHQ-9 is a diagnostic measure for clinical depression. A score of ≥15 on the PHQ-9 is associated with a 68% sensitivity and 95% specificity for diagnosing “major depressive disorder” based on the DSM-IV criteria (20).
Standard protocol approvals, registrations, and patient consents
The study protocol was approved by institutional review board at University of South Carolina. Written informed consent was obtained from all participants.
MR imaging
All participants were scanned on a Siemens 3T scanner located at the McCausland Center for Brain Imaging (Columbia, South Carolina). Participants were instructed to keep their eyes closed, stay awake, relax, and think of “nothing in particular” during the resting state scan. All conditions and lighting were consistent throughout the entire study for all participants. Some participants (five CM and five controls) were scanned prior to a system hardware upgrade; however, any variance due to this upgrade was controlled for in our analysis by adding this as a nuisance regressor variable to the general linear model (GLM) using the Freedman-Lane approach (21).
The imaging parameters for the Trio (12 channel head coil) system consisted of a 6 minute high-resolution T1 weighted magnetization-prepared rapid gradient echo (MP-RAGE) scan (repetition time [TR] = 2250 ms, echo time [TE] = 4.15 ms, 192 slices, 50% slice gap, flip angle = 9°, voxel size = 1.0 mm3, Field of View [FOV] = 256 mm2, iPAT factor of 2, and using a sagittal, ascending acquisition) and a 15 minute resting state functional imaging scan using a T2* weighted BOLD contrast-sensitive sequence ([TR] = 1550 ms, [TE] = 34 ms, 42 slices, 20% slice gap, flip angle = 71°, voxel size = 2.5 mm3, FOV = 215 mm2, and using a transversal, descending acquisition).
The imaging parameters for the Prisma (20 channel head coil) system included an acquisition of 6 minute high resolution T1 weighted MP-RAGE scan (same parameters as Trio, except that TE = 4.11 ms) and a 15 minute resting state functional imaging scan using a T2* weighted BOLD contrast-sensitive sequence (TR = 1100 ms, TE = 35 ms, 56 slices, 20% slice gap, flip angle = 72°, voxel size = 2.4 × 2.4 × 2.0 mm3, FOV = 216 mm2, and using a transversal acquisition).
MRI preprocessing
Rs-fMRI preprocessing was completed using a combination of Statistical Parametric Mapping 12 (SPM12) software and custom MATLAB scripts (https://github.com/neurolabusc/nii_preprocess). The pipeline consisted of standard procedures including motion correction, coregistration, normalization, frequency filtering (0.01 to 0.1 Hz bandpass), and spatial smoothing (8 mm full width at half maximum). For each network, a connectivity atlas was constructed using spherical (15 mm diameter) regions of interest (ROIs) centered on the peak Montreal Neurological Institute (MNI) coordinates for the a priori network of the salience network (12). The salience network (Figure 1) includes the bilateral dorsal anterior cingulate cortex (dACC), bilateral dorsolateral prefrontal cortex (dlPFC), left anterior PFC (aPFC), bilateral hypothalamus, bilateral orbital frontal insula (OFI), paracingulate cortex, bilateral extended amygdala (BA 34), bilateral supramarginal gyrus (SMG), left periaqueductal gray (PAG), bilateral supplementary motor area (SMA)/pre SMA, bilateral ventral tegmental area/substantia nigra (VTA/SN), bilateral superior temporal, bilateral temporal pole, bilateral ventral striatum/pallidum (vSP), and right ventrolateral PFC (vlPFC). The ROIs (nodes) and their MNI coordinates used in the salience network are reported in the supplemental Table 1.
Axial and sagittal view of the salience network. Locations: (1) Left dlPFC, (2) Right dlPFC, (3) Right vlPFC, (4) Left anterior PFC, (5) Left OFI, (6) Right OFI, (7) Left temporal pole, (8) Right temporal pole, (9) Left SMA/preSMA, (10) Right SMA/preSMA, (11) Paracingulate cortex, (12) Left dACC, (13) Right dACC, (14) Left extended amygdala, (15) Right extended amygdala, (16) PAG, (17) Left hypothalamus, (18) Right hypothalamus, (19) Right dorsomedial thalamus, (20) Left VTA/SN, (21) Right VTA/SN, (22) Left vSP, (23) Right vSP, (24) Left superior temporal, (25) Right superior temporal, (26) Left SMG, (27) Right SMG. Node depth is illustrated by transparency. Images were made with “Surf Ice” (https://www.nitrc.org/projects/surfice/) using the exact MNI coordinate locations. Demographic and clinical features (CM subgroup only) of subjects based on all subgroups. Values represent mean ± standard deviation or number (% of total). Significance indicated by asterisks. Abbreviations: CM: chronic migraine; CMMOH: Chronic migraine with medication overuse headache; C: controls; BMI: body mass index; HA: moderate to severe headache days; ASC-12: allodynia symptoms checklist; HIT-6: headache impact test; PHQ-9: patient health questionnaire-9.

Statistical analysis
Using the generated ROIs (“nodes”), functional connectivity matrices were created by extracting the mean BOLD time course from each node in the salience network, and Pearson correlation (r) coefficients were calculated between each of those nodes. Next, Pearson correlation coefficient values were Fischer’s Z transformed (to convert to a normal distribution) to produce the “functional connectivity strengths” that are used in the analysis. Comparison groups were formed to investigate the relationship between the following groups: CMMOH versus controls, CM versus controls, and CMMOH versus CM. The intranetwork NTNC (also referred to as “edge”) strengths are defined as the correlation coefficient generated between each pair of ROIs.
When comparing each subgroup to their matched controls, we used a one tailed, two sampled t-test. When comparing between the CM subgroups (CMMOH vs. CM), a two tailed, two sampled t-test was used. Corrected p values of <0.05 were considered significant for intranetwork NTNC strength differences (CMMOH vs. CM). Corrected p values of <0.01 were considered significant for intranetwork NTNC strength differences (CM subgroups vs. controls). A larger threshold was used when comparing between the two CM groups, because differences may be subtle compared to the CM versus controls. All statistical tests were conducted using permutation thresholding (10,000 permutations) to control for multiple comparisons (22).
To assess associations between intranetwork functional connectivity strength and clinical parameters, Pearson correlations were conducted between the shared significant intranetwork NTNC strengths and clinical parameters, including years with migraine, years with CM, number of moderate to severe headache per month, PHQ-9, HIT-6, and allodynia symptoms checklist scores (ASC 12). Correlations were corrected for multiple comparison using the Bonferroni correction (n = 351; p = 0.00014).
Results
Participants
Differences of intranetwork connections in the salience network for each CM subgroup as compared to controls.

These intranetwork connections are similarly disrupted in both CM subgroups.
Values represent the average Fischer Z transformed correlational efficiency (“Functional connectivity strength”) between the two stated nodes ± standard deviation.
Abbreviations: CM: chronic migraine; CMMOH: Chronic migraine with medication overuse headache; dm: dorsomedial; dACC: dorsal anterior cingulate cortex; dlPFC: dorsolateral prefrontal cortex; aPFC: anterior PFC; OFI: orbital frontal insula; SMG: supramarginal gyrus; SMA: supplementary motor area; VTA/SN: ventral tegmental area/substantia nigra; vSP: ventral striatum and pallidum; vlPFC: ventrolateral PFC; × = connectivity between the pair of ROIs.

Significant intranetwork connectivity difference in salience network between CMMOH versus controls (top) and CM versus controls (bottom). Colored line represents a significant intranetwork connectivity difference (respectively) between the CM groups versus their matched controls. Green represents a unique difference to that comparison group and blue represents a shared difference in both comparison groups. Colored bars represent limbic associations: Blue = corticolimbic, green = limbic, and purple = paralimbic. Images were generated using the Matlab application “Circro” (https://github.com/bonilhamusclab/circro). Abbreviations: CM: chronic migraine; CMMOH: chronic migraine with medication overuse headache; dm-: dorsomedial; PFC: prefrontal cortex; SMA: supplementary motor area; SN/VTA: substantia nigra/ventral tegmental area.
Differences of intranetwork connections when comparing CMMOH versus CM.
Intranetwork connection was found to be significant in CMMOH versus control group.
Intranetwork connection was found to be significant in CM versus control group.
Values represent the Fischer Z transformed correlational efficiency (“Functional connectivity strength”) between the two stated nodes ± standard deviation.
Abbreviations: CM: chronic migraine; CMMOH: chronic migraine with medication overuse headache; dACC: dorsal anterior cingulate cortex; dlPFC: dorsolateral prefrontal cortex; aPFC: anterior PFC; OFI: orbital frontal insula; SMA: supplementary motor area; VTA/SN: ventral tegmental area/substantia nigra; vSP: ventral striatum and pallidum; vlPFC: ventrolateral PFC; ×: connectivity between the pair of ROIs.

Significant intranetwork connectivity difference in CMMOH compared to CM in the salience network. Colored line represents an intranetwork connectivity difference between CMMOH versus CM. Green represents that the intranetwork connection was lower in CM patients (CMMOH > CM) and blue represents that the intranetwork connection was lower in CMMOH patients (CMMOH < CM). Colored bars represent limbic associations: Blue = corticolimbic, green = limbic, and purple = paralimbic. Abbreviations: CM: chronic migraine; CMMOH: chronic migraine with medication overuse headache; dm-: dorsomedial; PFC: prefrontal cortex; SMA: supplementary motor area; SN/VTA: substantia nigra/ventral tegmental area.
Numerically, we did find increased intranetwork connectivity; however, it did not meet our statistical threshold. We were expecting this, based on our methods, as a “decrease” is best described as dyssynchrony between regions. In a pre-defined network, such as the salience network, if a problem exists within a network, it is more likely to see a “decrease” (or dyssynchrony) of connectivity using this method. In order to see two regions that are significantly “increased”, they would have to be afflicted in the exact same manner and therefore become more synchronized to each other. In other words, to see an “increase”, two regions would have to either be functionally firing together more, or less, at the same time as each other.
Discussion
In this study, we identified the patterns of NTNC disruptions within the salience network in the interictal state of CM and CMMOH. We observed significant desynchronization between right dlPFC to ipsilateral vlPFC and SMG, as well as between right vlPFC to ipsilateral SMA/preSMA regions in both subgroups. Medication overuse did not have any impact on these changes. These findings suggest that the shared aberrant intranetwork connectivity may represent common efferent pathways in CM and CMMOH. Furthermore, both CM and CMMOH presented with disruptions in various regions of the cortico-limbic system. The involvement of the limbic system is not surprising, as it is vital in regulating emotional saliency and pain (23–25). Conventionally, the limbic system consists of mesocortical and mesolimbic pathways. The structures comprising the limbic system overlap with the majority of regions within the salience network, such as ACC, OFI, regions of the PFC, thalamus, hypothalamus, VTA/SN, extended amygdala, PAG, and vSP (26–29). Even though both CM subgroups revealed decreased synchronization in regions of the mesocorticolimbic system, it cannot be assumed that similar disruptions are related to the same pathophysiology. In fact, the differences identified when comparing between subgroups provided more insight into the pathogenesis behind CM and CMMOH.
Salience network intranetwork functional connectivity in CM
One of the most intriguing findings in this study is the prominent desynchronization between the bilateral extended amygdala, and extended amygdala to ipsilateral temporal pole and OFI, in CM compared to controls. Moreover, when comparing between CM subgroups, the disruption between bilateral extended amygdala was an obvious delineator, representing a potential unique type of neuroplasticity in CM that is not seen in CMMOH. The extended amygdala, otherwise known as the sublenticular extended amygdala or subcallosal region (as illustrated in Figure 4), is defined by its neural connections and immunohistochemical staining. This macrostructure (which includes central and medial amygdaloid nuclei, substantia innominata, the nucleus accumbens shell, and bed nuclei of the striaterminalis) plays an important role in regulating nociception, aversive motivational state, reward, memory, and learning (30–32). In animal models, excitotoxic lesions in the central amygdaloid nucleus inhibited aversive responses (33) and administration of NMDA into the amygdala elicited cortical spreading depression (34), implicating that the amygdala may serve as an important link between migraine pain and emotional affect. Given the aberrant increase of functional activity in the amygdala during the ictal stage of episodic migraine (35), it is plausible that the increased frequency of migraine may induce excitotoxicity of the amygdala due to over-sensitization after the transition from episodic migraine to CM in susceptible patients.
Anatomical location (MNI: −28, 4, −18) of left extended amygdala using MRIcron (https://www.nitrc.org/projects/mricron).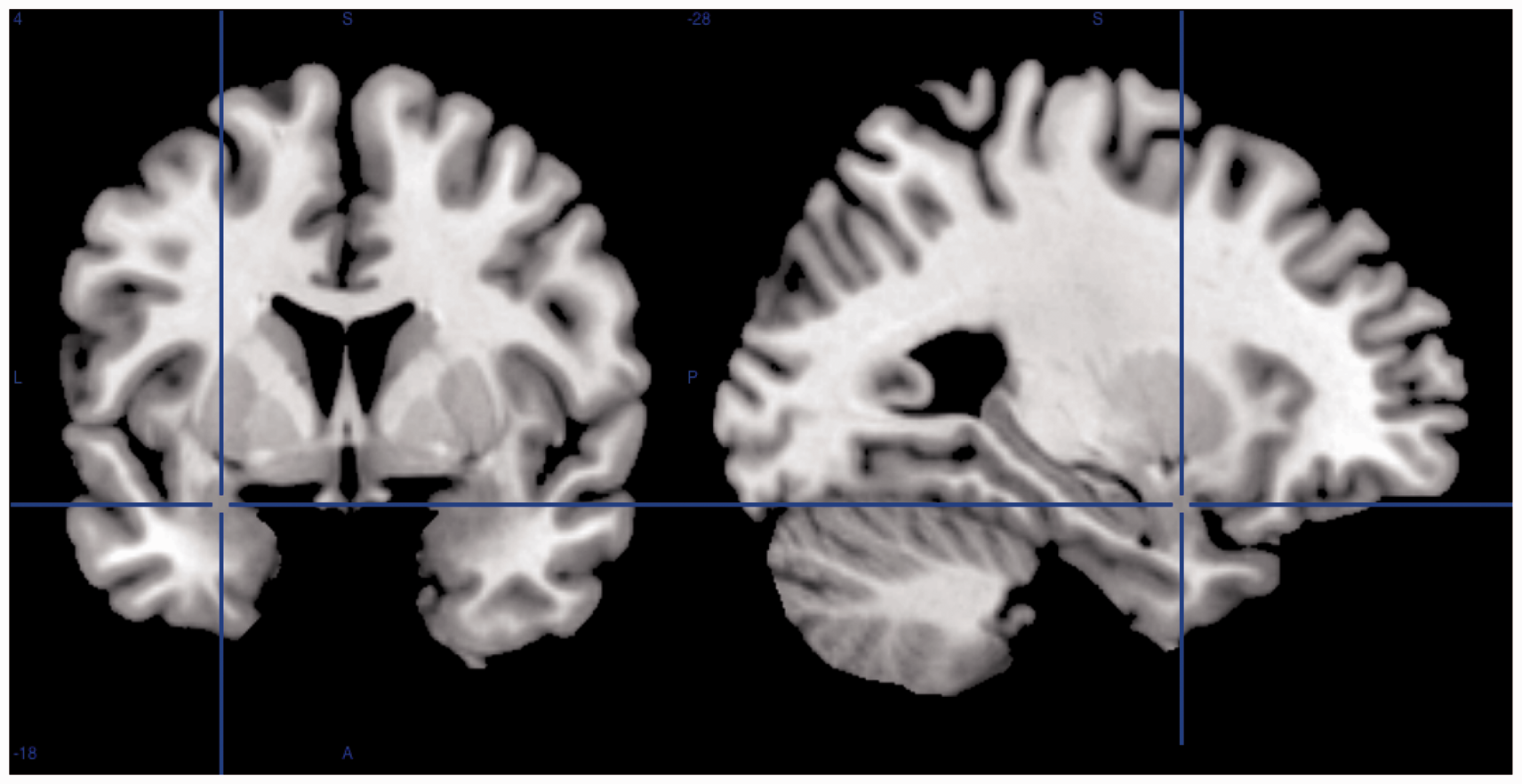
Substantial evidence in structural and functional brain imaging research supports the notion that lack of habituation may be important in migraine pathogenesis (36). In contrast to the basolateral amygdala, which has abundant excitatory neurons, the extended amygdala is rich in GABAergic neurons and endogenous opioids, such as enkaphalins, dynorphin, and endorphins (37,38). Moreover, the extended amygdala has strong interconnections with subcortical (i.e. the thalamus, hypothalamus, and brainstem) and cortical regions (i.e. the auditory, olfactory, and visual cortex) (31,32,39–42). Given the intricate neuroanatomical and neurochemical relationship between the extended amygdala and pain processing/modulation in migraine, we postulate that the decreased functional coactivation of the bilateral extended amygdala contributes to lack of inhibition in pain processing/modulation; that is, the inability to habituate to excessive external or internal stimuli. We speculate that the bilateral extended amygdala serves as the main “hub” for habituation, and deficiencies with this area may result in the misinterpretation of salient input in CM.
Salience network intranetwork functional connectivity in CMMOH
This study revealed widespread disruptions between cortical to limbic structures in CMMOH compared to healthy controls. This is consistent with the findings that recurrent negative reinforcement of overusing analgesic medication triggers metabolic changes in the cortico-limbic systems, which contributes to the development of addictive and compulsive traits, as well as a decreased pain threshold (43–47). These decreased connectivity patterns were not as extensive in the CM subgroup when compared to healthy controls. It is plausible that the more prominent disruption in CMMOH is largely attributed to the consequence of medication overuse. Addiction-related plasticity in the CNS is observed in our CMMOH subgroup, as evident by dysfunctional connectivity involving dlPFC and aPFC, similar to what has been reported (25,43,46,48–51). The left dlPFC appears to be a key region preferentially affected in CMMOH, as decreased functional coactivation was seen predominantly between the left dlPFC to bilateral dACC and vSP. It is conceivable that functional desynchronization of the left dlPFC with other regions of this network contributes to lack of cognitive control in CMMOH and is associated with increased saliency of pain fueled by the negative reinforcement of analgesic medication overuse. Taken together, we observed a “shift in paradigm” in the functional desynchronization of NTNC within the salience network when comparing CM versus CMMOH, reflective of the reorganization in neural resource within the salience network. Specifically, the CM cohort revealed a lack of functional coactivation among mesolimbic structures centered around the bilateral amygdala, whereas similar aberrant connectivity among cortical to limbic structures centered around the left dlPFC was observed in CMMOH.
Strengths and limitations of the study
The strength of this study is our relatively large scale systematic approach in examining intranetwork NTNC (351 pairs) within the salience network. Previous resting state fMRI studies of CM often examined focal regions of interest and their relationship with the whole brain, rather than connectivity changes. The methodology used in this project allowed us to understand how CM may affect this intrinsic network important for the evaluation and interpretation of salient information in chronic migraneurs. In this cohort, we did not include any patients who started out with severe intractable headaches with prolonged use of acute pain medications. This type of patient does exist, but was not included as part of our study. Therefore, the difference we saw in CM vs CMMOH is suggestive of disruption of the reward/addiction pathway due to overuse of medications, essentially rewiring the brain circuits of the chronic migraineurs. We also acknowledge that there are several limitations to this study. We cannot be certain that the results are consequences or causes of CM (predisposition to CM); however, this is a common limitation in similar studies. We cannot be certain that the results are free from confounders; we did attempt to minimize any potential bias. Some CM participants (n = 15) were on daily migraine prophylaxis medications, but we do not believe this impacted our results based on our previous analysis (16). Participants were not matched exactly according to the anxiety/depression scale, therefore we cannot exclude the possibility that some of the changes in connectivity may be related to anxiety/depression. The prominent desynchronization in the bilateral amygdala observed in CM is unlikely to be attributed to mood disorders, given higher PHQ-9/HIT 6 in CMMOH. Furthermore, using Pearson’s correlation, we did not find PHQ-9 to be associated with any of the intranetwork NTNC connections in the salience network. Additionally, differences in using acute pain medications (i.e. type, dosing) may impact the functional connectivity. Lastly, even though we did not find any correlation to clinical characteristics, this may be caused by using multiple comparison data in Pearson analysis, this is known to be associated with a higher type II error rate (false negative). A larger sample size is needed to examine the correlational relationship of clinical parameters to NTNC changes using linear regression and multiple comparisons.
In contrast to the whole brain analysis used to investigate the regional reorganization, our methods are based on a pre-defined network with the intention of using this specific network as a “standard” to examine whether the underlying changes are related to CM. We believe a network approach is more appropriate when studying a complex neurological disorder such as CM and CMMOH. In addition, it is premature to suggest that increased connectivity (that is, rs-fMRI connectivity, not task-based connectivity), or decreased connectivity can be extrapolated to hyperexcitability or hypoexcitability states. Hopefully, new methodology in fMRI in the near future can address this issue. Future studies would benefit by utilizing Granger causality to predict directionality (52).
Footnotes
Clinical implications
We suggest that the aberrant assignment of salience to external and internal stimuli plays an important role in CM and CMMOH interictally, mostly involving mesolimbic pathways (especially centered on the bilateral extended amygdala) in CM and prefrontal-subcortical limbic pathways in CMMOH.
The aberrant NTNC has the potential to be used as a potential target for therapeutic intervention, or as a non-invasive marker for evaluating clinical outcomes in management of CM and CMMOH.
Acknowledgements
The authors would like to acknowledge Taylor Hanayik (University of South Carolina), for his assistance in analysis and interpretation of data, and Charmaine Jenkins (University of South Carolina), for her technical assistance.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
